Abstract
Pharmacodynamic behavior of branded and biosimilar enoxaparin was compared in a crossover study in primates. Blood samples collected at baseline and at 1, 4, 6, and 28 hours post-subcutaneous administration of Lovenox or Fibrinox were evaluated using clot-based and amidolytic assays. Anti-Xa levels following Fibrinox and Lovenox administration were not different. Anti-IIa levels were significantly higher in Lovenox-treated animals 1 to 6 hours post-administration. Higher drug levels were measured by Heptest in Fibrinox-treated animals from 4 to 6 hours. Pharmacokinetic differences were not observed using anti-Xa or Heptest assays. The area under the curve (anti-IIa) following Lovenox treatment was significantly larger than following Fibrinox treatment. When drug levels (anti-IIa) were plotted against anti-Xa or Heptest drug levels, a hysteretic relationship which was distinct for Fibrinox- and Lovenox-treated primates was observed suggesting a lack of bioequivalence for the low-molecular-weight heparin tested. In vivo behavior is an important consideration for defining pharmacoequivalence of complex biologic drugs.
Introduction
Low-molecular-weight heparins (LMWHs) have been shown in a number of clinical trials to be safe and effective in the prevention and treatment of venous thrombosis 1 and in the treatment of patients with pulmonary embolism or acute coronary syndrome.2,3 Although smaller than unfractionated heparin, LMWHs maintain a high degree of structural heterogeneity. Commercially available LMWHs are distinct agents when compared in terms of their molecular weight/molecular weight distribution and in vitro potency. 4 Molecular weight influences the pharmacokinetics and pharmacodynamics of heparin oligosaccharides. It is known that there is a minimal chain length required to promote thrombin inhibition; heparin chains below this molecular weight threshold retain their ability to inhibit factor Xa. It is also known that smaller heparin chains are better absorbed.
There continues to be interest in developing biosimilar versions of LMWHs. Defining biosimilarity for LMWHs has been difficult because chemical means of measuring plasma heparin levels do not exist, potency measures typically only measure antithrombin (AT)-dependent activities, bioequivalence in terms of one activity does not ensure identical pharmacokinetic behavior in terms of other biological activities, and identification and quantification of the various oligosaccharide components in an LMWH preparation is difficult. 5 Currently there are a number of biosimilar LMWHs available in South America and Asia. Studies have shown that these agents can differ considerably from the branded product as well as from each other.5,6
When administered to healthy volunteers, the pharmacodynamic activity of branded LMWHs (anti-Xa activity and anti-IIa activity), its duration in the circulation, and the rate of absorption are product specific.7,8 There is one report describing a single dose, randomized sequence, open label, two period crossover study comparing the pharmacodynamic activity of branded and biosimilar enoxaparin in Chinese individuals. 9 In this study, based on 90% confidence intervals, the two formulations met the regulatory requirements for bioequivalence. In the current study, the pharmacodynamic behavior of branded and biosimilar enoxaparin was compared in a crossover study following a single subcutaneous dose to nonhuman primates.
Materials and Methods
Two formulations of enoxaparin were tested. Lovenox (lot #514956) was obtained from Sanofi-aventis, Paris, France. Fibrinox (lot#0002) was obtained in pre-filled syringes at a concentration of 100 mg/mL from Sandoz S.A., Buenos Aires, Argentina.
Non-human primates (Macaca mulatta) were anesthetized with an intramuscular injection of ketamine (Ketaset, Fort Dodge Animal Health, Fort Dodge, Iowa). Upon anesthesia, the primates were weighed and a baseline blood sample was collected by venipuncture. A site on the abdomen was shaved and cleansed by alternate wiping with betadine and ethanol prior to the subcutaneous administration of LMWH. Blood samples were collected at 1, 4, 6, and 28 hours postadministration. All blood samples were centrifuged within 15 minutes of collection to make platelet-poor plasma. Platelet-poor plasma was stored in aliquots at −70°C until analysis. Primates were randomly dosed with one of the 2 test agents. After a minimum of 1 week washout, the same primates were dosed with the other test agent.
Plasma samples were evaluated using clot-based and amidolytic assays. Activated partial thromboplastin time was determined using reagents obtained from Trinity Biotech, Berkeley Heights, New Jersey on a fibrometer (BBL, Cockeysville, Maryland). Heptest clotting time was determined using reagents obtained from Haemachem (St Louis, Missouri) on a fibrometer. Anti-Xa and anti-IIa activities were determined using amidolytic assays as described previously by our group. Human factor Xa and thrombin were obtained from Enzyme Research Laboratories (South Bend, Indiana). Spectrozyme Xa and Spectrozyme TH were obtained from American Diagnostica (Greenwich, Connecticut).
Concentration–response curves were made by supplementing Lovenox or Fibrinox to pooled baseline plasma over a concentration range of 0.6 to 10 µg/mL. Best-fit curves were made and the LMWH concentration in each primate blood sample was determined by extrapolation using the measured clotting times or percent inhibition of thrombin or factor Xa. The plasma concentrations determined using each assay were used to calculate values for pharmacokinetic parameters. 10 The elimination rate constant (ke) was determined by calculating the slope of the linear portion of the natural log concentration versus time curve. Elimination half-life was calculated as 0.693/ke. Area under the curve (AUC) was determined from the concentration versus time curve using the trapezoidal rule and was extrapolated to infinity by adding a value equal to the last measured concentration divided by the slope of the elimination curve. Systemic clearance (Clsys) was calculated as dose/AUC. Volume of distribution (Vd) was calculated as Clsys/ke.
Results are presented as the mean ± standard error of the mean (SEM). Two-way repeated measures analysis of variance was carried out to determine whether differences between treatments were statistically significant (SigmaStat v. 3.11; Systat Software, Inc, San Jose, California). P < 0.05 was considered statistically significant.
Results
The time course of plasma LMWH levels was assessed using 3 functional measures. Levels of LMWH determined by circulating anti-Xa activity following Fibrinox and Lovenox administration were not observed to be significantly different (Figure 1 ). Circulating anti-IIa levels, however, were significantly higher in Lovenox-treated animals at 1, 4, and 6 hours postadministration (Figure 2 ). Using the Heptest assay, higher circulating drug levels were measured in Fibrinox-treated animals at 4 and 6 hours (Figure 3 ).
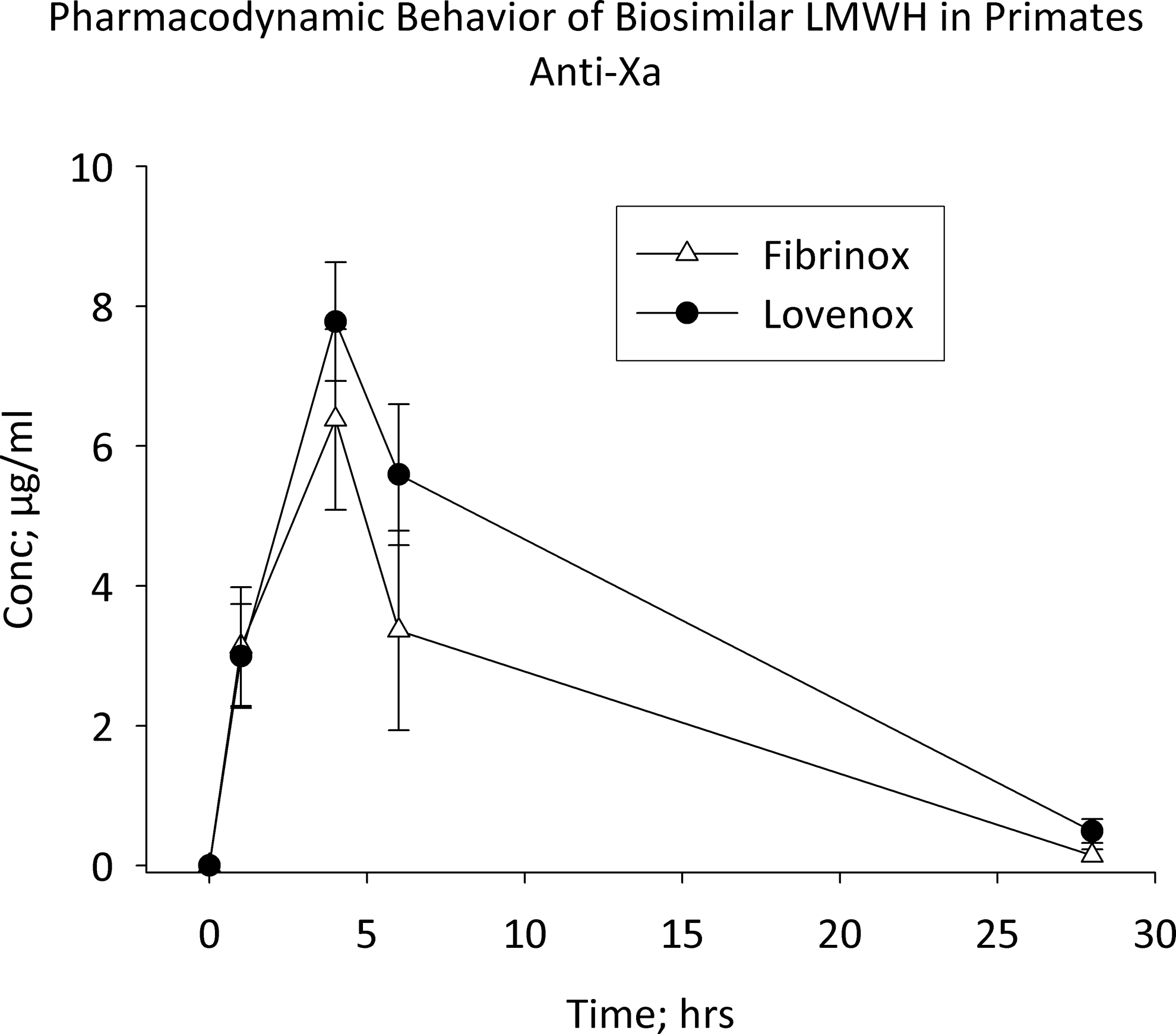
Pharmacodynamic behavior of LMWH formulations in primates. Macaca mulatta were treated with Lovenox and Fibrinox at a dose of 1 mg/kg subcutaneously (SC) in a crossover fashion. Blood samples were collected prior to dosing and at 1, 4, 6, and 28 hours post-dosing. Platelet-poor plasma was analyzed for anti-Xa activity using an amidolytic assay. Results represent the mean ± SEM of 8 primates/treatment group. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
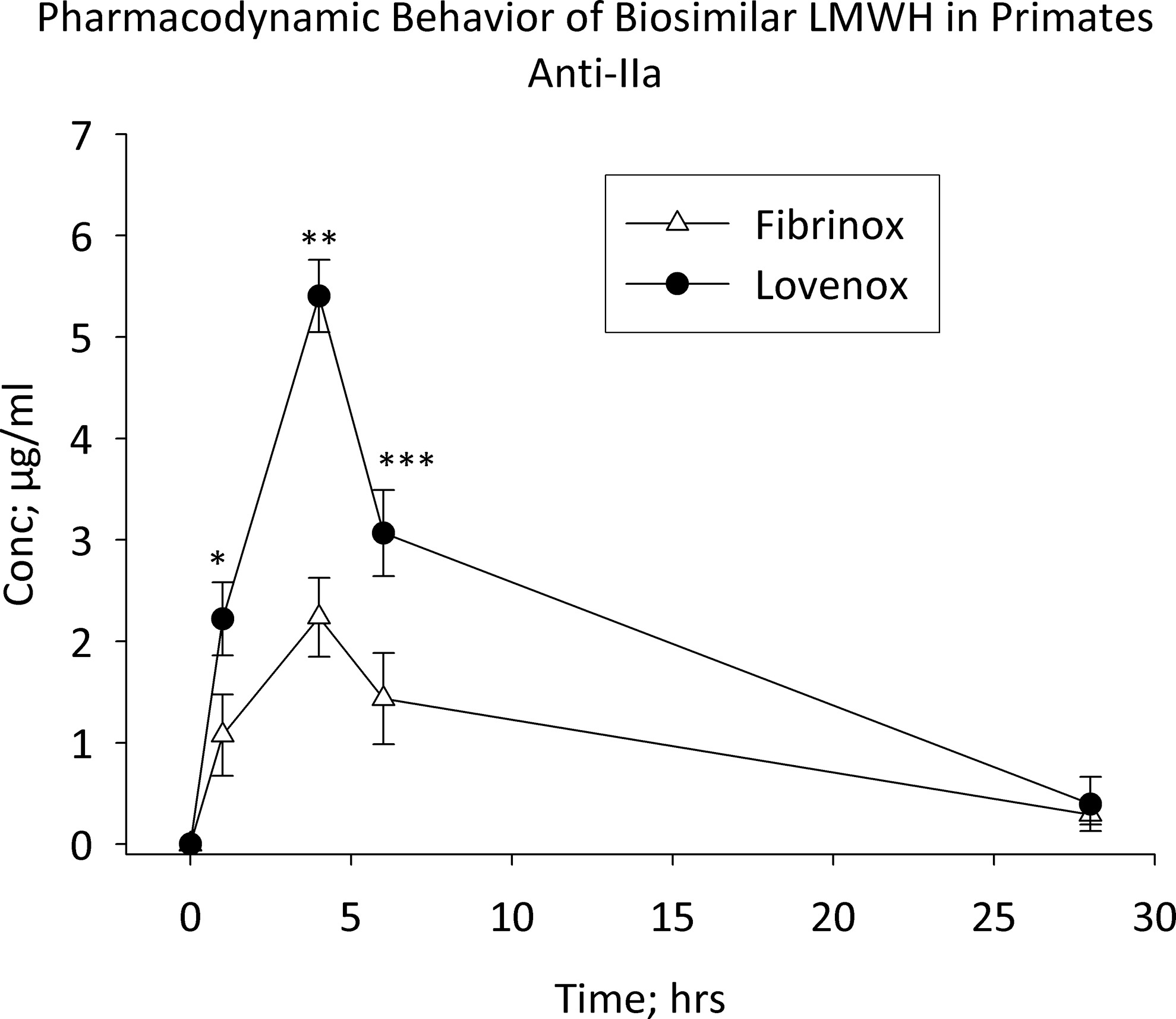
Pharmacodynamic behavior of LMWH formulations in primates. Macaca mulatta were treated with Lovenox and Fibrinox at a dose of 1 mg/kg subcutaneously (SC) in a crossover fashion. Blood samples were collected prior to dosing and at 1, 4, 6, and 28 hours post-dosing. Platelet-poor plasma was analyzed for anti-IIa activity using an amidolytic assay. Results represent the mean ± SEM of 8 primates/treatment group. *P = 0.017, **P < 0.001, ***P = 0.001 versus Fibrinox. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
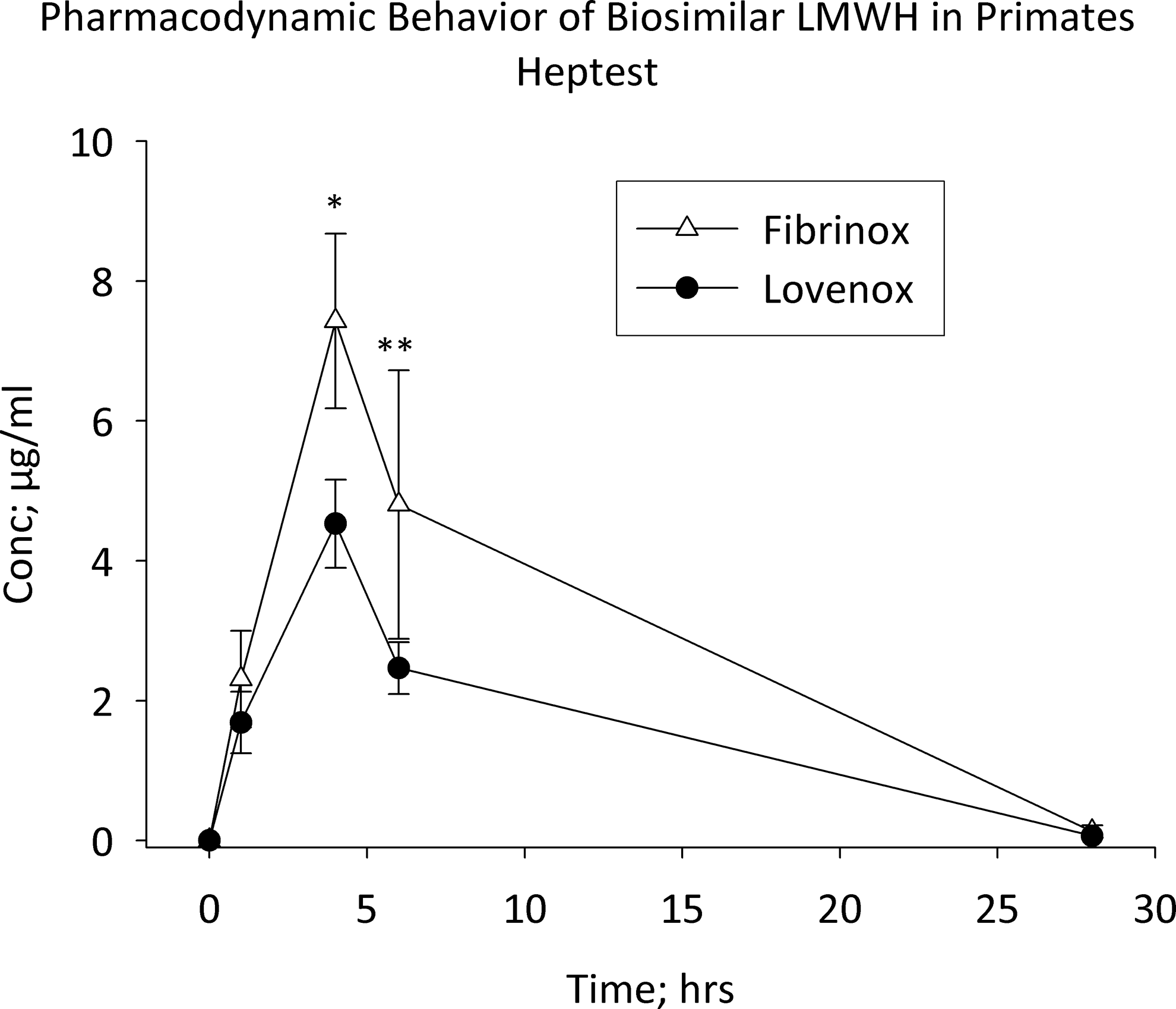
Pharmacodynamic behavior of LMWH formulations in primates. Macaca mulatta were treated with Lovenox and Fibrinox at a dose of 1 mg/kg SC in a crossover fashion. Blood samples were collected prior to dosing and at 1, 4, 6, and 28 hours post-dosing. Platelet-poor plasma was analyzed using the Heptest assay. Results represent the mean ± SEM of 8 primates/treatment group. *P = 0.009, **P = 0.031 versus Lovenox. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
The circulating drug concentrations were used to estimate pharmacokinetic parameters. Table 1 summarizes the pharmacokinetic behavior of Fibrinox and Lovenox following subcutaneous administration to primates. A high degree of variability was observed in the absorption and elimination of the LMWHs. Statistically significant differences in pharmacokinetic behavior was not observed when circulating drug concentrations were determined using anti-Xa activity or Heptest prolongation. Using drug concentrations determined from anti-IIa activity, the AUC following Lovenox treatment was significantly larger than following Fibrinox treatment.
Estimation of Pharmacokinetic Parameters
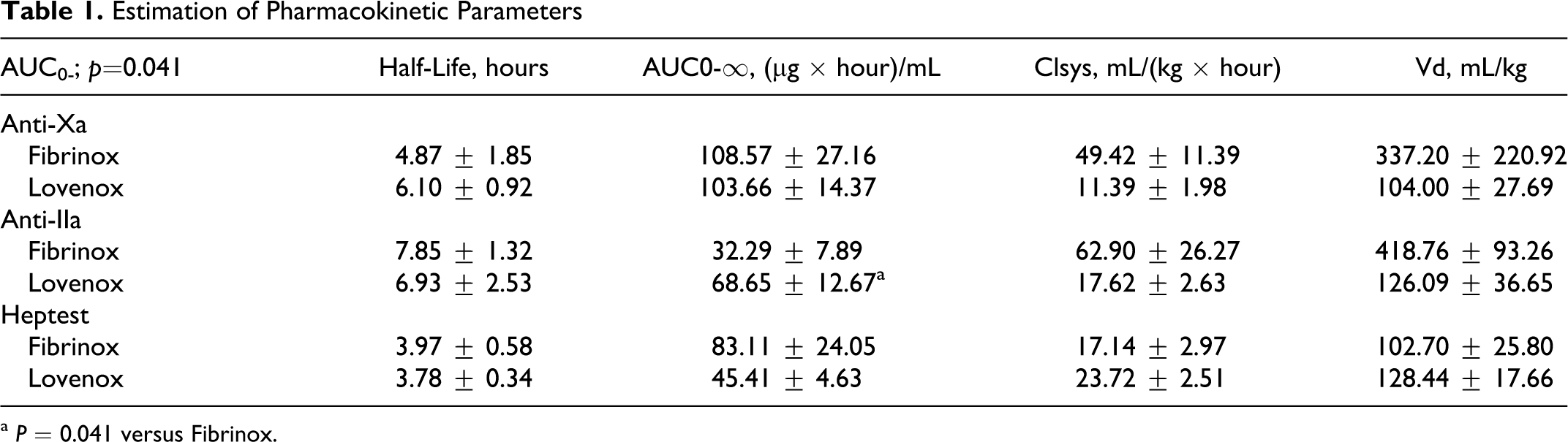
a P = 0.041 versus Fibrinox.
Plotting mean drug levels based on anti-IIa activity versus mean drug levels based on anti-Xa activity at the various sample times indicated a hysteretic relationship (Figure 4 ). Importantly, the hysteresis loops obtained by analyzing plasma samples from Fibrinox- and Lovenox-treated primates using common assays were distinct. Clear differentiation of the 2 LMWHs was also observed when drug levels measured by anti-IIa activity were plotted against those determined using the prolongation of the Heptest (Figure 5 ).
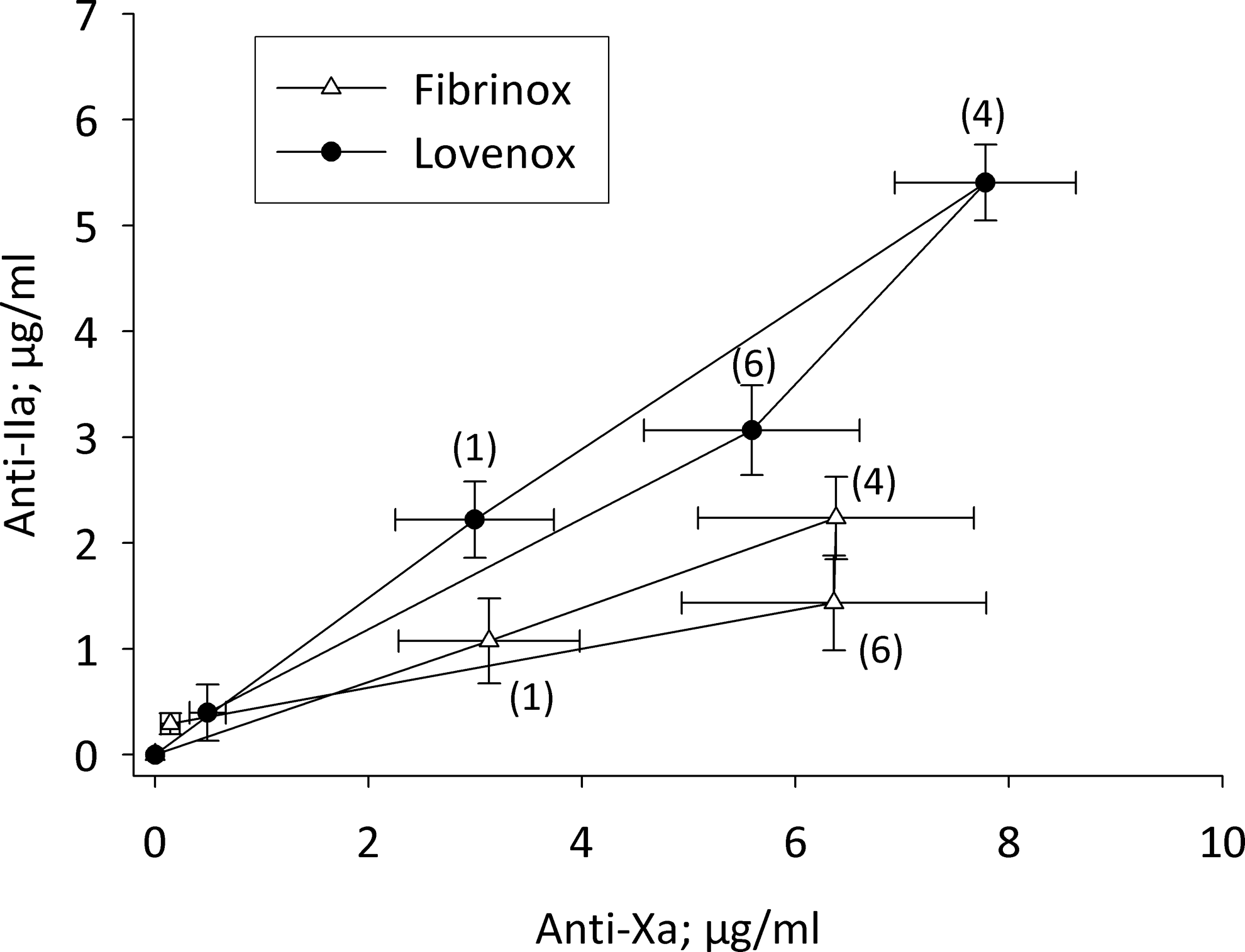
Pharmacodynamic behavior of LMWH formulations in primates. Macaca mulatta were treated with Lovenox and Fibrinox at a dose of 1 mg/kg SC in a crossover fashion. A clockwise hysteresis is observed when plasma drug concentrations determined based on anti-IIa activity are plotted against those determined using anti-Xa activity. The numbers in parentheses on the graph indicate the time of sample collection in hours post-LMWH administration. Results represent the mean ± SEM of 8 primates/treatment group. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
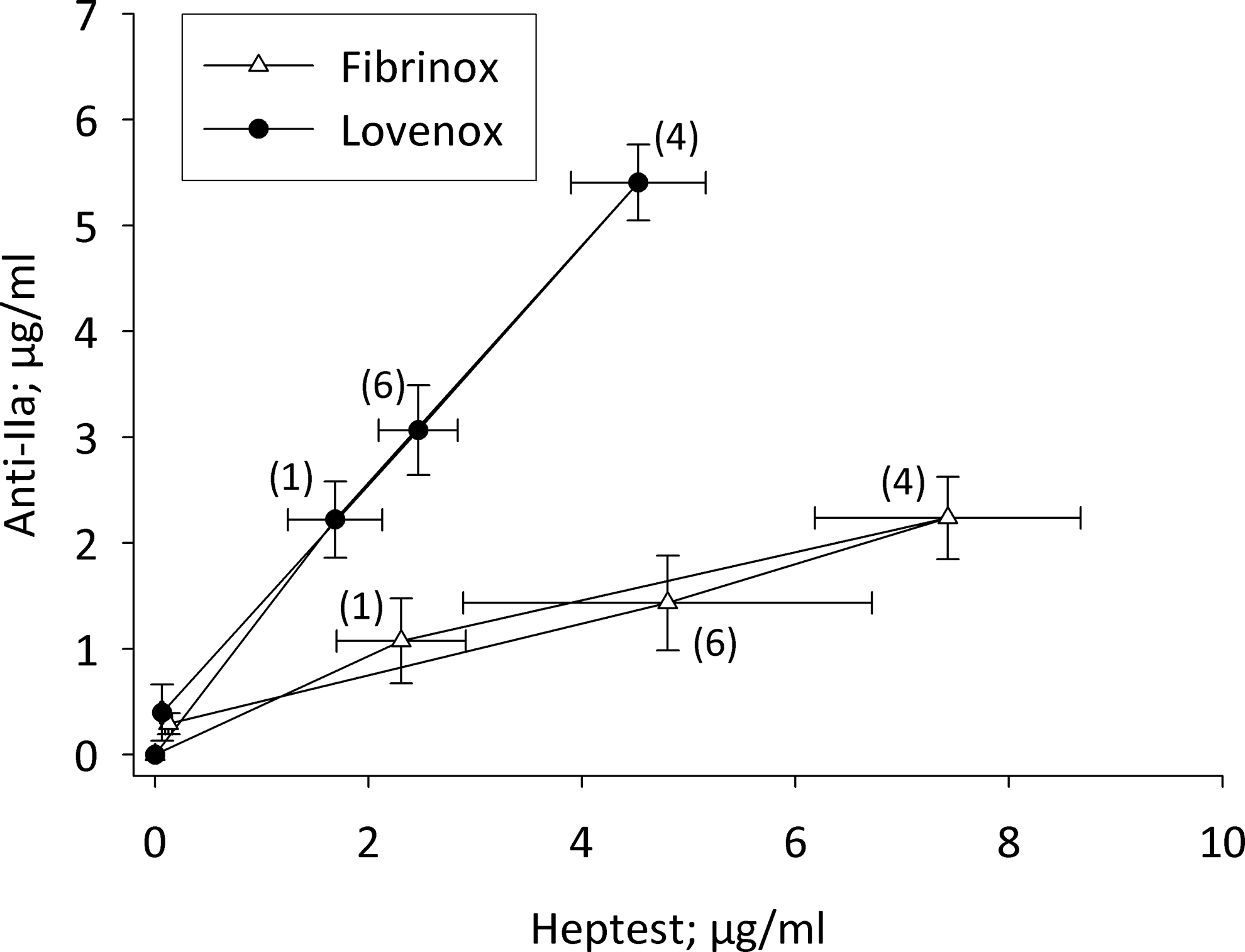
Pharmacodynamic behavior of LMWH formulations in primates. Macaca mulatta were treated with Lovenox and Fibrinox at a dose of 1 mg/kg subcutaneously (SC) in a crossover fashion. A clockwise hysteresis is observed when plasma drug concentrations determined based on anti-IIa activity are plotted against those determined using Heptest prolongation. The numbers in parentheses on the graph indicate the time of sample collection in hours post-LMWH administration. Results represent the mean ± SEM of 8 primates/treatment group. LMWH indicates low-molecular-weight heparin; SEM, standard error of the mean.
Discussion
Defining generic equivalence requires the demonstration of identical chemical structure and pharmacokinetic/pharmacodynamic behavior. This is difficult for biological drugs such as LMWHs because chemical means of measuring plasma heparin levels do not exist, pharmacokinetic behavior needs to be quantified in terms of biological activity, heparins are multicomponent and efficient means of identifying and quantifying the various oligosaccharide components in an LMWH preparation have not been available. In addition, potency can be expressed in terms of a number of AT-dependent and AT-independent activities, which may not be proportional. Guidelines from both the US Food and Drug Administration and European Medicines Agency for defining biosimilarity of LMWHs call for performing pharmacokinetics and pharmacodynamics studies.
The pharmacokinetic behavior of LMWHs is different when the drug levels are determined using distinct biological activities. 11 This results from LMWHs being multicomponent drugs. It is known that smaller heparin chains are absorbed to a greater extent than longer chains. This is observed most clearly in the nearly complete bioavailability of fondaparinux and LMWHs compared to a subcutaneous bioavailability of approximately 30% for unfractionated heparin. It is also known that heparin chains of at least 18 saccharides are required to produce anti-thrombin activity. 12 Therefore, it is expected that the time course of various heparin activities in the plasma will be different.
The measured concentration of a drug and its corresponding effect can be asynchronous, that is, peak drug levels may not occur at the same time as peak effect levels. This phenomenon can be due to a number of reasons including the development of tolerance, the formation of agonistic or antagonistic metabolites, up- or downregulation of receptors, response delay, or the slow distribution of drug from the sampling site to the effect site. 13 In these cases, when concentration is plotted against effect, a hysteretic loop is formed. Such an effect has previously been reported when anti-Xa levels were measured following the subcutaneous administration of tritiated tinzaparin to dogs. 14 In this study, the clockwise hysteresis loop is believed to be a result of rapid absorption of smaller, relatively inactive components and slower absorption of larger, more biologically active heparin chains.
In our study, plotting drug concentrations determined by one functional parameter against those derived from a different parameter provided clear visual evidence that the 2 LMWHs tested exhibit distinct pharmacodynamic behavior and, therefore, do not appear to be bioequivalent. This investigation underscores the earlier observation where differences between generic and branded products have been reported. The pharmacodynamic analysis using the hysteresis plots clearly suggests that compositional differences between generic and branded products which are not detectable by pharmacopoeial methods may exist. Therefore, to demonstrate bioequivalence between generic and branded products, pharmacodynamic studies are warranted. Measuring additional functional parameters such as prolongation of the prothrombinase-induced clotting time, inhibition of thrombin generation, or the release of tissue factor pathway inhibitor (TFPI) may also be valuable.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
