Abstract
Objective:
To evaluate the efficacy and safety of darexaban (YM150) in Asian patients undergoing total hip or total knee arthroplasty.
Methods:
In 2 phase II/III multicenter, randomized, double-blind, placebo-controlled, parallel-group studies, patients were randomized to oral darexaban 15 mg twice daily (bid), darexaban 30 mg bid, oral placebo bid, or subcutaneous enoxaparin 20 mg bid. Primary efficacy outcome for both studies was total venous thromboembolism (VTE) incidence.
Results:
Both darexaban doses were statistically significantly superior to placebo for total VTE incidence (hip study: darexaban 15 mg bid [2.9%] vs placebo [17.1%], P < .001; darexaban 30 mg bid [5.2%] vs placebo [17.1%], P = .003; and knee study: darexaban 15 mg bid [27.2%] vs placebo [52.8%], P = .002; darexaban 30 mg bid [15.5%] vs placebo [52.8%], P < .001). In both studies, the incidence of bleeding events was low across all treatment groups.
Conclusion:
Darexaban is effective and well tolerated as VTE prophylaxis in Asian patients undergoing elective major orthopedic surgery.
Keywords
Introduction
The risk of venous thromboembolism (VTE) is increased in patients undergoing major orthopedic surgery and is associated with a significant increase in mortality. 1,2 Epidemiological studies have reported that in the United States, 2 the VTE incidence, in the absence of thromboprophylaxis, in orthopedic surgery patients was 40% to 60% and in Europe 3 35% to 45%. International guidelines, such as the American College of Chest Physicians (ACCP), recommend that patients undergoing total hip arthroplasy (THA) or total knee arthroplasy (TKA) receive thromboprophylaxis as subcutaneous administration of a low-molecular-weight heparin (such as enoxaparin), subcutaneous administration of an indirect factor Xa (FXa) inhibitor (fondaparinux), or oral administration of a vitamin K antagonists (VKAs, such as warfarin or other coumarins). 2 However, such guidelines may not be universally applicable to Asian patients, owing to different risk factor profiles. 4
Current guidelines in Japan 5 are based upon the seventh ACCP guidelines and recommend thromboprophylactic approaches according to the level of estimated risk of VTE in certain patient groups. Mechanical intervention, that is, intermittent pneumatic compression (IPC), alone, is recommended for moderate-risk patients. For high-risk patients (including those undergoing THA/TKA), IPC or one of low-dose unfractionated heparin (UFH), warfarin, enoxaparin, or fondaparinux is recommended.
Several specific and novel targets within the coagulation cascade have been the focus of more recent therapeutic developments for novel anticoagulants. 6 In particular, FXa has emerged as an attractive target, as it acts at the convergence point of the intrinsic and extrinsic coagulation pathways. 7,8 Indeed, both direct and indirect inhibitors of FXa are available or undergoing clinical development for a wide range of indications. 9 –11 One such direct, oral, FXa inhibitor is darexaban (YM150). The potential of darexaban as a clinically effective and well-tolerated antithrombotic agent for VTE prophylaxis has previously been demonstrated in 3 studies in primarily Caucasian patients following elective THA. 12 –14 The objective of the 2 phase II/III studies presented here was to evaluate the efficacy and safety of darexaban administered at oral doses of 15 or 30 mg twice daily (bid), in Asian patients undergoing THA or TKA.
Methods
Patients
Adult male and female patients (aged 20 years or older) scheduled for elective THA or TKA were enrolled in these 2 studies. Major exclusion criteria were a history of deep vein thrombosis (DVT) or pulmonary embolism (PE); expected confinement to bed for at least 7 days prior to surgery; diagnosed with hemorrhagic or coagulation disorders, or idiopathic-/heparin-induced thrombocytopenia; concomitant use of antiplatelet or anticoagulant therapy, or planned treatment with these agents during the period from 1 week prior to surgery until the end of venography; history of major trauma, major surgery, or eye, spinal cord, or brain surgery within 90 days prior to surgery; other scheduled lower extremity surgery during the study period; clinically significant bleeding within 90 days prior to the screening visit; use of an intrathecal or epidural catheter that could not be removed 2 hours prior to the start of treatment; diagnosed uncontrolled, moderate or severe hypertension (systolic and/or diastolic blood pressure ≥160 and ≥100 mm Hg, respectively); a gastrointestinal ulcer within 180 days prior to the screening visit; myocardial infarction or stroke within 180 days prior to surgery; weight <40 kg at screening; diagnosed bacterial endocarditis; diagnosed hepatic impairment, and/or renal impairment (defined as serum creatinine 1.5 times the upper limit of normal [ULN] and/or creatinine clearance <30 mL/min); diagnosed retinopathy; and malignancy currently under treatment.
Study Design
These were 2 phase II/III multicenter, randomized, double-blind, placebo-controlled, parallel-group studies conducted at centers within Japan, Taiwan, and Thailand (and in Korea for the THA study). Patients undergoing THA or TKA were randomized to oral darexaban 15 mg bid, darexaban 30 mg bid, oral placebo bid, or subcutaneous enoxaparin 20 mg bid. Darexaban and placebo were used under double-blind conditions, and enoxaparin was used under open-label conditions.
The first dose of oral study medication (darexaban or placebo) was administered 12 to 24 hours after completion of THA or TKA (Figure 1). Subsequent doses of study medication were administered as 1 tablet bid, usually in the morning and evening, with at least 6 hours between doses. Study medication was administered for 10–14 days after surgery. Enoxaparin sodium (Clexane; Sanofi-aventis, Paris, France) was administered initially as a subcutaneous injection of 20 mg (2000 IU), 24–36 hours after completion of THA or TKA. Subsequently, enoxaparin 20 mg was administered every 12 hours by subcutaneous injection, for 10–14 days. The concomitant use of any nonpharmacologic therapy for VTE prophylaxis, for example, IPC and compression stockings, was permitted.
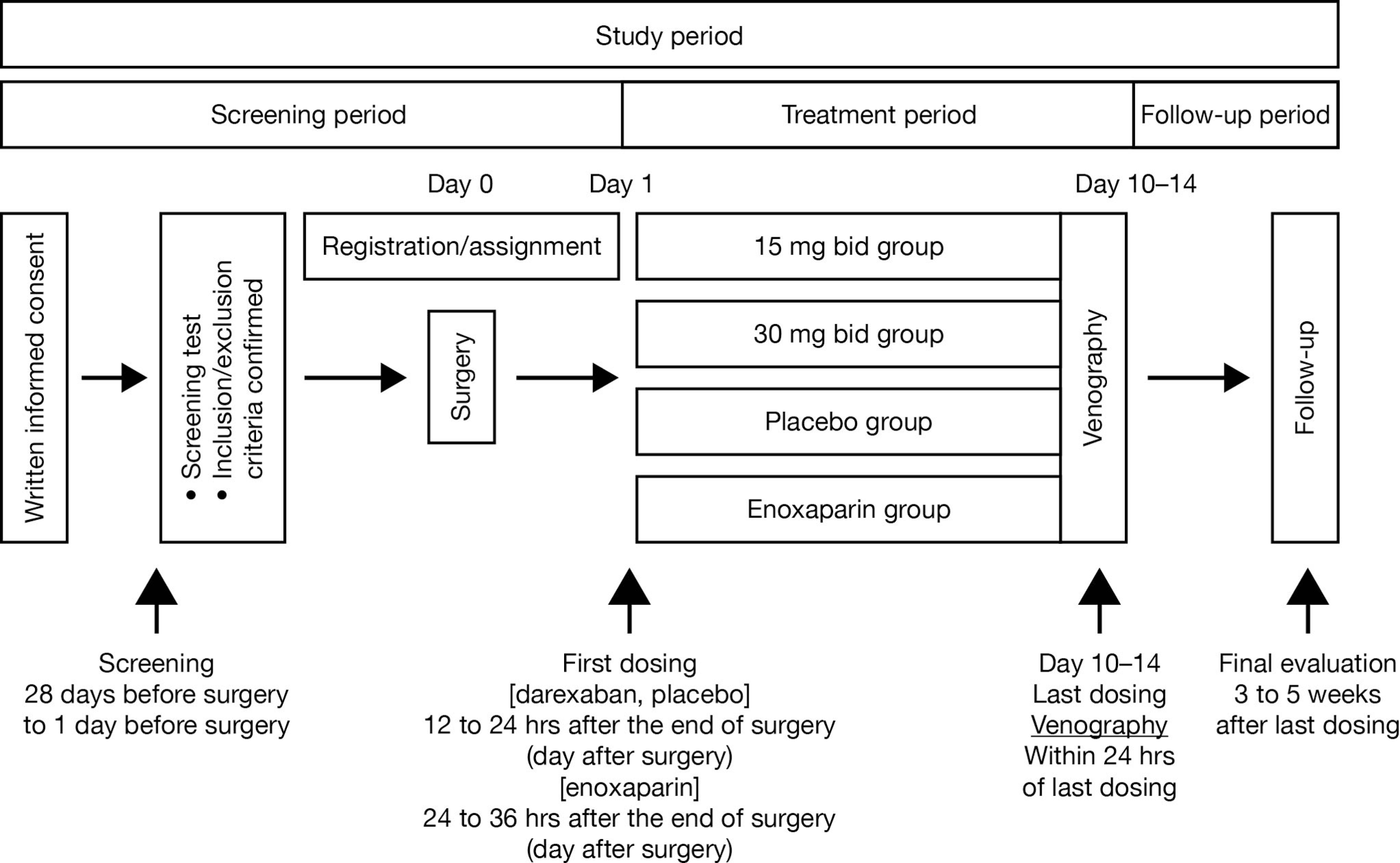
Design of the total hip arthroplasty and total knee arthroplasty studies. bid indicates twice daily.
Both studies were reviewed and approved by the institutional review board and independent ethics committee and were conducted in accordance with the ethical principles set out in the Declaration of Helsinki, the applicable Good Clinical Practice guidelines, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use Guidelines, and all applicable local laws and regulations. Written informed consent was obtained before participation.
Assessments
During the screening visit, patients underwent measurement of weight, height, vital signs, electrocardiogram (ECG), laboratory tests (blood biochemistry, urinalysis, and cardiac markers), and prothrombin time – international normalized ratio (PT-INR), together with a detailed medical history. On the first day of study drug administration (day 1), 12-lead ECG, cardiac markers, and hemoglobin levels were assessed. Evaluations of vital signs, ECG, laboratory tests, cardiac markers (troponin-T and creatine kinase myocardial band), and hemoglobin levels were repeated on days 2 and 7 and also on the last day of dosing (days 10-14). For determination of darexaban glucuronide concentrations and PT-INR evaluation, blood samples were obtained on day 7 (1–3 hours after dosing) and again on the last day of dosing (within 1 hour before dosing) in order to achieve a steady-state concentration at both peak and trough level. Bilateral venography of the lower extremities was performed within 24 hours of the final dose of study medication. A follow-up visit was scheduled 3–5 weeks after the last dose of study medication.
Outcomes
Primary efficacy outcomes
The primary efficacy outcome for both studies was the number of patients who experienced at least 1 of the following VTE events (defined as total VTE incidence), DVT (proximal and/or distal) proven by venography at the end of study drug administration; symptomatic DVT or PE confirmed by imaging during the treatment period; and death due to any cause during the treatment period (including death from PE).
Secondary efficacy outcomes
The secondary efficacy outcomes for both studies were the numbers of patients who experienced an occurrence of any of the following VTE events during the treatment period: (i) total DVT incidence (proximal and/or distal) confirmed by venography and/or symptomatic DVT; (ii) DVT by proximal and distal location; (iii) symptomatic DVT; (iv) symptomatic PE; (v) all deaths, regardless of cause; (vi) major VTE: (one or more) proximal DVT, proximal symptomatic DVT, symptomatic PE, and death due to any cause; (vii) asymptomatic DVT confirmed by venography; and (viii) asymptomatic DVT by proximal and distal location.
Pharmacodynamics
The potential relationship between PT-INR and darexaban glucuronide concentration was also explored in both the studies.
Safety outcomes
Adverse events (AEs) were recorded throughout both the studies. Among these AEs, bleeding events were classified as major bleeding, clinically relevant nonmajor (CRNM) bleeding, or minor bleeding. The incidence of all bleeding events and bleeding events resulting in discontinuation of study medication were also recorded throughout these 2 studies. Other safety variables included laboratory tests, vital signs, and 12-lead ECG.
Statistical Analysis
Based upon the estimated incidences of VTE events observed in previous studies, the planned number of patients for randomization in the THA study was 160 for the darexaban 15 mg bid, darexaban 30 mg bid and placebo groups, and 100 for the enoxaparin group; the planned number of patients for randomization in the TKA study was 90 in each treatment group.
In both studies, 4 analysis populations were defined. The full analysis set (FAS) comprised all patients who had undergone planned THA or TKA and received at least 1 dose of study medication. The modified FAS (mFAS) included patients in the FAS, who had a venograph that could be evaluated within 24 hours of the last dose of study medication, and/or patients with symptomatic DVT or PE and/or participants who died due to any cause. The pharmacodynamic analysis set (PDAS) comprised all patients who received at least 1 dose of study medication and had pharmacodynamic (PD) results for at least 1 time point. Finally, the safety analysis set, consisted of all patients who received at least 1 dose of study medication.
The following closed procedure testing was used to analyze the primary efficacy end point (total VTE) by comparing the darexaban groups, in order from the highest to lowest dose, to the placebo group. First, the incidence of VTE in the darexaban 30 mg bid group was compared with placebo, using the 2-sided Fisher exact test with a 5% significance level. If the difference between darexaban 30 mg bid and placebo was statistically significant, the differences between the incidence of VTE in the darexaban 15 mg bid and placebo groups were then compared using the same methodology. Secondary efficacy end points were analyzed for the FAS, with incidences and corresponding 95% confidence intervals (CIs) calculated for the darexaban and placebo treatment groups. The darexaban and placebo arms were compared using the 2-sided Fisher exact test with a 5% significance level. The enoxaparin group was not statistically compared with the darexaban groups and placebo group because enoxaparin was included as a benchmark drug in these studies. The incidences of the different categories of bleeding events were calculated for each treatment group, together with their 95% CIs. Other AEs were described using the number and percentages of patients. Results for PT-INR were tabulated and summary statistics calculated by each evaluation time point. Plots of the darexaban glucuronide plasma concentration and the PT-INR were prepared in order to explore the relationship between the darexaban glucuronide plasma concentration and this coagulation parameter. Descriptive statistics were used to present the baseline demographics.
Results
Total Hip Arthroplasty Study
Patient disposition and baseline characteristics
This study was conducted from May 2009 to March 2010 at 81 centers in Japan, Taiwan, Korea, and Thailand. A total of 622 patients were randomized to treatment, of whom 610 patients received at least 1 dose of study medication. From this population, 564 patients completed study medication and 46 discontinued prematurely (Figure2A). The mFAS comprised 481 patients.
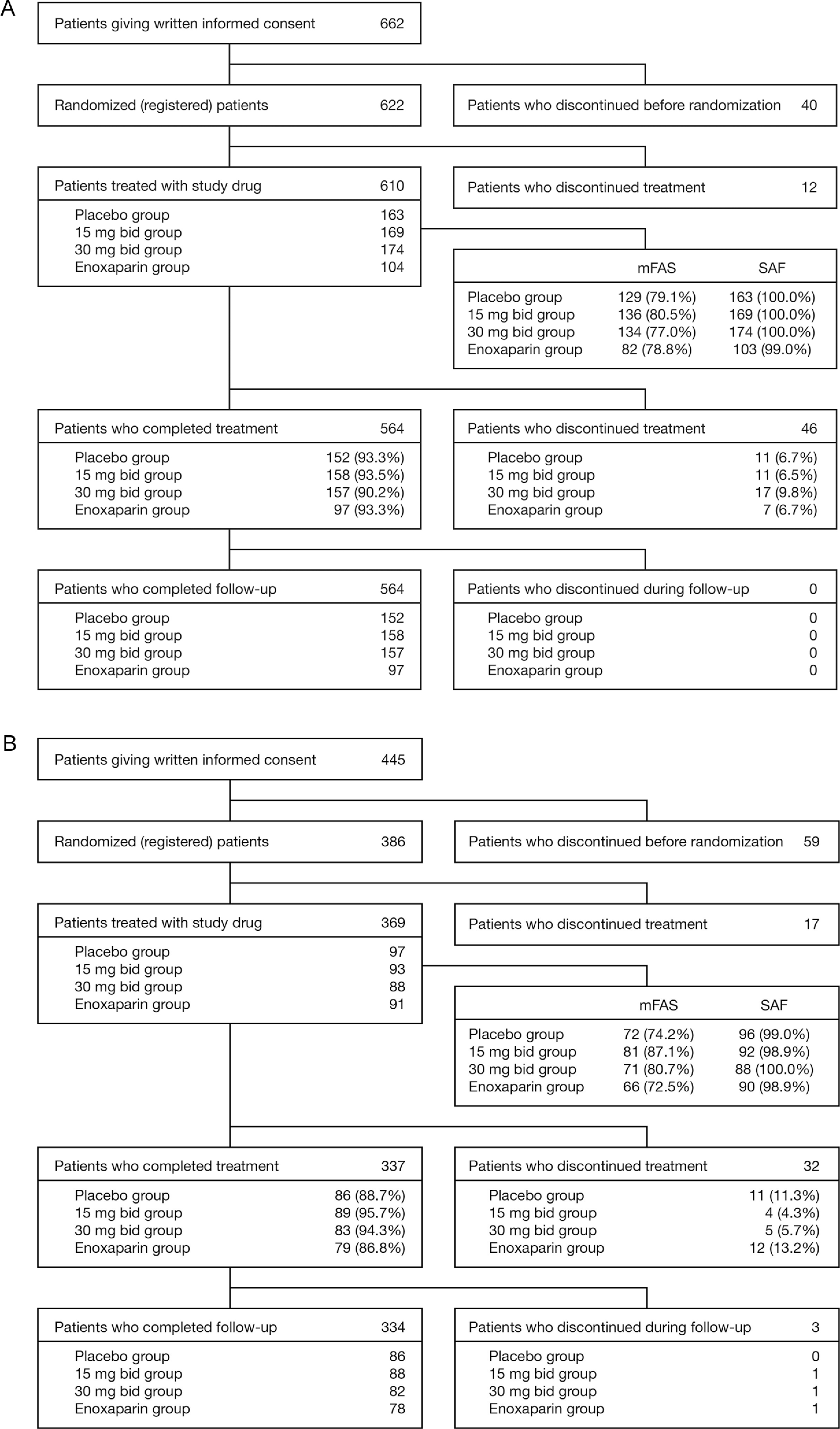
Patient disposition in A, the total hip arthroplasty and B, the total knee arthroplasty studies. bid indicates twice daily; mFAS, modified full analysis set; SAF, safety analysis set.
Baseline demographics and characteristics for the mFAS were similar across treatment groups (Table 1). The average duration of surgery in each group was less than 2 hours (1.68-1.84 hours), and patients were able to walk 2 or more steps on average on days 2.2–2.5 after surgery. The use of prior or concomitant therapies was comparable between the treatment groups. Concomitant mechanical prophylaxis was received by 94.8%–98.8% of patients across all treatment groups. Patients in each group were treated for an average of between 11.7 and 12.1 days.
Baseline Characteristics and Demographics in the Total Hip Arthroplasty Study (Modified Full Analysis Set).
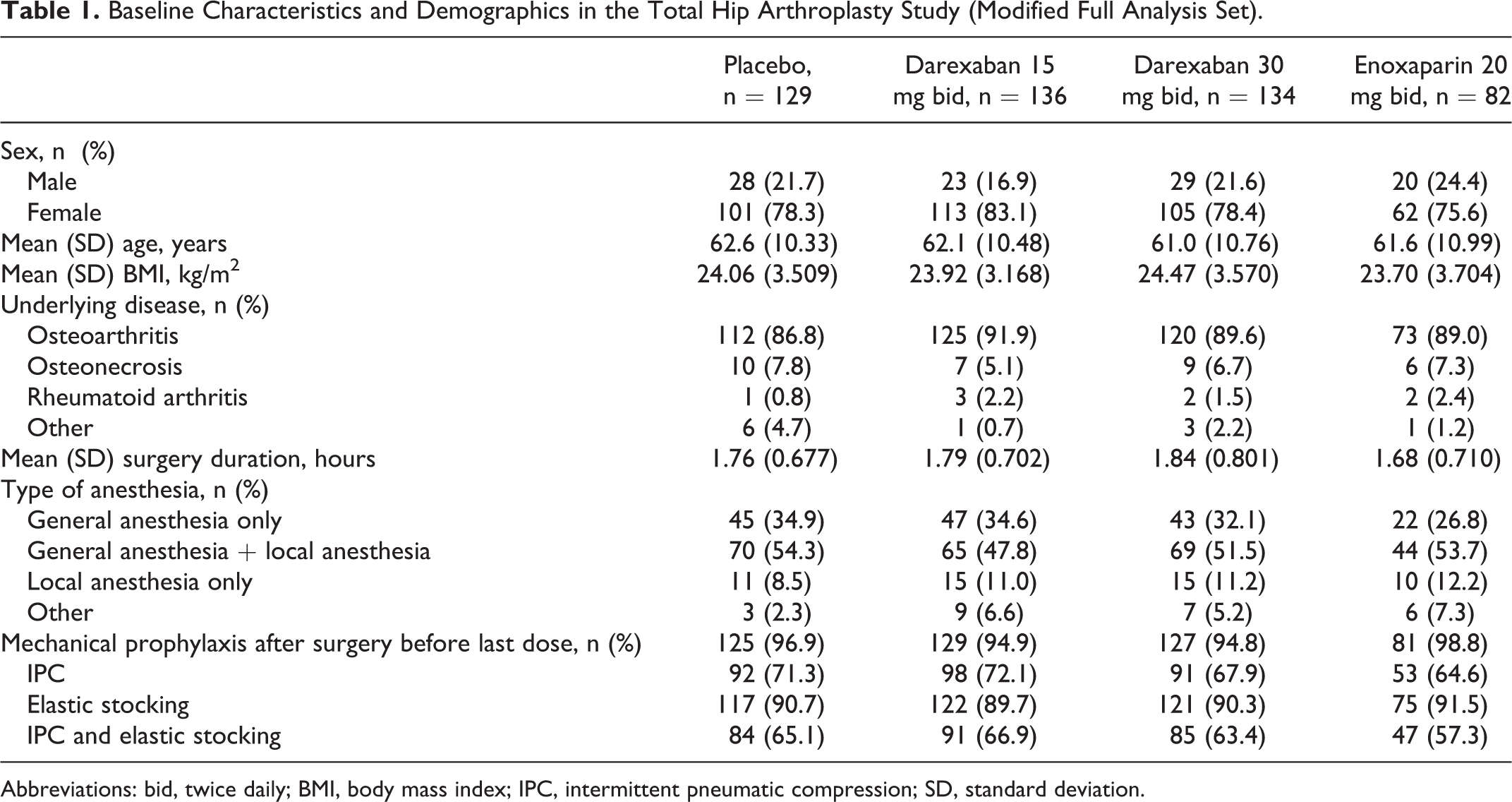
Abbreviations: bid, twice daily; BMI, body mass index; IPC, intermittent pneumatic compression; SD, standard deviation.
Efficacy
Primary efficacy outcome
The incidence of total VTE in the mFAS was 17.1% in the placebo group (95% CI: 11.01-24.67), 2.9% in the darexaban 15 mg bid group (95% CI: 0.81-7.36), 5.2% in the darexaban 30 mg bid group (95% CI: 2.13-10.47), and 2.4% in the enoxaparin 20 mg bid group (95% CI: 0.30-8.53). Both doses of darexaban were significantly superior to placebo for this outcome measure (darexaban 15 mg bid vs placebo, P < .001; darexaban 30 mg bid vs placebo, P = .003). The incidence of total VTE in the darexaban 15 mg bid and 30 mg bid groups was numerically similar to the enoxaparin group (Table 2).
Incidence of Venous Thromboembolism in the Total Hip Arthroplasty Study.a
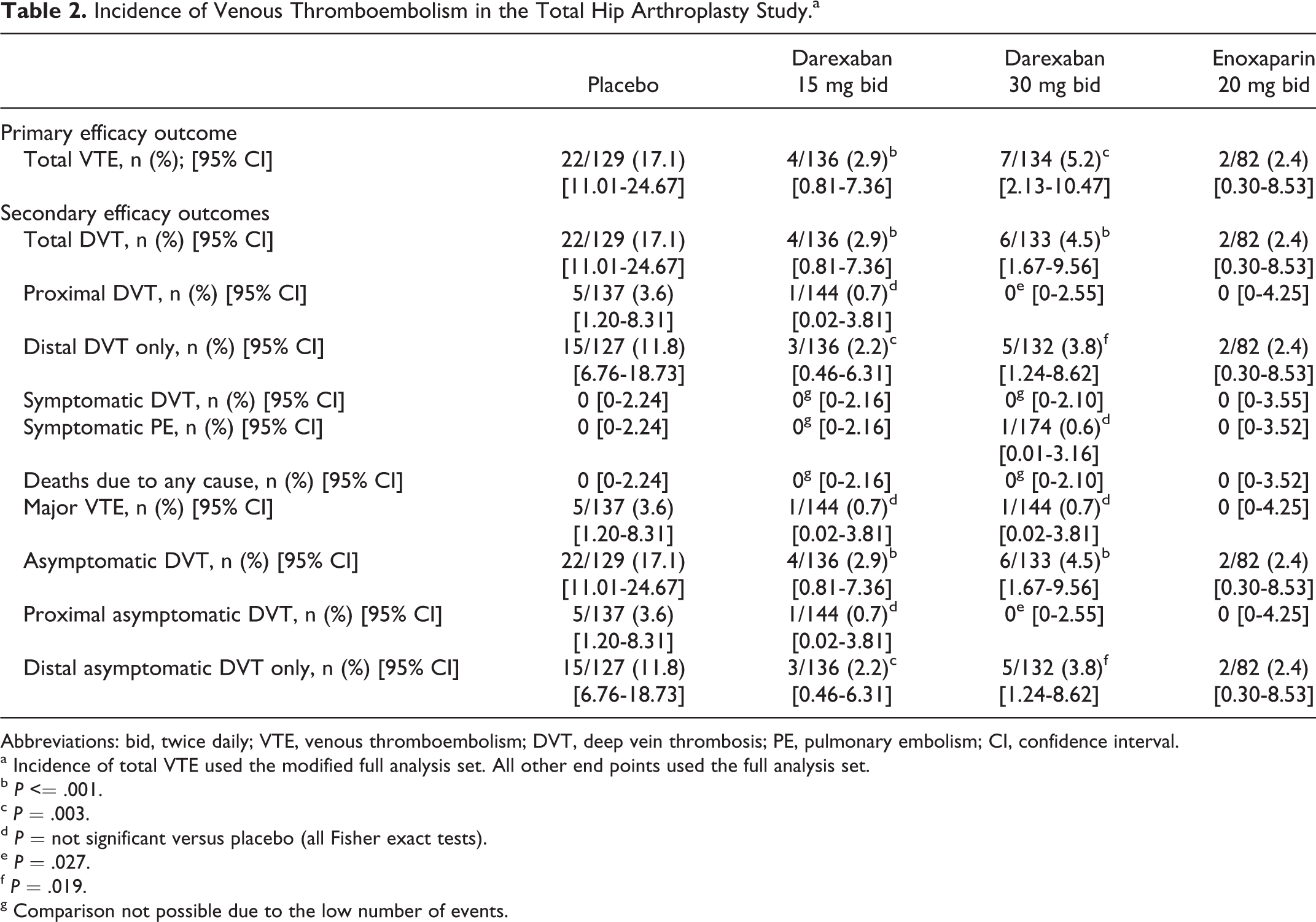
Abbreviations: bid, twice daily; VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; CI, confidence interval.
a Incidence of total VTE used the modified full analysis set. All other end points used the full analysis set.
b P <= .001.
c P = .003.
d P = not significant versus placebo (all Fisher exact tests).
e P = .027.
f P = .019.
g Comparison not possible due to the low number of events.
Secondary efficacy outcomes
Significant differences between darexaban at both doses and placebo were observed for the incidences of total DVT, distal DVT only, asymptomatic DVT, and distal asymptomatic DVT only in the FAS (Table 2). Darexaban 30 mg bid was also superior to placebo for the incidences of proximal DVT and proximal asymptomatic DVT. Symptomatic PE occurred in one participant in the darexaban 30 mg group (Table 2).
Pharmacodynamics
The PDAS comprised 609 patients (placebo, n = 163; darexaban 15 mg bid, n = 169; darexaban 30 mg bid, n = 174; and enoxaparin 20 mg bid, n = 103). The mean PT-INR 1-3 hours after dosing on day 7 (corresponding with peak plasma concentrations of darexaban glucuronide) was 1.0 for placebo, 2.0 for darexaban 15 mg bid, 2.7 for darexaban 30 mg bid, and 1.0 for enoxaparin 20 mg bid, indicating increases in the darexaban groups compared to the placebo and enoxaparin groups. The mean PT-INR prior to the last dose of study medication (corresponding with trough plasma concentrations of darexaban glucuronide) was 1.0 for placebo, 1.6 for darexaban 15 mg bid, 2.0 for darexaban 30 mg bid, and 1.0 for enoxaparin 20 mg bid, again indicating increases in the darexaban groups compared to the placebo and enoxaparin groups. Darexaban glucuronide plasma concentrations and PT-INR were plotted and an exploratory analysis revealed that there was a correlation between PT-INR and darexaban glucuronide concentration (Figure 3A). No different noteworthy trends were found between Japanese and other Asian patients (Figure 3A).
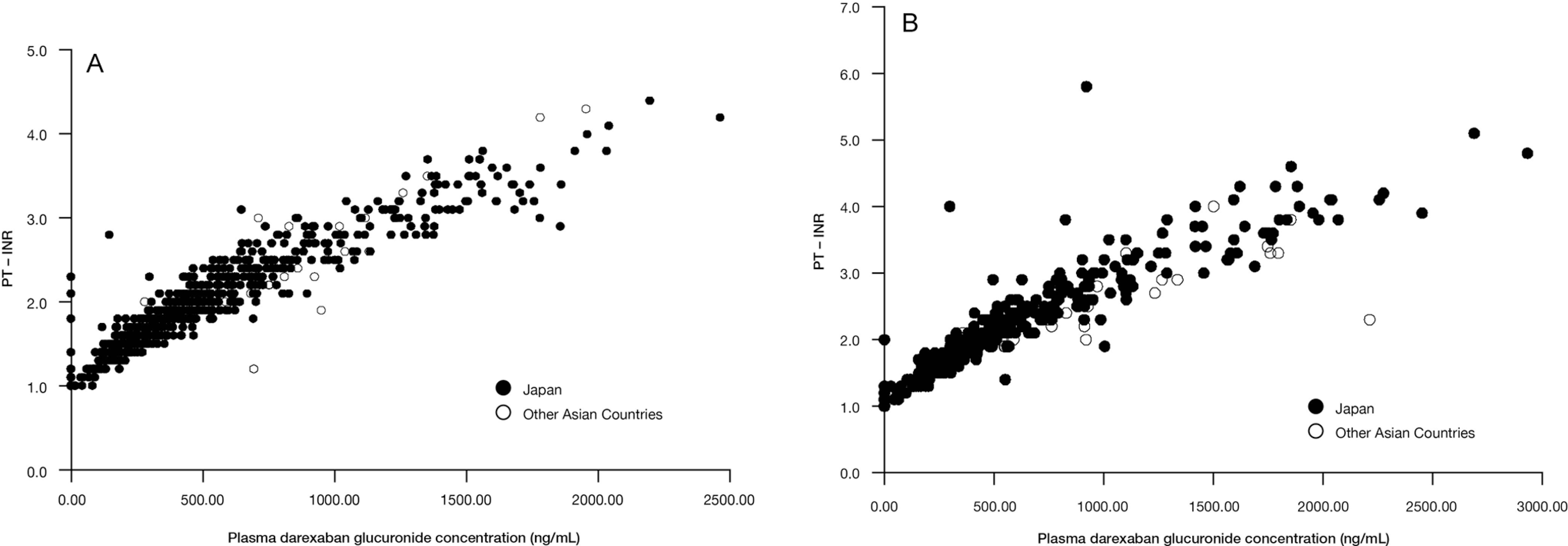
The PT-INR values and corresponding darexaban glucuronide plasma concentrations. A, Total hip arthroplasty study. B, Total knee arthroplasty study. PT-INR indicates prothrombin time –International Normalized Ratio.
Safety
Bleeding events
The incidence of major and/or CRNM bleeding events during the treatment period was low across all treatment groups (Table 3). There were no major bleeding events in any treatment group. Three CRNM bleeding events occurred in 3 patients in the darexaban 15 mg bid group (hematochezia, hematuria, and subcutaneous hematoma), in 2 patients in the darexaban 30 mg bid group (hematuria and subcutaneous hematoma), and in 2 patients in the enoxaparin 20 mg bid group (hematuria and subcutaneous hematoma). The incidence of all bleeding events was comparable between the treatment groups (Table 3). One patient in the darexaban 30 mg bid group discontinued due to the occurrence of 2 minor bleeding events (subcutaneous hemorrhage and wound hemorrhage). There were no noteworthy differences between the incidence of bleeding events during the treatment period and the end of the study (Table 3).
Bleeding Events in the Total Hip Arthroplasty Study (Safety Analysis Set).
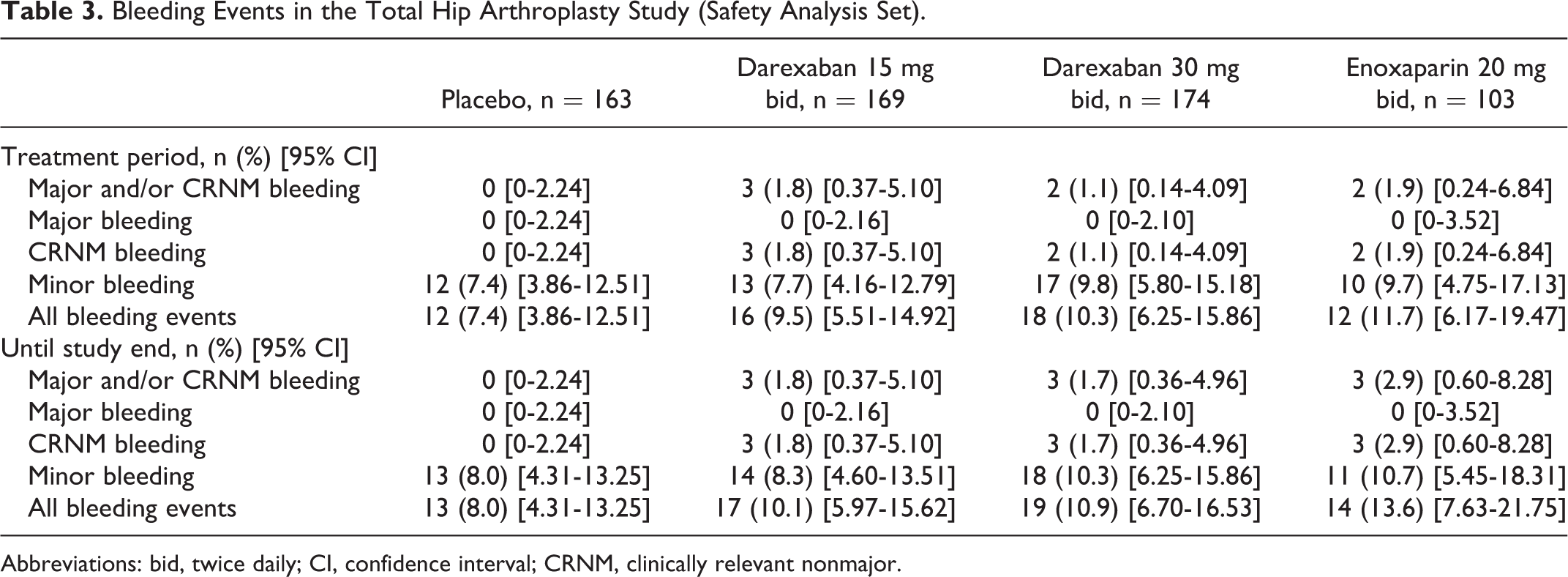
Abbreviations: bid, twice daily; CI, confidence interval; CRNM, clinically relevant nonmajor.
Adverse events
The incidence of AEs during the treatment period is summarized in Table 4. Overall, both the total incidence of AEs and the incidence of AEs other than VTE were highest in the enoxaparin 20 mg bid group and comparable between the darexaban and placebo groups (Table 4). The majority of AEs were of mild severity (70.4%-82.5% of patients across all treatment groups). The incidence of AEs resulting in discontinuation was generally low, although somewhat higher in the darexaban 30 mg bid group (Table 4). The incidence of serious AEs (SAEs) was low in all groups (0.6%-2.9%). There were no deaths during the treatment period.
Adverse Events Reported During the Treatment Period in the Total Hip Arthroplasty Study (Safety Analysis Set).
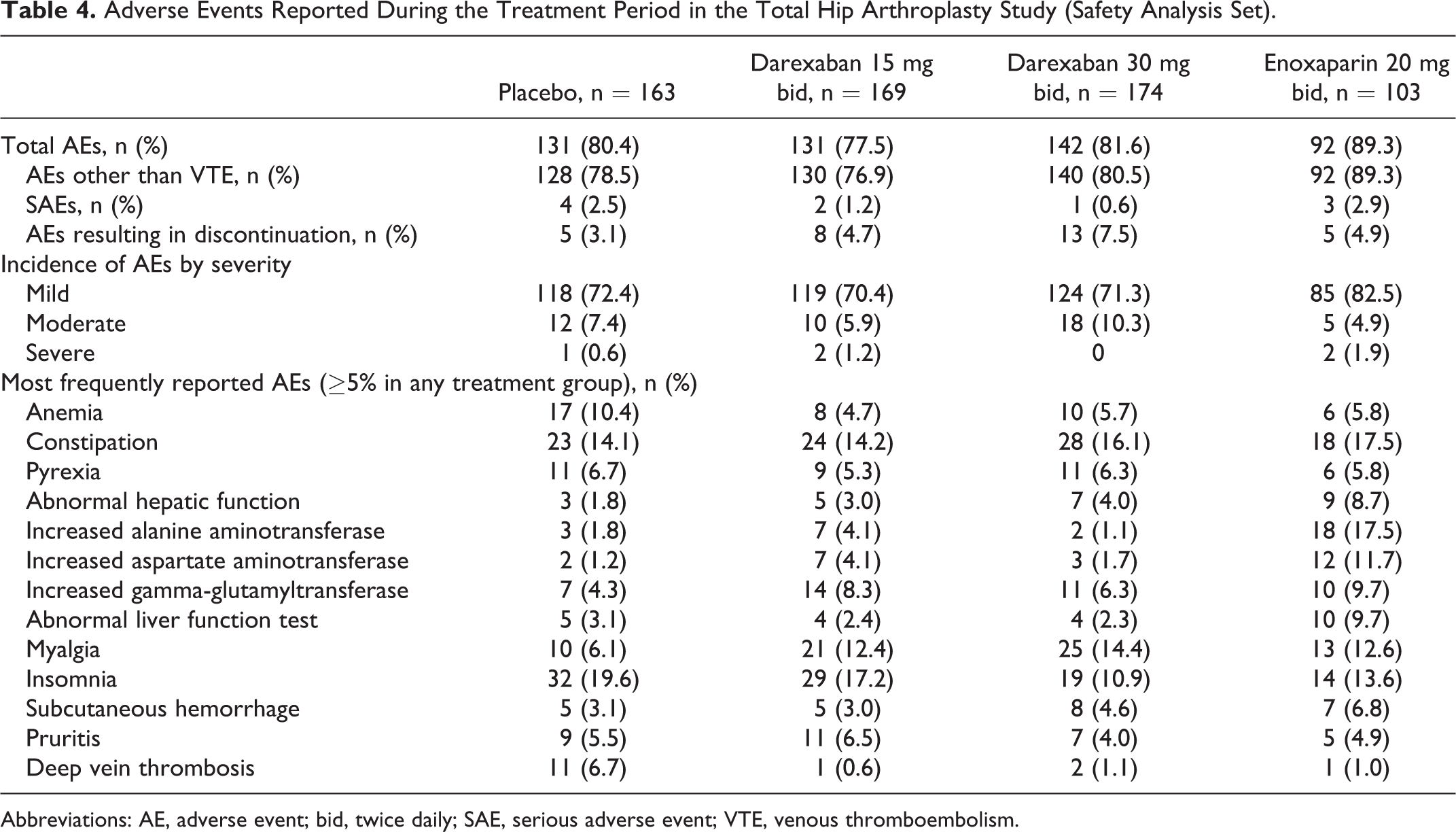
Abbreviations: AE, adverse event; bid, twice daily; SAE, serious adverse event; VTE, venous thromboembolism.
Laboratory assessments
The incidences of abnormal liver function tests was low in all treatment groups during the treatment period (Table 5). In only 1 patient (0.6%), in the darexaban 30 mg bid group, alanine aminotransferase (ALT) or aspartate aminotransferase (AST) increased to >3 times the ULN and total bilirubin increased to >2 times the ULN on the same day (same sample) during the treatment period (Table 5). The investigator determined that this event was most likely related to a concomitant antibiotic (clarithromycin) but that a causal relationship to the study drug could not be ruled out. There were no clinically significant changes in vital signs during the study.
Percentage of Patients With Liver Function Abnormalities During the Treatment Period (Safety Analysis Set) in Total Hip Arthroplasty Study.
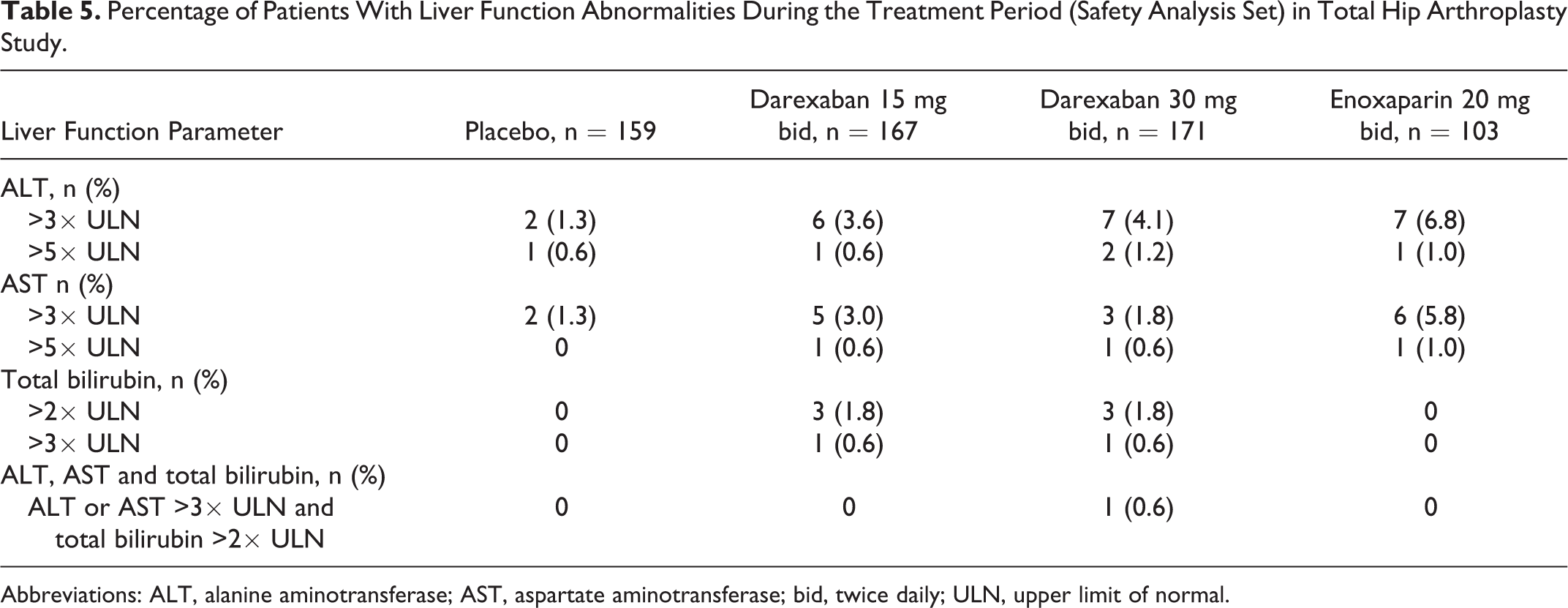
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; bid, twice daily; ULN, upper limit of normal.
Total Knee Replacement Study
Patient disposition and baseline characteristics
This study was conducted from June 2009 to January 2010 at 67 centers in Japan, Taiwan, and Thailand. A total of 386 patients were randomized to treatment, of whom 369 patients received at least 1 dose of study medication. From this population, 337 patients completed study medication and 32 discontinued prematurely (Figure 2B). The mFAS comprised 290 patients.
Baseline demographics and characteristics for the mFAS were similar across treatment groups (Table 6). The average duration of surgery in each group was less than 2 hours (1.80-1.85 hours), and patients were able to walk 2 or more steps on average, on days 2.4–3.0 after surgery. The use of prior or concomitant therapies was comparable between treatment groups. Concomitant mechanical prophylaxis was received by 87.7%–90.3% of patients across all treatment groups. Patients in each group were treated for an average of between 11.5 and 11.9 days.
Baseline Characteristics and Demographics in the Total Knee Arthroplasty Study (Modified Full Analysis Set).
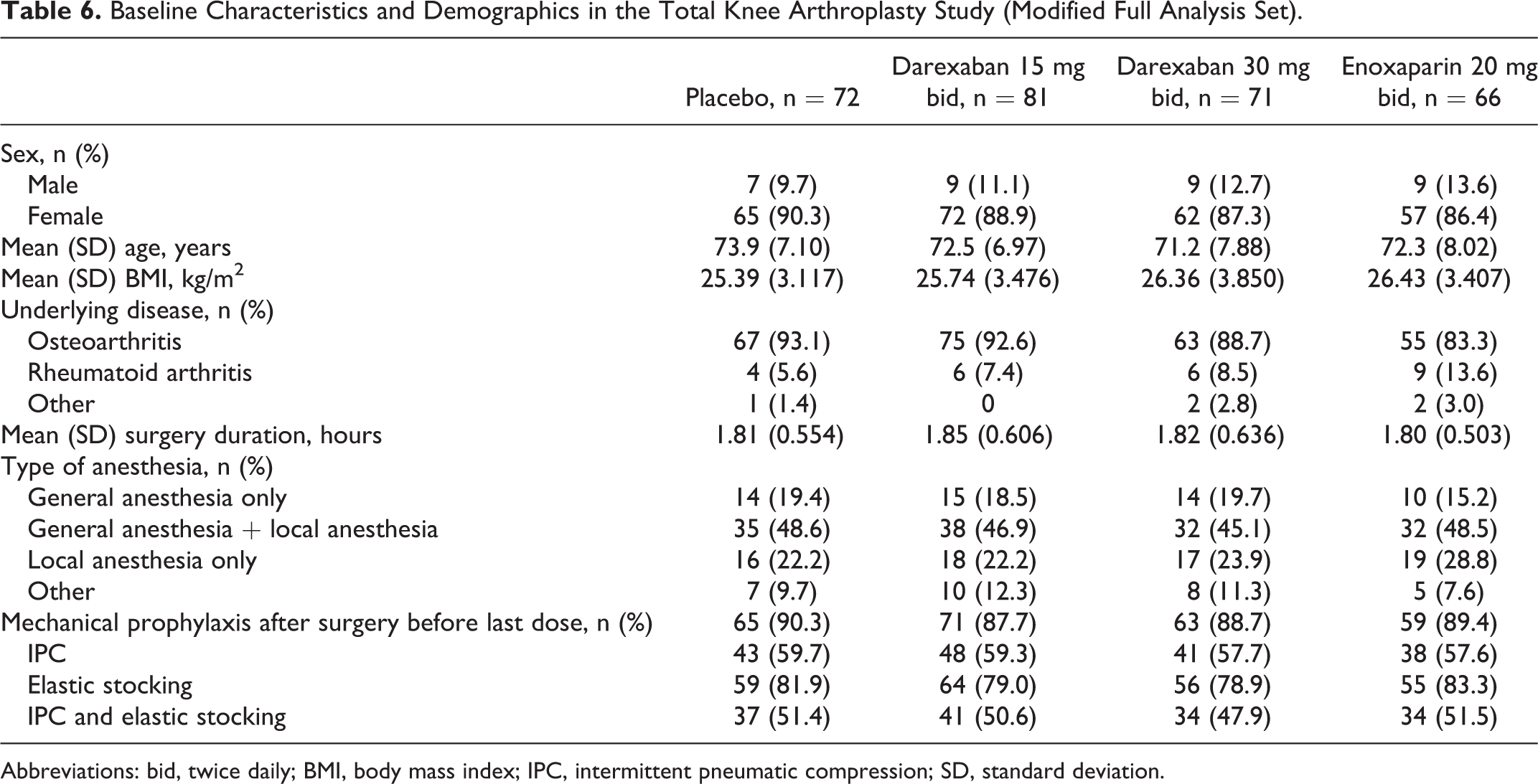
Abbreviations: bid, twice daily; BMI, body mass index; IPC, intermittent pneumatic compression; SD, standard deviation.
Efficacy
Primary efficacy outcome
The total VTE incidence in the mFAS was 52.8% in the placebo group (95% CI: 40.65-64.67), 27.2% in the darexaban 15 mg bid group (95% CI: 17.87-38.19), 15.5% in the darexaban 30 mg bid group (95% CI: 8.00-26.03), and 22.7% in the enoxaparin 20 mg bid group (95% CI: 13.31-34.70). Both doses of darexaban were significantly superior to placebo for this outcome measure (darexaban 15 mg bid vs placebo, P = .002; darexaban 30 mg bid vs placebo, P < .001). The incidence of total VTE in the darexaban 15 mg bid and 30 mg bid groups was numerically similar to the enoxaparin group (Table 7).
Incidence of Venous Thromboembolism in the Total Knee Arthroplasty Study.a
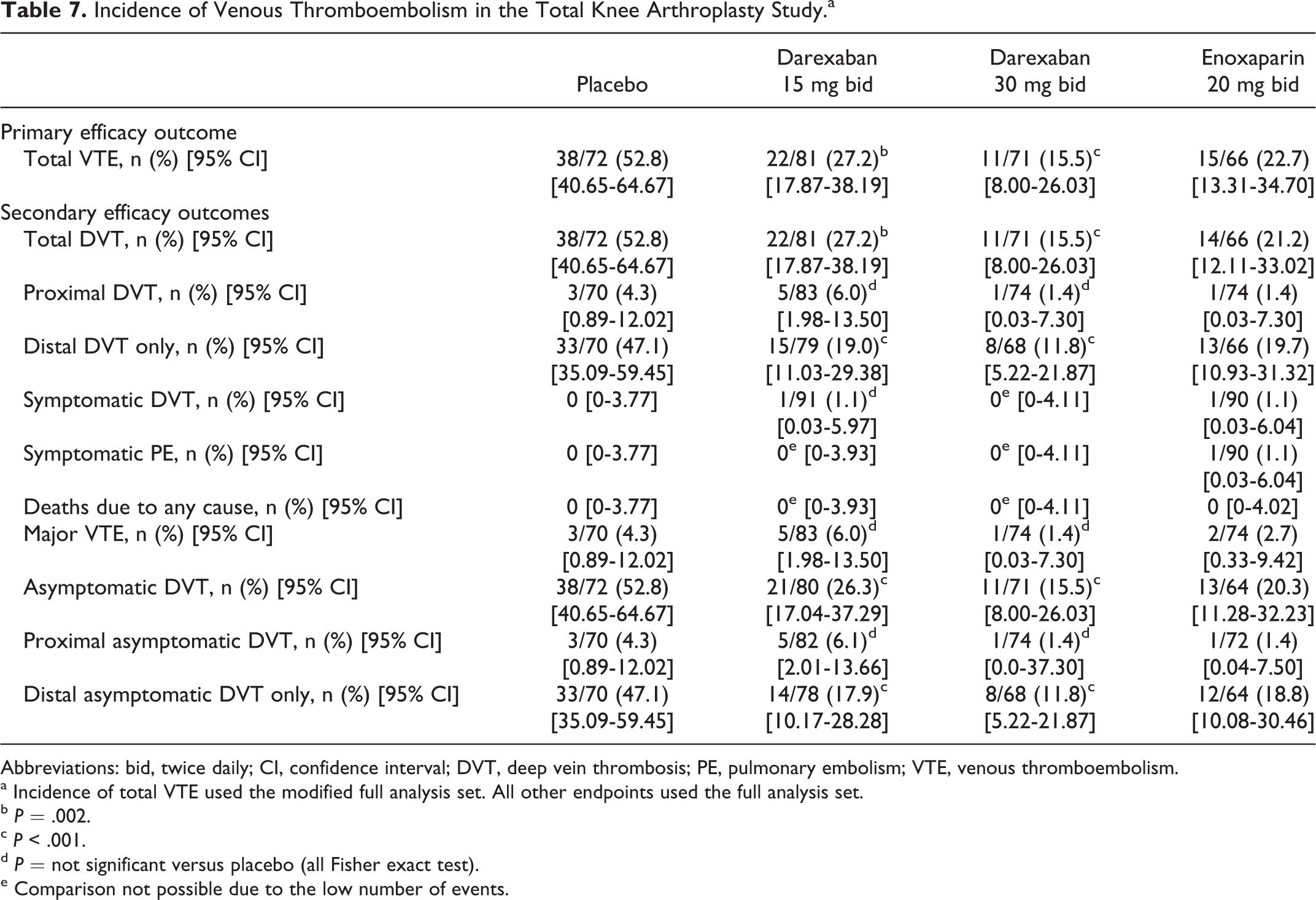
Abbreviations: bid, twice daily; CI, confidence interval; DVT, deep vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
a Incidence of total VTE used the modified full analysis set. All other endpoints used the full analysis set.
b P = .002.
c P < .001.
d P = not significant versus placebo (all Fisher exact test).
e Comparison not possible due to the low number of events.
Secondary efficacy outcomes
Significant differences between darexaban at both doses and placebo were observed for the incidences of total DVT, distal DVT only, asymptomatic DVT, and distal asymptomatic DVT only (Table 7). There were no differences between darexaban and placebo for the incidences of proximal DVT, symptomatic DVT, major VTE, and proximal asymptomatic DVT.
Pharmacodynamics
The PDAS comprised 366 patients (placebo, n = 96; darexaban 15 mg bid, n = 92; darexaban 30 mg bid, n = 88; enoxaparin 20 mg bid, n = 90). The mean PT-INR 1–3 hours after dosing on day 7 was 1.1 for placebo, 2.2 for darexaban 15 mg bid, 3.1 for darexaban 30 mg bid, and 1.0 for enoxaparin 20 mg bid. The mean PT-INR prior to the last dose of study medication was 1.0 for placebo, 1.7 for darexaban 15 mg bid, 2.3 for darexaban 30 mg bid, and 1.0 for enoxaparin 20 mg bid. An exploratory analysis revealed that there was a correlation between PT-INR and darexaban glucuronide concentration (Figure 3B). No different noteworthy trends were found between Japanese and other Asian patients (Figure 3B).
Safety
Bleeding events
The incidence of major and/or CRNM bleeding events during the treatment period was low across all treatment groups (Table 8). There was 1 major bleeding event in the darexaban 30 mg bid group (wound hemorrhage). One CRNM bleeding event occurred in 1 patient in the placebo group (upper gastrointestinal hemorrhage) and 4 CRNM bleeding events occurred in 3 patients in the enoxaparin group (2 each of subcutaneous hematoma and hematuria). The incidence of all bleeding events during the treatment period was highest in the enoxaparin 20 mg bid group and comparable between the darexaban and placebo groups (Table 8). Five patients withdrew from the study due to bleeding events (placebo, n = 1; darexaban 30 mg bid, n = 1; enoxaparin 20 mg bid, n = 3). There were no noteworthy differences among the incidences of bleeding events during the treatment period and the end of the study (Table 8).
Bleeding Events in the Total Knee Arthroplasty Study (Safety Analysis Set).
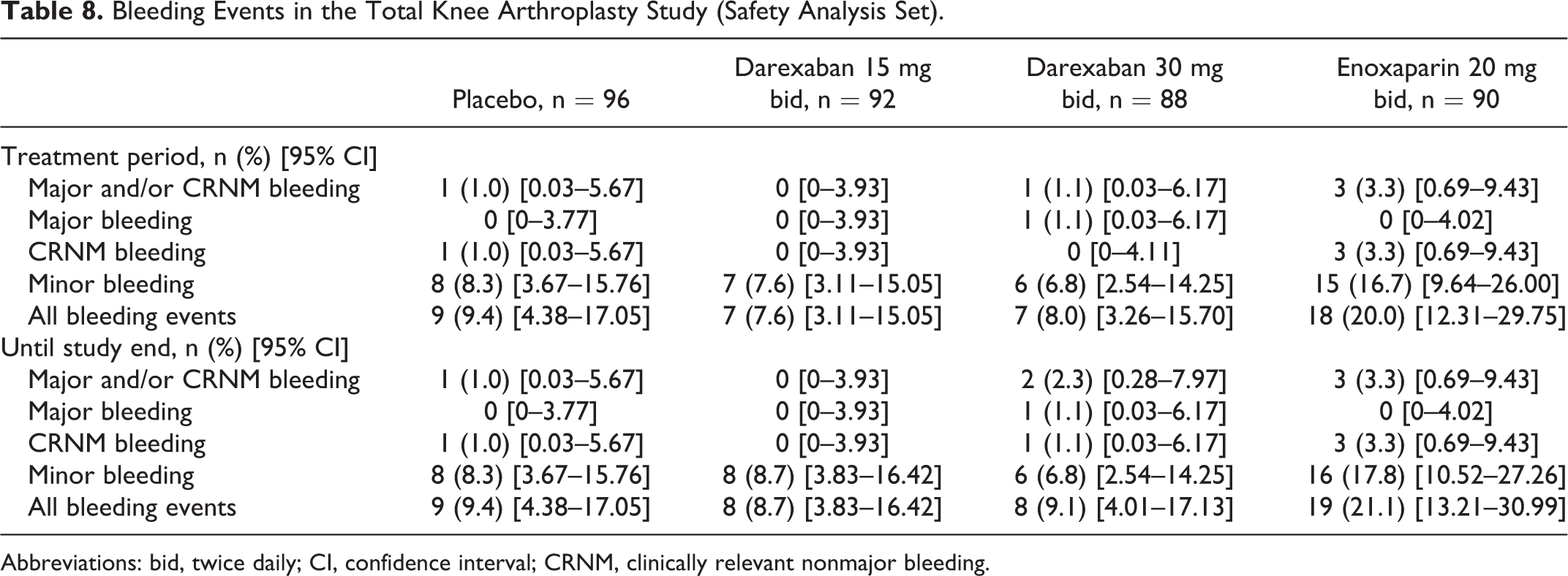
Abbreviations: bid, twice daily; CI, confidence interval; CRNM, clinically relevant nonmajor bleeding.
Adverse events
Overall, the total incidence of AEs during the treatment period was comparable across treatment groups (Table 9). The majority of AEs were of mild severity (58.0%-66.3% of patients across all treatment groups). The incidence of AEs resulting in discontinuation was highest in the enoxaparin 20 mg bid and placebo groups (Table 9). The incidence of SAEs was low in all groups (1.0%-3.4%). There were no deaths during the treatment period. During the follow-up period, 1 participant in the darexaban 30 mg bid group died because of PE occurring on day 24 (13 days after last dosing). The investigator attributed the PE to lack of efficacy of the drug.
Adverse Events Reported During the Treatment Period in the Total Knee Arthroplasty Study (Safety Analysis Set).
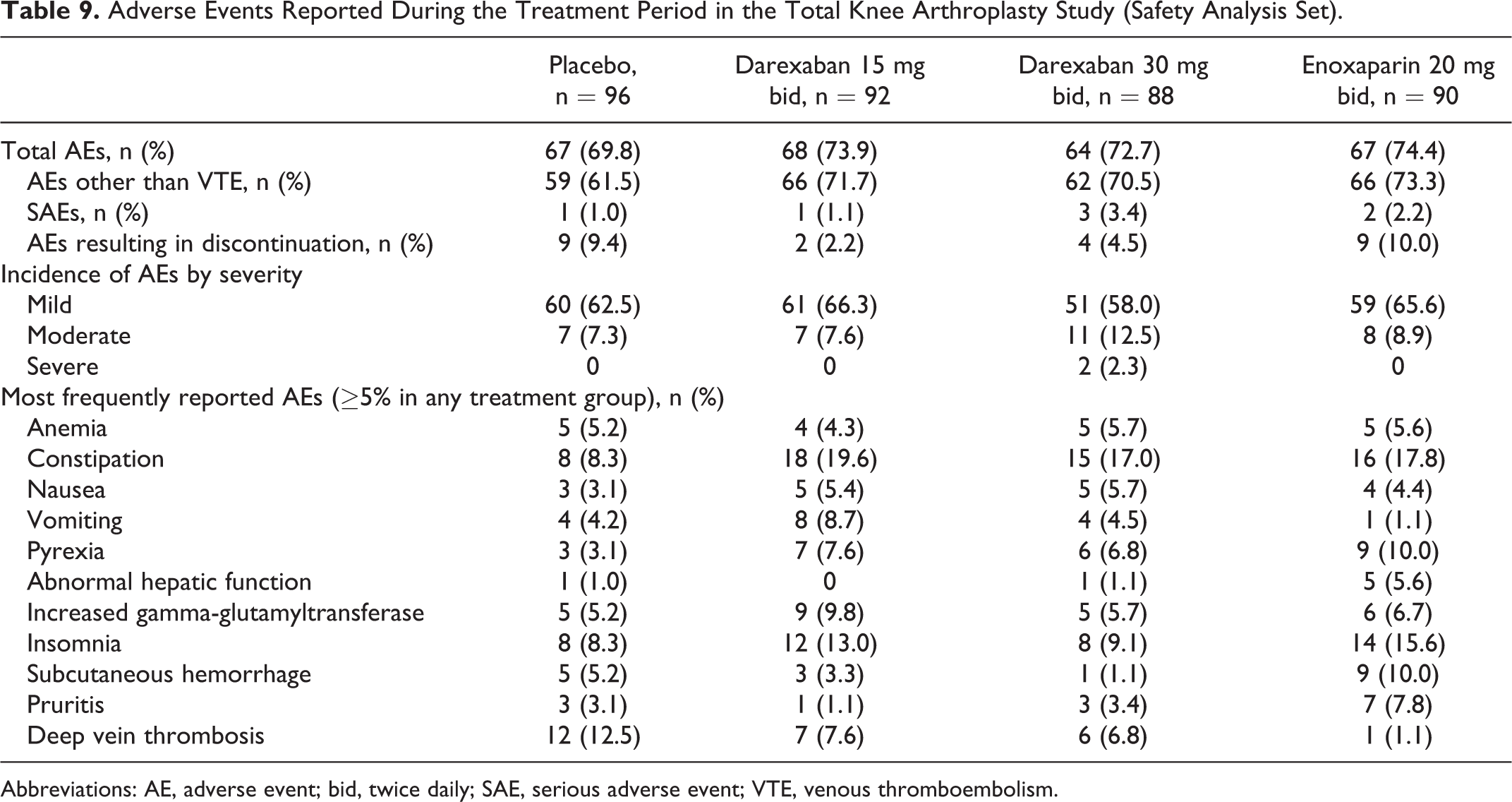
Abbreviations: AE, adverse event; bid, twice daily; SAE, serious adverse event; VTE, venous thromboembolism.
Laboratory assessments
The incidence of abnormal liver function tests was low in all treatment groups during the treatment period (Table 10). There were no patients in whom ALT or AST increased to more than 3 times the ULN and total bilirubin levels increased to more than 2 times the ULN on the same day during the treatment period. There were no clinically significant changes in vital signs during the study.
Percentage of Patients With Liver Function Abnormalities During the Treatment Period (Safety Analysis Set) in Total Knee Arthroplasty Study.
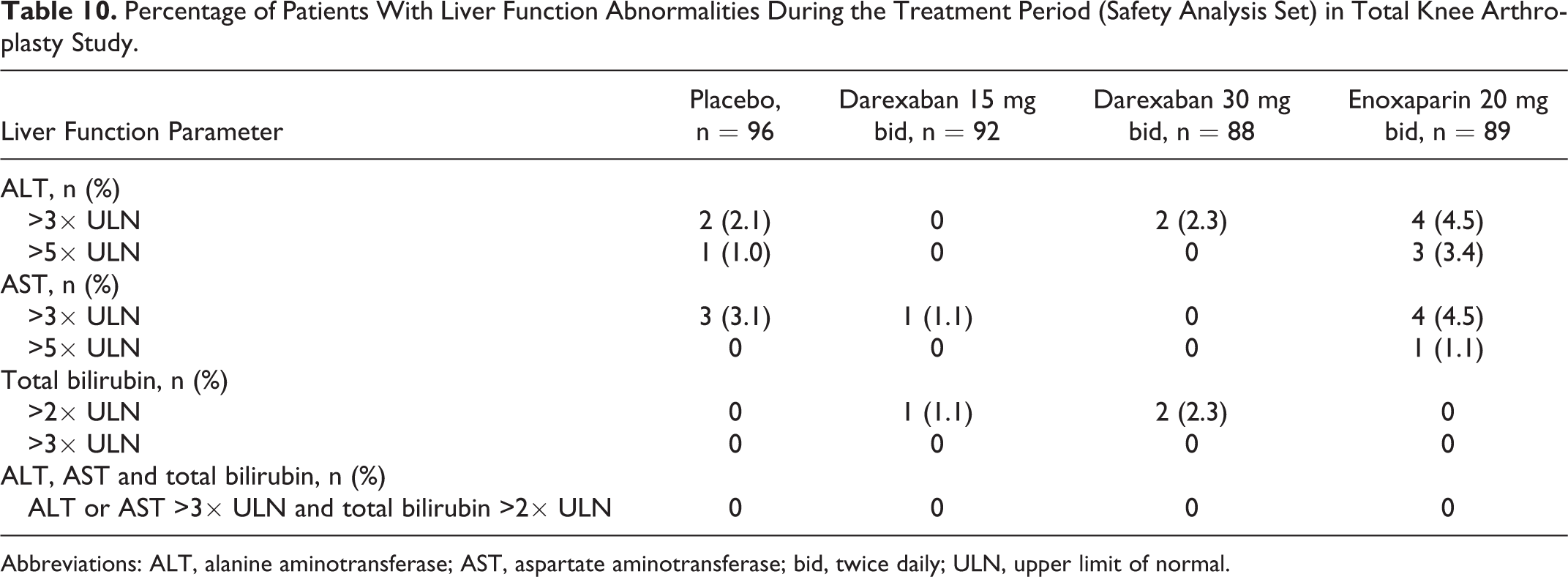
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; bid, twice daily; ULN, upper limit of normal.
Discussion
These 2 studies assessed the efficacy and safety of darexaban, administered at oral doses of 15 or 30 mg bid, in Asian patients undergoing THA or TKA. Enoxaparin was also used as a standard drug in these studies. It was administered at a dosage of 20 mg bid which was the approved dosage in Japan although it was not the customary dose of 30 mg bid, given in the United States for VTE prophylaxis in elective orthopedic cases. A clinical study of enoxaparin showed the mean body weight of Japanese patients was approximately 55 kg and it was two-thirds that of their Caucasian counterparts, therefore, the enoxaparin dosage in Japan was thought to be equivalent to that in the United States. 15 The dose of darexaban for Caucasian patients needs to be higher, in order to have effectiveness, than the doses in the present studies. In line with other clinical trials investigating thromboprophylaxis in THA and TKA, 6 –8,16 the majority of patients participating in these 2 studies were women. Notably, few placebo-controlled studies have currently been undertaken in this area; as such, our findings represent the true effect of darexaban in this patient population.
Results presented here demonstrate the superiority of darexaban over placebo for the prevention of VTE in patients undergoing major orthopedic surgery, who were also receiving nonpharmacologic VTE prophylaxis. In addition, there were relative risk reductions in the total incidence of VTE (primary efficacy outcome) with darexaban 15 mg bid and 30 mg bid versus placebo of 82.8% and 69.4%, respectively, for patients undergoing THA, and of 48.5% and 70.6%, for patients undergoing TKA. These findings compare favorably with the results from 2 randomized, double-blind, placebo-controlled trials in Japanese patients undergoing THA or TKA, who received the same dosing regimen of enoxaparin. In those studies, the relative risk reduction versus placebo for VTE was 52% and 51% for the patients with THA and TKA, respectively. 7 The incidence of total VTE in the placebo arms of our 2 studies (THA = 17.1%, TKA = 52.8%) was relatively low but may be attributed to a number of factors, including the recognition of the risk of VTE in Japanese patients after orthopedic surgery and the subsequent introduction of guidelines in 2004 for its prevention, 17 recent advancements in surgical techniques and postoperative care, the use of IPC, and earlier postoperative ambulation. In both studies, statistically significant differences were also observed in the incidence of total DVT, distal DVT only, asymptomatic DVT, and distal asymptomatic DVT only (secondary efficacy outcomes) in both the darexaban 15 and 30 mg bid groups compared with placebo. During the treatment period, the PT-INR increased in the darexaban groups around peak and trough; this was greater in the darexaban 30 mg bid group than the darexaban 15 mg bid group. A clear pharmacokinetics (PK)/PD relationship was observed between plasma darexaban glucuronide concentration and PT-INR.
Overall, darexaban, administered at the doses of 15 and 30 mg bid, was well tolerated. The incidences of bleeding events were comparable across all treatments groups in the THA study, with very low rates of major and CRNM bleeding, and no major bleeding events reported in any treatment group. Similar results were obtained in the TKA study, with the exception of a higher rate of minor bleeding events in the enoxaparin group relative to both placebo and darexaban. Other studies in Japanese patients undergoing THA or TKA have also reported higher rates of minor bleeding with enoxaparin 20 mg bid than with placebo. 7 In the THA study, the incidence of AEs was highest in the enoxaparin 20 mg bid group and comparable in the darexaban and placebo treatment groups. In the TKA study, the incidence of AEs was comparable across all treatment groups. Notably, increases in the dose of darexaban did not appear to result in an increase in the incidence of AEs. Although transient elevations of liver transaminases and/or bilirubin have been widely reported with enoxaparin and novel oral anticoagulants, including darexaban, 12,13, 18, 19 in the current 2 studies, laboratory evaluations revealed no clinically relevant adverse effects on liver, renal, or cardiac function.
Current Japanese guidelines recommend that patients undergoing THA or TKA should receive IPC or specific pharmacological therapy with UFH, enoxaparin, or fondaparinux. 5, 17, 20 However, all these products need to be administered parenterally, which is inconvenient for patients and can have negative impact on the quality of life. These limitations can result in poor anticoagulant control which can lead to both bleeding complications 21 and the underuse of appropriate anticoagulation therapy. 22 –25 Although there is no antidote for darexaban, darexaban offers patients anticoagulant therapy without the requirement for frequent monitoring and dose adjustment. Furthermore, results from these 2 studies demonstrate that darexaban effectively prevents VTE without increasing the risk of bleeding or other AEs in patients undergoing THA or TKA.
Conclusion
Treatment with oral darexaban 15 mg or 30 mg bid for 10 to 14 days after elective THA or TKA is associated with a significantly reduced rate of VTE when compared with placebo and a similar rate to that observed with enoxaparin 20 mg bid. The incidence of AEs and bleeding events in both darexaban groups was comparable with that in the placebo group, and no new safety concerns were identified. Based on these findings, darexaban is effective and well tolerated when administered as VTE prophylaxis in Asian patients undergoing elective major orthopedic surgery.
Footnotes
Acknowledgments
The adjudication committee and the principal investigators are as follows: the adjudication committee members for VTE: S. Fujita, H. Nakamura, K. Nakamura, and S. Nakata; the adjudication committee members for safety: S. Ozeki and S. Hasegawa; the principal investigators for total hip arthroplasty study: H. Yajima, S. Miyano, M. Inoue, S. Abe, K. Kawasaki, M. Maruo, H. Kakizaki, S. Harada, H. Mishima, S. Iida, Y. Harada, J. Chiba, T. Ishii, R. Izumida, T. Okuaki, F. Sukezaki, T. Matsuda, S. Hasegawa, M. Matsubara, Y. Yabuki, M. Kosugi, J. Mibe, M. Sakamoto, K. Sugimoto, T. Mori, S. Kondo, M. Nakamura, K. Hirakawa, M. Ito, T. Matsumoto, S. Iwasada, S. Mori, A. Sano, A. Sudo, Y. Okuda, S. Yamazoe, H. Iwaki, H. Ohashi, K. Fujiwara, M. Saito, T. Nishimura, T. Nonaka, M. Kim, M. Matsuura, N. Shibanuma, B. Wadayama, T. Matsubara, K. Kawate, N. Ikeda, T. Sato, S. Mitani, T. Ozaki, S. Miyoshi, M. Matsushita, S. Kuratsu, J. Arima, Y. Yasunaga, S. Nakano, J. Nakashiro, T. Mori, Y. Ito, H. Miura, Y. Nakashima, F. Higuchi, M. Mawatari, K. Akasaki, N. Kaku, Ching-Jen Wang, Mel. S. Lee, Robert Wen-Wei Hsu, Yi-Chin Fong, Tai-Sheng Tan, Cheng-Yo Yen, Youn Soo Park, Taek Rim Yoon, Shin-Yoon Kim, Sattaya Rojanasthien, Vatanachai Rojvanit, Thanainit Chotanaphuti, Yongsak Wangroongsub; the principal investigators for total knee replacement study: H. Yajima, K. Ishizaki, S. Miyano, F. Oha, M. Inoue, S. Nagasaki, H. Kakizaki, Y. Onuma, T. Kikuchi, K. Okuma, S. Iida, J. Chiba, T. Ishii, S. Katoh, T. Nakamura, F. Sukezaki, T. Matsuda, T. Nagase, T. Tateishi, M. Kosugi, J. Mibe, M. Sakamoto, K. Sugimoto, A. Hyoudo, K. Nishimoto, K. Hirakawa, T. Matsumoto, S. Tsuboi, S. Mori, A. Sano, T. Sato, J. Kusada, A. Sudo, Y. Okuda, H. Ohashi, K. Fujiwara, S. Nozaki, M. Saito, T. Nishimura, K. Imoto, M. Akagi, Y. Kadoya, N. Shibanuma, T. Matsubara, A. Okuda, N. Ikeda, T. Sato, N. Abe, S. Miyoshi, M. Matsushita, S. Kuratsu, J. Arima, M. Deie, H. Miura, F. Higuchi, S. Ide, E. Nakamura, Yi-Chin Fong, Cheng-Yo Yen, Ing-Lin Chang, Tai-Sheng Tan, Sattaya Rojanasthien, Vatanachai Rojvanit, Thanainit Chotanaphuti, Yongsak Wangroongsub. The authors thank Frances Gambling from Medicus International for editorial assistance. Editorial support was funded by Astellas Pharma.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Takeshi Fuji has received consulting fees from Astellas, Bayer and Daiichi-Sankyo, and royalties from Showa Ikakogyo. Mashio Nakamura has received consulting fees from Astellas, and consulting fees and honoraria from GSK. Masahiro Takeuchi has received consulting fees from Astellas.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were funded by Astellas. Editorial support was provided by Medicus International and funded by Astellas.
