Abstract
Background:
Idiopathic pulmonary fibrosis (IPF) is associated with a prothrombotic state.
Aim:
To study mean platelet volume (MPV) and Platelet Distribution Width (PDW) as markers of platelet activation and their potential association with lung function in patients with recently diagnosed IPF.
Materials and Methods:
This study included 56 patients with IPF (age 64.9±7.4 years) and 79 controls (age 64.2 ± 5.9 years).
Results:
An inverse relation was demonstrated between platelet count and MPV in the control group but not among patients with IPF. Platelet count was significantly lower in patients with IPF compared with controls (230 ± 60 vs 256 ± 75 × 103/μL, P = .038). Conversely, MPV was higher in patients versus controls (10.3 ± 1.2 vs 9.8 ± 1.2 fl, P = .024), while there was no difference between the groups in PDW. Respiratory function was, as expected, significantly impaired in patients with IPF versus controls in terms of forced expiratory volume in first second (FEV1; 67.2 ± 23.1 vs 102.6 ± 15.9% of predicted value, P < .001), forced vital capacity (FVC; 65.3 ± 21 vs 95.2 ± 16.1% of predicted value, P < .001), FEV1/FVC (83.1 ± 15 vs 87.5 ± 6.4%, P = .041) and partial pressure of oxygen in arterial blood (PaO2; 67.1 ± 10.3 vs 81.5 ± 15.2 mm Hg, P < .001). No significant correlation was seen between MPV and FVC (r = −.1497, P = .275), MPV and lung diffusion capacity for carbon monoxide (r = .035, P = .798) and total lung capacity (r = .032, P = .820).
Conclusions:
Patients with IPF exhibit higher MPV values and lower platelet count. Further studies are needed to assess the clinical implications of these findings.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a rare, debilitating disease, characterized by progressive lung parenchymal scarring, which leads to impaired gas exchange and progressive dyspnea. 1 Prognosis is poor with a 5-year survival comparable with that of many cancers. 2 The underlying pathophysiological mechanisms are poorly understood. 1 Aberrant and incessant proliferation of fibroblasts and abnormal re-epithelization due to a triggering event are considered to play a key pathogenic role. 3
Other mechanisms, studied in animal models, including activation of the coagulation cascade, may also play a contributory role. 4,5 Additionally, population studies report increased incidence of acute coronary syndrome and venous thromboembolism (VTE) among patients with IPF. 6 –8 In tissue samples displaying usual interstitial pneumonia on histopathology, the absence of blood vessels is striking within the fibroblast foci, while marked increase in vascularization is prominent in the adjacent normal tissue. 8,9 Recently, a trial of warfarin in IPF was discontinued due to increased death rate among treated patients that was not related to anticoagulation complications 10 ; this may suggest a positive correlation between anticoagulation and progressive fibrosis. Furthermore, a UK population study clearly documented a prothrombotic state in IPF. 11
We assessed the mean platelet volume (MPV) and platelet distribution width (PDW) as markers of platelet activation and their potential association with lung function (forced vital capacity [FVC], forced expiratory volume in first second [FEV1], FEV1/FVC, total lung capacity [TLC], and lung diffusion capacity for carbon monoxide [DLco]).
Patients and Methods
Study Participants
We included 56 patients with recently diagnosed IPF, according to the American Thoracic Society/European Respiratory Society/Japanese Respiratory Society/Latin American Thoracic Association criteria. 1 The control group consisted of 79 healthy participants, matched for age and body mass index (BMI), none of whom was a current smoker. The study was approved by the local institutional committee, and patients gave their informed consent.
Exclusion criteria were as follows: recent thromboembolic events, hematogical disease, major comorbidities with known association to a prothrombotic state (cancer and active infection), current smoking, and comorbidities requiring antiplatelet or anticoagulant treatment (ie, atrial fibrillation).
Participant Evaluation
Blood samples were collected from antecubital vein of patients and controls so as to perform full blood count analysis. Additionally, arterial blood gas analysis was conducted. (ABL3000, Radiometer Co, Tokyo, Japan) as well as lung function tests (Chest Co, Tokyo, Japan), according to the published guidelines. 12 –14
Both MPV and PDW were measured in EDTA tubes within 30 minutes after sampling by an automated blood cell counter (Sysmex XE 2100; Kobe, Japan). 15,16
The control group consisted of a population without active disease who were evaluated for a health certificate. Data for this group were collected on a retrospective basis, and there were no body plethysmography and single-breath test data.
Statistical Analysis
Statistical analysis was performed using SPSS 17.00 software. Normality was assessed with the Kolmogorov-Smirnoff test, and differences between groups were evaluated by the Student t test. Correlations of MPV and PDW with indices of lung function were evaluated using the Pearson correlation coefficient and linear regression analysis. Data are expressed as mean ± standard deviation, unless otherwise indicated. A 2-tailed P < .05 was considered significant.
Results
A significant inverse relation between MPV values and platelet count was demonstrated by linear regression analysis in the entire study sample (R 2 = .038, β = −.195, and P = .026) and in controls (R 2= .055, β = −.235, and P = .041) but not among patients with IPF (R 2 = .002, β = −.045, and P = .745). Mean platelet count of patients with IPF was significantly lower when compared to controls (230 ± 60 vs 256 ± 75 × 103/μL, P = .038; Figure 1). Conversely, MPV was significantly higher in patients versus controls (10.3 ± 1.2 vs 9.8 ± 1.2 fl, P = .024; Figure 2). Additionally, platelet mass (MPV × platelet count) exhibited no difference between patients and controls (2387 ± 681 vs 2512 ± 708, P = .314). Mean values of PDW did not differ in a statistically significant way between the 2 groups (13.5 ± 2.0 vs 13.3 ± 2.4 fl, respectively, P = .766; Figure 3).
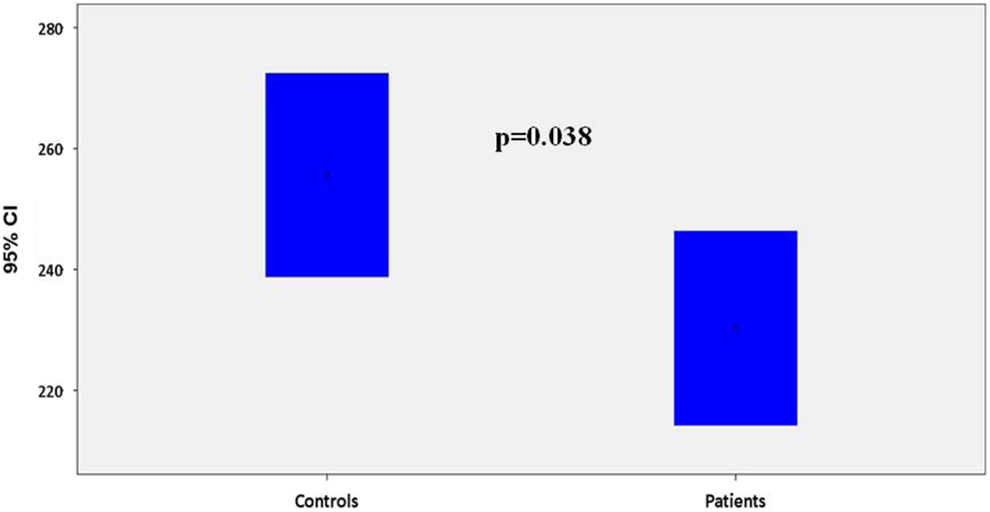
Platelet count (×103/μL) in controls and patients with idiopathic pulmonary fibrosis (IPF; bars represent 95% confidence intervals [CIs]). (bars represent 95% confidence intervals [CIs]).
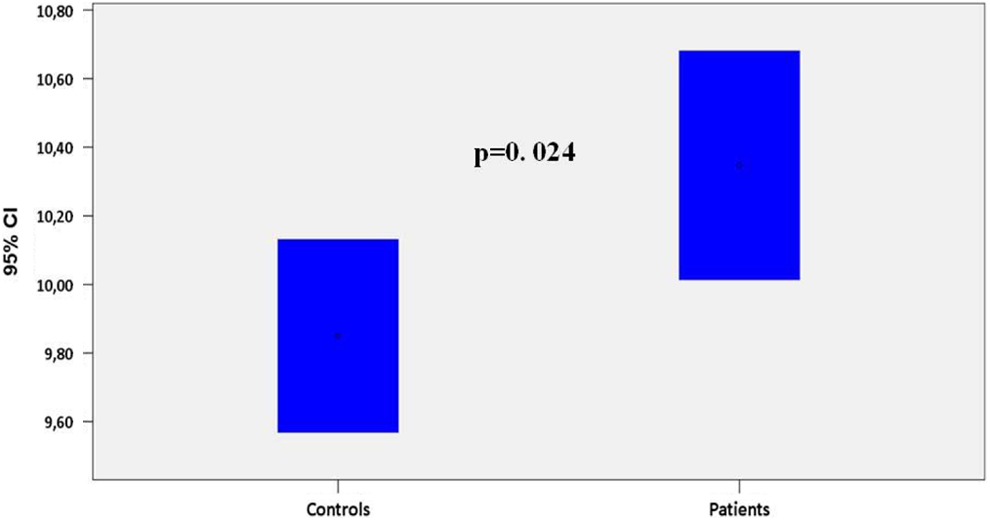
Mean platelet volume
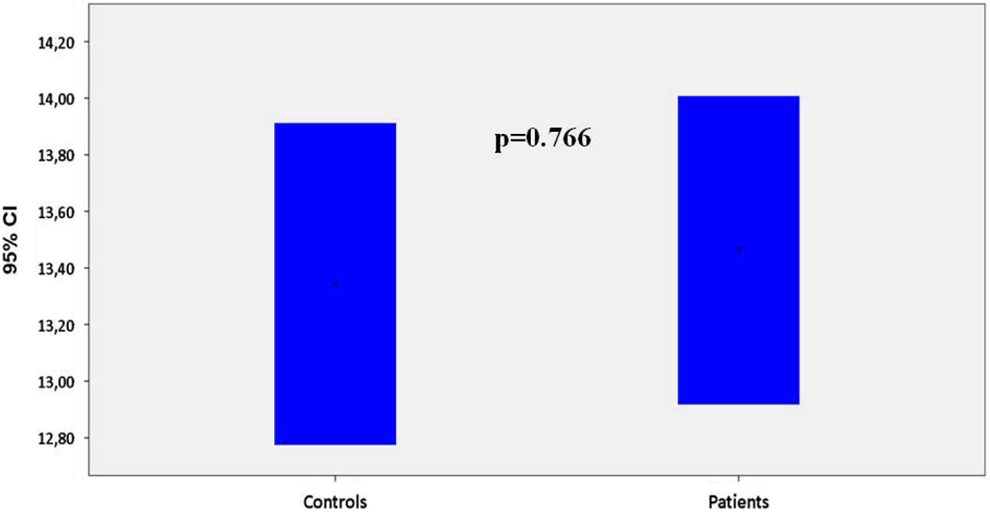
Platelet distribution width (PDW, fl) in controls and patients with idiopathic pulmonary fibrosis (IPF; bars represent 95% confidence intervals [CIs]).
As expected, respiratory function was significantly impaired in patients with IPF versus controls in terms of FEV1 (67.2 ± 23.1 vs 102.6 ± 15.9% of predicted value, P < .001), FVC (65.3 ± 21 vs 95.2 ± 16.1% of predicted value, P < .001), FEV1/FVC (83.1 ± 15 vs 87.5 ± 6.4%, P = .041) and PaO2 (67.1 ± 10.3 vs 81.5 ± 15.2 mm Hg, P < .001). The DLCO and TLC measurements were performed only in patients with IPF, revealing a generally impaired respiratory function (DLCO 36.4 ± 16.3% and TLC 61.8 ± 16.6% of predicted value). Table 1 displays the comparison of lung function tests between patients with IPF and controls.
Epidemiological Characteristics and Lung Function Test in Patients With Idiopathic Pulmonary Fibrosis (IPF) and Controls.
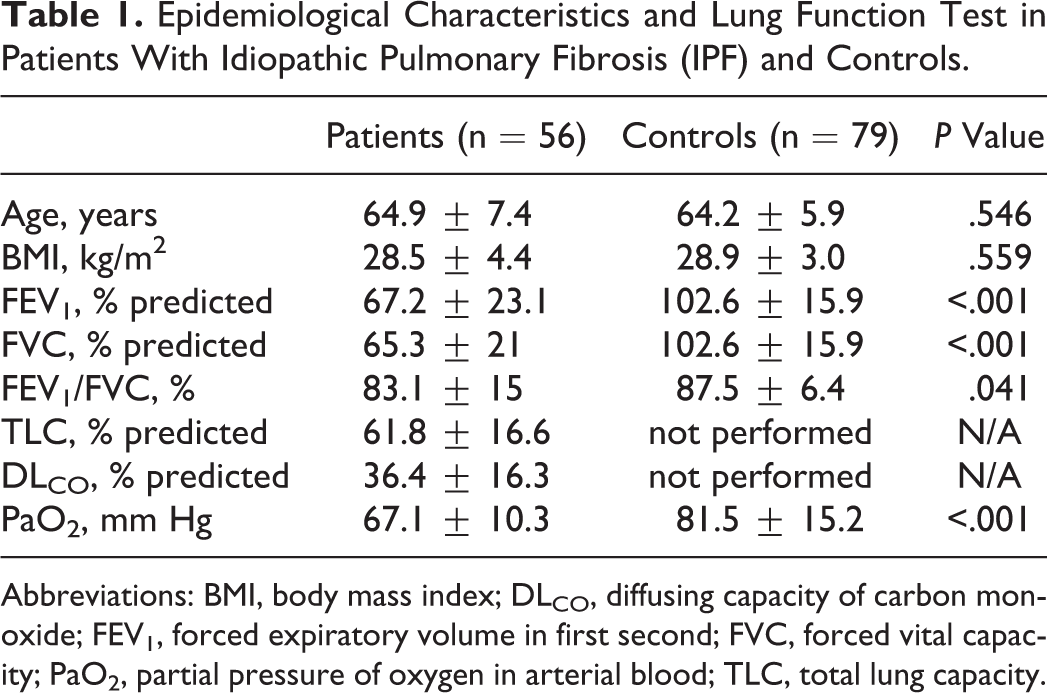
Abbreviations: BMI, body mass index; DLCO, diffusing capacity of carbon monoxide; FEV1, forced expiratory volume in first second; FVC, forced vital capacity; PaO2, partial pressure of oxygen in arterial blood; TLC, total lung capacity.
Finally, no statistically significant correlation was observed between MPV and the following indices: FVC (r = −.1497, P = .275), DLCO (r = .035, P = .798), and TLC (r = .032, P = .820).
Discussion
In comparison with controls, patients with IPF exhibit significantly higher mean MPV value and lower mean platelet count, whereas no difference between the 2 groups is observed in the PDW values. However, there was no correlation between MPV and lung function tests among patients with IPF. To the best of our knowledge, this is the first study evaluating platelet function indices in IPF. Similar findings have been reported in another lung disease, chronic obstructive pulmonary disease (COPD). 15,16 In obstructive sleep apnea, MPV is also increased, but a correlation with parameters of breathing function during sleep was established. 17
The pathogenesis of lung fibrosis in IPF currently remains elusive. The activation of the coagulation cascade is thought to play an important role. 18 Animal models of lung fibrosis have revealed formation of a provisional fibrin matrix in response to increased vascular permeability resulting from microinjuries. 19,20 This could result in epithelial to mesenchymal transition (EMT). 19,20 The EMT might be initially induced by thrombin and coagulation factor X through proteinase-activated receptor 1 induction of transforming growth factor β and sustained by the differentiation of fibroblasts into myofibroblasts. 20,21
There is evidence of both VTE and coronary thrombosis in IPF. 6 –8,23,24 Recently, a UK population study reported that patients with IPF are more likely to have an inherited or acquired clotting defect or fibrinolytic dysfunction, 11 providing evidence of an association between IPF and a prothrombotic state. Arguably, our findings suggesting platelet activation add another piece in the puzzle of hemostatic disorders in patients with IPF.
A prothrombotic state might either result from IPF or be another manifestation of the same underlying pathogenic mechanisms that drive fibrosis rather than play a role in the disease pathogenesis. Moreover, the trial paradoxically showing higher mortality and morbidity rates in warfarin-treated patients with IPF 10 suggests that the coagulation cascade might be activated in response to fibrosis and that it might exert a protective rather than causative role in the course of the disease.
We found that MPV showed no correlation with IPF severity as evaluated by lung function tests. A potential explanation is that platelet activation might be the result of hypoxemia and increased bone marrow activity (also present in other lung diseases such as COPD 15,16 ) rather than related to fibrosis per se. As already mentioned, platelet activation and fibrosis could be 2 independent manifestations of the same pathogenic background, and the prothrombotic milieu in IPF might remain stable regardless of disease progression. Clearly, more work is needed.
This study has certain limitations. First of all, as mentioned previously, no causal relationship between platelet activation and lung fibrosis was documented. Additionally, we did not measure other biomarkers of prothrombotic state (eg,
In conclusion, patients with IPF had a higher MPV and lower platelet count. Given that the MPV is an index of vascular disease, 27,28 as well as an important predictor of acute coronary events 29 –31 and of venous thrombosis, 32,33 our findings may prove promising in terms of identifying potential biomarkers of cardiovascular morbidity and of venous thrombosis in IPF. However, further studies are needed to replicate these findings, identify further and more specific markers of platelet activation in this disease, and delineate their clinical implications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NP has been an advisory board member of TrigoCare International; has participated in sponsored studies by Novo Nordisk and Novartis; has received honoraria as a speaker from Novo Nordisk and Pfizer; has attended conferences sponsored by TrigoCare International, Novo Nordisk, Sanofi-Aventis and Pfizer. DPM has given talks and attended conferences sponsored by Merck, Sharp & Dohme, AstraZeneca and Libytec. PS has been an advisory board member of Novartis and Menarini, received honoraria as a speaker for AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Menarini, Novartis and Takeda.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
