Abstract
We examined the hypothesis that the factor V Leiden (FVL) and G20101A prothrombin gene mutations are commonly associated with hip osteonecrosis. We prospectively evaluated 244 consecutively referred adults with osteonecrosis (ON), 161 idiopathic and 83 secondary. Cases (n = 244) did not differ from 104 normal controls by race. Of the 244 patients, 23 (9.4%) were FVL heterozygotes versus 2 of 104 controls (1.9%), P = .013, risk ratio (RR) = 4.90, 95% confidence interval (CI) 1.18 to 20.4. Of the 161 patients with idiopathic ON, 15 (9.3%) were FVL heterozygotes versus 2 of 104 normal controls (1.9%), P = .017, RR = 4.84, 95% CI 1.13 to 20.8. Of the 83 patients with secondary ON, 8 (9.6%) FVL heterozygotes versus 2 of 104 normal controls (1.9%), P = .024, RR = 5.01, 95% CI 1.09 to 23.0. Prothrombin gene heterozygosity in normal controls (2.9%) did not differ from ON cases (3.4%), P = 1.0. The thrombophilic FVL mutation is commonly associated with and may be pathoetiologic for hip osteonecrosis.
Introduction
Osteonecrosis of hips in adults 1 –13 and Legg-Calve-Perthes disease in children 14,15 is commonly associated with familial thrombophilia–hypofibrinolysis. It has been hypothesized that the development of osteonecrosis (ON) is mediated by venous thrombosis in the femoral head, leading to increased intraosseous venous pressure, and thence to impaired arterial flow, osseous hypoxia, and bone death. 1 –4,6,16 Bjorkman et al have reported that the factor V Leiden (FVL) and 20210A prothrombin gene (PTG) mutations were more frequent in patients with idiopathic or secondary ON of the hips 12 or knees 17 than in a population of healthy controls. However, in both studies by Bjorkman et al, 12,17 when the contributions of the FVL and PTG mutations were assessed separately, the FVL mutation was the predominant contributor to case–control differences.
The associations of the FVL and PTG mutations with ON are important for 2 reasons. First, the diagnosis provides an opportunity for anticoagulation with enoxaparin with a goal of stopping the progression of idiopathic Ficat stages I and II ON of the femoral head which decreases the frequency of hip replacement. 6,16 Second, particular thromboprophylactic precautions and observation are appropriate because of the increased likelihood of thromboembolism when undertaking total hip or knee replacement in patients with the FVL or PTG mutations and familial thrombophilia. 18 –22
In the current report, we examined the hypothesis that the thrombophilic FVL and PTG mutations are commonly associated with and may be a treatable 6,16 pathoetiology for ON of the hip.
Materials and Methods
Study Design
Our institutional review board approved the research protocol. Signed informed consent was obtained from each patient. The diagnosis of ON was verified by history and physical examination, anteroposterior and frog-leg lateral radiographs, and magnetic resonance imaging scans of the hips. 6,16
Patients
In this prospective cohort study, we studied 244 patients with ON of one or both hips, consecutively referred by orthopedic surgeons to our center between 1997 and 2011 from the United States. We characterized the ON as idiopathic when there was no known underlying etiology, or secondary, associated with long-term and/or high-dose corticosteroids (total dose 3000-4000 mg), 23 alcoholism (>400 mL alcohol/wk), 12 or hip dislocation or fracture. 1,8,23 –25
Normal Control Participants
For comparison of polymerase chain reaction (PCR) measures of the FVL and PTG mutations, our previously described 3 normal controls included 44 healthy adult men (17 hospital personnel and 27 being evaluated for hyperlipidemia) and 60 healthy adult women (23 hospital personnel and 37 being evaluated for hyperlipidemia).
Laboratory Methods
The PCR methods 4,14 were used to identify FVL and PTG mutations and wild-type normal alleles.
Statistical Analyses
The study was powered a priori to examine a well-defined hypothesis, 12 so that the prevalence of the FVL gene mutation in patients with ON would be 15% versus 3% in healthy normal controls. With α = .05 and β = .2, 89 patients and 89 controls would be needed to demonstrate that the FVL mutation differed significantly comparing patients with the controls.
In 2 × 2 tables, patients with ON were compared with normal controls. Differences were assessed by chi-square tests or Fisher exact tests when at least 1 expected cell size was <5. Risk ratios (RRs) with 95% confidence intervals (CIs) were calculated.
Results
Patients did not differ (P > .05) from controls by race, with 219 of 244 patients (90%) white, versus 91 of 104 (88%) controls. Patients’ and controls’ race, gender, and age are displayed in Table 1.
Factor V Leiden Mutation in 244 Patients With Idiopathic and Secondary Osteonecrosis and in 104 Healthy Adult Controls.a
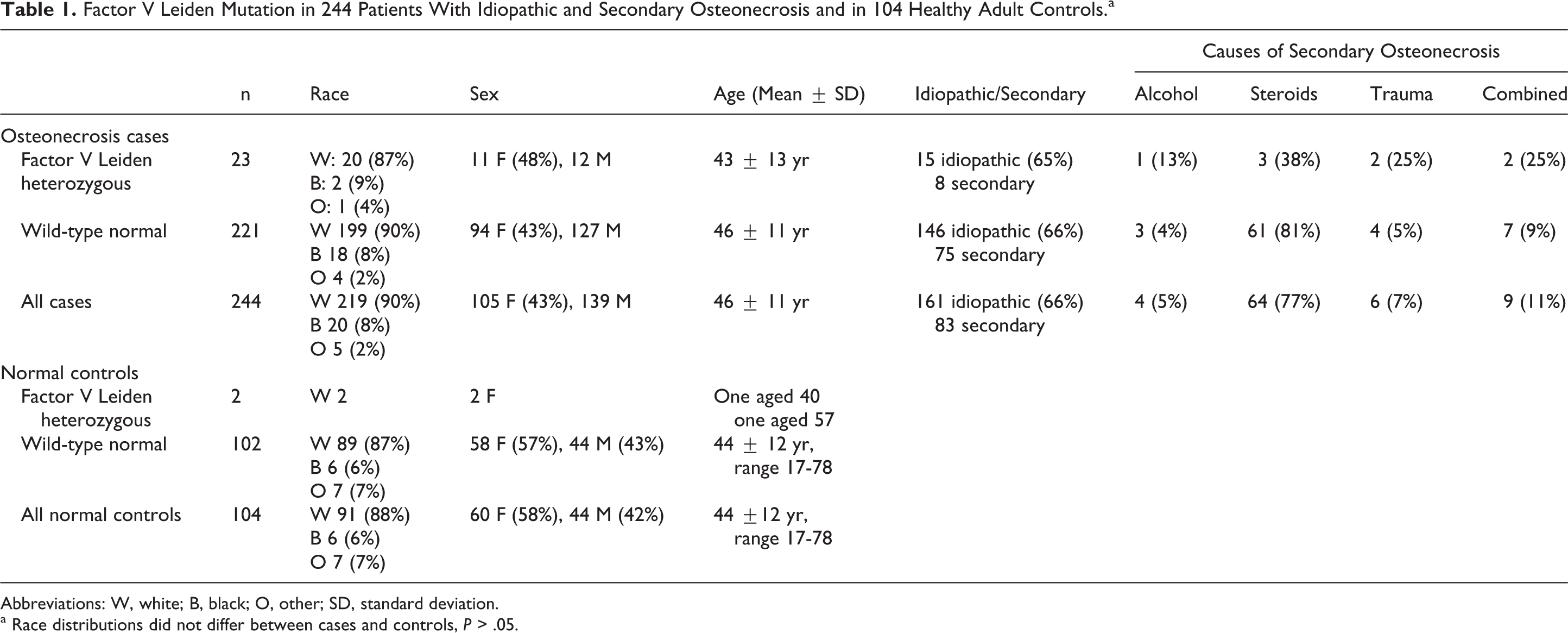
Abbreviations: W, white; B, black; O, other; SD, standard deviation.
a Race distributions did not differ between cases and controls, P > .05.
Of the 244 patients with ON who were referred to our center, ON was idiopathic in 161 and secondary in 83 (Table 1). Use of long-term and/or high-dose corticosteroid was the most common pathoetiology for secondary ON (Table 1).
Of the 244 patients with ON, 23 (9.4%) were heterozygous for the FVL mutation versus 2 of 104 normal controls (1.9%), RR for being FVL = 4.90, 95% CI 1.18 to 20.4, P = .013 (Table 2). Of the 161 patients with idiopathic ON, 15 (9.3%) were heterozygous for the FVL mutation versus 2 of 104 normal controls (1.9%), RR = 4.84, 95% CI 1.13 to 20.8, P = .017 (Table 2). Of the 83 patients with secondary ON, 8 (9.6%) were heterozygous for the FVL mutation versus 2 of 104 normal controls (1.9%), RR = 5.01, 95% CI 1.09 to 23.0, P = .024 (Table 2).
Factor V Leiden Mutation in 244 Patients With Osteonecrosis Compared With 104 Healthy Adult Controls.
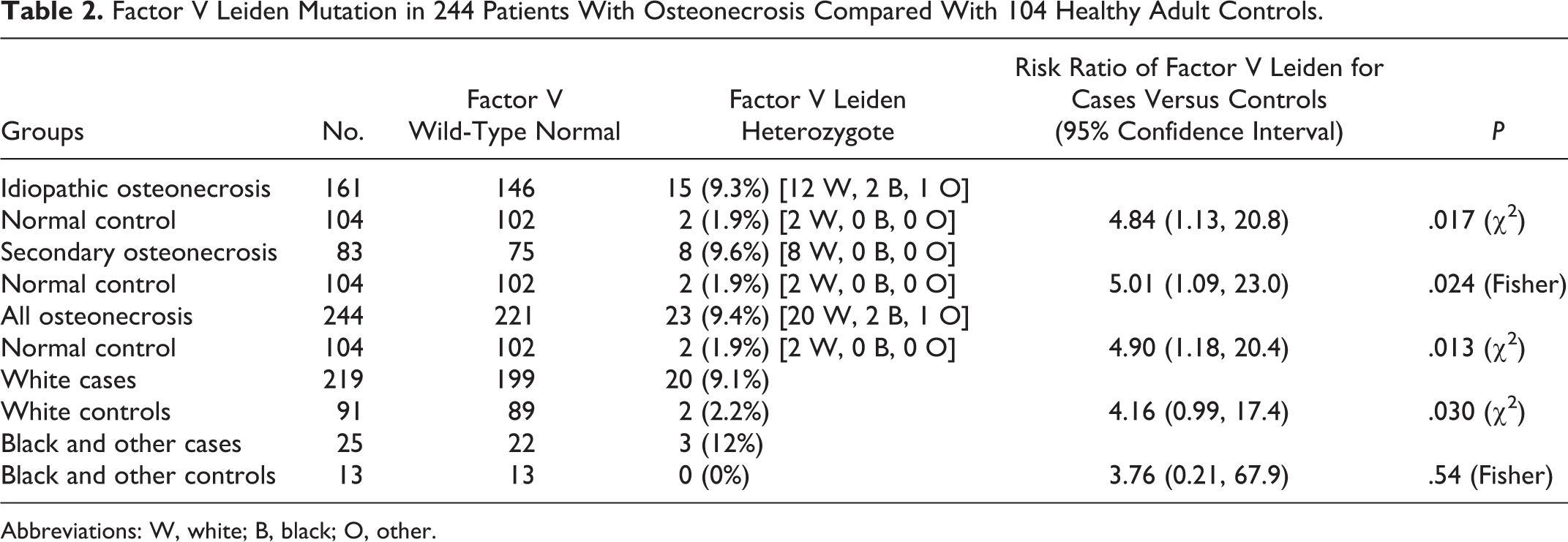
Abbreviations: W, white; B, black; O, other.
The FVL mutation differed in white cases versus white controls, 9.1% versus 2.2% (P = .03). Although the FVL mutation was more common in black/other race cases than in controls, 12% versus 0%, the difference was not significant due to the small number of participants (Table 2).
Of the 235 patients having measures of PTG, 8 (3.4%) were heterozygous for PTG, not different than 3 of 104 controls (2.9%), RR = 1.18, 95% CI 0.32 to 4.36, P = 1.0 (Table 3).
The PTG Mutation in 235 Patients With Osteonecrosis Compared With 104 Healthy Adult Controls.
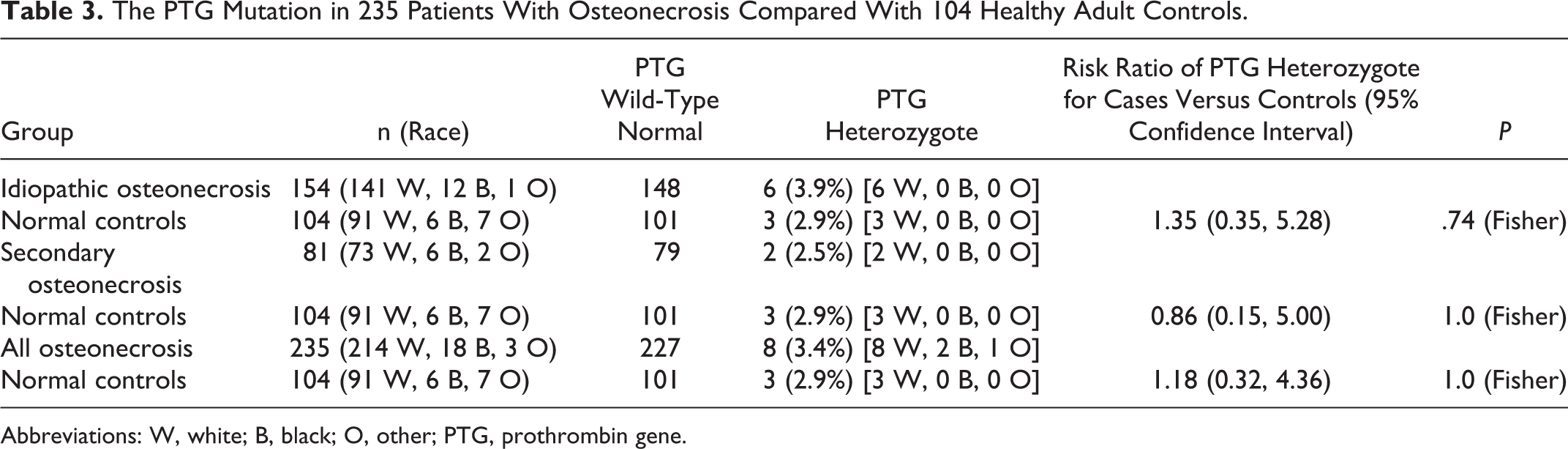
Abbreviations: W, white; B, black; O, other; PTG, prothrombin gene.
Discussion
In adults, the FVL mutation or PTG or both have been reported to be significantly more common in patients with ON of the hip 12 and knee 17 than in healthy control participants. In children with Legg-Calve-Perthes disease, Balasa et al 14 reported that the FVL mutation was more common in cases than in controls, odds ratio (OR) 3.39, 95% CI of 1.18 to 9.73. Vosmaer et al 15 reported that the incidence of Legg-Calve-Perthes disease was increased in the presence of the FVL mutation (OR, 3.3; 95% CI: 1.6-6.7).
As has been shown with other gene mutations which cause thrombophilia, 12,17,26 –28 hypofibrinolysis, 26 –29 or reduction of nitric oxide production, 3,30,31 the association of the FVL or PTG mutations with ON is important because it provides an opportunity for medical treatment. Anticoagulation with enoxaparin 6,16 in patients with thrombophilia can stop the progression of idiopathic Ficat stages I 32 and II ON of the femoral head, thus decreasing the frequency of hip replacement.
Our study affirms the hypothesis that the FVL mutation is a risk factor for ON of the femoral head, present in 9.3% of 161 patients with idiopathic ON and in 9.6% of 83 patients with secondary ON. Similar to the reports of Bjorkman et al in ON of the hip 12 or knee, 17 in the current study, the PTG mutation was not by itself a significant risk factor for ON of the femoral head, present in 3.4% of 235 patients with ON versus 2.9% of 104 normal controls.
The prevalence of FVL mutation in our control population, 2.2% in Caucasians, was lower than our previously reported 3.5% (6 of 170) in healthy Cincinnati Caucasian children, 14 and lower than reported by Ridker et al, 33 5.27% in Caucasian Americans, 2.21% in Hispanic Americans, 1.23% in African Americans, 0.45% in Asian Americans, and 1.25% in Native Americans. Gregg et al 34 reported that the FVL mutation was present in 4.4% of California Caucasians, and the prevalence varies widely in the Mediterranean region, as low as 2%. 35 Hence, there is considerable variance in the prevalence of the FVL mutation related to geographic area, ethnic group, and the degree of ethnic mixture within ethnic groups. 14,33 –35
Diagnosis of the presence of the FVL mutation can facilitate a medical, anticoagulant approach to stop the progression of ON before collapse of the head of femur (Ficat stages I and II) 6 16 and enables aggressive peri-postoperative thromboprophylaxis to manage the increased likelihood of deep venous thrombosis and pulmonary embolus. 18 –22 Diagnostic focus on first-degree family members, half of whom (on average) will have the same gene mutation with increased risk of ON and thrombosis 36 –38 may also prove a useful product of this investigation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: in part by the Lipoprotein Research Fund of the Jewish Hospital of Cincinnati.
