Abstract
The long-term outcomes of pulmonary thromboembolism (PTE) and/or incidence of chronic thromboembolic pulmonary hypertension (CTEPH) are not well documented. Three hundred twenty-five consecutive cases objectively diagnosed with PTE monitored for an average 16.3 months (6-50.7 months) were investigated. Data concerning recurrence, residual thrombus, mortality, and CTEPH in particular were collected. Chronic thromboembolic pulmonary hypertension frequency acute first episode of PTE was 4.6%. Chronic thromboembolic pulmonary hypertension developed within 12 months after PTE attack in approximately 80% of patients and it did not occur after 2 years. Residual chronic thrombus was determined at the 3rd month in 48% of cases post-PTE, at the 6th month in 27.4%, and at the 12th month in 18.2%. At multivariate regression analysis, systolic pulmonary arterial pressure > 50 mm Hg, hazard ratio: 10.1 (95% confidence interval: 4.1-71.2) were predictors of CTEPH. Chronic thromboembolic pulmonary hypertension develops as a serious complication in a significant number of cases with PTE. Closer monitoring of high-risk cases in particular is important in terms of early diagnosis and treatment.
Introduction
The most important long-term complication of pulmonary thromboembolism (PTE) is chronic thromboembolic pulmonary hypertension (CTEPH) that is associated with considerable morbidity and mortality. Although CTEPH had been thought to develop in relatively few patients post-PTE, the latest data suggest an unexpectedly high incidence of approximately 4%.1–5 However, the long-term outcomes of PTE or CTEPH incidence are not well documented.6–8 In addition, although there are well-defined risk factors for PTE, the risk factors for CTEPH have not yet been sufficiently defined. 9 For that reason, the determination of risk factors is of great importance for the early diagnosis of patients with CTEPH, with its high mortality and morbidity.
We were aimed to determine the incidence of symptomatic CTEPH after acute PTE, frequency of residual thrombus, and to investigate the risk factors that may influence CTEPH development in the study.
Materials and Methods
Study Design
The study was approved by our local ethical committee, and written informed consent was obtained from all patients. The study, retrospective between January 2006 and October 2008 and prospective between November 2008 and November 2009, was conducted at a university hospital, a tertiary care hospital that serves as a primary referral center for patients with suspected PTE.
All cases diagnosed with objectively confirmed acute PTE (using computed tomography pulmonary angiography [CTPA] or high-probability pulmonary perfusion scintigraphy, or medium-probability perfusion scintigraphy in the presence of deep vein thrombosis [DVT] at Doppler ultrasonography or magnetic resonance angiography) were enrolled in the study. Cases with any other comorbid condition apart from thromboembolic disease that might lead to pulmonary hypertension (PH) (such as chronic obstructive pulmonary disease [COPD], diffuse interstitial pulmonary disease [DPLD], collagen tissue disease, congenital coronary disease, left heart failure, or medium-severe coronary valve disease [CVD]) were excluded from CTEPH evaluation. Cases undergoing PTE for the first time were included in CTEPH frequency calculation. However, patients with a history of venous thromboembolism (VTE) were included for CTEPH risk factors.
Patients
Patients diagnosed with pulmonary embolism (PE) had received standard anticoagulant treatment consisting of unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) and were potential candidates for thrombolytic therapy. In the initial treatment of PE, UFH was administered as a bolus of 80 U/kg followed by a continuous infusion of 18 U/kg per h. The target activated partial thromboplastin time is 1.5 to 2.5 times that of a normal control sample. However, some patients were treated with LMWH 100 IU/kg twice daily. Thrombolytic therapy used an intravenous tissue plasminogen activator (100 mg for 2 hours). Oral anticoagulant was given for at least 3 months, with the aim of achieving an international normalized ratio of 2.0 to 3.0 during follow-up. A vena cava filter was installed in patients in whom the anticoagulant therapy was contraindicated or with a high risk of recurrent VTE. Patients were monitored and followed up on a regular basis during and after treatment.
Demographic characteristics thought to be potential risk factors for PTE and thrombus localization and spread, and treatment techniques, systolic pulmonary arterial pressure (sPAP) and accompanying DVT during an acute event, comorbid conditions, a previous history of VTE, and laboratory findings were recorded.
Patients were classified as having PE associated with risk factors in 3 groups: transient (recent trauma, fracture, surgical intervention, hospitalization, pregnancy, and the use of oral contraceptives or hormone-replacement therapy) or permanent (antithrombin deficiency, protein C, or protein S; mutation in the factor V Leiden or prothrombin gene; and the presence of lupus anticoagulants, active cancer, immobilization due to chronic medical illness, or 2 or more first-degree relatives with VTE) and all other patients were classified as having idiopathic PE.
Pulmonary thromboembolism observed at CT during an acute event was examined in 2 different ways, depending on thrombus spread and thrombus localization. (1) Thrombus localization indicates main pulmonary artery, lobe, and segment/subsegment. (2) Thrombus spread was defined as very severe, severe, medium, or slight (very severe = located in the main pulmonary artery + lobe + segment/subsegment; severe = only located in the main pulmonary artery; medium = located in the lobe ± segment/subsegment; and mild = located in the segment/subsegment).
Observation and Investigation
Patients were monitored for CTEPH-associated symptoms and findings (exercise or resting dyspnea, syncope, chest pain, etc) with polyclinic checkups during and after anticoagulant therapy for at least 3 to 6 months. All cases were investigated in terms of CTEPH, residual thrombus, recurrent VTE, and cause and date of death.
At long-term follow-up after acute PTE, patients with symptoms suggestive of PH were investigated in terms of CTEPH in the light of the contemporary literature and algorithms recommended in PH guidelines.2,7,10 Following detailed medical questioning and physical examination, individuals were evaluated using pulmonary imaging, pulmonary function test, blood arterial gases, and transthoracic Doppler echocardiography (ECHO).
Echocardiography examination was performed with two-dimensional, conventional and tissue Doppler echocardiography on a Vivid 7 system (GE Vingmed Ultrasound, Horten, Norway) with a 3.4 MHz transducer probe. Systolic pulmonary arterial pressure >35 mm Hg at rest with Doppler ECHO was regarded as CTEPH.7,11 The sPAP was calculated from the tricuspid regurgitation pressure gradient (TIPG = 4 × tricuspid regurgitation velocity 2 ) using the modified Bernoulli equation with the addition of right atrial pressure (5 mm Hg). Echocardiography was performed independently in duplicate by 2 operators blinded to the results of other operator and unaware of the clinical characteristics of the patients.
Patients with no other pathology that might account for PH etiology were examined for CTEPH using pulmonary perfusion scintigraphy. A macro field-of-view gamma camera (Siemens E-cam Dual-Head; Siemens, Hoffman Estates, USA) equipped with a low-energy, parallel-hole, high-resolution collimator was used to confirm lung perfusion imaging. Images were obtained at 4 standard projections (anterior, posterior, right posterior oblique, and left posterior oblique). Chronic thromboembolic pulmonary hypertension was excluded in the event of normal or low-probability perfusion scintigraphy.7,10,12 Chronic thromboembolic pulmonary hypertension was considered in cases with at least one segmental or wider perfusion defect (medium- or high-probability scintigraphy), after which these patients were administered contrast CTPA.11,12
Computed tomography pulmonary angiography was determined using 16-channel multislice scanner (Somatom Volume Zoom and Sensation 16; Siemens, Erlangen, Germany). A 100-mL dose of iopromide (Ultravist 370; Schering, Berlin, Germany) was then injected intravenously using a power injector via the right antecubital vein at a rate of 2 mL/s. Chronic thromboembolic pulmonary hypertension was confirmed in the case of organized thrombus occluding the pulmonary arteries completely, and/or else eccentrically and/or concentrically, and/or a mosaic perfusion picture in the parenchyma with reduced vascular calibration in distal regions (variations in the size of lobe and segmental-level vessels) and pulmonary artery dilatation at CTPA.9,11 Other pathologies involved with the differential diagnosis of CTEPH at detailed examination of the parenchyma and mediastinum at CT were also evaluated.7,11 The severity of symptomatic impairment was classified according to the New York Heart Association (NYHA) classification of heart failure.
Pulmonary angiography and right catheterization were performed before surgery on patients diagnosed with CTEPH and evaluated as operable at the surgical treatment center.2,12
Statistical Analysis
The Kolmogorov-Smirnov test was used to test for normal distribution of continuous variables. Data characterized by normal distribution were expressed as mean values followed by standard deviation. Parameters without such a distribution were expressed as a median with range. Student test (normal distribution) or Mann-Whitney (nonnormal distribution) test were used to compare the 2 groups. The investigation for a diagnostic cutoff value is based on receiver–operating characteristic (ROC) curves. Multivariate regression analysis was used in a stepwise descending method from prognostic factors with significance <.1 at univariable analysis. Results are given as odds ratio (95% confidence interval [CI]), and P <.05 was considered statistically significant. Data were analyzed using SPSS statistical software (version 13.01, serial number 9069728, SPSS Inc, Chicago, Illinois).
Results
Patient Characteristics
Three hundred twenty-five consecutive patients objectively diagnosed with PTE between January 2006 and November 2009 were identified. Persistent dyspnea was determined in 102 of these during monitoring, and pulmonary artery pressures were measured using transthoracic ECHO. Under the study design, a total of 34 patients were excluded from CTEPH investigation, 32 of the PTE patients for comorbid condition that might lead to PH other than thromboembolic disease (11 moderate-to-severe dilate cardiomyopathy, 6 severe COPD, 4 CVD, 3 severe sleep apnea syndrome, 3 end-stage DPLD, and 5 patients with uncommon causes) and 2 patients lost to follow-up. Symptomatic cases among the remaining 291 patients were investigated in terms of CTEPH.
Of the 291 acute PTE cases, 259 had developed it for the first time, while 32 had a previous history of VTE (16 patients with previous PTE and 16 with previous DVT). Only the 259 cases with first episode PTE were included in CTEPH frequency calculations. The study flowchart is given in Figure 1 . However, patients with a previous history of VTE were included in the risk factor investigation. Patients were monitored for at least 6 months, with an average duration of 16.3 (6-50.7) months. Characteristics of the cases developing CTEPH (n = 19) and without CTEPH (n = 272) are given in Table 1 .
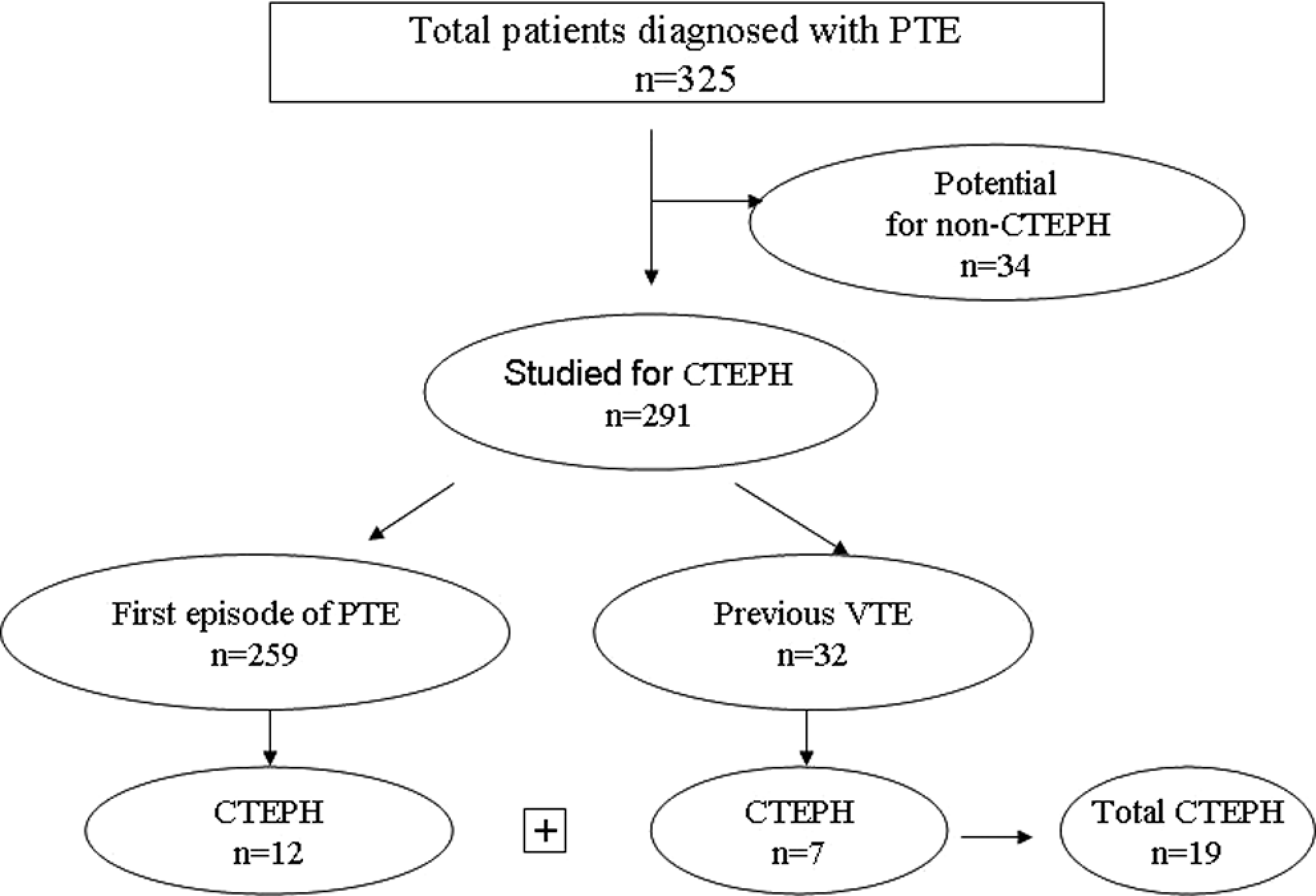
Flow diagram for study selection.
Demographic and Clinical Characteristics of Patients a
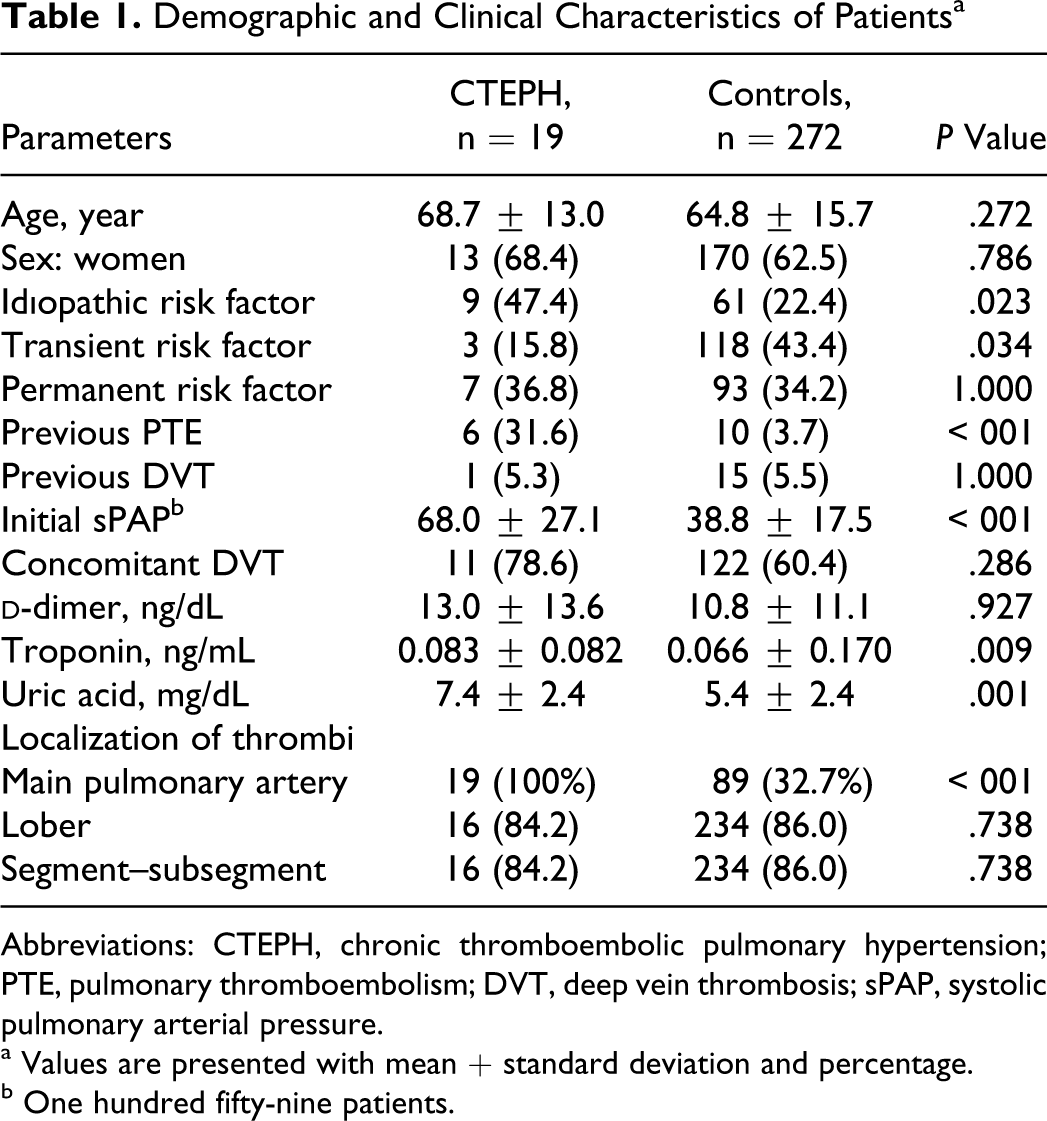
Abbreviations: CTEPH, chronic thromboembolic pulmonary hypertension; PTE, pulmonary thromboembolism; DVT, deep vein thrombosis; sPAP, systolic pulmonary arterial pressure.
a Values are presented with mean + standard deviation and percentage.
b One hundred fifty-nine patients.
Long-Term Results
Mortality
A PTE-associated fatality level of 8.9% was determined in a 1-month period after acute events compared to a general long-term mortality rate of 24.6% during monitoring. In order of frequency, 36.3% of deaths were associated with PTE, 22.5% with malignity, 12.5% with COPD/respiratory failure, 7.5% with heart failure, and 7.5% with CTEPH. Malignity was diagnosed post-PTE attack in 6 (33.3%) of the 18 patients who died due to malignity, the commonest cause of death after thromboembolic disease.
Thrombus resolution
At CTPA investigation for thrombus resolution in the 3- to 12-month period after an acute event, residual chronic thrombus was determined in 48% of cases (58 of 121) at the 3rd month, 27.4% (40 of 146 cases) at the 6th month, and 18.2% (29 of 159) at the 12th month. However, persistent PH was determined in 19 of the 29 cases with persistent chronic residual thrombus. Pulmonary hypertension did not develop in 2 cases, despite the persistence of residual chronic thrombus in the lobe ± segmental branches at 27 and 36 months.
Recurrent thromboembolism
Recurrent thromboembolism was determined in 19 (5.8%) of the 325 PTE cases during monitoring. Recurrence took place in 13 (5%) of the cases undergoing PTE attack for the first time, and re-embolism occurred in 4 (25%) of the 16 cases with a previous history of PTE. No re-embolism was determined in any of the 16 cases with a previous history of isolated DVT.
In terms of permanent and temporary risk factors, recurrence was greater in idiopathic PTE cases, although the difference was not statistically significant (in 10% of the 70 idiopathic PTE cases, P = 138; 4.1% of the 121 cases with temporary risk factors, P = 215; and 5% of the 100 cases with permanent risk factors, P = 195); 71.4% of the recurrences in the idiopathic PTE cases developed within approximately 6 months of the end of treatment. In terms of the relation between delayed thrombus resolution and re-embolism, the recurrent embolism level was significantly high in cases in which thrombus resolution lasted more than 3 months (in 11 [19%] of the 58 cases with residual thrombus at the third month and 1 [1.6%] of the 63 cases with complete resolution; P = 002).
Chronic thromboembolic pulmonary hypertension
Symptomatic CTEPH was determined in 12 (4.6%) of the 259 with first episode of PTE. Of the 32 cases with a previous history of VTE, symptomatic CTEPH developed in 37.5% of those with a history of PTE (6 of 16) and in 1 (6.3%) of the 16 cases with a history of DVT. Chronic thromboembolic pulmonary hypertension thus developed in a total of 19 patients, 12 after first PTE attack and 7 with a previous history of VTE. All patients’ functional capacities were between NYHA classes II and IV. Six (31.6%) of the 19 cases developing CETPH died in 6 to 18 months after diagnosis. Two of the cases with functional classes III to IV were operated on. The postoperative functional capacities of one of these rose to NYHA class II at ninth month follow-up and the other at the sixth month. Other patients were monitored with conventional treatment (Table 2 ).
Characteristics of Patients With Symptomatic CTPH
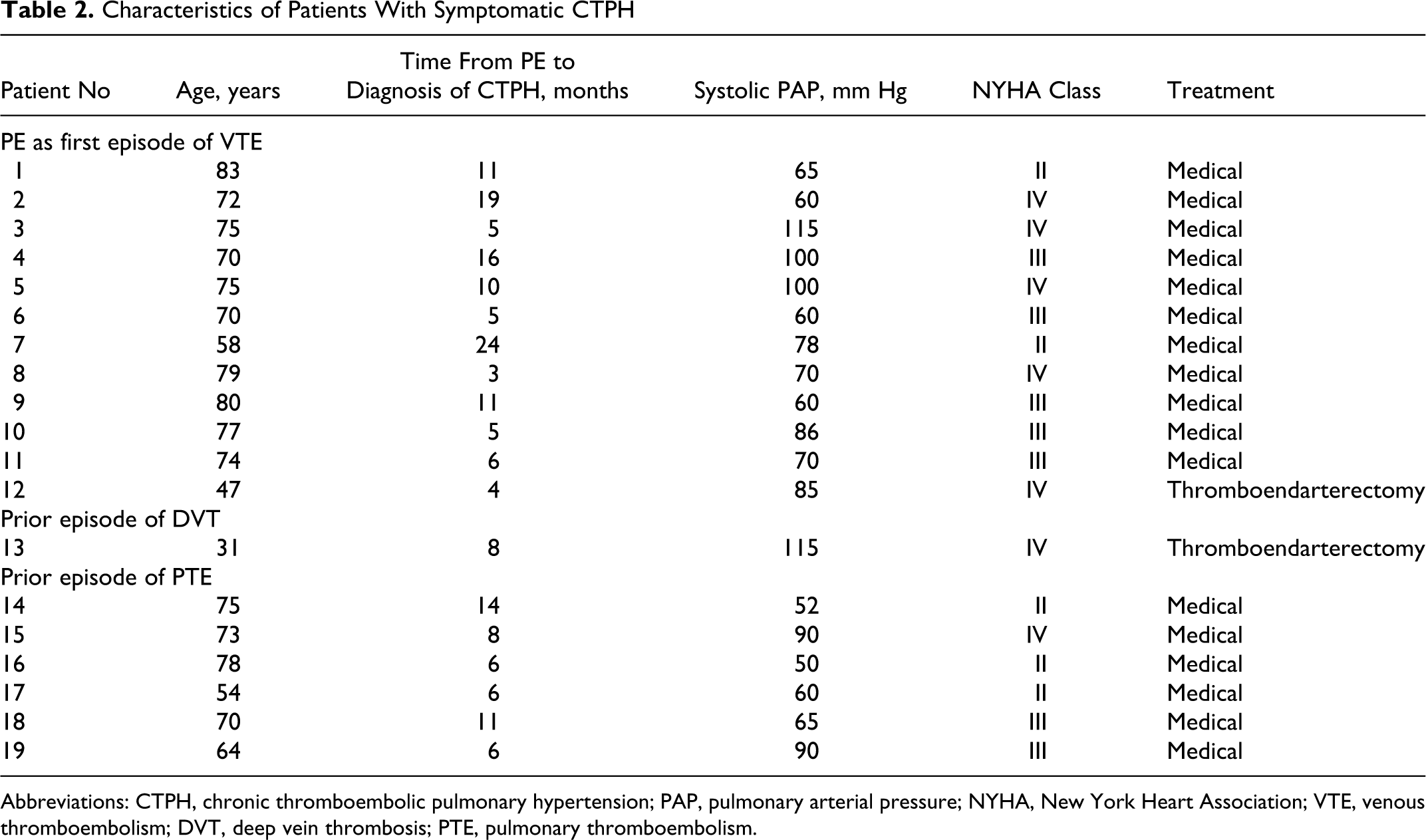
Abbreviations: CTPH, chronic thromboembolic pulmonary hypertension; PAP, pulmonary arterial pressure; NYHA, New York Heart Association; VTE, venous thromboembolism; DVT, deep vein thrombosis; PTE, pulmonary thromboembolism.
Patients were diagnosed with CTEPH on an average of 9.4 months after the first episode of PTE; 47% (9 patients) of the CTEPH were diagnosed 3 to 6 months after PTE, 32% (6 patients) in 6 to 12 months, and 21% (4 patients) in 12 to 24 months. Chronic thromboembolic pulmonary hypertension developed within 12 months after PTE attack in approximately 80% of patients, no cases occurred after 2 years among the patients with more than 2 years of follow-up data. The cumulative incidence of venous thromboembolism after a first episode of pulmonary embolism is given in Figure 2 .
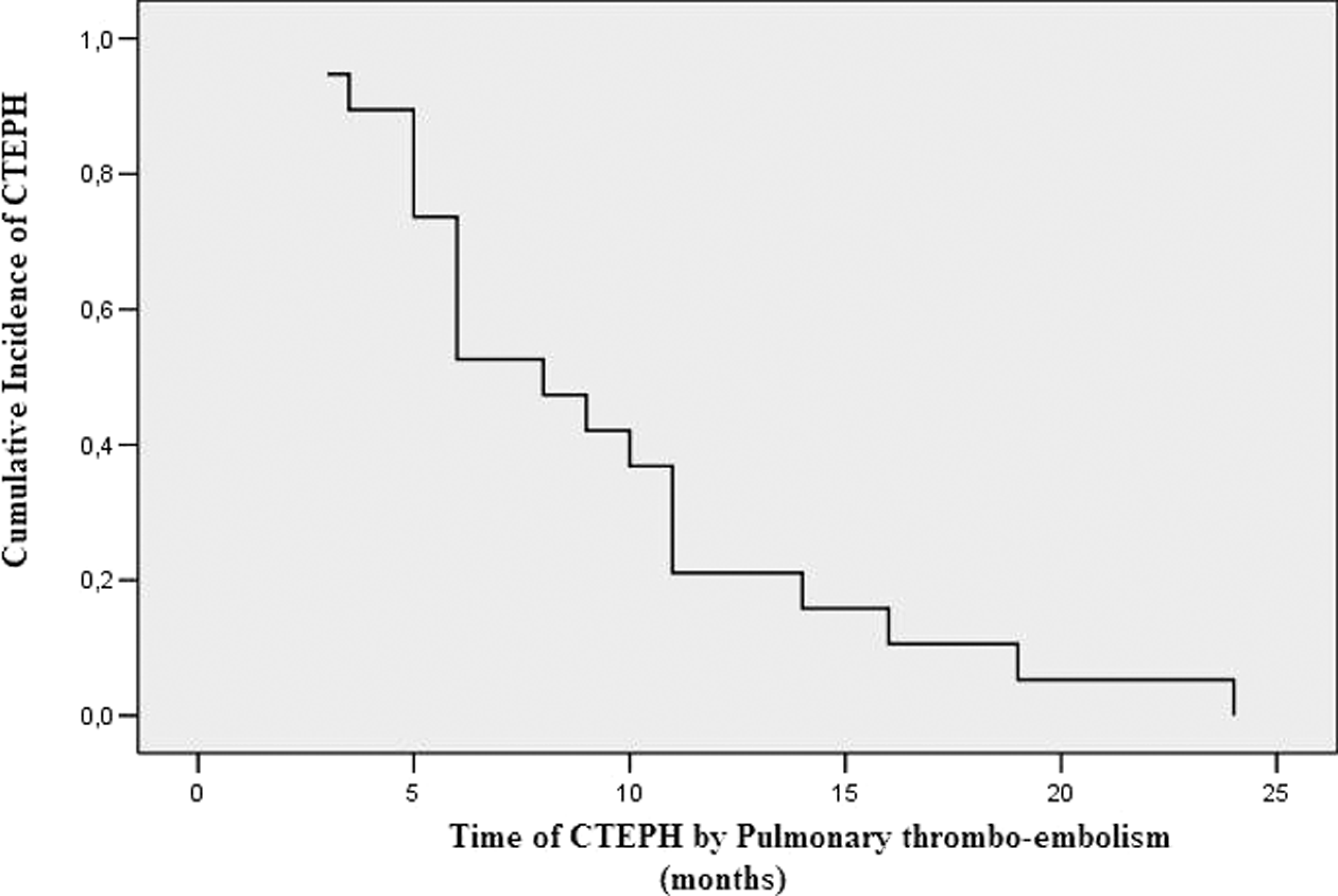
The cumulative incidence of venous thromboembolism after a first episode of pulmonary embolism.
Risk Factor Analysis
Receiver–operating characteristic analysis was used to identify the optimal serum troponin and uric acid cutoff value and average sPAP during PTE diagnosis in order to predict CTPEH. In the ROC analysis, the area under the curve (AUC) was 0.776 (95% CI: 0.667-0.885) of troponin levels for CTEPH. Serum troponin of 0.07 ng/mL showed high sensitivity and specificity for CTEPH of 60% and 82%, respectively. Area under the curve was 0.713 (95% CI: 0.480-0.945) uric acid levels for CTEPH. In cases with uric acid levels >7 mg/dL, the specificity and sensitivity for CTEPH were 72% and 80%, respectively. The AUC was 0.854 (95% CI: 0.705-1.0) sPAP for CTEPH. In acute events, sPAP > 50 mm Hg had a specificity and sensitivity of 75% and 85%, respectively, for prediction of CTEPH.
In univariate analysis, a significant correlation was determined between CTEPH development and previous/recurrent PTE, elevated uric acid, sPAP >50 mm Hg in acute events, widespread severe thrombus, and idiopathic PTE (Table 3 ).
Risk Factors to Predict Symptomatic CTEPH in Univariate Analysis
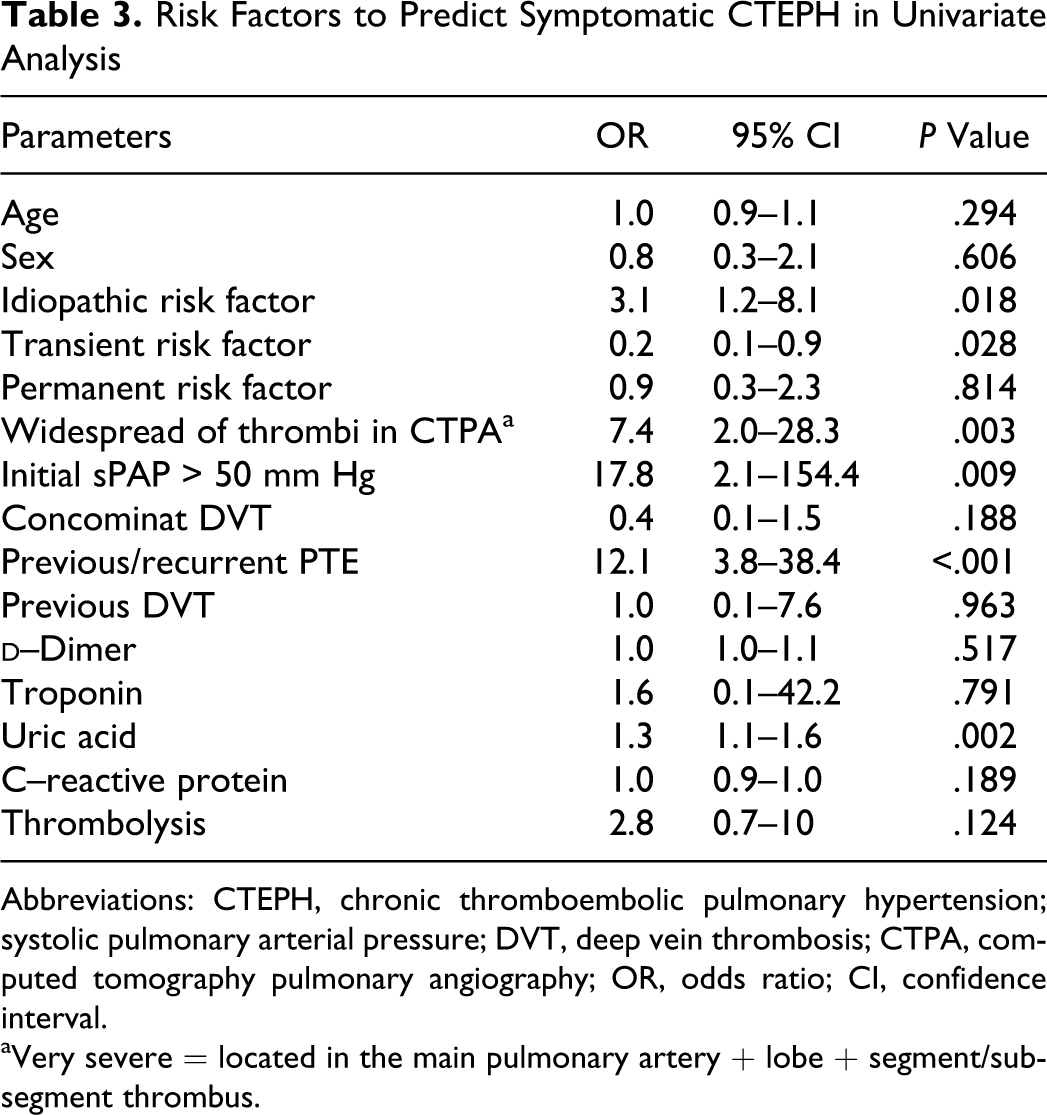
Abbreviations: CTEPH, chronic thromboembolic pulmonary hypertension; systolic pulmonary arterial pressure; DVT, deep vein thrombosis; CTPA, computed tomography pulmonary angiography; OR, odds ratio; CI, confidence interval.
aVery severe = located in the main pulmonary artery + lobe + segment/subsegment thrombus.
At multivariate regression analysis, sPAP >50 mm Hg was associated with a 10.1-fold (95% CI: 4.1-71.2, P = 001) increase in the important finding for predicting CTEPH (Table 4 ).
Risk Factors to Predict Symptomatic CTEPH in Multivariate Analysis
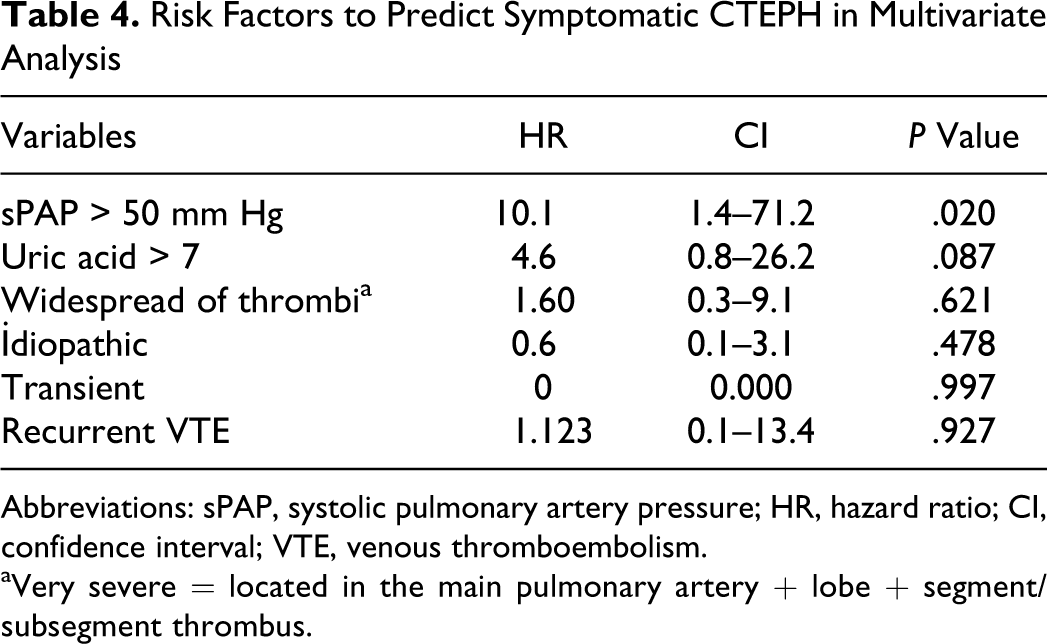
Abbreviations: sPAP, systolic pulmonary artery pressure; HR, hazard ratio; CI, confidence interval; VTE, venous thromboembolism.
aVery severe = located in the main pulmonary artery + lobe + segment/subsegment thrombus.
Discussion
We found that the incidence of symptomatic CTEPH was 4.6% during the average 16.3-month (range 6-50.7 months) monitoring period after the first episode of PTE. This result challenges the contemporary belief that CTPH is rare after an episode of PE and develops long after the acute first PTE.
The CTEPH was traditionally thought to develop in the long-term post-PTE.2,13 However, in the present study, the majority (80%) of cases developing CTEPH was diagnosed within the first year after an acute event, and no new CTEPH diagnosis was determined after the second year. The results of our study are in agreement with those of Pengo et al. 3 In our opinion, CTEPH is more likely to exhibit clinical symptoms within the first year and is unlikely to follow a silent course for several years. Chronic thromboembolic pulmonary hypertension developing after more than 2 years is probably associated with recurring asymptomatic re-embolisms rather than first PTE attack. Our study thus showed that, contrary to the current opinion, the development of CTEPH after acute PTE is more common and generally takes place within a shorter period.
Complete thrombus resolution and full restoration of the pulmonary vascular bed following PTE attack takes 4 to 8 weeks. 13 However, full resolution within this time frame is reported in only 50% to 80% of cases.14–16 One study involving 157 patients with diagnosed PTE determined residual perfusion defect in 66% of cases following 3-month anticoagulant therapy at monitoring with perfusion scintigraphy. 17 Sanchez et al reported residual perfusion defect in 29% of 254 patients with symptomatic PTE at ventilation/perfusion scintigraphy 1 year after diagnosis. 18 Full resolution was observed after 4 weeks in 81% of 69 patients monitored with CT angiography. 16 Remy-Jardin et al used CT to evaluate 62 cases with central PTE alone, approximately 11 months after diagnosis and observed full resolution in 48%, endovascular abnormality in 52%, and incomplete resolution in 39%. 19 Unexpectedly, however, persistent PH was only determined in 8% of these patients at ECHO.
Residual chronic thrombus was determined at the 3rd month in 48% of cases administered CT, 27.4% at the 6th month, and in 18.2% at 12 months. However, persistent PH was only determined at ECHO in 11.9% of these cases. Our results, which are in agreement with the literature, thus show that residual thrombus remains in a significant number of cases post-PTE and that fewer of these lead to PH. The lower 3- to 12-month residual chronic thrombus levels in particular in our study may be attributed to Remy-Jardin et al including only central thromboembolisms in their study performed with CT and to residual thrombus being shown using perfusion scintigraphy in other studies.17–19 Because perfusion defects associated with parenchymal scarring develop in the infarct regions, post-PTE may have increased the perfusion defect levels in these studies to some extent. In addition, residual chronic thrombus is evident in many patients in the late period post-PTE without PH developing. However, the long-term clinical significance of this is unclear. 8
Recurrent VTE develops at a level of 2% to 7% during 3 to 6 months of anticoagulant therapy.8,20 The first 6 to 12 months after cessation of anticoagulant therapy represent the highest risk of recurrence and there is known to be a higher rate of recurrence in idiopathic VTE cases.3,8,13 We determined a recurrence level of 5.8%, and 71% of idiopathic recurrence took place within the first 6 months after the end of treatment. We therefore think that although the length of treatment in cases of idiopathic PTE is a matter for debate, anticoagulant therapy should be administered for a longer term or even for life. In this way, both recurrence-associated mortality and CTEPH, a mortal condition, may be prevented.
It is still unclear whether there is a correlation between recurrent PTE and persistent residual thrombus in the late period after PTE. In our study, the level of recurrent PTE in cases where the thrombus resolution lasted longer than 3 months was statistically significantly higher compared to cases with complete resolution. It may be that residual thrombus observed at CT might be confused with recurrent thromboembolism materials. However, bearing in mind that the residual chronic thrombus in our cases was determined using control CT with no clinical symptoms and that the recurrent thromboembolic events were identified at CT performed due to clinical symptoms, this idea can be excluded as improbable. Moreover, the residual chronic thrombus and acute thrombus material in recurrent PTE were differentiated by radiological appearance. The clinical significance of residual chronic thrombus not leading to PH after PTE attack can be attributed to an increased risk of recurrence in these patients.
On the other hand, while the risk factors constituting a predisposition to acute PTE have been well defined, our knowledge of conditions influencing the development of CTEPH is limited. 9 For instance, the correlation between age and CTEPH is uncertain in the literature. A correlation between CTEPH and advanced age was reported in 1 study, while others have reported a correlation with youth.3,5 As in the study by Bonderman et al, we found no correlation between CTEPH and age. 21
In two previous study, CTEPH was more frequent diagnosed with idiopathic PTE.3,22 This may be because for thrombus formed is unknown and the underlying disease therefore is not treated. Recurrent thromboembolic events are known to play a role in CTEPH development.3,9,21 In the present study, the frequency of cases with previous PTE was significantly higher in the CTEPH group.
In our study, a significant correlation was determined between CTEPH development and widespread thrombus load, while none was determined between thrombus localization and CTEPH development. Widespread perfusion defect was reported as a risk factor for CTEPH development in 2 previous studies.3,23 Our findings, compatible with those in the literature, show that widespread thrombus significantly increases the risk of CTEPH.
Elevated PAP and right ventricular dysfunction are present in approximately half of PTE cases, in such a way as to be correlated with early stage mortality.5,14,23 Pulmonary arterial pressure >50 mm Hg in an acute event is correlated with the development of persistent PH in the late period.5,23 In the present study, right ventricular dysfunction and elevated sPAP (>35 mm Hg) was present in 57% of the cases examined using ECHO. Systolic PAP was higher in the cases developing CTEPH compared to those not. At the time of PTE diagnosis, sPAP >50 mm Hg was considered a significant risk factor for CTEPH, in the study.
High serum acid levels have long been known to indicate poor prognosis in any acute disease. 24 Levels of uric acid, the final oxidation product of purine metabolism, may rise under conditions of oxidative metabolism impairment. High uric acid levels have been shown to be correlated with disease severity and mortality in primary PH.11,25 The mean uric acid levels were higher in patients with CTEPH than in patients without it. Uric acid levels >7 mg/dL have a 72% specificity and 80% sensitivity in predicting CTEPH.
This study has various limitations. The first is that PH was diagnosed using ECHO rather than pulmonary artery catheterization. However, sPAP >50 mm Hg (sPAP > 60 mm Hg in approximately 90% of CTEPH cases) was determined in all of cases developing CTEPH. In addition, there is a powerful correlation between sPAP>50 mm Hg at ECHO and pulmonary catheterization. 7 Moreover, all cases determined with PTE were confirmed again at least twice by individuals unaware of one another. On the other hand, we do not know the number of asymptomatic cases. However, these patients are frequently reported to be diagnosed after becoming symptomatic and the clinical significance is debatable. In addition, asymptomatic CTEPH is not an uncommon finding after PTE. 4 Although part of the study was retrospective, CTEPH was diagnosed with ECHO and scintigraphy findings and chronic thrombus at CT. Moreover, PTE was diagnosed with CTPA in 98% of cases. Our results are therefore quite objective.
In conclusion, CTEPH develops after PTE in a significant number of patients. Close monitoring diagnosed with PTE (especially in the first year in high-risk patients) and the long-term anticoagulant therapy, especially in patients with idiopathic PTE, is of vital importance in terms of preventing re-embolism and CTEPH.
Footnotes
This study was conducted at Farabi Hospital, Trabzon, Turkey.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
