Abstract
This is the first study from north India that investigated the association of thrombomodulin (TM) polymorphisms with acute myocardial infarction (AMI) in 350 patients (≤ 40 years, n = 184 and ≥ 60 years, n = 166) and 350 matched-controls. The TM polymorphisms were determined by polymerase chain reaction–single-stranded conformational polymorphism and DNA sequencing. The TM 1418TT genotype (odds ratio [OR] 2.8; 95% confidence interval [CI] 1.3-6.4; P = .012) was independent risk predictor of young AMI as were hypertension (OR 3.3; 95% CI 1.8-5.9; P < .001), diabetes mellitus (OR 14.3; 95% CI 2.9-44.6; P = .001), smoking (OR 13.8; 95% CI 7.7-24.7; P < .001), family history (OR 1.8; 95% CI 1.1-3.3; P = .045), high body mass index (OR 2.2; 95% CI 1.3-3.6; P = .002), and high waist–hip ratio (OR 4.1; 95% CI 2.4-7.1; P < .001). Mean plasma TM also showed association with young AMI (P < .001). Smoking carriers of TM 1418CT + TT genotype had significantly higher risk of AMI (OR 12.8; 95% CI 6.0-27.3; P < .001) when compared with nonsmoking noncarriers. In conclusion, TM 1418C/T polymorphism is independent predictor of AMI and synergies with smoking.
Introduction
Acute thrombosis at the site of a ruptured, lipid-rich atherosclerotic plaque in coronary artery is the most important pathogenetic mechanism in acute myocardial infarction (AMI). 1 Although several traditional coronary risk factors such as hypertension, diabetes mellitus, smoking, alcohol intake, family history of coronary artery disease (CAD), and obesity play roles in the development of AMI, there is an increased awareness of the contribution of polymorphic variants of genes encoding proteins involved in the hemostatic system as risk factors. 2 –4 Thrombomodulin (TM) is an endothelial cell surface glycoprotein receptor for thrombin. It suppresses blood coagulation by decreasing thrombin procoagulant activities, including fibrinogen clotting and platelet activation. The TM-thrombin complex also activates protein C that catalyzes the proteolytic degradation of blood clotting factors V and VIII. 5 Therefore, TM acts as an important physiological anticoagulant and deficiency of this protein could result in excessive thrombus formation. Studies have suggested that dysfunction of TM plays an important role in the pathogenesis of MI. 6
Human TM gene, located at chromosome 20p11.2, is a single copy gene that contains 6 epidermal growth factor (EGF)-type repeats, lack introns, and pseudogenes. 7 Recent studies have identified proximal promoter regions in TM gene that are important transcriptionally, providing positive and negative regulatory elements for constitutive and modulated expression. 8 Several polymorphisms in coding and promoter region of the TM gene have been identified which have predisposing effects in MI. 9 –12 The influence of these gene polymorphisms over the level or activity of plasma TM is unclear. There are no reports from India on the role of TM gene polymorphisms and plasma TM levels in association with AMI which raises the question of whether these have predisposing effects on AMI in our population. This prompted us to study the TM gene polymorphisms and plasma TM levels in north Indian patients with AMI for the first time. In India, CAD resulting in AMI is one of the leading causes of premature death among the middle aged. Identification of a genetic predisposition to AMI would thus have a beneficial impact on its prevention in the north Indian population.
The aim of the present study was to investigate whether TM gene polymorphisms (–33G/A, GG–9/–10AT, and 1418C/T [Ala455Val]) are associated as independent risk factors for AMI in north Indians. Correlation of the TM gene polymorphisms with plasma TM levels was also investigated.
Materials and Methods
Participants
A total of 350 patients were recruited from the Department of Cardiology, Postgraduate Institute of Medical Education and Research, Chandigarh, India. Two age groups of patients were studied (≤ 40 years, n = 184 and ≥ 60 years, n = 166) to determine whether the genetic influences play a greater role in young patients compared with the elderly patients. Diagnosis of definite AMI was based on World Health Organization criteria, 13 that is, patients should have any 2 of the following 3 conditions: resting anginal chest pain lasting more than 30 minutes, ST segment elevation more than 1 mm above baseline in at least 2 leads of a standard 12-lead electrocardiogram and creatinine phosphokinase (CPK), and CPK-MB elevation to at least twice the upper limit of the normal range. Controls comprised of 350 random unrelated young (≤ 40 years) healthy participants from north India and matched with patients in terms of geographic origin and ethnicity. None of the individuals reported history of any cardiac event or hospitalization. The sample size was determined using EPIINFO statistical package. The patients and controls were enrolled between July 2006 and December 2009. The ethics committee of the institute approved the study. All the participants gave their informed written consent prior to their inclusion in the study.
The data on demographic characteristics and the presence of traditional coronary risk factors were collected from all study participants, which included hypertension, diabetes mellitus, family history of CAD, obesity, smoking, and alcohol intake. The participants were considered to have hypertension if elevated blood pressure (>140/90 mm Hg) was measured or if they were already being treated with antihypertensive agents. They were defined as having diabetes mellitus if they had a fasting blood glucose level >140 mg/dL or were already being treated for diabetes. A positive family history was defined as the presence of at least one first- or second-degree relative with a history of CAD. In terms of smoking and alcohol intake, the participants were classified on the basis of self-report. Body mass index (BMI) was categorized as normal weight (18.5-22.9 kg/m2), overweight (23-27.5 kg/m2), obese (27.6-40 kg/m2), and morbidly obese (>40 kg/m2). Normal waist–hip ratio (WHR) for males is ≤0.8 and for females is ≤ 0.95. Normal plasma TM levels according to the TM enzyme-linked immunosorbent assay (ELISA) kit ranges from 2.39 to 7.9 ng/mL. Blood sampling was performed at least 4 weeks after the onset of coronary event when the patient had been hemodynamically stabilized. Aseptic venous blood samples were obtained after overnight fasting and collected in EDTA-containing vials for genetic analysis and plasma TM estimation.
Estimation of Plasma TM Levels
Samples were assayed for TM antigen using a commercially available ELISA (Immubind TM, American Diagnostica Inc, Greenwich, Connecticut) that uses 2 monoclonal antihuman TM antibodies. The capture antibody is directed against EGF1-EGF2 and the horseradish peroxidase-conjugated antibody is directed against EGF5-EGF6.
Genetic Analysis
Genomic DNA was isolated from white blood cells by phenol–chloroform extraction and ethanol precipitation. The TM promoter fragment containing –33 nucleotide and –9, –10 nucleotide was amplified from genomic DNA by a polymerase chain reaction (PCR) in a final volume of 25 μL containing 50 ng of DNA, 0.5 mmol/L deoxyribonucloetide triphosphate (dNTP) mix (Fermentas Life Sciences, New Delhi, India), 1× PCR buffer (10 mmol/L Tris-HCl [pH 9], 1.5 mmol/L MgCl2, 50 mmol/L KCl, and 0.01% gelatin), 1 U of Taq DNA polymerase (Bangalore Genei, Bangalore, India) and 0.1 μmol/L of each primer (MWG Biotech, Bangalore, India). The sequences of the primers 9 were 5′-CAGCAATCCGAGTATGCGG-3′ and 5′-CTCCTGTCCGTCCCAGCC-3′ (corresponding to nucleotides –112 to –94 and +69 to +52, respectively; nucleotide number according to Wen et al 8 ). The PCR was carried out for 35 cycles, with each cycle consisting of 1 minute of denaturation at 94°C, 1 minute of annealing at 63°C, 45 seconds of extension at 72°C, and a final extension at 72°C for 10 minutes. A 181 base pair (bp) DNA fragment was obtained. The TM –33G/A polymorphism was detected by single-strand conformation polymorphism (SSCP). Three microliter aliquots of PCR product were denatured by the addition of 3 μL denaturing solution containing 95% formamide, 0.05% bromophenol blue, 0.05% xylene cyanol, and 20 mmol/L EDTA and heated to 95°C for 10 minutes, followed by rapid cooling on ice before loading on gel. The electrophoresis was performed on 15% polyacrylamide gels with 10% glycerol at 4°C, 20 mA for 18 hours. The SSCP was performed using the DCode Universal Mutation Detection System from BioRad Laboratories, Gurgaon, India. Gels were visualized using silver staining.
The PCR-SSCP was done to determine 1418C/T (Ala455Val) polymorphism in exon of TM gene. Amplification of DNA was carried out by PCR in a final volume of 25 μL containing 50 ng of DNA, 0.5 mmol/L dNTP mix, 1× PCR buffer (10 mmol/L Tris-HCl [pH 9], 1.5 mmol/L MgCl2, 50 mmol/L KCl, and 0.01% gelatin), 1 U of Taq DNA polymerase and 0.1 μmol/L of each primer. The following primer set resulted in 153 bp fragment 5′-CGGTACCTTCGAGTGCATCT-3′ and 5′-ACGG-CCGGAGGAGTCAAGGT-3′. 10 The PCR was carried out for 35 cycles consisting of 1 minute of denaturation at 94°C, 1 minute of primer annealing at 63°C and 45 seconds of extension at 72°C, and final extension at 72°C for 10 minutes. The SSCP was performed on 12% polyacrylamide gel with 10% glycerol using the DCode Universal Mutation Detection System from BioRad. Gels were visualized using silver staining. Samples showing shifts in the mobility pattern on the gel were analyzed by DNA sequencing (ABI 3130 Genetic Analyzer, Applied Biosystems, Gurgaon, India). In sequence chromatogram where the sequence was neat with no background, sequencing in both directions was not performed. However, in all cases where the reads were not neat, resequencing from an independent PCR reaction was performed for confirmation of the results.
Statistical Methods
Plasma TM levels were expressed as mean value ± standard deviation. Mann-Whitney U test was used to compare plasma TM levels among patients and controls. The TM genotype and allele frequencies were estimated by gene counting and expressed as percentages of the totals. Comparison of prevalence of TM gene polymorphisms among patients and controls was done by chi-square (χ 2 ) test. To analyze the traditional risk factors stratified according to TM genotypes, χ 2 test was performed. Mann-Whitney U test and Kruskal-Wallis test were used to investigate the correlation between plasma TM levels with TM gene polymorphisms. Multinominal logistic regression analysis was used to study synergistic effects of TM gene polymorphisms and smoking on the occurrence of AMI. Multiple logistic regression analysis was performed to adjust for the confounding effects of traditional coronary risk factors. The strength of association of TM gene polymorphisms with the occurrence of AMI was estimated by calculation of the odds ratio (OR) and the Cornfield method for the calculation of 95% confidence intervals (CIs). A P value of <.05 was considered to be statistically significant. All the computations were carried out using the program, Statistical Package for the Social Sciences (SPSS), version 13.0, Chicago for windows.
Results
Demographic Characteristics and Traditional Coronary Risk Factors
The study population comprised of 350 controls (mean age 31.1 ± 6.0 years, 75.7% males) and 350 patients, of which there were 184 patients surviving a first AMI at the age of ≤40 years (mean age 36.4 ± 4.5, 96.2% males) and 166 patients surviving a first AMI at the age ≥60 years (mean age 64.1 ± 5.1 years, 74.7% males).
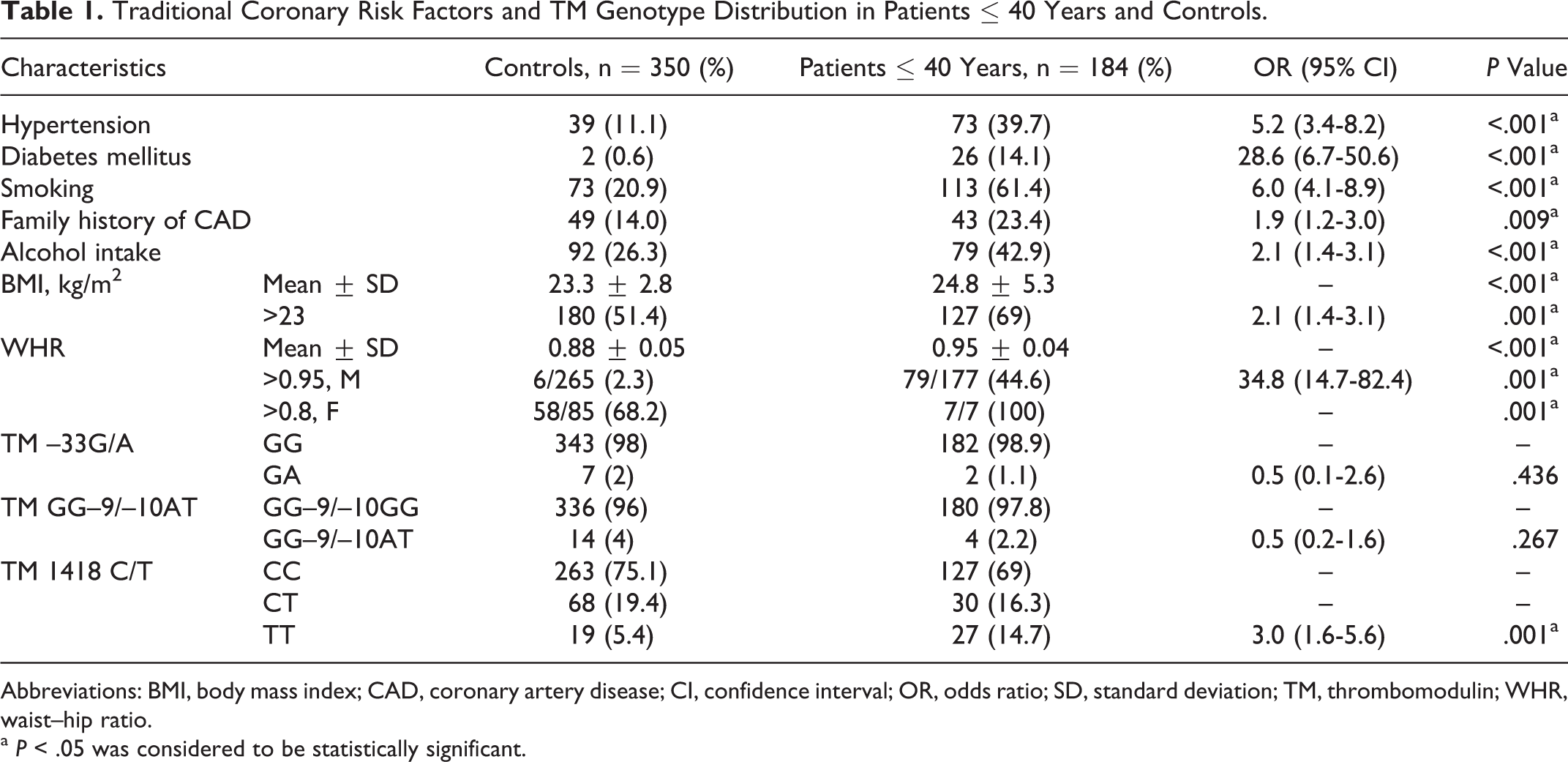
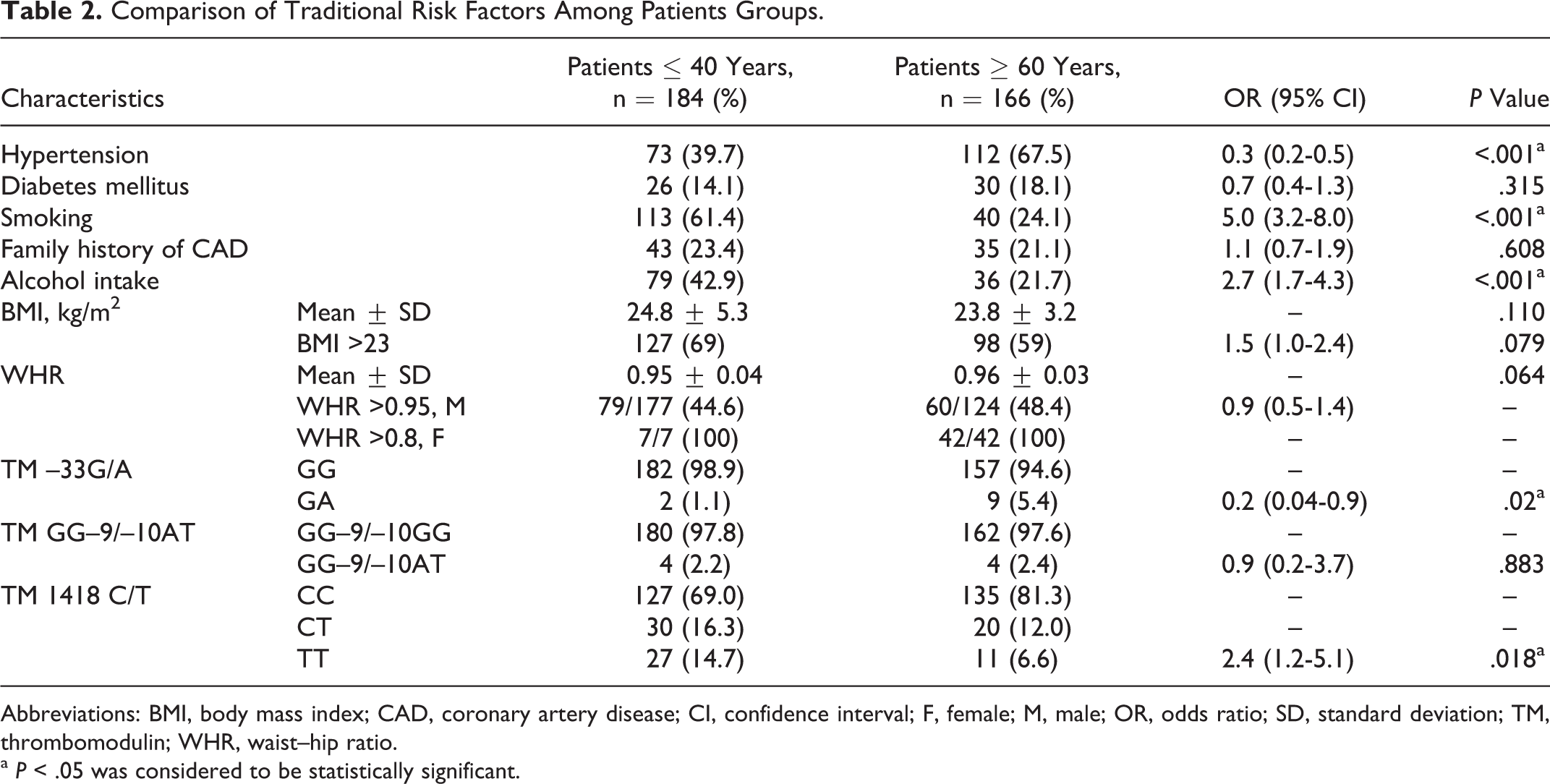
Comparison of traditional risk factors among patients aged ≤ 40 years and controls is shown in Table 1. Hypertension (P < .001), diabetes mellitus (P < .001), smoking (P < .001), family history of CAD (P = .009), alcohol intake (P < .001), high BMI (P < .001), and high WHR (P < .001) were more frequent in young patients with AMI than controls. When the two patient groups were compared (Table 2), hypertension was more prevalent in elderly patients while smoking and alcohol intake were more prevalent in young (P < .001).
Traditional Coronary Risk Factors and TM Genotype Distribution in Patients ≤ 40 Years and Controls.
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CI, confidence interval; OR, odds ratio; SD, standard deviation; TM, thrombomodulin; WHR, waist–hip ratio.
a P < .05 was considered to be statistically significant.
Comparison of Traditional Risk Factors Among Patients Groups.
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CI, confidence interval; F, female; M, male; OR, odds ratio; SD, standard deviation; TM, thrombomodulin; WHR, waist–hip ratio.
a P < .05 was considered to be statistically significant.
Plasma TM Levels
Plasma TM levels were in the range of 1.0 to 18 ng/mL in controls, 1.25 to 15.4 ng/mL in young patients, and 2.5 to 16.8 ng/mL in patients ≥ 60 years. Mean plasma TM level was significantly higher (P < .001) in young patients (5.2 ± 2.5 ng/mL) than controls (4.7 ± 2.8 ng/mL). It was even higher (P = .001) in patients aged ≥60 years (6.0 ± 2.7 ng/mL) than patients aged ≤40 years (5.2 ± 2.5 ng/mL).
TM Gene Polymorphisms
Comparison of TM gene polymorphisms among patients with AMI and controls is shown in Table 1. The variant 1418T (455Val) allele in TM gene occurred with higher frequency in young patients as compared to that in the controls (P = .001). However, the prevalence of –33G/A polymorphism (GA genotype; P = .436) and GG–9/–10AT polymorphism (–9GA, –10GT genotypes; P = .267) showed no significant difference between patients ≤ 40 years and controls. When the two patient groups were compared (Table 2), frequency of TM –33G/A polymorphism (GA genotype) was significantly higher in elderly patients than young (P = .02), while that of TM 1418TT (455Val) genotype was significantly higher in young patients than elderly patients (P = .018). The TM GG–9/–10AT polymorphism (–9GA, –10GT genotypes) did not show significant difference in the distribution of their genotypes among the two patient groups (P = .883).
Identification of Independent Risk Factors for AMI
Multiple logistic regression analysis showed that hypertension (OR 3.3; 95% CI 1.8-5.9; P < .001), diabetes mellitus (OR 14.3; 95% CI 2.9-44.6; P = .001), smoking (OR 13.8; 95% CI 7.7-24.7; P < .001), family history of CAD (OR 1.8; 95% CI 1.1-3.3; P = .045), high BMI (OR 2.2; 95% CI 1.3-3.6; P = .002), high WHR (OR 4.1; 95% CI 2.4-7.1; P < .001), and TM 1418TT (455Val) genotype (OR 2.8; 95% CI 1.3-6.4; P = .012) were independent risk predictors of AMI in young.
When the two patient groups were compared, hypertension (OR 0.3; 95% CI 0.2-0.6; P < .001) and TM –33GA genotype (OR 0.07; 95% CI 0.01-0.4; P = .003) were more prevalent in elderly patients while smoking (OR 3.8; 95% CI 2.2-6.5; P < .001) and TM 1418TT (455Val) genotype (OR 3.5; 95% CI 1.5-8.1; P = .004) were more prevalent in young patients.
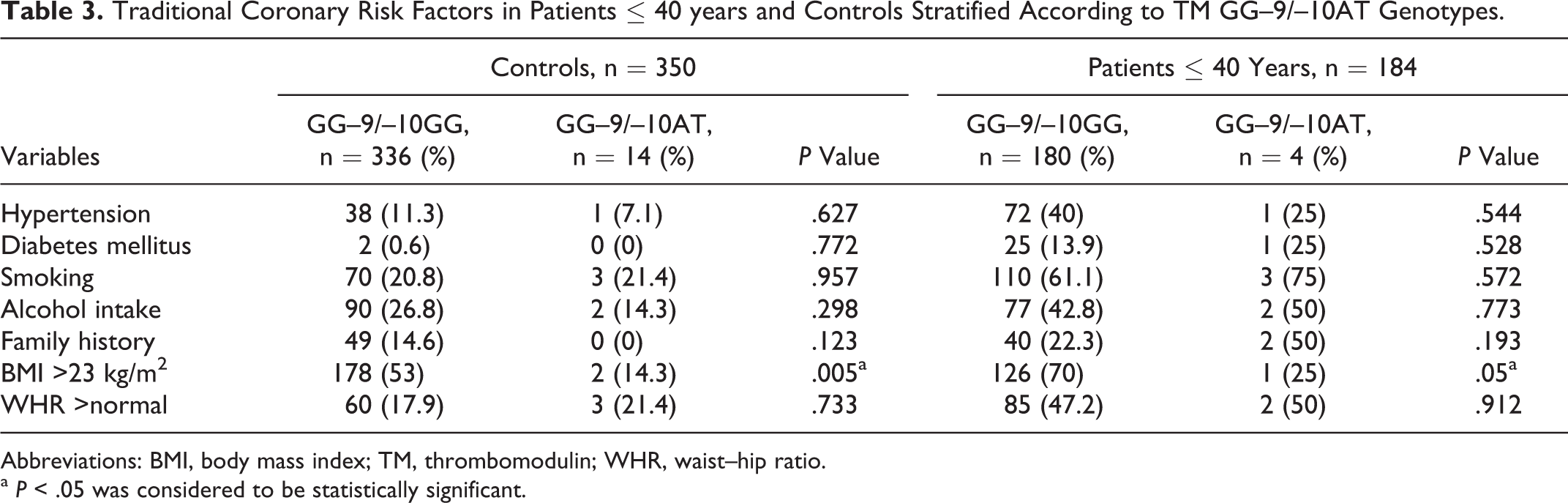
TM Genotypes Stratified According to Traditional Coronary Risk Factors
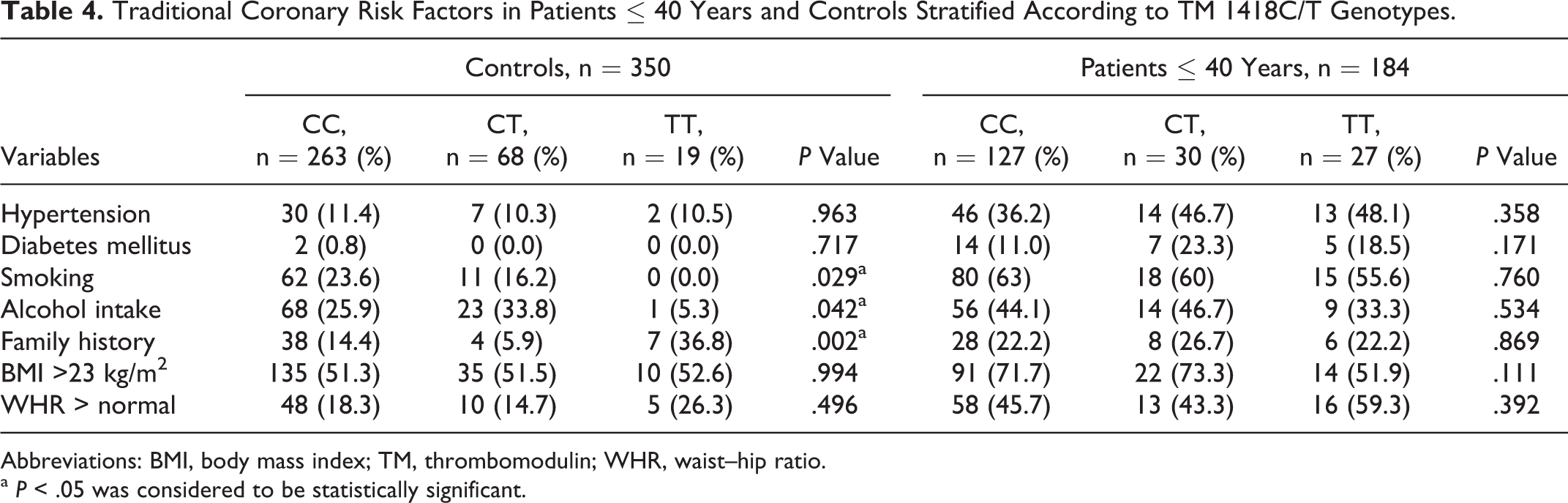
Analyzing the traditional coronary risk factors in young patients with AMI and controls stratified by the TM GG–9/–10AT genotypes (Table 3), significant statistical difference with high BMI in controls (P = .005) as well as young patients was observed (P = .05). When analysis was done with TM 1418C/T genotypes (Ala455Val; Table 4), prevalence of smoking (P = .029) and alcohol intake (P = .042) was higher in controls with TM 1418CC (Ala455) genotype while that of family history of CAD (P = .002) was higher in controls with 1418TT (455Val) genotypes. Similar analysis on TM –33G/A genotypes showed no significant difference (P > .05, data not shown).
Traditional Coronary Risk Factors in Patients ≤ 40 years and Controls Stratified According to TM GG–9/–10AT Genotypes.
Abbreviations: BMI, body mass index; TM, thrombomodulin; WHR, waist–hip ratio.
a P < .05 was considered to be statistically significant.
Traditional Coronary Risk Factors in Patients ≤ 40 Years and Controls Stratified According to TM 1418C/T Genotypes.
Abbreviations: BMI, body mass index; TM, thrombomodulin; WHR, waist–hip ratio.
a P < .05 was considered to be statistically significant.
Interaction of TM Genotypes With Smoking
Significant interaction between smoking and TM 1418C/T polymorphism (1418 CT + TT genotype) on the occurrence of young AMI was observed. Among patients who did not smoke, TM 1418C/T polymorphism (1418 CT + TT genotype) was not associated with an increased risk of AMI (OR 1.4; 95% CI 0.8-2.4; P = .291). Smoking carriers of TM 1418C/T polymorphism (1418 CT + TT genotype) had a significantly higher risk of AMI (OR 12.8; 95% CI 6.0-27.3; P < .001) when compared with patients who were nonsmoking noncarriers. No interaction of smoking with TM GG–9/–10AT and TM –33G/A polymorphism on the occurrence of AMI in young was observed.
Correlation of TM Levels With Genotypes
There was no correlation between plasma TM levels and TM –33G/A, TM GG–9/–10AT, and TM 1418C/T (Ala455Val) genotypes (P > .05) in young patients with AMI as well as controls.
Discussion
This is the first study from India that has investigated the role of TM gene polymorphisms and plasma soluble TM levels in association with AMI in north Indians. Mean plasma soluble TM level was significantly higher in patients as compared with the controls, showing its association with risk of AMI in young. In our study, mean plasma soluble TM level were even higher in elderly patients than that of young. In contradiction to our study, Salomaa et al found that the high concentration of soluble TM is independently associated with decreased risk of coronary heart disease (CHD) in a large population-based case-cohort study among initially healthy individuals. 6 Several studies in literature, however, support the results from present study. Blann et al reported that among 54 survivors of MI, soluble TM was directly related to the risk of a recurrent cardiovascular event during 49 months of follow-up. 14 Cross-sectional study by Blann et al has also reported a positive association between soluble TM and different manifestations of atherosclerosis, such as CHD and peripheral occlusive arterial disease. 15 Li et al found in Han Chinese that soluble TM level was significantly higher in patients with CAD than in controls (P < .05), 16 which is similar to that of present study. Soluble TM in plasma is a degradation product of endothelial TM. Results from a recent Atherosclerosis Risk in Communities Study (ARIC) report by Salomaa et al in 1997 suggested that plasma concentrations of soluble TM may also be affected by increased proteolytic activity that cleaves soluble TM from endothelial TM. 6 The proteolytic enzymes that catalyze the cleavage are thought to be released from activated white blood cells, so it is possible that a high concentration of soluble TM may be related to inflammation. In people with existing atherosclerosis, soluble TM concentration may reflect the degree of endothelial damage and associated inflammation, rather than expression of TM. 6 This clearly explains our results of higher plasma soluble TM levels in patients with AMI as compared to the controls. Elderly patients with AMI have higher degree of atherosclerosis than younger patients, which explains high plasma soluble TM levels in our elderly patients with AMI.
In the present study, TM 1418C/T (Ala455Val) polymorphism was an independent risk factor for AMI as were hypertension, diabetes mellitus, smoking, family history of CAD, and obesity. The influence of TM 1418C/T (Ala455Val) polymorphism on AMI was further enhanced by smoking. The TM residue 455 is located at the EGF-like domain of the extracellular region of endothelial membrane TM. 7 There are 6 EGF-like repeats, and the last 3 repeats are functionally important for protein C activation and thrombin binding. The TM 455 is located in the last EGF-like repeat. A valine substitution for alanine was therefore considered to have the potential of altering the TM activity. Possibly, Ala455 to Val replacement might affect the function of the TM molecule and the activation of the protein C anticoagulant pathway. Several studies support the results from our study. Norlund et al reported the association (P = .035) of TM 1418C (Ala455) allele with premature MI in 97 Swedish MI survivors and 159 healthy controls. 10 In a large cohort study by Wu et al on 376 CHD cases (23% black, 77% white) showed that TM 455Val increased risk of CHD by 6.1-fold (risk ratio 6.1; 95% CI 1.7-22.9) and is an independent risk factor for CHD in blacks. 17 TM Ala455 genotype showed significant protection in noncases (P < .016). A study by Ranjith et al on 195 patients with AMI <45 years of Asian Indian origin in South Africa revealed that TM Ala455Val variant allele was a significant risk factor for CHD. 18 In contrast, no difference was observed between patients and control participants for the allelic frequency of TM 1418C/T (Ala455Val) with 33 of 104 individuals homozygous for 1418T (455Val) within both the patient and the control groups in a study by Ireland et al. 12 Park et al showed that TM C1418T (Ala455Val) was not independent risk factors for either the 1-vessel disease or the 2- or 3-vessel disease. 19 The TM 1418C/T polymorphism (CT + TT genotypes) was not associated with AMI onset in a study by Chao et al. 20
The TM –33G/A polymorphism was not associated with risk of AMI in young nor this polymorphism synergized with smoking. However, TM –33GA genotype was more prevalent in elderly patients than young in our study. The TM –33G/A polymorphism was first identified in the promoter region of the TM gene by Ireland et al. 12 Since then there are contradictory studies in literature, some showing and some not showing association between this polymorphism and risk of AMI. Li et al suggested that there was a significant association of the –33G/A mutation in TM gene with CAD. 16 Study by Li et al showed that TM –33G/A polymorphism (OR 2.3; 95% CI 1.3-4.1) was independent risk factor for young MI in Han Chinese population. 9 Moreover, smoking carriers of TM –33G/A polymorphism had a nearly 10-fold increased risk of young MI (OR 9.8; 95% CI 4.3-22.4) when compared with nonsmoking noncarriers. Park et al indicated that –33G/A polymorphism in TM gene is a genetic risk factor for MI in Koreans. 19 Chao et al in a study on 145 patients with premature AMI and 143 controls indicated that focus must be on TM promoter –33G/A polymorphism in terms of the role of TM gene mutation on premature AMI. 20 However, Zhao et al did not find significant difference in the frequency of the A allele of TM –33G/A polymorphism between patients with CAD and controls (P = .249), between patients with MI and controls (P = .163), or between premature patients with MI and controls (P = .265) in northern Han Chinese, which is consistent with our study. 21
No association of TM GG–9/–10AT polymorphism with AMI was observed in the present study or show synergistic effect with smoking. Ireland et al identified GG–9/–10AT in the 5′ untranslated region of the TM gene in 3 of 104 individuals (2 heterozygous and 1 homozygous Asian). 12 This polymorphism was present in low frequency (<3%) in Caucasian patients with AMI and absent in controls. There is currently no experimental evidence linking the –9/–10 GG to AT substitution to reduced gene transcription, but its proximity to the TATA box can be noted. Park et al could not identify the TM –9/–10AT allele in 190 Korean individuals either by SSCP or by direct sequencing, suggesting that this substitution is very rare in Koreans. 19 This polymorphism was absent in Oriental populations in patients with AMI as well as controls in a study by Chao et al. 20
In the present study, traditional risk factors did not predominate among the TM GG–9/–10AT genotypes, except for high BMI (P = .005) in controls and young patients (P = .05). In controls with TM 1418CC (Ala455) genotypes, prevalence of smoking and alcohol intake was higher while family history of CAD showed higher prevalence with 1418TT (455Val) genotypes. This indicates that interaction of environmental factors with genotypes may exist. However, none of the traditional coronary risk factors had significant statistical difference for TM –33G/A genotypes in patients and controls. Synergistic effect of smoking and TM 1418TT (455Val) polymorphism on the occurrence of AMI was observed. Complex multifactorial nature of AMI is indicated from these observations.
Our data showed that the plasma TM levels were not correlated with TM –33G/A, TM GG–9/–10AT, and TM 1418C/T (Ala455Val) genotypes. In a similar study by Norlund et al on Swedish patients, mean plasma TM levels were not statistically different between TM 1418CC, CT and TT genotypes. 10 However, in contradiction to our study, Li et al 16 showed that soluble TM level was significantly higher (P < .05) in patients with TM GG genotype than in those with TM GA + AA genotypes.
Conclusion
The TM 1418 C/T (Ala455Val) polymorphism is an independent risk factor for AMI in north Indians. Effect of this polymorphism on the risk of AMI is further enhanced by smoking. The TM 1418TT (Ala455Val) genotype is, however, not associated with increase in plasma soluble TM level. The TM –33GA genotype (OR 0.07; 95% CI 0.01-0.4; P = .003) was more prevalent in elderly patients while TM 1418TT (455Val) genotype (OR 3.5; 95% CI 1.5-8.1; P = .004) was more prevalent in young patients. This indicated that the 1418C/T (Ala455Val) dimorphism in TM gene might be etiological factor involved in the pathogenesis of AMI in our young population. The results from the present study supported that some genetic polymorphisms are unique to particular ethnic groups. Ethnic background plays an important role in studying the clinical significance of genetic variation and more such studies from India to validate this finding are needed.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from Department of Science and Technology, India (SR/SO/HS-72/2005) and Council of Scientific and Industrial Research, India [09/141(0164)/2006].
