Abstract
Although it was thought that platelets did not play a significant role in the pathogenesis of venous thromboembolism (VTE), several studies demonstrated that a marked activation of platelets occurs in patients with VTE. We carried out a case–control study to investigate the effect of the T744C P2RY12 polymorphism on the risk of VTE in the Korean population. We enrolled 154 consecutive patients with VTE and 415 healthy controls. Genotype frequencies for patients with TT, TC, and CC were 71.4%, 24.7%, and 3.9% and in the controls, 68.2%, 30.1%, and 1.7%, respectively. T744C P2RY12 polymorphism did not significantly affect the risk of VTE. Our study shows that T744C P2RY12 polymorphism did not significantly affect the risk of VTE in the Korean population.
Introduction
Platelet aggregation plays a key role in arterial thrombosis and its complications. Adenosine diphosphate (ADP) is a critical molecule in platelet aggregation. Adenosine diphosphate is released from activated platelets and binds to 2 G protein-coupled receptors P2RY1 and P2RY12. 1 P2RY12 is particularly important in platelet aggregation, since its coupling to a GI protein is responsible for stabilizing platelet aggregates and amplifying aggregation induced by ADP and other agonists. 2 P2RY12 is the target of thienopyridine antiplatelet agents, such as clopidogrel and ticlopidine. 3 Stimulation of this receptor facilitates fibrinogen binding to the glycoprotein (GP) IIb-IIIa receptor, leading to reversible primary platelet aggregation and resulting in irreversible secondary platelet aggregation. 4
The H2 haplotype of the P2RY12 ADP receptor was shown to be significantly associated with increased ADP-induced platelet aggregation in healthy participants. 5 Following that, polymorphisms of P2RY12 were sought as the key to explaining the different responses to thienopyridine antiplatelet agents seen in patients with cardiovascular disease.6-9 Until now, the association between polymorphisms of P2RY12 and variability in platelet response to thienopyridine antiplatelet agents has been inconsistent.
Although it was thought that platelets did not play a significant role in the pathogenesis of venous thromboembolism (VTE), several studies have reported shortened platelet survival in patients with recurrent VTE.10,11 Recently, Chirinos et al demonstrated that a marked activation of platelets occurs in patients with VTE. 12 Venous thromboembolism and arterial thrombosis are usually considered distinct disease entities because of their different anatomic location, risk factors, clinical presentation, and modalities for prevention and treatment. However, recent studies have uncovered an association between VTE and atherosclerosis.13–16 These observations have revived an interest in the possible pathogenetic relationship between P2RY12 polymorphisms and VTE. To address this issue, we carried out a case–control study to investigate the effect of the T744C P2RY12 polymorphism on the risk of VTE in the Korean population.
Patients and Methods
Study Population
Consecutive patients with recent (<6 months) objective diagnosis of deep vein thrombosis or pulmonary embolism who visited to the Bundang CHA hospitals (Seungnam, Korea) between May 2005 and December 2009 were enrolled in the study. Venous thromboembolism was defined as provoked or unprovoked depending on the presence or absence of any of the following risk factors: recent surgery (<3 months), recent trauma/fracture (<3 months), immobilization (>7 days), malignancy, stroke, severe medical disease, autoimmune disease, pregnancy, use of oral contraceptives, and known hypercoagulable disease. Venous thromboembolism was classified as provoked if there was at least 1 of these risk factors. The control group was selected among patients visiting the Bundang CHA Health Promotion Center for periodic health examination with no medical history of VTE. The Institutional Review Board of Bundang CHA Hospital approved the research protocol and written informed consent was obtained from all participating individuals.
T744C P2RY12 Genotyping
The P2RY12 T744C (rs2046934) genotypes were analyzed using the polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) method. 17 Primer sequences used for amplification of P2RY12 T744C were as follows: Forward 5′-TCA CTT ATC TCT GGT GAA ATA AAA AGA TTA CGT A-3′ and Reverse 5′-GTC AGA AAT GGC CTG TGT ATA TAT GGT CAT GAG TTG GCG TAC C-3′. Amplification of P2RY12 T744C was performed using Maxime PCR premix (iNtRON, Seongnam, Korea). The annealing and extension temperatures were 60°C and 72°C, respectively. The PCR products were digested at 37°C for 16 hours by Rsa I (New England BioLabs, Beverly, Massachusetts). The P2RY12 744T allele produced 2 fragments of 190 and 40 base pairs (bp), whereas the 744C allele produced 3 fragments of 157, 40, and 33 bp. The reaction products (10 µL) were loaded on 3% agarose gel, stained with ethidium bromide, and directly visualized under UV illumination.
Statistical Analysis
Statistical analyses were conducted using SPSS 13.0. Differences between the patients with VTE and controls were assessed using the Student t test. Categorical variables were compared using the chi-square test. The distribution of allele frequencies for the T744C P2RY12 gene polymorphism was calculated by chi-square test to determine whether the observed genotype distributions conformed to the expected Hardy-Weinberg equilibrium. Logistic regression analyses were performed to calculate the relative risk of VTE associated with each genotype. Multivariate analysis was performed to select independent risk factors for VTE among genotypes and clinical variables using logistic regression analysis. Odds ratios and corresponding 95% confidence intervals were calculated. Statistical significance was determined to be P < .05.
Results
Population
Baseline characteristics of patients with VTE and controls are shown in Table 1 . A total of 154 patients with VTE and 415 controls were enrolled in the study from January 2006 to June 2009. The median age of the patients with VTE was 54, and 79 patients were male (51.3%). Among the clinical variables compared with patients with VTE, controls had more frequent hypertension history (P = .005). Other than the history of hypertension, there was no significant difference between patients with VTE and controls. Venous thromboembolism was unprovoked in 60 patients and provoked in 94. Among the provoked VTE cases, the VTE in 56 patients (59.6%) were related to recent surgery, recent trauma/fracture or immobilization, malignancy (n = 9), stroke (n = 17), severe medical disease (n = 4), autoimmune disease (n = 4), pregnancy (n = 4), use of oral contraceptives (n = 5), hypercoagulable disease (n = 6), and others (n = 10).
Baseline Characteristics of VTE Patients and Controls
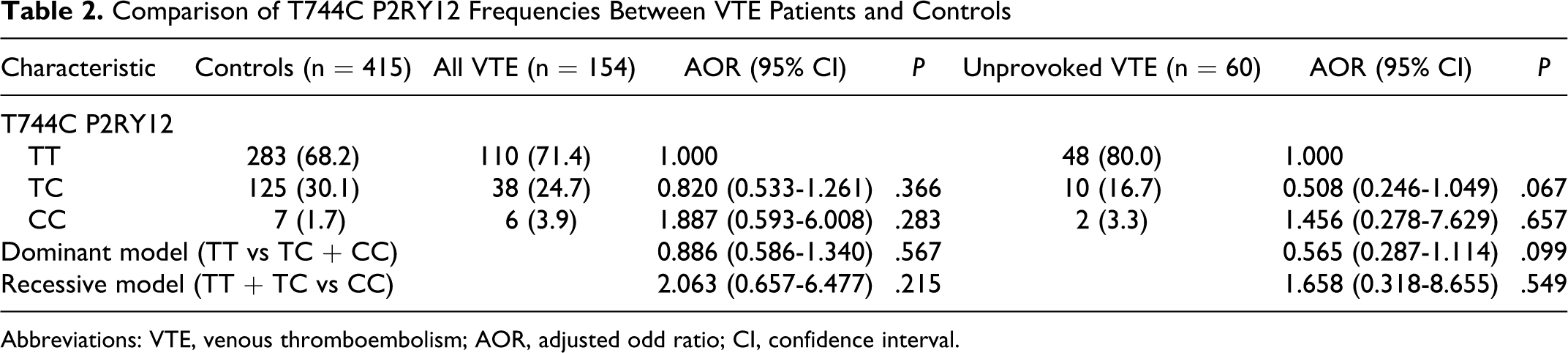
Abbreviation: VTE, venous thromboembolism.
Genotype Frequency of T744C P2RY12 Gene Polymorphism
In the patients with VTE, genotype frequencies for patients with TT wild type, TC heterozygous, and CC homozygous for the T744C polymorphism were 71.4% (n = 110), 24.7% (n = 38), and 3.9% (n = 6), and in the controls, 68.2% (n = 283), 30.1% (n = 125), and 1.7% (n = 7), respectively. The frequencies of each genotype also conformed to a Hardy-Weinberg equilibrium (P > .05). In multivariate analysis, after adjusting for age, sex, and clinical variables (hypertension, diabetes mellitus, hyperlipidemia, and smoking) in the comparisons between VTE and controls, the frequency of the TC heterozygous and CC homozygous genotype was not significantly higher in patients with VTE than in controls. When CC homozygous and TC heterozygous patients were combined for statistical analyses (dominant model), genotype distributions were not significantly different between patients with VTE and controls. Also, when TC heterozygous and TT wild-type patients were combined for statistical analyses (recessive model), the frequency of the CC homozygous genotype was not significantly higher in patients with VTE than in controls (Table 2 ). We classified VTE into provoked or unprovoked VTE cases. We performed same analysis in the unprovoked VTE cases. There was no significant genotype difference between unprovoked VTE and controls. However, in the unprovoked patients with VTE, the frequency of the TC heterozygous genotype was marginally lower in patients with VTE than in controls (odds ratio [OR] = 0.508; 95% confidence interval [CI] = 0.287-7.629, P = .067).
Comparison of T744C P2RY12 Frequencies Between VTE Patients and Controls
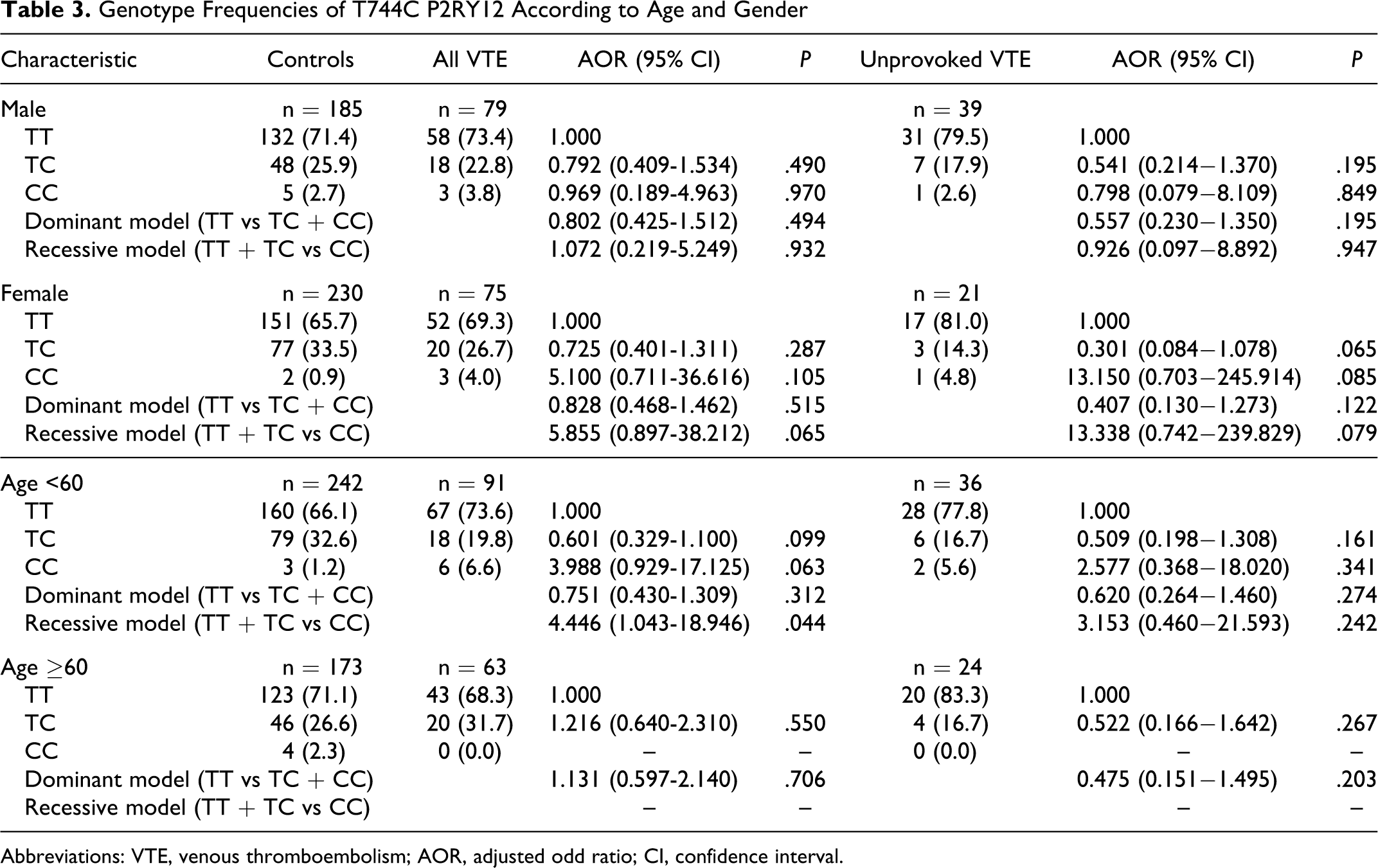
Abbreviations: VTE, venous thromboembolism; AOR, adjusted odd ratio; CI, confidence interval.
Genotype Frequency of T744C P2RY12 Gene Polymorphism According to Age and Gender
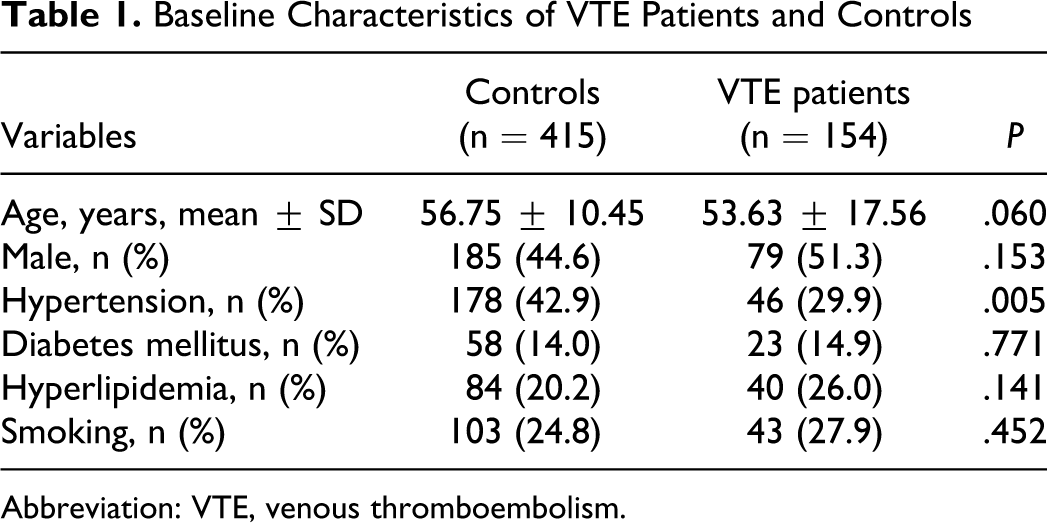
In the gender analysis, the frequencies of the each genotype were not significantly different between patients with VTE and controls in both males and females. The same result was shown in the recessive model. In the subset analysis by age, the frequency of the CC homozygous genotype was significantly higher in patients with VTE than in controls (OR = 4.446, 95% CI = 1.043-18.946) <60 years. However, genotype distributions were not significantly different between VTE and controls >60 years (Table 3 ). In the Table 4 , we summarized the genotype and allele frequencies of T744C P2RY12 polymorphism in different populations. Also, in the subset analysis by hypertension, diabetes mellitus, hyperlipidemia, and smoking, genotype distributions were not significantly different between VTE and controls (data not shown).
Genotype Frequencies of T744C P2RY12 According to Age and Gender
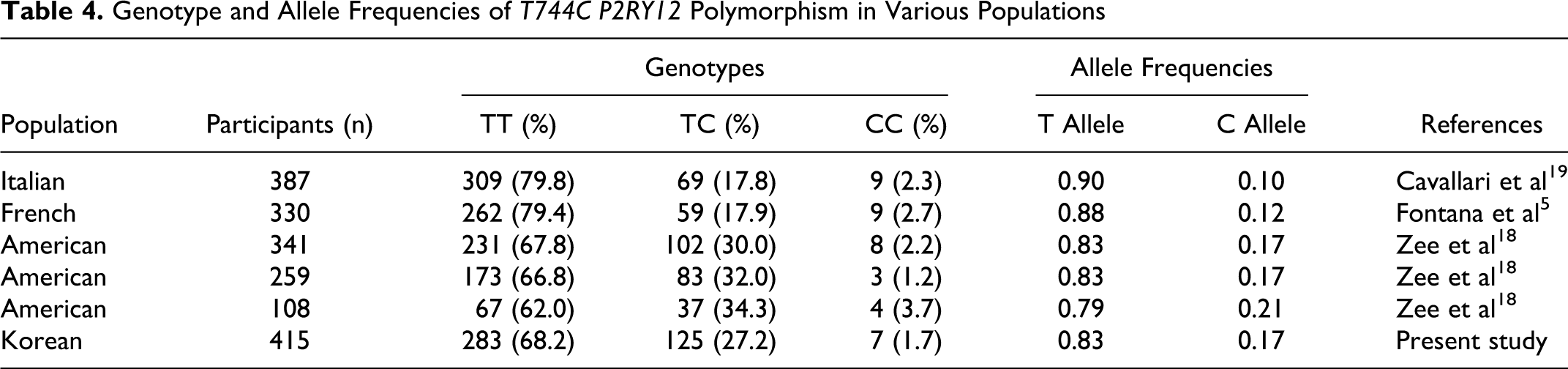
Abbreviations: VTE, venous thromboembolism; AOR, adjusted odd ratio; CI, confidence interval.
Genotype and Allele Frequencies of T744C P2RY12 Polymorphism in Various Populations
Discussion
This is the first study to evaluate an association between the T744C P2RY12 gene polymorphism and VTE in an Asian population. However, we did not find any associations between the T744C P2RY12 gene polymorphism and VTE. In the unprovoked VTE patients, we just found the frequency of the TC heterozygous genotype was marginally lower in patients with VTE than in controls (OR = 0.508, 95% CI = 0.287-7.629, P = .067). In the subset analysis by age, patients with VTE <60 years of age, but not those >60 years of age, had a significantly higher prevalence of the CC homozygous genotype than healthy control participants in the recessive model (TT + TC vs CC).
The P2RY12 gene is located in the P2 receptor gene cluster on chromosome 3q24-q25. The P2RY12 gene spans 47 kb and comprises 3 exons (http://www.ncbi.nlm.nih.gov). The mechanisms through which these gene sequence variations lead to increased atherothrombotic risk have not been well established. Fontana et al first reported a genetic effect of P2RY12 gene polymorphism on the platelet aggregation response to ADP, which is a key function in arterial and VTE. At present, 9 common polymorphisms in the P2RY12 gene have been described. Among them, C139T, T744C, ins801A, and G52T polymorphisms were in complete linkage disequilibrium. So, Fontana et al defined H1 as the major haplotype (a C in position 139, a T in position 744, and absence of the i-ins801A in the intron, as well as a G in position 52 of exon 2) and H2 as the minor haplotype (a T in position 139, a C in position 744, presence of the i-ins801A in the intron, and a T in position 52 of exon 2). 5 They demonstrated that the H2 haplotype is more frequently associated with patients with peripheral arterial disease. 17 Based on these results, the P2RY12 gene have been considered possible candidate in the pathogenesis of vascular diseases. In this respect, several studies have asked whether the thienopyridine response may be influenced by the P2RY12 gene polymorphism. However, these studies concluded that there is no relationship between P2RY12 polymorphisms and platelet response to clopidogrel, as defined by ADP-induced aggregation.6–9 Thus, we can postulate that genetic variation of the P2RY12 gene does not modulate the platelet response to clopidogrel and multiple factors may influence the variability in platelet response to this agent.
One previous study measured the association between the T744C P2RY12 gene polymorphism and VTE. However, in contrast to our results, the results of that earlier study showed that the P2RY12 gene haplotype H2 is associated with a lower risk of VTE. 18 Interestingly, genotype distributions were significantly different for C139T, T744C, ins801A, and G52T polymorphisms between VTE cases and controls in a dominant model. In this study, these polymorphisms showed same effect, we performed only T744C polymorphism in our study. There are several possible explanations for these disparate results. First, because these studies were conducted on a small number of patients, there were not enough data to generalize the genetic effect of T744C P2RY12 gene polymorphism on the risk of VTE. Second, the role of the T744C P2RY12 gene polymorphism in the risk of VTE may vary between populations due to differing racial backgrounds. Table 4 shows genotype and allele frequencies of the T744C P2RY12 gene polymorphism in different populations. The 744C allele frequencies of the T744C P2RY12 polymorphism in patients with VTE in the present study and an American study were 1.7 and 1.7 to 2.1, respectively. However, the 744C allele frequency of patients with VTE in our study was similar to the controls in the American study.
Although our study populations are too small to generalize our results, it is the first to investigate the role of the T744C P2RY12 polymorphism on the risk of VTE in Asians. In conclusion, the minor allele frequency of the T744C P2RY12 gene polymorphism in healthy Korean population was measured as 1.7%. Our findings suggest that the T744C P2RY12 polymorphism is not associated with the risk of VTE in the Korean population. However, larger studies are needed to generalize these results.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Priority Research Center Program through the National Research Foundation of Korea Grant funded by the Korean Government (2009-0093821), partly supported by Korea Research Foundation (KRF-2008-313-E00281), and partly supported by the research fund from the Korean Ministry of Science and Technology (R01-2006-000-10654-0).
