Abstract
Familial Mediterranean fever (FMF) is an autosomal recessive disorder characterized by periodic provocative attacks of fever with peritonitis, pleuritis, arthritis, or eriseplemya. Tumor necrosis factor-α (TNF-α) plays an important role in the regulation of the immune response as a part of the cytokine network, including activation of macrophages and apoptosis. We investigated the possible association of TNF-α promoter −1031T/C and −308G/A polymorphisms in 86 FMF patients carrying M694 V homozygous mutation and 100 matched healthy controls both from Iranian Azeri Turks. Our data showed that patients with TNF-α −308 GG are more susceptible to the development of amyloidosis and arthritis (P value <.05). These data also showed that the frequency of TNF-α −308 A allele is considerably low among patients with amyloidosis, and it may have protective role among them (odds ratio [OR] = 0.083, χ2 = 5.46, P value = .003). Further evaluation of this polymorphism may be important and need further studies.
Keywords
Introduction
Familial Mediterranean fever (FMF) is an autosomal recessive disorder that was characterized by recurrent inflammatory attacks of fever and serositis 1 and belongs to a group of rare hereditary recurrent unprovoked inflammation called autoinflammatory diseases (AIDs). 2 Familial Mediterranean fever is the most frequent of the hereditary fevers, which include the hyperimmunoglobulinaemia D with periodic fever syndrome (HIDS; MIM 260920), and autosomal-dominant recurrent fevers such as tumor necrosis factor receptor-1–associated periodic syndrome (TRAPS; MIM 142680 and 134610). 3 It affects populations from Mediterranean ethnics, mainly non-Ashkenazi Jews, Arabs, Armenians, Turks 4 , and Iranian Azeri Turks.5,6 MEFV is the gene responsible for FMF located on the short arm of chromosome 16 (16p13.3) and encodes a protein named marenostrin or pyrin. 4 Pyrin is produced in certain white blood cells (neutrophils, eosinophils, and monocytes) that play a role in inflammation and in fighting infections. More than 190 variants, primarily clustered in exon 10, have been identified so far. 7 The pathogenesis of the disease is still unclear. Different studies have shown the increased levels of interleukin 6 (IL-6), IL-10, serum soluble IL-2 receptor (sIL-2R), and tumor necrosis factor-α (TNF-α) during or between the attacks of FMF,8,9,10 indicating the activation of cytokine network during these periods. Tumor necrosis factor -α is a multifunctional, proinflammatory cytokine implicated in various pathophysiological processes and plays an important role in the regulation of the immune response as a part of the cytokine network, including activation of macrophages and apoptosis. 11 During inflammation, TNF-α stimulates the expression of IL-1 and other cytokines that promotes leukocyte migration and increases vascular permeability. In recurrent episodes of FMF, all these manifestations exist, therefore TNF-α may have an important role in the severity of attacks in FMF disease. 8 TNF-α is located within the Major histocompatibility complex (MHC) region on chromosome 6p21.3. This region is highly polymorphic, and the TNF-α has a large number of polymorphisms wherein most of them are located in its promoter region and some of them have effect on the expression level of the gene. 12 Most popular single-nucleotide polymorphisms (SNPs) in TNF-α are −1031(T>C), −863 and −857(C>A), −851(C>T), −419(G to C inversion), −376, −308, −238, −163, and −49 (G>A). The association of some of these polymorphisms with susceptibility to several AIDs like systemic lupus erythematosus, insulin-dependent diabetes, inflammatory bowel disease,13 –15 Behçet’s disease (BD), 16 and other infections has been shown. 12 The aim of this study was to find the possible association of TNF-α −1031T/C and TNF-α −308G/A polymorphisms with the severity of clinical manifestations of FMF disease in patients from Iranian Azeri Turkish ethnic group carrying M694 V homozygous mutation for MEFV gene.
Material and Methods
Participant
Our case–control study included 86 Iranian Azeri Turkish FMF patients with homozygous M694V mutation in MEFV gene. These patients were also clinically diagnosed, according to the diagnostic criteria of FMF. 17 One hundred healthy, unrelated, age- and sex-matched individuals from the same geographical area without any familial history of FMF or other inflammatory diseases were selected as the control group.
Genotyping
Genomic DNA was extracted from peripheral blood cells using simple salting out procedure. 18 Both polymorphisms in the promoter region of the TNF-α gene at positions −1031T/C and −308G/A were detected by cleaved amplified polymorphic sequence (CAPS) called as polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) assay. For the amplification of −1031T/C polymorphism site via PCR, we used the forward (5′-GGGGAGAACAAAAGGATAAG-3′) and reverse (5′-CCCCATACTCGACTTTCATA-3′) primer pair. Initially, the PCR was subjected to denaturation for 5 minutes at 95°C, followed by 30 cycles of amplification (30 seconds at 95°C, 30 seconds at 55°C, and 30 seconds at 72°C). A final elongation step (5 minutes at 72°C) was applied at the end of the 30 cycles. The PCR product is 270 bp. Then PCR followed by an overnight digestion with the restriction enzyme BbsI (C allele, 159 and 111 bp; T allele, 270 bp) at 37°C. Digested PCR fragments were separated by a 10% polyacrylamid gel electrophoresis and visualized by ethidium bromide staining. Forward primers (5′-AGGCAATAGGTTTTGAGGGCCAT-3′) and reverse (5′-TCCTCCCTGCTCCGATTCCG-3′) were used to amplify the 107-bp DNA fragment of the TNF-α −308A>G polymorphism. Polymerase chain reaction conditions included 5 minutes for initial denaturation at 95°C; 35 cycles at 95°C for 1 minute for denaturation, 30 seconds at 65°C for annealing, and 30 seconds at 72°C for extension, followed by 5 minutes at 72°C for final extension. After amplification, the PCR products were digested (at 37°C) by restriction endonuclease NcoI (G allele, 87 and 20 bp; A allele, 107 bp) for 16 hours. Digested PCR products were electrophoresed in a 10% polyacrylamide gel and visualized by ethidium bromide staining.
Statistical Analysis
Allelic and genotypic association between patients and healthy controls was carried out using chi-square test with Yates correction or Fisher exact test, where appropriate. Probability values of .05 or less were regarded as statistically significant. Differences in haplotype frequencies of each polymorphism between patients and healthy controls were also analyzed using chi-square test. The odds ratio (OR) and the 95% confidence intervals (CI) were calculated for all data and used in analysis. We calculated the frequencies of TNF-α −1031 and TNF-α −308 polymorphisms for each clinical symptom separately, and the possible differences between patients with and without a specific clinical feature was studied using chi-square test and comparing confidence intervals.
Results
The distribution of genotype and allele frequencies of TNF-α −1031 T>C and TNF-α −308 G/A polymorphisms showed no significant difference between FMF patients and healthy controls in this cohort (Table 1 ).
TNF-α Polymorphisms in Iranian Azeri Turkish Patients With FMF and Healthy Controls
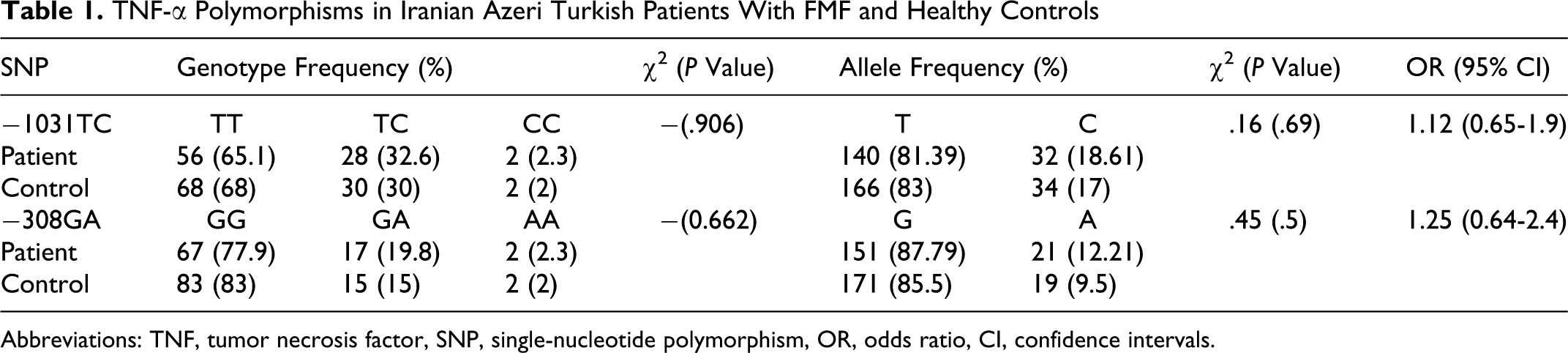
Abbreviations: TNF, tumor necrosis factor, SNP, single-nucleotide polymorphism, OR, odds ratio, CI, confidence intervals.
We could identify the haplotype for all individuals except 4 of them that were heterozygous in both TNF-α −1031 and TNF-α −308 polymorphisms. None of the identifiable haplotypes showed significant differences between controls and FMF patients (data not shown).
Clinical analysis of FMF patients showed that 89% (73 of 80) had fever, 89.4% (76 of 85) had peritonitis, 50% (41 of 82) had pleuritis, 50% (42 of 84) had arthritis, 35% (28 of 80) developed amyloidosis, 25% (19 of 76) had myalgia, and 21.4% (15 of 70) of them had erisiplemya.
In order to study the possible phenotype–genotype correlations among patients with FMF, different genotypes and alleles in correlation with their phenotypes were statistically analyzed (Tables 2 and 3 ). The frequencies of TNF-α −308 GG, GA, and AA genotypes in patients with amyloidosis were 0.964, 0.036, and 0.0, and in patients without amyloidosis were 0.673, 0.288, and 0.038, respectively (P value = .0059). The frequencies of TNF-α −308 GG, GA, and AA genotypes in patients with arthritis were 0.881, 0.071, and 0.048 and in patients without arthritis were 0.690, 0.310, and 0.0, respectively, (P value = .0062). None of the other clinical features showed statistically significant difference between different genotypes.
Tumor Necrosis Factor-α Genotypes Distributions and Clinical Features
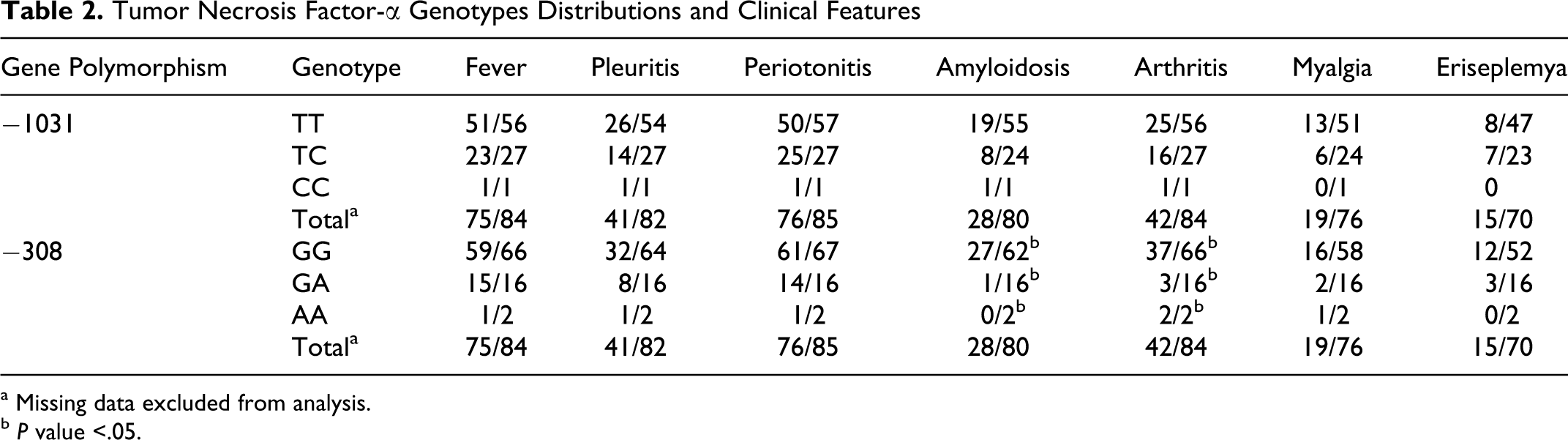
a Missing data excluded from analysis.
b P value <.05.
Tumor Necrosis Factor-α Allele Distribution and Clinical Features
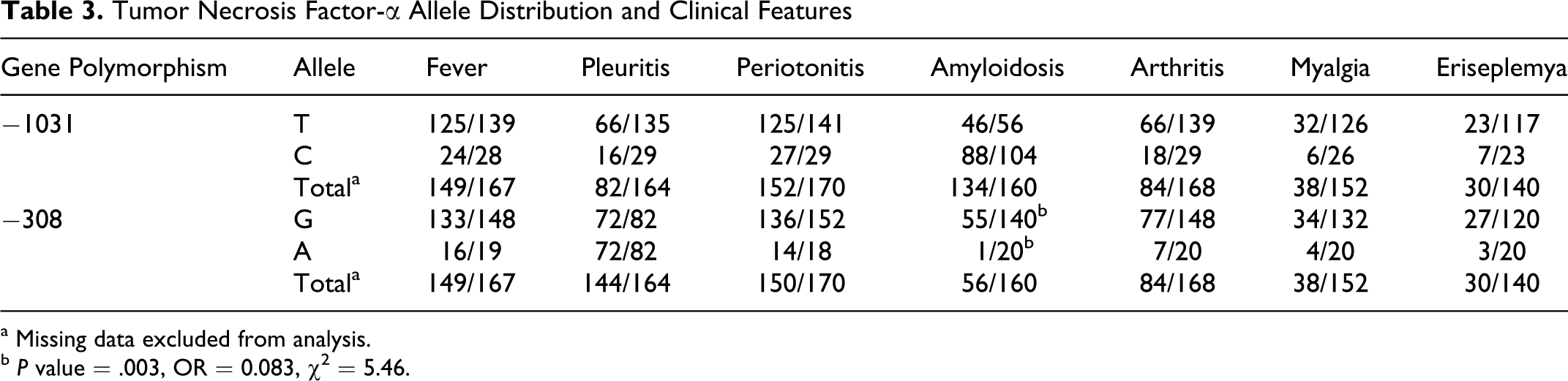
a Missing data excluded from analysis.
b P value = .003, OR = 0.083, χ2 = 5.46.
The numbers of G and A alleles of TNF-α −308 in patients with amyloidosis were 55 (98%; 95% CI = 90.5-99.6) and 1 (2%; 95% CI = 0.03-0.94), respectively. These numbers for patients without amyloidosis were 87 for G (82%; 95% CI = 73.7-88.2) and 19 for A (18%; 95% CI = 11.7-26.3) alleles. These data showed significant difference between these 2 groups of patients with FMF (OR = 0.083, χ 2 = 5.46 and P value = .003). Allelic analysis of data among patients with FMF with and without arthritis showed no significant differences (Table 3).
Discussion
Familial Mediterranean fever is a hereditary AID characterized by recurrent episodes of inflammation attacks, fever, and seroitis.19 -21 Against other inflammatory diseases in FMF, patients return to the clinically normal conditions when the attack disappears. 22
Mainly in genetic disorders, it is well known that patients with one and the same mutations may vary clinically. These notifications reflect the existence of extra environmental factors or possible modifier genes that affect the disease phenotypes. In the case of FMF, there is variable clinical profile for patients carrying the same MEFV gene mutations. It strongly suggests a possible role for other genetic and/or environmental factors in disease progression. These potential factors can include those genes that participate in inflammation such as IL-6, IL-10, sIL-2R, and TNF-α. Therefore, we theorized that TNF-α as a proinflammatory cytokine may be a modifier gene and have an effect on the progression and severity of FMF disease. Several reports have shown that the levels of cytokines increase during and between the FMF attacks.20,21 There are several studies that investigate the association between cytokines and inflammatory diseases.8,23 -32 Akar et al. showed that the SAA1 −13T genotype may have an effect on the development of amyloidosis. 27 Some other studies suggest a protective effect of the SAA1 beta and gamma alleles on the development of amyloidosis.28,29,31 Touitou et al presented Country of recruitment, rather than MEFV genotype, as a key risk factor for renal amyloidosis in FMF. 30 Karahan et al showed that IL-6 −174 G>C polymorphism had not been associated with FMF and amyloidosis. 25 Kobak et al reported that there had been no significant differences between patients with FMF from Turks and controls for the TNF-α −1031 T>C and −238 G>A polymorphisms, 8 and Akar et al showed that there was no significant difference between controls and patients with FMF with and without amyloidosis for the TNF-α −308 G/A allele. 24 Existence of coagulation abnormalities were reported in patients with FMF so far. 32 In many studies, the relationship between inflammation and coagulation response has been described.32 -34 Production of both IL-6 and IL-8 was considerably upregulated by activated protein C. Thrombin enhanced the migration of leukocyte induced by IL-1 and TNF-α.33,34 These data indicate the role of proinflammatory cytokines (like TNF-α) in the activation of coagulation. In a study, Bonyadi et al reported that TNF-α −1031 T>C showed significant association with BD in Iranian Azeri Turks but TNF-α −308 G>A did not. 16 This is the first report of TNF-α gene promoter polymorphisms association with FMF and its clinical manifestations in patients from Azeri Turks living in northwestern Iran. We had 86 patients with FMF carrying M694 V homozygous mutation and 100 healthy samples for control group both from Azeri population. Our data showed no association between cases and controls for both TNF-α −1031 T>C and TNF-α −308 G>A genotypic and allelic in this cohort. These data support previous findings.
All recorded clinical symptoms of the patients were statistically analyzed regarding TNF-α polymorphisms (−1031 T/C and −308 G/A) to find the possible role of them on progression and severity of disease by comparing the genotypic and allelic frequencies of patients with and without specific symptom. Although the analysis of TNF-α −1031 T/C polymorphism showed no significant association in the development of the clinical features, statistically significant difference was found between patients with and without amyloidosis and arthritis for TNF-α −308 G/A polymorphism. Overall these findings indicate that patients with TNF-α −308 GG genotype are more susceptible to amyloidosis and arthritis, and TNF-α −308 A allele shows considerably lower sensitivity to amyloidosis and may have protective role against amyloidosis in patients with FMF carrying M694V mutation from Azeri Turkish population. TNF-α −308 A allele showed no association with arthritis.
Footnotes
Acknowledgments
This project was financially supported by Center of Excellence for Biodiversity (University of Tabriz) and center of Liver & Gastrointestinal Disease Research Center, Tabriz University of Medical Sciences.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
