Abstract
Venous thromboembolism (VTE), including pulmonary embolism (PE) and deep vein thrombosis (DVT) occurs more frequently in cancer patients than in the general population. A retrospective cross-sectional study was carried out in patients with solid tumor complicated with VTE admitted to the Cancer Center, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology between January 1st, 2008 and December 31th, 2017. The incidence of VTE in hospitalized cancer patients was 1.8%, twice the incidence of VTE in hospitalized non-cancer patients. The annual incidence of cancer-associated VTE in our center varied between 1.6% in 2015 and 0.4% in 2009 with an overall average incidence of 1.3% over the research decade. BMI values of 549(67.7%) cancer patients were within the normal range, but none of patients had BMI greater than 35 kg/m2. 747(92.1%) cancer patients had ECOG PS score ≤ 2 and 481(59.3%) had distant metastasis. Patients with pancreatic, bladder, ovarian and endometrial cancer had the highest incidence of VTE. Upper extremity DVT (47.2%) was more common in cancer patients and might be closely associated with CVC (74.9%), while lower extremities DVT (36.1%) intended to PE development (15.0%). The annual incidence rates showed a fluctuating and upward trend over the research decade. VTE occurrence was closely related to tumor stage, tumor site, catheterization and anti-neoplasm therapy in cancer patients.
Introduction
Venous thromboembolism (VTE) is a common complication in the natural history of malignancies. Cancer patients are at a high risk of VTE, account for approximately 20% of all vein thrombosis patients 1 and have a 4- to 6-fold increased risk of developing vein thrombosis compared to patients without cancer. VTE, which comprises deep vein thrombosis (DVT) and pulmonary embolism(PE), is responsible for 9% of death in cancer patients, making it the second leading cause of death for cancer patients. 2 Acute idiopathic VTE can even present as the first manifestation of an occult malignancy when it develops in less common sites such as the neck, the vena cavae, or the visceral. 3 Additionally, the development of VTE in a cancer patient always portends a worse prognosis. 4,5
Despite the severity of VTE, few studies provide information on VTE specific to the Chinese cancerous population. In fact, the sensitivity and specificity of the present scales for VTE assessment (eg. Caprini Risk Score, Padua Prediction Score and Khorana Risk Scor) are not high enough to predict VTE among the hospitalized cancer population. 6 In this retrospective analysis, records of patient admitted to Cancer Center, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (HUST) between 2008 and 2017 were analyzed to estimate the incidence of VTE in patients with malignancies. In addition, the characteristics of patients, medication and thrombosis were further analyzed to reflect the incidence and regularity of VTE among the Chinese cancer population, to arouse the highlight of oncologist on the VTE risk in the long lasting anti-neoplasm treatment.
Materials and Methods
Patients and Data Sources
The retrospective population based cross-sectional study was defined as hospitalized patients with a confirmed diagnosis of solid malignancies admitted to the Cancer Center, Union Hospital, Huazhong University of Science and Technology (HUST) between January 1st, 2008 and December 31th, 2017. Patients was screened and diagnosis information was abstracted from medical records of hospital admission through International Statistical Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9 CM) code (C00-89, 97). The cancer stage was established based on clinical assessment and imaging following the sixth Edition of the UICC TNM classification of malignant tumors.
Definition of Deep Venous Thrombosis and Pulmonary Embolism
Deep veins referred to veins accompanying the arteries of the same name. DVT was diagnosed according to ultra-sonographic result, and PE was radiologically diagnosed by computerized tomographic pulmonary angiography (CTA). Patients were excluded if they: had superficial venous thrombosis, solid cancer diagnosis was not confirmed, diagnosed of VTE 1 year prior to first cancer diagnosis, 3 had thrombosis resulting from direct tumor invasion.
Statistical Analysis
The data was summarized using standard descriptive statistics for demographic characteristics and clinical variables. Incidence was calculated as the proportion of patients developing VTE or DVT or PE in each year or the 10-year studying period. The denominator was the total number of solid cancer inpatients for the specified time period. In analyses, we stratified for BMI (<18, 18∼25, 25∼30, ≥30), Eastern Cooperative Oncology Group-Performance Status (ECOG-PS) scores (0∼1, 2, 3∼4). Miss value was filled with interpolation method. Chi-square or Fisher test was used to calculate the statistical differences among categorical variable. A
Results
Annual Incidence of Venous Thromboembolism in Cancer Patients
Among 45,315 solid cancer patients admitted to the Cancer Center, Union Hospital, Tongji Medical College, HUST, between January 1st, 2008 and December 31th, 2017, 811 cases (1.79%) were diagnosed with VTE during their hospitalization period. Among these patients, 776 cases (1.71%) were diagnosed with DVT and 103 patients were (0.23%) diagnosed with PE (including 66 cases complicated with DVT and 37 cases without).
The annual incidence of cancer-associated VTE (CAT) in our cancer center varied between 1.62% in 2015 and 0.35% in 2009, with an overall average incidence of 1.30% over the research decade. The incidence rate of VTE showed a general ascending trend from 2008 to 2015, but revealed a descending trend from 2016 to 2017. Incidence rates of both DVT and PE showed similar trend as that of VTE.(Supplementary Figure 1) Annual incidences of VTE in non-cancer patients ranged from 0.75% to 0.99% with an overall incidence rate at 0.91%. Patients hospitalized with solid malignancies had twice the incidence of VTE as in patients without cancer.
Incidence of Venous Thromboembolism by Subtype of Cancer
Patients with lung cancer, cervical cancer, colorectal carcinoma, breast cancer and lymphoma accounted for 32.4%, 9.3%, 8.6%, 8.4%, and 6.4% of all VTE patients respectively (Figure 1A).

Clinical features of VTE: A. composition of cancer subtypes inVTE patients with malignancy. clinical features of VTE: B The blue ellipse represented DVT developed in the upper limbs, the yellow ellipse represented DVT developed in the lower limbs, the green ellipse represented DVT developed in the splanchnic veins and the red ellipse represented DVT developed in the neck or thorax. The overlap area represented that the site of DVT located in 2 anatomic parts of the body. (ULDVT: upper limb DVT, LLDVT: lower limb DVT, SPDVT: splanchnic DVT, N&T DVT: neck and thoracic DVT).
The incidence rate of VTE of the most common cancer were shown in Table 1. According to the VTE incidence rate, the cancers were divided into 3 groups. No statistical difference was found within each group, while there was a prominent difference (p < 0.01) between different risk groups. A relatively high risk (4.3%) of developing VTE was found in patients with cancers of pancreas, bladder, uterus endothelial and ovary cancer were featured with relatively high risk. Lung, testis, colorectal, cervical, stomach, cervical, breast, prostatic cancers and lymphoma were found to have a moderate risk (2.3%) of VTE development. Low risk (1.1%) was observed in patients with breast, esophagus, hepatic carcinoma and brain cancer. (Table 2)
Site distribution, PE and Central Venous Catheter (CVC) of cancer patients With DVT diagnosis.

Incidence Rates of VTE in Several Common Types of Solid Cancer.
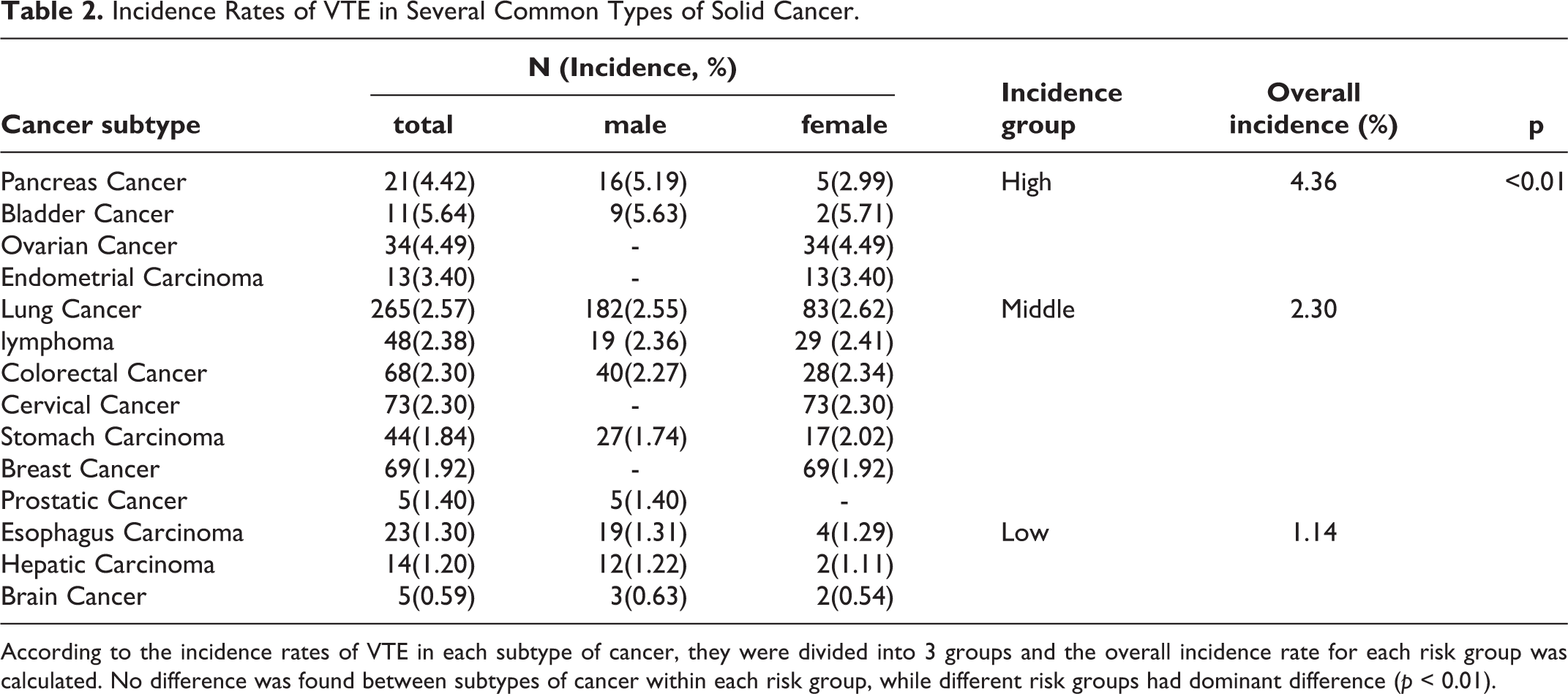
According to the incidence rates of VTE in each subtype of cancer, they were divided into 3 groups and the overall incidence rate for each risk group was calculated. No difference was found between subtypes of cancer within each risk group, while different risk groups had dominant difference (
Epidemiology of Venous Thromboembolism in Cancer Patients
Among all the 811 VTE patients complicated with cancer, 393 were female (1.9%) and 418 were male (1.7%) with no statistical difference between the 2 sexes (χ2 = 0.69, p = 0.41). The mean age at onset of VTE was 54.8 ± 11.5 years old.
The average Body Mass Index (BMI) of patients was 20.8 ± 3.3 kg/m2. The lowest BMI value was 13.8 kg/m2 and the highest 33.3 kg/m2. A majority of patients (67.7%) had a BMI within the normal range of 18 to 25 kg/m2. Only 3.1% of the patients were obese or with a BMI over 30 kg/m2. (Table 3)
Summary of Demographic and Clinical Characteristics of Patients at Time of Venous Thromboembolism (VTE) Diagnosis, and Therapeutic Treatment of Patients Before VTE Diagnosis. All the Anti-Cancer Therapies were Within 6 Month Before the VTE Diagnosis, the Supportive Treatment were within 1 Month Before the VTE Diagnosis.

747 (92.1%) patients had ECOG-PS score ≤ 2, and only 64 of them (7.9%) couldn’t live on their own with ECOG PS score more than 2. The TNM classification distribution of cancer patients with VTE was 6.7%, 12.6%, 20.0%, 59.3% for stage I, II, III, IV respectively. (Table 3)
44 patients (5.4%) were diagnosed with VTE before their diagnosis of malignancy. VTE occurred in 392 patients (48.3%) simultaneously or within 3 months of their initial diagnosis of cancer. There were 91(11.2%), 79(9.7%), 89(11.0%), and 75(9.3%) patients diagnosed with VTE between 3 to 6 months, 6 months to 1 year, 1 to 2 years, 2 to 5 years of cancer diagnosis. Only 41(5.1%) cases had VTE onset 5 years after their first cancer diagnosis. (Table 3)
Therapeutic Treatment of Cancer Patients With Venous Thromboembolism
Two-thirds of patients (n = 544) had received chemotherapy within 6 months prior to the diagnosis of VTE. Among these patients, 41.8% (n = 339) of them were on a platinum-containing regimen, 18.7% (n = 152) on the taxane-containing regimen, 14.1% (n = 114) on pyrimidine derivatives, 12.8% (n = 104) on cytosine derivatives, and 11.3% (n = 92) on alkylating agent regimen. 102 patients (12.6%) were being treated with or had received target therapy within half year before the VTE confirmation. 6.7% (n = 54) of them were treated with anti-VEGF agents. 44(5.4%) were on anti-EGFR/TKI containing regime. Only 26.6% of the patients (n = 216) were under radiotherapy 6 months before the diagnosis of VTE. Postoperative patients were at a high risk of VTE 6 months after surgery, and accounted for 34.8% of patients with VTE. (Table 3)
Clinical Characteristics of Venous Thromboembolism
Of the 811 VTE patients, 708(87.3%) developed only DVT without PE, 35(4.3%) developed PE without DVT, and 68(8.4%) developed both DVT and PE. Among the 776 patients with DVT, the most commonly affected sites were the upper extremities (47.2%), lower extremities (36.1%), thoracic and neck veins (including jugular vein, superior vena cava and right atrium) (9.0%) and splanchnic veins (2.1%). Due to the network of blood vessels in the body, 34 patients (4.4%) were found to have developed DVT in the upper limb as well as the neck and thoracic veins, 2(0.3%) had developed DVT both in splanchnic, neck and thoracic veins. One patients had developed DVT in the lower extremities and splanchnic veins. Only 7(0.9%) patients were found to have developed DVT in discontinuous veins (6 cases with DVT in both upper and lower extremities, 1 with DVT in lower extremities, thorax and neck veins). (Figure 1B and Table 1)
The incidences of patients with DVT in the lower extremities and splanchnic veins developing PE were 15.4%, 12.5% respectively, which were higher than the risk of PE developing with DVT in other sites (8.8%). The risk of developing PE was lowest in patients with thrombosis of upper extremity (3.5%). Among 366 patients with DVT in the upper extremities, 274(74.9%) had been inserted with a central venous catheter (major peripheral central venous catheter). However, only 39 out of 280 (13.9%) with DVT in the lower extremities were inserted with a central venous catheter. This implied that the major cause of DVT development in the upper extremities might be the insertion of central venous catheter. (Table 1). Upper extremity DVT was more commonly diagnosed in cancer patients, but less likely to cause severe complications than lower extremity DVT.
Discussion
Cancer is a well-recognized high risk factor of VTE, and VTE is the most common complication in cancer patients. Cancer alone is associated with a 4 to 7 fold risk of thrombosis. 8,9 Stein has reported that incidence of VTE in patients hospitalized with common cancer also had twice the incidence of VTE in patients without cancer. Our data was similar to Stein’s result in the United State, the risk of VTE in hospitalized patients with solid cancer (1.79%) was nearly twice in hospitalized patients who were not diagnosed with malignancy (0.91%), and was much lower than the risk as compared with the normal person. The difference might attributed to that control groups in our research and Stein’s research both were hospitalized patients without cancer, who had relatively higher risk of VTE as compared with the general population.
The prevalence of VTE in Chinese cancerous population still lacks large-scale cohort studies, only a single-center retrospective analysis in China has reported the incidence of pulmonary embolism in hospitalized patients with solid malignancies to be 0.27%. 10 Our research intended to provide this data, the incidence of VTE, PE and DVT among the admission solid cancer patients between 2008 and 2017 in our center were 1.79%, 0.23%, 1.71%. As the observed patients in this study were followed up to 2018, it might be appropriate to calculate VTE incidence in events per 1,000 person-years (p-y), for easy comparison of the incidence rate acquired in this research with those in previous studies. The incidence rates calculated in this study was 13 events per 1,000 p-y.
According to data of previous research, VTE prevalence ranged from 8/1000p-y in Denmark between 1997 and 2006 to 58/1000p-y in United Kindom between 2001 and 2011. Our data showed that incidence of VTE in Chinese patients with cancer was at a relative low level. In the United State, incidence was 20/1000p-y according to Stein’s research between 1979 and 1999, however, it decreased to 12/1000p-y between 2005 and 2014, presented a non-significant downward trend and declined by 60% over this period. Although VTE prevalence was relatively low among solid cancer population in our center, the incidence in China had exceeded that in the United State. The upward trend implied that race factor might not be a crucial factor in the thrombosis generation, and VTE occurrence should be regarded as the increasing caner prevalence and the widely used target drug in recent several years.(Supplementary figure 2)
Cancer cells activate the clotting system by releasing procoagulant properties, making it the main cause of hypercoagulability of cancer patients. Rates of VTE varied in different cancer sites, and the largest proportion of VTE patients with cancer did not necessarily have the highest VTE risk. Patients with tumors of pancreas, ovarian and brain cancer are generally reported to be most strongly associated with VTE. 3,11 Digestive system cancers (e.g., stomach, colon rectum), kidney, bladder cancer, and lung adenocarcinoma showed moderate risk of VTE. Relatively low risks were seen in patients with breast or prostate cancer. 12
Most common cancer types bar brain tumor showed similar incidence rates in this research and previous studies. The incidence of VTE in brain tumors especially glioblastoma (GBM) is controversial. Shlomit et al. had reported that the incidence of VTE in GBM patients was as high as 22% in M. D. Anderson Center. 13 Another cohort showed that VTE occurs in 20% of gliomas in Southern Alberta of Canada. 14 In our center, only 6 out of over 800 (0.59%) GBM patients were diagnosed with VTE. This difference might due to racial difference, genetic variation, admission bias and anti-cancer therapies. The admission GBM patients in our center had a relatively high ECOG PS score and their admission were mainly for radiotherapy, so their VTE risk during admission was not as high as the risk during the long-term follow-up.
Obesity is an independent factor for venous thrombosis. 15 A majority of Chinese cancer patients (67.7%) had a normal BMI (18 to 25 kg/m2), and only few patients (3.1%) had a BMI ranging between 30 and 35 kg/m2. None of the patients had a BMI over 35 kg/m2. Nevertheless, less than half of the patients had a BMI below 25 kg/m2, and 23% larger than 30 kg/m2 in westerners 15,16 This could be attributed to the differences in physicality, nutrition status and dietary concept. Our previous study had shown that setting the BMI of tumor patients at 24kg/m2 as a benchmark during anti-cancer therapy could improve the predictive value of Khorana scale. 6 It implied that the current cut-off value of BMI ≥35 kg/m2 in Khorana scale might not suitable for Chinese cancer population. Whether modifying BMI cut-off value could improve the sensitivity and specificity of the existing VTE assessment scales was worthy further prospective study.
Blood stasis and hypercoagulability status is considered more crucial than endothelial damage in the pathogenesis of VTE in cancer patients. 17 It is widely accepted that bed rest and long journey are common reasons for the formation of VTE especially lower limbs DVT. Therefore, bed rest for over 72 hours is an important item in the commonly used venous thromboembolism prediction scores. However, our results demonstrated that 92.1% VTE patients had relative good physical status with ECOG PS equal or less than 2, implying a closer relationship between hypercoagulability of cancer patients and VTE occurrence.
In several cohort studies focusing on cancer patients, the risk of VTE increased significantly with the advancing tumor stage. 11,18 Our cross-sectional study showed that 59.3% of VTE patients with cancer were in stage IV, indicating the relatively higher risk of VTE in advanced cancer patients.
In the previous literature, 1 of 10 persons presenting with idiopathic VTE is diagnosed with cancer afterward within 5 to 10 years’ time, and the diagnosis is confirmed within 1 year in 75% of cases. 3 5.4% of subjects in our study found VTE 1 year before their first diagnosis of cancer. More than half of the patients developed VTE within 6 months after their initial cancer diagnosis, in consistent with Blom and Horsted’s conclusion that risk of VTE was highest in the first 6 months since the diagnosis of malignancy. 19,20 The incidence decreased significantly after 5 years of first tumor diagnosis (5.1%).
The risk of VTE should be increasing gradually as the progress of cancer, contrary to the phenomenon that VTE risk was highest in the first few months after the diagnosis of cancer. There must exist other factors resulting in VTE occurrence. Considering this, we traced back to the anti-cancer therapies half year before VTE diagnosis. Chemotherapy, widely used in 67.1% patients, has been reported to be associated with a 2- to 6-fold increased VTE risk. Although only 12.53% patients had received targeted drugs in this research, oncologists should pay more attention to the potential VTE risk 21 with the gradual popularization of the targeted therapy(Supplementary figure 2).
Supportive therapies also increase the risk of VTE. Erythropoietin (EPO) and G-CSF are reported to increase the risk of VTE by 13-27% and 109% respectively. 22 Synthetic progestogens such as medroxyprogesterone are still recommended for the management of anoxia and weight loss in cancer patients despite their increased risk of VTE. 23 Anemia is very common among cancer patients due to long-lasting chemotherapy, radiotherapy and nutrition consumption by tumor cells. Transfusion of blood cell and administration of EPO are effective methods to ameliorate anemia but also work as risk factors responsible for VTE. 24 Dexamethasone, a drug for relieving vomiting and radiation inflammation, also promoted the development of VTE. 22 Before the palliative treatment, doctors should evaluate the balance between the risk of VTE and the supportive therapeutic effect, as well as prepare for the VTE prevention and treatment plan.
Central venous catheter (CVC) especially peripheral insertion central catheter (PICC) is increasingly used in the management of cancer patients for their convenience in chemotherapy and the protection of blood vessels. Despite of its benefits, insertion of PICC increases the rate of both PICC-related deep vein thrombosis (PRDVT) and distant DVT (lower limb DVT and PE). 25 Nearly 70% of upper extremities DVT were PRDVT while only 15.7% of lower extremity patients had PICC. PICC might be the main cause of upper extremity DVT among cancer patients. Despite of the common occurrence of upper extremity DVT (47.2%), it seldom developed into PE (3.6%). Lower extremities DVT were much closely related with PE (15.0%) as the common cognition. Hence, clinicians should attach more importance to the high rate of PRDVT and the severe consequence of lower extremity DVT.
In conclusion, we have described and analyzed the VTE occurrence among Chinese cancer population. There are several drawbacks in our study. Our study focused on data collected from merely 1 hospital, and we mainly attributed the increase of VTE rate to increase caner prevalence and the widely used target drug. More multi-center, large-population investigations are still needed to substantially affirm the results. Our previous study has evaluated the performance of the Caprini and Padua scores among Chinese hospitalized patients, 6 but the predictability of these scales is still limited. More detailed research on VTE patients in Chinese cancer population is required to develop a more comprehensive and inclusive prediction scale. Surgical instrumentation, anti-cancer therapy and several other factors make inpatients of the oncology department on high risk of VTE. Further investigation is required to be able to distinguish VTE patients precisely from the general cancerous population.
Supplemental Material
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620975484 - Solid Tumor Complicated With Venous Thromboembolism: A 10-Year Retrospective Cross-Sectional Study
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620975484 for Solid Tumor Complicated With Venous Thromboembolism: A 10-Year Retrospective Cross-Sectional Study by Miao Peng, Shengli Yang, Guiling Li, Tao Zhang, Xiaojuan Qin, Chen Shi, Jian Chang, Mengni Chen, Chen Chen, Bingjie Li, Sihang Cao, Ting Li, Renwang Chen, Prapti Bakhshi, Min Jin, Gang Wu and Jianli Hu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors Note
Miao Peng and Shengli Yang contributed equally to this paper. They analyzed, interpreted data and wrote the paper. Guiling Li, Tao Zhang, Xiaojuan Qin and Chen Shi provided raw data. Jian Chang, Mengni Chen, Chen Chen, Bingjie Li, Sihang Cao, Ting Li and Renwang Chen performed data collection. Prapti Bakhshi and Min Jin revised the paper. Gang Wu and Jianli Hu designed and conducted the research and revised the paper. All authors approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by undergraduate innovation and entrepreneurship training program of Hubei province (S202010487043 and S202010487044) and Huazhong University of science and technology undergraduate innovation and entrepreneurship training program (2019A0036, DYLC2020040 and DYLC 2020050).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
