Abstract
Mutations of hemostasis/coagulation-related genes have been speculated to cause recurrent spontaneous abortion (RSA). This study investigated the genetic association between the polymorphisms of factor V (F5), factor II (F2), antithrombin (SERPINC1), protein C (PROC), protein S (PROS1), protein Z (PROZ), factor XIII (F13A1), and carboxypeptidase B2 (CPB2) genes and RSA. The 426 patients with RSA and 444 controls were recruited in this study, and single-nucleotide polymorphisms (SNPs) were analyzed by using SNPscan technology. Genotype and allele frequencies of rs3136520 in F2, rs3024731 in PROZ, and rs1050782 in F13A1 showed statistically significant differences between the 2 groups. TT genotype of rs3136520 (P = .031, odds ratio [OR] = 0.986, 95% confidence interval [CI] = 0.976-0.997) and AA genotype of rs2069906 in PROC (P = .021, OR = 0.114, 95% CI = 0.014-0.902) in their recessive models and AG + GG variants of rs1050782 (P = .007, OR = 0.681, 95% CI = 0.516-0.899) in the dominant model might be associated with the reduced risk of RSA. AT + TT variants of rs3024731 (P = .010, OR = 1.479, 95% CI = 1.098-1.994) may increase disease susceptibility in dominant model. Haplotype analysis of rs3024731 and rs3024735 in PROZ displayed that the AA and TG haplotype were inclined to decrease and increase the risk of RSA, respectively. These results suggested that rs3136520, rs2069906, rs3024731, and rs1050782 may have a significant association with the genetic susceptibility of RSA in Chinese Han women.
Keywords
Introduction
Recurrent spontaneous abortion (RSA) is defined as 2 or more consecutive miscarriages with the same partner in reproductive-age women, 1 affecting approximately 1% to 5% of females. 2,3 Recurrent spontaneous abortion is a complex, multifactorial condition caused by several factors, such as uterine anatomic anomalies, immune and endocrine disorders, chromosome aberrations, and infections. 4 –6 However, the pathogenesis remains unexplained in approximately 40% to 50% of cases with RSA. 7,8
In recent decades, thrombophilia has been suggested that it could be a risk factor in women with RSA. 5,9 –11 Thrombophilia could be classified into 2 categories, hereditary and acquired, presenting a blood hypercoagulable state and could result in RSA and other obstetrical complications such as placental abruption, severe preeclampsia, and intrauterine growth restriction. 10,12 –14 In the RSA research field, antiphospholipid syndrome belongs to the acquired thrombophilia and is the most established reason for thrombophilia causing recurrent miscarriage. 15 However, the role of inherited thrombophilia resulting in RSA was ambiguous. Maternal genetic variants of relevant genes that were involved in the process of blood coagulation and fibrinolysis may give rise to RSA. Previous studies suggested that factor V (F5) Leiden mutation (G1691A, rs6025) and prothrombin (factor II [F2]) G20210A mutation (rs1799963) could be risk factors for RSA in Caucasians. 16 –19 However, for other relevant genes such as antithrombin (SERPINC1), protein C (PROC), protein S (PROS1), protein Z (PROZ), factor XIII (F13A1), and carboxypeptidase B2 (CPB2), the exact role of genetic alterations in each of these genes in the onset of RSA in Chinese Han women remains unclear.
Therefore, the goal of this study is to investigate the association between single-nucleotide polymorphisms (SNPs) of F5, F2, SERPINC1, PROC, PROS1, PROZ, F13A1, and CPB2 genes and RSA in Chinese Han women.
Materials and Methods
Participants
This case–control study enrolled 426 women with a history of 2 or more spontaneous abortion prior to 12 weeks who were evaluated in the reproductive medicine center, the First Affiliated Hospital of Anhui Medical University, and 444 healthy women who had at least 1 live birth and no abortion history were recruited as a control group. All patients and controls belong to Chinese Han population, and they all had natural pregnancies. The participants with obesity (BMI > 30) were excluded in both groups. Patients with RSA having known causes including chromosomal abnormalities, anatomical anomalies, autoimmunity diseases, diabetes mellitus, thyroid dysfunction, and infections were also excluded in this research. All participants signed the informed consent, and the study protocol was approved by the local ethics committees.
Selection of SNPs
The selection criteria of SNPs included tag SNPs, which were chosen from the Chinese Han Beijing population of the International HapMap Consortium with the minor allele frequency greater than 0.05 and r 2 > 0.8 and SNPs that have been reported in the previous literature. Therefore, the total of 21 SNPs selected in this study including rs6025 in F5 (Leiden mutation), rs1799963 (20210 G>A), and rs3136520 in F2; 4 SNPs (rs2227589, rs2227612, rs5877, and rs941988) in SERPINC1; 3 tag SNPs (rs1799810, rs2069906, and rs5936) in PROC; 6 tag SNPs (rs13062355, rs4857037, rs6123, rs6803590, rs7609644, and rs8178607) in PROS1; rs3024731 and rs3024735 in PROZ; rs1050782 in F13A1; and rs1926447 and rs3742264 in CPB2.
Genotyping Technique
Peripheral blood samples were collected from all patients with RSA and controls. Genomic DNA was isolated by using DNA purification kits according to the manufacturer’s instructions. SNPscan technology (Genesky Biotechnologies Inc, Shanghai, China) which is based on double ligation and multiplex fluorescence polymerase chain reaction was used for SNP genotyping. Thirty-five random samples from cases and controls were used for repeated genotyping of all SNPs to achieve quality control, and repeated results were consistent with the original ones.
Statistical and Bioinformatics Analyses
SHEsis software was used to test Hardy-Weinberg equilibrium in controls, to calculate linkage disequilibrium (LD), and to inferred haplotypes of SNPs, and frequency <0.03 was ignored for the analysis. 20,21 If the selected SNPs were not conformed to Hardy-Weinberg equilibrium in controls, the locus should be excluded in further analysis. The statistical analyses were performed using SPSS 13.0 software. The comparison of genotype and allele frequencies between patients with RSA and controls were calculated by χ2 test or Fisher's exact test. Multifactor dimensionality reduction software (MDR, version 3.0.2) was used to predict and analyze the gene–gene interactions. Values of P < .05 were regarded statistically significant.
Results
All patients with RSA who participated in this study were with 2 or more early RSA (≤12 weeks). The mean age of 426 patients was 29.26 ± 4.294 years (range, 20-44), and the mean number of miscarriages was 2.40 ± 0.710 (range, 2-5). Among them, 301 (70.7%) women had 2 abortions, 89 (20.9%) women had 3 abortions, 26 (6.1%) women had 4 abortions, and 10 (2.3%) women had 5 abortions. The mean age of controls was 34.50 ± 4.895 years (range, 21-46), statistically older than the RSA patients group.
Among the selected 21 SNPs, genotype distributions of rs2227589 and rs941988 of SERPINC1 in the control group were not consistent with Hardy-Weinberg equilibrium, so these loci were excluded for further analysis. For 19 SNPs, the comparison of genotype and allele frequencies between the 2 groups is reported in Table 1. Genotype distributions and allele frequencies of rs3136520 in F2, rs3024731 in PROZ, and rs1050782 in F13A1 were all showed the statistical significant differences between cases with RSA and controls. The frequencies of rs3136520 T allele (P = .029, odds ratio [OR] = 0.657, 95% confidence interval [CI] = 0.449-0.961) and rs1050782 G allele (P = .006, OR = 0.761, 95% CI = 0.628-0.923) were decreased, whereas rs3024731 T allele (P = .028, OR = 1.236, 95% CI = 1.023-1.493) was increased in patients with RSA. Regarding rs2069906 in PROC, the distribution of genotype reached statistical significance with a value of P = .046. However, the allele frequencies between the 2 groups failed to show significant differences. In addition, no other association was found between the rest 15 SNPs and the risk of RSA in genotypic or allelic analyses.
Comparison of Genotype and Allele Frequencies of 19 SNPs Between the 2 Groups.a
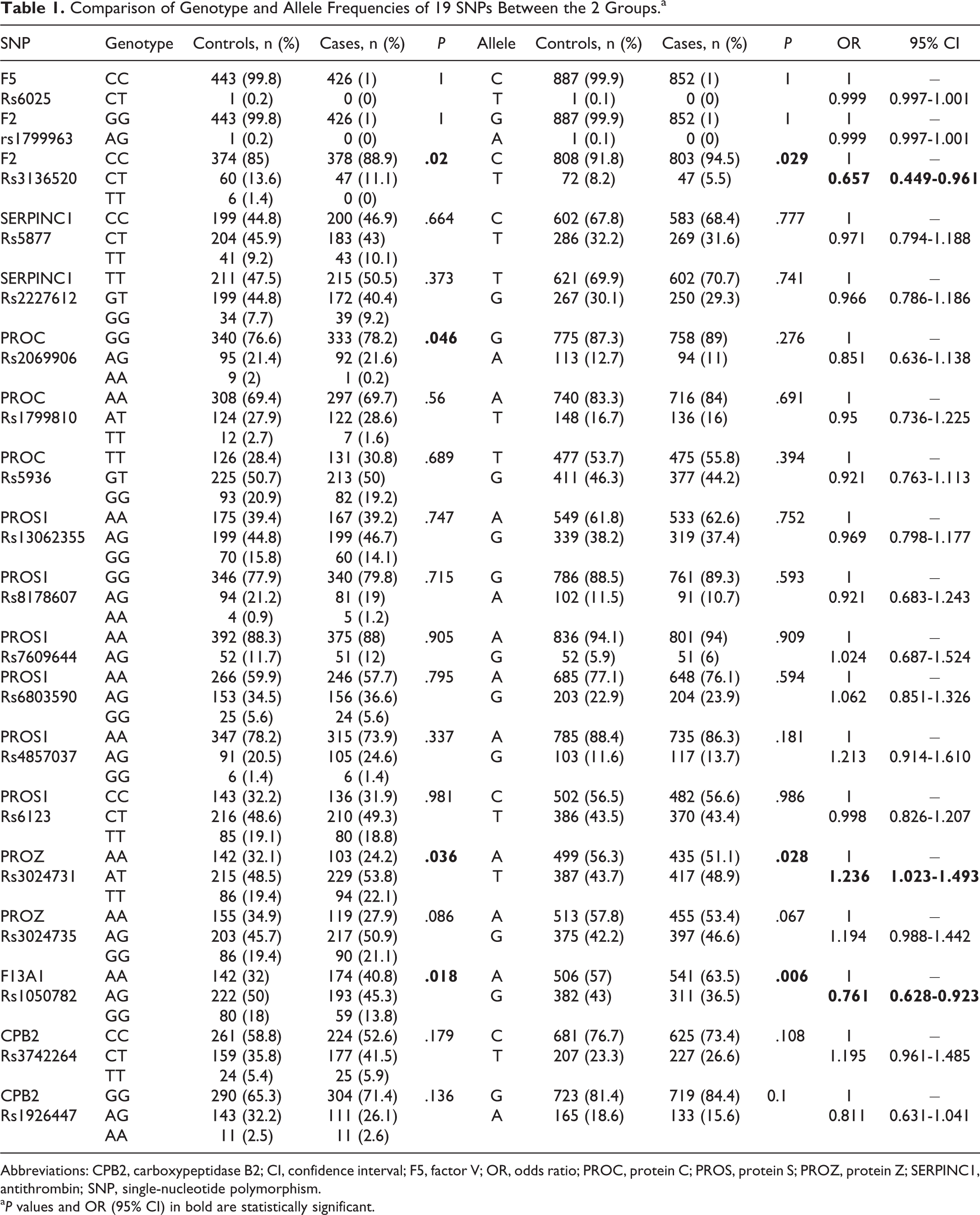
Abbreviations: CPB2, carboxypeptidase B2; CI, confidence interval; F5, factor V; OR, odds ratio; PROC, protein C; PROS, protein S; PROZ, protein Z; SERPINC1, antithrombin; SNP, single-nucleotide polymorphism.
a P values and OR (95% CI) in bold are statistically significant.
The genetic model analysis of rs3136520, rs2069906, rs3024731, and rs1050782 revealed statistical significance (Table 2). TT genotype of rs3136520 (P = .031, OR = 0.986, 95% CI = 0.976-0.997) and AA genotype of rs2069906 (P = .021, OR = 0.114, 95% CI = 0.014-0.902) in their recessive models and AG + GG variants of rs1050782 (P = .007, OR = 0.681, 95% CI = 0.516-0.899) in the dominant model were associated with the reduced risk of RSA. However, in the dominant model, compared to the AA genotype, AT + TT variants of rs3024731 were inclined to confer the susceptibility of RSA (P = .010, OR = 1.479, 95% CI = 1.098-1.994).
Genetic Model Analysis of the 4 SNPs.a
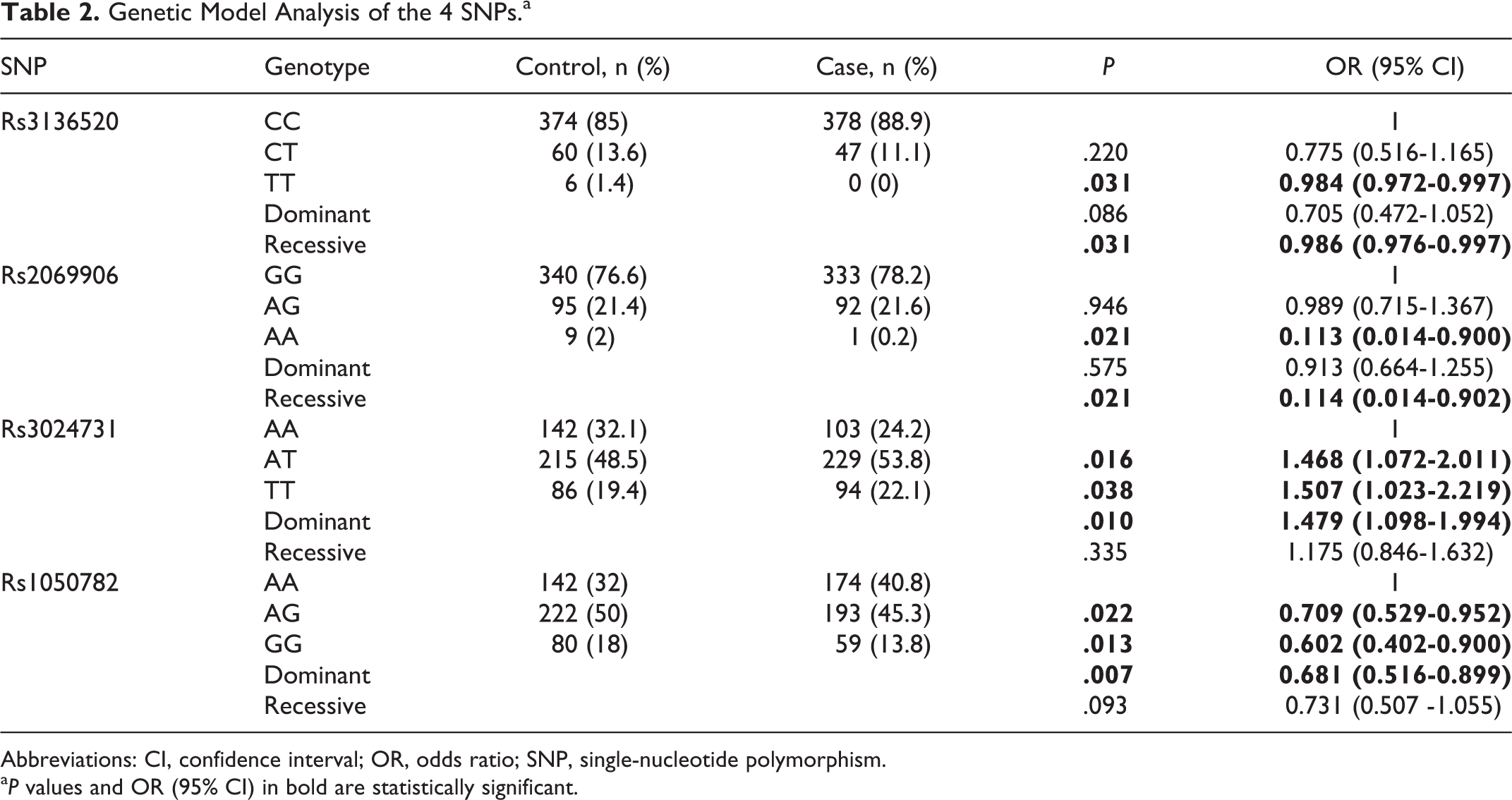
Abbreviations: CI, confidence interval; OR, odds ratio; SNP, single-nucleotide polymorphism.
a P values and OR (95% CI) in bold are statistically significant.
Rs3024731 and rs3024735 in the PROZ gene displayed a high degree of LD (D’= 0.945, r 2 = 0.825). The results of haplotype analysis with these 2 SNPs were listed in Table 3. The AA haplotype was associated with decreased risk of RSA (P = .037, OR = 0.817, 95% CI = 0.676-0.988), whereas TG haplotype tended to increase the risk of disease (P = .039, OR = 1.222, 95% CI = 1.010-1.480).
Haplotype Analysis With Rs3024731 and Rs3024735 in PROZ Gene.a
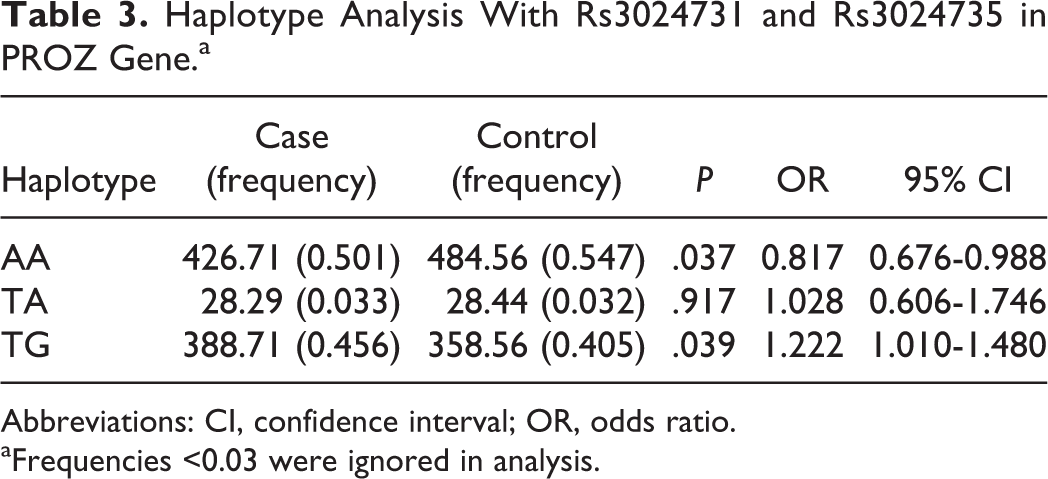
Abbreviations: CI, confidence interval; OR, odds ratio.
aFrequencies <0.03 were ignored in analysis.
Genetic interaction analysis among rs3136520 (F2), rs3024731 (PROZ), and rs1050782 (F13A1) were performed. The 3 best models were predicted from MDR software, and the prediction errors and cross-validation consistency for each of the models are displayed in Table 4. However, testing P value of these models did not show statistical significance and failed to find an association with the 3 SNPs between cases with RSA and controls.
The Best Gene–Gene Interactions Models Predict for the Association With RSA.

Abbreviations: CI, confidence interval; CVC, cross-validation consistency; OR, odds ratio; RSA, recurrent spontaneous abortion.
Discussion
Considerable researches have been done to identify possible relationship between the mutations of genes that may be involved in the inherited thrombophilia and RSA. But as far as we know, the positive results of researches were not much in Chinese Han women who experienced RSA. In this case–control study, the possible association were explored between the 19 SNPs from several genes that play vital roles in coagulation/fibrinolytic system and the risk of RSA.
F5 Leiden mutation and F2 G20210A mutation were the most studied thrombophilic gene mutations in RSA. However, this study indicated that they were not related to RSA in Chinese Han people because of the similar low mutant allele frequencies in patients and controls. Other studies did not find connections with RSA after these 2 mutation analyses in north western Iranian 22 and Israeli individuals. 23 Rs3136520 was one of the tag SNPs selected from the HapMap database and located on the intron region of the F2 gene. This study showed that TT genotype and T allele of rs3136520 were significantly lower in women with RSA than in controls and may act as the protective factor to decrease susceptibility to RSA. There have been limited investigations about this tag SNP of F2 gene in RSA research, and further explorations could focus on the effect of combining with other tag SNPs in Chinese Han women experiencing RSA.
Deficiencies of antithrombin, protein C, and protein S were regarded as risk factors for adverse pregnancy outcomes including fetal loss. 24,25 Mutations of these anticoagulation genes may influence their activities, and some variants have been investigated in RSA. GA + AA genotypes of rs2227589 in SERPINC1 gene might be a risk factor for RSA were reported in a Brazilian study, 26 whereas an Indian study 27 and a Chinese study 28 failed to find the relationship between this polymorphisms and RSA. In the current study about the 3 genes, rs2227589 and rs941988 of SERPINC1 were excluded because of the unbalanced Hardy-Weinberg equilibrium in controls. Therefore, 11 SNPs of these 3 genes were analyzed and found that AA genotype in a recessive model of rs2069906 located on the intron region of PROC gene may have a connection to the reduced risk of RSA. Just like rs3136520 in F2 gene, not much is known about the potential function of rs2069906 in PROC and its role in the etiology of RSA.
Lack of concentration of protein Z was also considered to have an association with various thrombotic diseases including venous/arterial thrombosis 29 and pregnancy complications. 30 Genetic variants may be an important reason for the deficiency of protein Z, and previous studies have investigated the association with several polymorphisms and RSA. Rs3024735 was reported to have a connection with RSA, 31,32 whereas 1 study also demonstrated that low protein Z levels could induce the unexplained pregnancy loss but not because of the rs3024735 variants. 33 A Bahrain study 34 found the allele frequencies of rs3024731 and rs3024735 with significant difference between patients with RSA and controls. The authors further reported that GA and AA genotypes of rs3024735, GA and AA genotype of rs3024719, and AA genotype of rs3024731 were linked with the decreased protein Z levels and demonstrated that GAGAAG and AGGTAG haplotypes may increase the disease susceptibility and AGGTGG haplotypes may reduce the risk of RSA with the 6 SNPs (rs3024718, rs3024719, rs3024778, rs3024731, rs3024735, and rs3024772) of PROZ gene. 34 In this study, rs3024731 and rs3024735 were investigated in RSA with Chinese Han women. In patients with RSA, the distributions of AT + TT genotypes and T allele of rs3024731 in PROZ were higher than in the control group and may increase the risk of RSA. But it failed to find an association between the rs3024735 and the onset of RSA. However, haplotype analysis with rs3024731 and rs3024735 indicated that AA and TG haplotype could decrease and confer the genetic susceptibility to RSA, respectively. Through comparison, these results were not concordant with earlier studies. These conflicting results may be explained by the different ethnic and genetic backgrounds, the size and selection of participants, and the change in environment. Further studies are needed to investigate the association between the polymorphisms of PROZ gene and RSA.
Coagulation factor XIII (FXIII) is known as the last zymogen and formerly named fibrin-stabilizing factor that could stabilize the fibrin clot after series of actions. 35 Factor XIII consists of 2 A subunits and 2 B subunits. The A subunits were encoded by the F13A1 gene and possess catalytic ability; the B subunits may only act as carriers. 36,37 Several studies have investigated the association between Val34Leu mutation in exon 2 of F13A1 gene and RSA. 22,38,39 However, the present study selected another polymorphism, rs1050782, which was chosen from one of the tag SNPs of F13A1 gene for analysis. Rs1050782, located in the 3′ untranslated region of F13A1, may as a functional site influence the expression of F13A1, although the specific effect of this variants remains unknown. In the current study, the frequencies of AG + GG genotypes and G allele in cases were lower than in controls, which all showed statistical significance, indicating that AG + GG genotypes and G allele of rs1050782 may be associated with decreased risk of RSA.
Carboxypeptidase B2 gene, also called thrombin-activatable fibrinolysis inhibitor gene, could inhibit fibrinolysis, and several polymorphisms including rs3742264 (505G/A) and rs1926447 (1040C/T) might influence the occurrence of venous thrombosis. 40,41 In an aspect of RSA, 1 study indicated that rs3742264 A allele was associated with RSA as a protective factor. 42 However, this study failed to reveal the connection between this SNP and RSA. Regarding rs1926447, some authors reported that this polymorphism was not related to RSA, 42,43 which is similar to the present results.
Because RSA is a complex multifactorial disease, the individual genetic variants were difficult to detect completely and may only account for a minor role in the occurrence of RSA. In the current study, 19 SNPs were analyzed and found 3 SNPs (rs3136520 of F2, rs3024731 of PROZ, and rs1050782 of F13A1) may be associated with RSA, with significant difference both in allele and in genotype distributions between the 2 groups. Therefore, the study chose these 3 SNPs and attempted to identify the gene–gene interactions using the MDR method. However, the negative results of the genetic interactions indicated that F2, PROZ, and F13A1 may play important roles participating in different pathways involved in the pathogenesis of RSA, but the specific functions and interactions with other genes remain unclear.
There are some limitations that should be considered. The patients recruited in this study, all suffered in early period of RSA, and late miscarriages were not being researched. Coagulation and fibrinolysis status was regulated by lots of crucial genes; other candidate genes or polymorphisms were not examined in this study. Current results were identified based on the moderate sample size. Further studies should be conducted with more candidate polymorphisms of genes involved in thrombogenesis and enlarged sample size to validate our results or find new associations with RSA in Chinese Han women.
Conclusions
To the best of our knowledge, this study was the first to identify that the polymorphisms rs3136520 of F2, rs2069906 in PROC, rs3024731 of PROZ, and rs1050782 of F13A1 may be associated with the genetic susceptibility of RSA in Chinese Han women.
Footnotes
Acknowledgments
The authors are grateful to Yamei Gao and Siyun Ye from Reproductive Medicine Center, the First Affiliated Hospital of Anhui Medical University, for sample collection. The authors also express gratitude to all participants of this study. Zuying Xu and Ying Zhang contributed equally to the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was support by the National Natural Science Foundation of China (81501232) and Public Welfare Technology Application Research Linkage Project of Anhui Province (1704f0804022).
