Abstract
This study compared genetic polymorphisms (factor V Leiden [FVL] 1691G/A, factor VII [FVII] 10976G/A, FVII HVR4, platelet membrane glycoproteins GP1BA 1018C/T, GP1BA VNTR, integrin ITGB3 1565T/C, ITGA2 807C/T and methylenetetrahydrofolate reductase [MTHFR] 677C/T), biochemical (fibrinogen and homocysteine), and conventional risk factors in 184 young and 166 elderly north Indian patients with acute myocardial infarction (AMI). Univariate analysis revealed higher prevalence of hypertension and obesity in elderly patients while smoking, alcohol intake, and low socioeconomic status in young patients (P < .001). Although mean fibrinogen predominated (P = .01) in elderly patients, mean homocysteine was higher (P < .001) among young patients. Prevalence of hyperhomocysteinemia was greater in young than in elderly patients (odds ratio: 2.8, 95% confidence interval: 1.8-4.4, P < .001); however, genetic polymorphisms were equally prevalent in young and elderly patients. Multiple logistic regression analysis showed smoking (P < .001), alcohol intake (P = .046), and hyperhomocysteinemia (P = .001) to be associated with AMI in the young patients while hypertension (P = .006) in elderly patients. To conclude, smoking, alcohol intake, and elevated homocysteine are the risk factors for AMI among young while hypertension among elderly patients.
Keywords
Introduction
Acute myocardial infarction (AMI) is a leading cause of mortality worldwide which is being responsible for 12.6% of total deaths each year. 1 Based upon a survey from United Kingdom, the prevalence of AMI in 2006 was approximately 4.1% in men and 1.7% in women 2 and was higher in the elderly than in the young patients. In contrast, in South Asian countries (such as India, Pakistan, Bangladesh, Sri Lanka, and Nepal), the prevalence of AMI was greater in patients younger than 40 years of age than those older than 60 years. 3 The prevalence of MI was also high (20%) in the younger age-group in the South African Asian Indian population. 4 These observations reflect the development of risk factors at a younger age in South Asians and South African Asian Indians. 3,4 Furthermore, several studies have reported that young patients have a different risk factor profile when compared to older patients. 3 –10
The etiology of AMI is multifactorial which involves complex interactions between genetic and environmental risk factors, with each risk factor showing additive effect. The conventional risk factors include hypertension, smoking, diabetes, obesity, and sedentary lifestyle. In addition, biochemical factors like plasma fibrinogen 10 –12 and plasma homocysteine (Hcy) 10 –13 have been proposed as indicators of an increased predisposition to AMI. Although the role of conventional risk factors in AMI is well accepted, it is equally likely that there are genetic contributions to AMI that are independent of the conventional risk factors. Several case–control studies have documented many genetic risk factors that significantly contribute in the pathogenesis of AMI. 14 –22 Limited knowledge of genetic risk factors that are associated with young and elderly patients with AMI from north India prompted us to carry out this study.
In the present study, factor V Leiden (FVL) 1691G/A, factor VII (FVII) 10976G/A, FVII HVR4, platelet membrane glycoproteins GP1BA 1018C/T, GP1BA VNTR, integrin ITGB3 1565T/C, ITGA2 807C/T, and methylenetetrahydrofolate reductase (MTHFR) 677C/T gene polymorphisms, biochemical factors (fibrinogen and homocysteine [Hcy]), and conventional risk factors have been studied in young (≤ 40 years) and elderly (≥60 years) north Indian patients with AMI for the comparison. Knowledge of the differences in risk factors between the elderly and young patients with AMI in north Indian population will help in developing preventive strategies for the management of the disease.
Materials and Methods
Study Design and Participants
This study was undertaken in the Department of Hematology, Postgraduate Institute of Medical Education and Research (PGIMER), Chandigarh, India. EPIINFO (Epi InfoTM 3.3.2, CDC Atlanta) statistical package was used to determine the sample size. A total of 350 patients with definite AMI per the World Health Organization (WHO) criteria 23 were recruited, who were consecutive patients at the Department of Cardiology, PGIMER. These patients were divided into 2 age-groups of ≤40 years of age (n = 184) and ≥60 years of age (n = 166). The patients were enrolled between July 2006 and December 2009. The ethics committee of PGIMER approved the study, and the patients gave their informed written consent.
A detailed personal history was taken including demographic factors, lifestyle factors, and conventional risk factors such as hypertension, diabetes mellitus, family history of coronary artery disease (CAD), obesity, smoking, alcohol intake, socioeconomic status (SES; high/middle/low), and lifestyle (rural/urban). The patients were considered to be hypertensive if they were already being treated with antihypertensive agents or were found to have a systolic blood pressure (BP) of ≥140 mm Hg or a diastolic BP of ≥90 mm Hg. History of diabetes was considered to be present if the individual was receiving therapy with either insulin or an oral hypoglycemic agent or had symptoms of diabetes with a fasting blood glucose level >140 mg/dL or were already being treated for diabetes. A positive family history was defined as the presence of at least 1 first- or second-degree relative with a history of CAD. For smoking and alcohol consumption, patients were classified on the basis of self-report. According to WHO Expert Consultation 2004, 24 body mass index (BMI) was categorized as normal weight (18.5-22.9 kg/m 2 ), overweight (23-27.5 kg/m 2 ), obese (27.6-40 kg/m 2 ), and morbidly obese (>40 kg/m 2 ). Normal waist-hip ratio (WHR) for males and females were ≤0.8 and ≤0.95, respectively. Hyperfibrinogenemia was defined as an increased level of fibrinogen (>4 g/L) in the blood. Hyperhomocysteinemia was categorized as mild (15-30 μmol/L), moderate (30-100 μmol/L), and severe (>100 μmol/L) depending on the levels of plasma Hcy.
Blood samples of the patients were taken at least 4 weeks after the onset of coronary event when the patients had been hemodynamically stabilized. After overnight fasting, aseptic venous blood samples were obtained and collected in EDTA for genetic analysis and estimation of plasma Hcy and in 3.2% trisodium citrate (9:1 v/v) for the estimation of plasma fibrinogen.
Biochemical Analysis
Estimation of plasma fibrinogen was done using STA-Fibrinogen kits on STA-analyzers (Diagnostica Stago, France) by clotting method of von Clauss. 25 For calculating plasma fibrinogen levels, a calibration curve was prepared using clotting time of diluted plasma calibrators. Normal plasma fibrinogen in the adult population is in the range of 2-4 g/L.
BioRad Microplate Enzyme Immunoassay Homocysteine kits (BioRad, Oslo, Norway) were used to determine total plasma Hcy levels on Tecan Absorbance Reader (Tecan Austria GmbH, Austria, Europe) using Magellan CE software (Magellan.CE Tracker software package, Tecan Group Ltd., Männedorf, Switzerland). For calculating plasma Hcy levels, a calibration curve was plotted using concentration of the calibrators against their absorbance readings. The Hcy control set was also used as a control for increasing the accuracy of measurement of plasma Hcy level. The limit of quantification for plasma Hcy level was 1.0 μmol/L, and coefficient of variation was less than 20% per the kit. Normal plasma Hcy level in the adult population is in the range of 5 to 15 μmol/L.
Genetic Analysis
Extraction of genomic DNA from peripheral blood leukocytes was performed using phenol–chloroform method. 26 Genetic polymorphisms such as FVL 1691G/A (rs6025), 17 GpIIIa or CD61 or ITGB3 1565T/C (rs5918), 18 GpIbα or CD42-α or GP1BA HPA-2 (1018C/T) (rs6065), 19 FVII R353Q (10976G/A) (rs6046), 20 and MTHFR 677C/T (rs1801133) 21 were determined by polymerase chain reaction (PCR) restriction fragment length polymorphism, using site-specific restriction endonucleases. GpIa or CD49B or ITGA2 807C/T (rs1126643) 22 gene polymorphism was detected by allele-specific PCR using internal controls. Genetic polymorphisms such as GP1BA VNTR 19 and FVII HVR4 20 were determined by PCR amplification and 12% polyacrylamide gel electrophoresis.
Statistical Methods
The data were expressed as mean ± standard deviation (SD) for continuous variables and proportions (%) for discontinuous variables. Analysis of variance (ANOVA) with Bonferroni correction was used to compare mean BMI and mean WHR among the 2 patients groups. Mean plasma fibrinogen and Hcy levels were compared by Mann-Whitney U test. Conventional risk factors, genotype frequencies (prevalence), and deviation of allele frequencies from Hardy-Weinberg equilibrium (HWE) were compared using chi-square (χ 2 ) test. Multiple logistic regression analysis adjusted for age and sex was performed to adjust for the confounding effects of conventional coronary risk factors. Significant risk factors in univariate analysis (P < .05) were included in the multiple logistic regression analysis. Odds ratio (OR) and 95% confidence intervals (CIs) were calculated to determine the association of risk factors with the occurrence of AMI. A P value of <.05 was considered to be statistically significant. SPSS version 13.0, Chicago, was used for the analysis of the present study.
Results
Demographic Data
Of the 350 patients recruited for the present study, 184 (52.5%) patients were younger than 40 years and 166 (47.4%) patients were older than 60 years. The younger patient group had a mean age of 36.4 ± 4.5 years, while the elderly group had a mean age of 64.1 ± 5.1 years. Patients in both the age-groups were predominantly males, but this was more evident in the younger (96.2%) than in the elderly group (74.7%). The male–female ratio was 25:1 for the young patients, but this difference for the elderly group became less striking (3:1) as the proportion of affected females increased with age.
Conventional Risk Factors
The profiles of conventional risk factors differed significantly among the young and elderly patient groups. Smoking was the most important risk factor in young patients (61.4%) compared with 24.1% in elderly patients (OR 5.0, 95% CI 3.2-8.0, P < .001). The prevalence of alcohol intake was higher in young patients (42.9%) when compared to 21.7% in the elderly group (OR 2.7, 95% CI 1.7-4.3, P < .001). Greater number of the younger patients (38.6%) belonged to low SES than the elderly patients (24.1%) with AMI (OR 2.0, 95% CI 1.2-3.1, P < .001). Hypertension (39.7%) was less frequently prevalent in young patients than that in elderly patients (67.5%) with AMI (OR 0.3, 95% CI 0.2-0.5, P < .001). As expected, obesity (high mean WHR) also showed greater prevalence in the older age-groups (P = .034). Other risk factors such as diabetes mellitus (14.1% vs 18.1%, OR 0.7, 95% CI 0.4-1.3, P = .315), family history of CAD (23.4% vs 21.1%, OR 1.1, 95% CI 0.7-1.9, P = .608), rural lifestyle (43.5% vs 36.1%, OR 1.4, 95% CI 0.9-2.1, P = .162), and high BMI (69% vs 59%, OR 1.5, 95% CI 1.0-2.4, P = .079) showed no significant difference in the patient groups. Univariate analysis showed that smoking, alcohol intake, and low SES increased the risk of AMI in the young patients while hypertension and obesity in elderly patients.
Biochemical Risk Factors
The mean plasma fibrinogen level was significantly higher in elderly patients (4.0 ± 1.0 g/L) than in young patients (3.8 ± 1.0 g/L, P = .01), showing elevated plasma fibrinogen to be associated with increased risk of AMI in the elderly patients. However, the prevalence of hyperfibrinogenemia was not significantly different in young and elderly patients with AMI (33.1% vs 28.8%, OR 0.8, 95% CI: 0.5-1.3, P = .381).
Young patients had significantly higher mean plasma Hcy level (24.0 ± 23.9 μm/L) than elderly patients (16.1 ± 18.0 μm/L, P < .001). Moreover, hyperhomocysteinemia showed the higher prevalence in young than in elderly patients (54.9% vs 30.1%, OR 2.8, 95% CI 1.8-4.4, P < .001), showing elevated plasma Hcy to be significantly associated with increased risk of AMI in young patients.
Genetic Risk Factors
There was no statistical deviation from Hardy-Weinberg equilibrium for any polymorphism in the 2 groups of patients in this study. Comparison of genetic risk factors among the 2 patient groups is shown in Table 1. Univariate analysis revealed that there was no significant difference (P > .05) between the young survivors of AMI and the elderly patients in terms of genotype or allele frequencies for all the 8 gene polymorphisms: 1691G/A of the factor V gene or factor V Leiden, 10976G/A and HVR4 of the factor VII gene, 1018C/T and VNTR of the platelet membrane glycoprotein GP1BA gene, 1565T/C of the integrin ITGB3 gene, 807C/T of ITGA2 gene, and 677C/T of methylenetetrahydrofolate reductase gene.
Comparison of Genetic Risk Factors Among Patients With Acute Myocardial Infarction.a
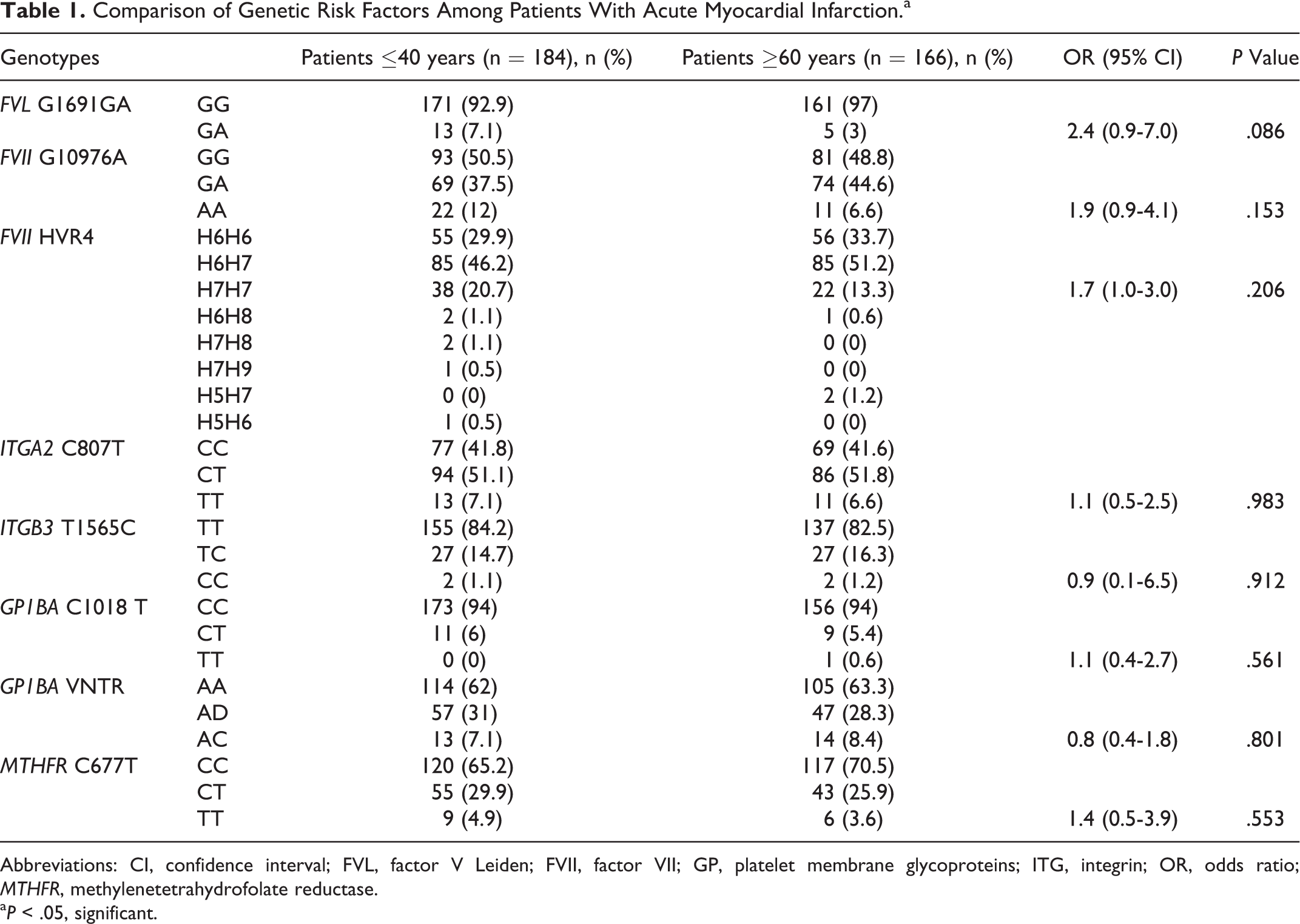
Abbreviations: CI, confidence interval; FVL, factor V Leiden; FVII, factor VII; GP, platelet membrane glycoproteins; ITG, integrin; OR, odds ratio; MTHFR, methylenetetrahydrofolate reductase.
a P < .05, significant.
Multiple Logistic Regression Analysis
Factors showing increased risk of AMI in univariate analysis were subjected to multiple logistic regression analysis to adjust for the confounding factors. Risk factors for patients with AMI following adjustment for confounders are shown in Table 2. Multivariate analysis revealed that smoking (OR 2.8, 95% CI 1.6-4.9, P < .001), alcohol intake (OR 1.7, 95% CI 0.9-3.0, P = .046), and hyperhomocysteinemia (OR 2.3, 95% CI 1.4-3.9, P = .001) were more prevalent in young patients, showing their association with AMI in the young patients. In contrast, higher prevalence of hypertension (OR 0.5, 95% CI 0.3-0.8, P = .006) in the elderly group showed it to be associated with increased risk of AMI in the elderly patients. Low SES (OR 0.6, 95% CI 0.4-1.0, P = .055) and high WHR (OR 0.7, 95% CI 0.4-1.2, P = .208), however, lost significance after adjusting for confounders.
Risk Factors for Patients With Acute Myocardial Infarction Following Adjustment for Confounders.
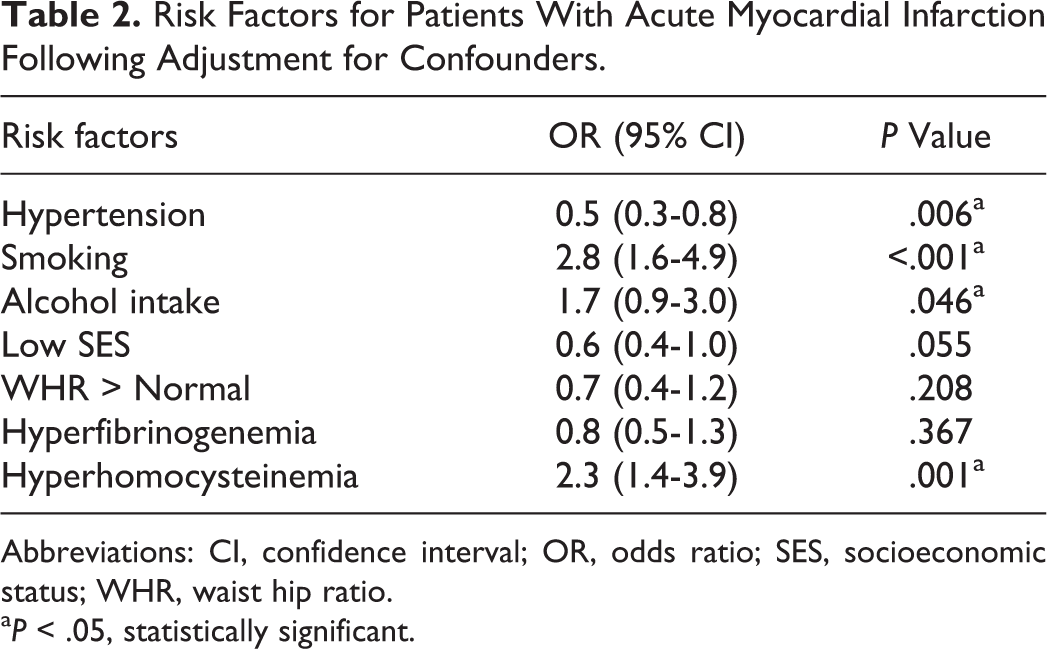
Abbreviations: CI, confidence interval; OR, odds ratio; SES, socioeconomic status; WHR, waist hip ratio.
a P < .05, statistically significant.
Discussion
Acute myocardial infarction occurs as a result of rupture of an atherosclerotic plaque in a coronary artery followed by acute thrombus formation. Both atherosclerosis and thrombosis contribute in the occurrence of AMI; however, the relative importance of these 2 processes varies from patient to patient and is different at different ages. Young patients with premature MI have less coronary atherosclerosis and involve more contribution of thrombosis than atherosclerosis in the development of AMI. However, in the elderly patients, coronary atherosclerosis is much more marked, and thrombosis plays a lesser role in the causation of AMI. Therefore, AMI in young patients when compared with the elderly patients provides a unique model to investigate the role of prothrombotic risk factors.
With this background, this study was designed to assess the degree of association between prothrombotic and hemostastic genetic polymorphisms, such as FVL 1691G/A, FVII 10976G/A, FVII HVR4, platelet membrane glycoproteins GP1BA 1018C/T, GP1BA VNTR, integrin ITGB3 1565T/C, ITGA2 807C/T, and MTHFR 677C/T, biochemical risk factors (fibrinogen and Hcy), and conventional risk factors with the occurrence of AMI in 184 young (≤40 years) and 166 elderly patients (≥60 years) from north India. None of the 8 polymorphisms deviated from Hardy-Weinberg equilibrium which provides evidence of no population stratification and/or genotyping error.
In this study, we found that in both the patient groups, males were more likely to have AMI than females. The male to female ratio was higher in the younger group (25:1) than in the older group (3:1), which is in agreement with previous studies. 22 –24 In a study by Dang et al 5 in south Indians, the male to female ratio was higher in the younger group (7.8:1) than in the older group (2.6:1). A similar trend in young patients with AMI was seen in the study by Siwach et al 6 in Haryana, India, where the male to female ratio was 20:1. The male–female ratio was 3.43:1 and 1.27:1 in young and elderly patients with AMI, respectively, in a study done by Bhatia et al in west Indian population. 24 In a study by Ranjith et al 4 in South African Asian Indians, patients in both the age-groups were predominantly males, but this was more evident in the younger (88%) patients.
Our study found that smoking was the most important risk factor in young patients with AMI, followed by alcohol intake, which is in line with the findings of other studies. 4 –7 A similar study by Ranjith et al 4 revealed that cigarette smoking was the most important risk factor in young patients (81%) compared with 49% for older patients (P < .0001). Smoking increases platelet adhesion and aggregation increasing the risk of thrombosis. It also damage vascular endothelium accelerating the development of coronary atherosclerosis responsible for AMI.
As expected, hypertension was shown to be less prevalent in our young patients with AMI than in elderly patients with AMI, which was also the finding in the other studies. 4,5,8 Hypertension (18%) was seen less frequently in young patients in a study by Ranjith et al. 4 Majority of our young patients with AMI belonged to lower socioeconomic group, which is consistent with the study by Siwach et al. 6 The prevalence of low SES was higher in young than in elderly patients in univariate analysis but lost its significance after adjusting for confounders in multivariate analysis.
In our study, WHR was lower in young patients with AMI than in elderly patients with AMI in univariate analysis. A similar finding in young patients with AMI from Haryana, India, was seen in the study by Siwach et al, 6 where majority of the young patients were thinly built. In contrast, obesity (BMI > 30 kg/m 2 ) was found to be more prevalent (18% vs13%) in the younger age-groups than in older age-groups in a study by Ranjith et al. 4 Other conventional risk factors such as diabetes mellitus, family history of CAD, rural lifestyle, and mean BMI were equally present in both the patient groups in our study, although these conventional risk factors show association with AMI in various studies. 7,8
Elevated plasma fibrinogen was associated with increased risk of AMI in our elderly patients with AMI when compared to the young group. In a study by Kumar et al 9 in 2009 on Indian population, serum fibrinogen was significantly higher in the elderly patients. Contrasting study by Mattioli et al in 2005 10 showed that acute-phase reactants like fibrinogen were quantitatively similar in young and old patients.
Our study showed that the young patients with AMI had significantly higher mean Hcy level than the elderly patients. Moreover, the prevalence of hyperhomocysteinemia was higher in young than in elderly patients, showing elevated plasma Hcy to be associated with increased risk of AMI in young patients. In a similar study by Mattioli et al in 2005, 10 Hcy is significantly higher in young patients than in old patients. Brattström et al in a study from Sweden had found the plasma homocysteine levels to increase with age which was linked to the use of multivitamins. 27 However, our study showed contrary results and we found younger patients to have higher levels and prevalence of hyperhomocysteinemia. An earlier study on normal controls in our population has detected a higher frequency of hyperhomocysteinemia and postulated it to be a major determinant leading to AMI in young patients and the levels decreased after vitamin and folate supplementation proving nutritional factors to be responsible. 28
Genotype frequencies in 1691G/A of the factor V gene (factor V Leiden), 10976G/A and HVR4 of the factor VII gene, 1018C/T and VNTR of the platelet membrane glycoprotein GP1BA gene, 1565T/C of the integrin ITGB3 gene, 807C/T of the ITGA2 gene, and 677C/T of the MTHR gene were compared in the young survivors of AMI and the elderly patients in our study. Our analysis revealed that all the 8 gene polymorphisms chosen for the present study were equally prevalent in both the patient groups. A genetic component is particularly important in young patients with AMI. Several studies in the literature have shown the association of FVL 1691GA, ITGB3 PlA1/A2 (1565 T/C), GP1BA HPA-2 (1018 C/T), FVII 10976 G/A (R353Q), FVII HVR4, and MTHFR 677 C/T gene polymorphism with risk of AMI in some ethnic groups, there are reports to the contrary. 14 –22 However, our study found no significant difference in the genotype frequencies (prevalence) of all the 8 gene polymorphisms.
In conclusion, the risk factor profiles differed significantly among the 2 patient groups. Smoking, alcohol intake, and elevated plasma Hcy are the risk factors for AMI among young patients. However, hypertension is the major risk factor for AMI among elderly patients. Absence of any difference in the genetic profile of the young and elderly patients with AMI was observed in our study. Knowledge of risk factors that selectively operate in young and elderly north Indians can help in planning appropriate preventive strategies that can target the different age-groups independently. This can also help to formulate investigations that can be done for the patients and their relatives. Strategies of smoking and alcohol cessation have to be targeted in the youth which can prevent the occurrence of AMI in the young patients. Diet rich in folate and vitamin B12, which can lower the plasma hcy, should be recommended. In addition, the preventive measures to control hypertension in the elderly patients should be emphasized in north Indian population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The experimental work of this study was supported by grants from the Council of Scientific and Industrial Research, New Delhi, India [grant number 09/141(0164)/2006] and the Department of Science and Technology, New Delhi, India [grant number SR/SO/HS–72/2005].
