Abstract
Despite the existence of antithrombotic guidelines, there is low compliance with these guidelines in clinical practice. Until now pharmacy interns (PIs) have not been involved in this process. The objectives were to involve PIs to evaluate compliance with antithrombotic guidelines for VTE prophylaxis in surgery patients, and in cases of noncompliance to carry out pharmaceutical interventions. The study was conducted in 7 hospitals in Germany involving 27 PIs within the project “Pharmacy interns on the ward” (P-STAT 2). Pharmacy interns determined the thromboembolic risk, documented antithrombotic medication, and checked the compliance with current antithrombotic guidelines. A total of 6491 patients were enrolled; 5695 patients received antithrombotic prophylaxis. Antithrombotic guideline was followed in 77.5% patients. Many patients are not receiving appropriate VTE prophylaxis or heparin bridging regimen despite the fact that evidence-based antithrombotic guidelines are available. Pharmacy interns may play an important role in antithrombotic management.
Introduction
Venous thromboembolism (VTE) is an important health care problem. Almost all hospitalized patients have at least 1 risk factor for VTE, and approximately 40% have 3 or more risk factors, for example surgery, immobility, increasing age, or obesity. Without thromboprophylaxis, the incidence of hospital-acquired deep vein thrombosis (DVT) is approximately 10% to 40% among medical or general surgical patients and 40% to 60% following major orthopedic surgery. Mortality, acute and long-term morbidities, and resource utilization related to unprevented VTE strongly support effective preventive strategies at least for moderate-risk and high-risk patients. There is sufficient evidence to recommend routine thromboprophylaxis for most hospitalized patient groups. Implementation of evidence-based thromboprophylaxis strategies provides benefit to patients. 1 Unfortunately, despite several randomized clinical trials demonstrating the benefit of thromboprophylaxis and practice guidelines recommending the use of thromboprophylaxis, there is low compliance of evidence-based thromboprophylaxis guidelines and their application in clinical practice. 1–4
Bridging therapy with low-molecular-weight heparin (LMWH) or unfractionated heparin (UFH) is usually recommended in patients who have to interrupt their medication with oral anticoagulants before surgical procedures. The rationale for bridging anticoagulation is to minimize the time before and after a procedure that patients are not receiving therapeutic anticoagulation and, thereby, minimize the risk of thromboembolism. 5 The interruption of oral anticoagulants minimizes the risk of bleeding, but exposes patients to an elevated thromboembolic risk. Hence, bridging therapy is essential and is accomplished either by treating hospitalized patients with intravenous UFH or by administering subcutaneous LMWH. 6
Clinical pharmacists can play an important role in the health care team. Since 2001 when the revised License Ordinance for Pharmacists came into effect in Germany, pharmacy interns (PIs) have the opportunity to spend part of their mandatory internship on hospital wards. Feasibility of PIs on hospital wards in Germany as well as high acceptance by physicians and nurses were shown in a pivotal study 7 in 2002. To our knowledge, PIs have not been involved in monitoring and improving the compliance with antithrombotic guidelines on hospital wards. Pharmacy interns are highly qualified although they are lacking practical experience. Therefore, supervision by an experienced clinical pharmacist is needed. Involving PIs gives them a chance to gain practical experience. Furthermore, personnel expenditures are low, this being a chance of involving qualified staff in improving quality in a key process.
The work presented here was part of the German multicenter study “pharmacy interns on the ward (P-STAT 2)”. In this study, PIs were also involved in medication reconciliation, identifying, resolving, and preventing drug-related problems, and evaluating the adherence to guidelines for antibiotic prophylaxis in surgery patients.
The overall objective of this study was to evaluate the feasibility of involving PIs in monitoring and improving compliance to antithrombotic guidelines.
Further objectives in terms of antithrombotic guidelines were to evaluate the following issues in surgery patients in German hospitals by PIs Thromboembolic risk of patients, Compliance with antithrombotic guidelines for prophylaxis of venous thromboembolism or heparin-bridging regimens, Pharmaceutical interventions in patients of noncompliance with antithrombotic guidelines, Patient without VTE prophylaxis, and Thromboembolic or bleeding events.
Materials and Methods
Setting
The study was conducted in 7 hospitals in Germany (5 community hospitals and 2 university hospitals) on 14 different wards. The clinics of the participating wards were general and visceral surgery (n = 5), gynecology (n = 1), orthopedic and accident surgery (n = 6), otolaryngology (n = 1), and urology (n = 1).
Recommendations for VTE prophylaxis and heparin-bridging regimens according to the current antithrombotic guidelines of each hospital ward were postulated for participating and checked by the study coordinator (Carina Hohmann) before initiation of the study.
Pharmacy Interns
Twenty-seven PIs (92.6% female) spent their 6-month mandatory internship on a hospital ward either from May to October 2008 or November 2008 to April 2009 within the project “Pharmacy interns on the ward” (P-STAT 2). 8 The objective of this project was to encourage patient-oriented education of PIs on wards. It is hypothesized that their work will result in an improvement in quality and safety in the medication use process. One part of this study was the evaluation of the VTE prophylaxis among others.
Each hospital pharmacy (n = 5) or hospital supplying community pharmacy (n = 2) attended 4 PIs in their internship (2 PIs every half year). One of the originally included PIs dropped out before the internship started, so that 1 pharmacy was supervising only 3 PIs.
Before study onset, both PIs and principal pharmacists were trained for 2 days in pharmaceutical care, medication reconciliation, and antithrombotic guidelines. Pharmacy interns were supervised by pharmacist with experience in clinical pharmacy practice.
Patients
Patients admitted to the hospital wards were consecutively recruited from June 2008 to April 2009. Demographic data, main diagnosis, indication for surgery, comorbidities, and renal function were documented. The level of thromboembolic risk (low, moderate, high risk) was determined according to the individual patient’s risk factors using the current German VTE guideline. 9 Furthermore, LMWH or UFH and the dosage of the heparin as well as renal function were documented from the medical records. Patients who did not receive any VTE prophylaxis during the hospital visit were documented as well. Compliance with the antithrombotic guideline was checked, considering both prescribed heparin, dosing regimen, thromboembolic risk, and renal function. Compliance with the guideline was defined as the consistency of the prescribed heparin with the standard implemented on the respective hospital ward. Every deviation of prescribed heparin, dosing regimen, or disregarded renal function was defined as noncompliance with antithrombotic guidelines. Pharmacy interns were instructed to intervene in cases of noncompliance with the existing guideline. Pharmacy interns worked under the responsibility and close supervision of a clinical pharmacist. After providing a pharmaceutical intervention, the adoption was checked by the PI.
All thromboembolic events such as DVT, pulmonary embolism (PE), ischemic stroke, and bleeding events were documented.
All data were checked for face validity by 2 clinical pharmacists with experience in thrombosis research and clinical pharmacy practice on the basis of the available documented patient data, main diagnosis, or operative indication, secondary diagnoses, and the thromboembolic risk.
Due the fact that this study was no prospective interventional study, and patient anonymity was ensured so that no patient-specific data was recorded; therefore, institutional review board approval was waived.
Statistical Analyses
Statistical analysis was performed using PASW 18 (Predictive Analytics Software, SPSS Inc., Chicago, IL, USA). Data are given as means, standard deviation, median, and interquartile range.
Results
Patient Population
From June 2008 to April 2009, a total of 6491 patients were enrolled for the evaluation of VTE prophylaxis or heparin-bridging regimens from 14 hospital wards. The patients’ characteristics and renal function are given in Table 1 . Average age of the patients was 58.3 ± 19.7 years; 52.4% were female.
Patients’ Characteristics Including Renal Function
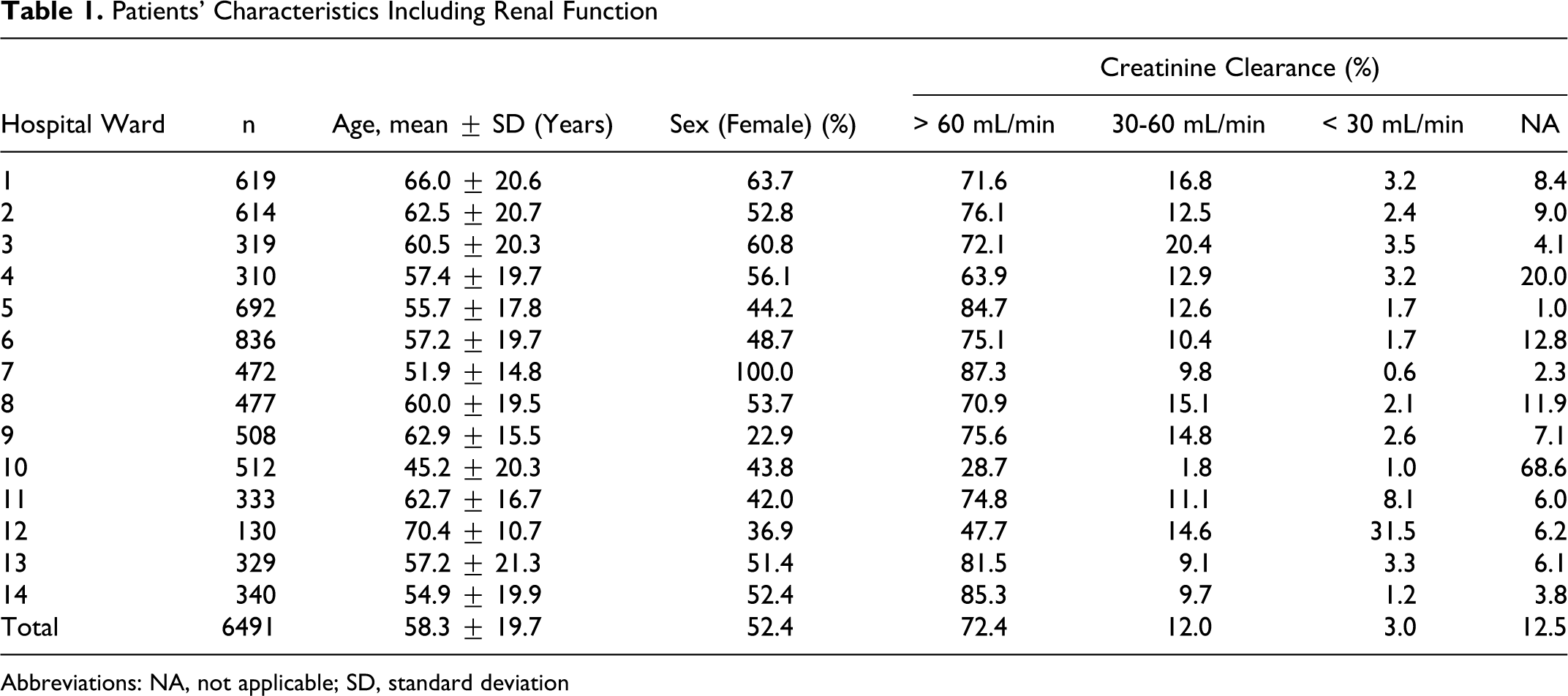
Abbreviations: NA, not applicable; SD, standard deviation
Thromboembolic Risk
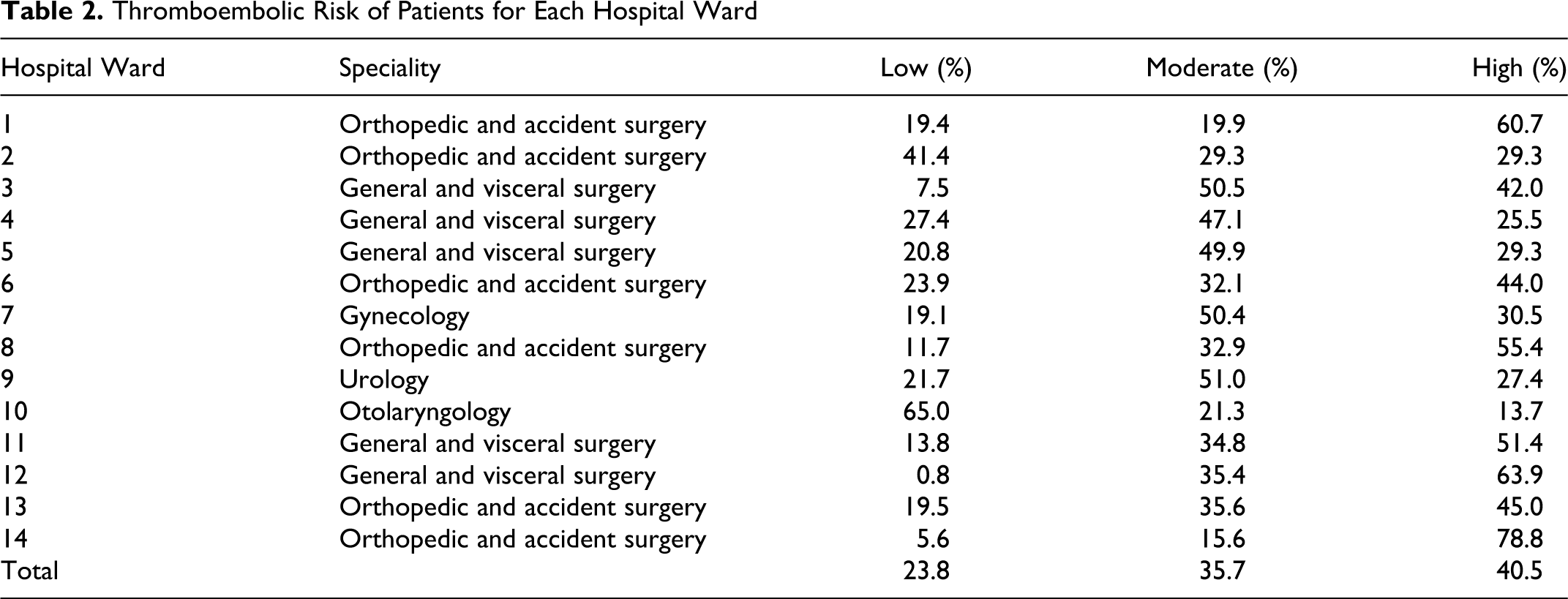
In total, 23.8% of patients had a low, 35.7% of patients had a moderate, and 40.5% of patients had a high thromboembolic risk (see Table 2 ). Patients with high thromboembolic risk were mainly in hospital wards with orthopedic and accident surgery and general and visceral surgery.
Thromboembolic Risk of Patients for Each Hospital Ward
Compliance With Antithrombotic Guidelines
Of the 6491 patients enrolled, 5695 received antithrombotic prophylaxis or heparin-bridging regimens and were available for evaluation. Results are summarized in Figure 1 . Antithrombotic guidelines were followed in 77.5% (interquartile range [IQR] 63.5-86.3) of the documented cases with a minimum of 43.8% (hospital ward 7) and a maximum of 95.7% (hospital ward 14).
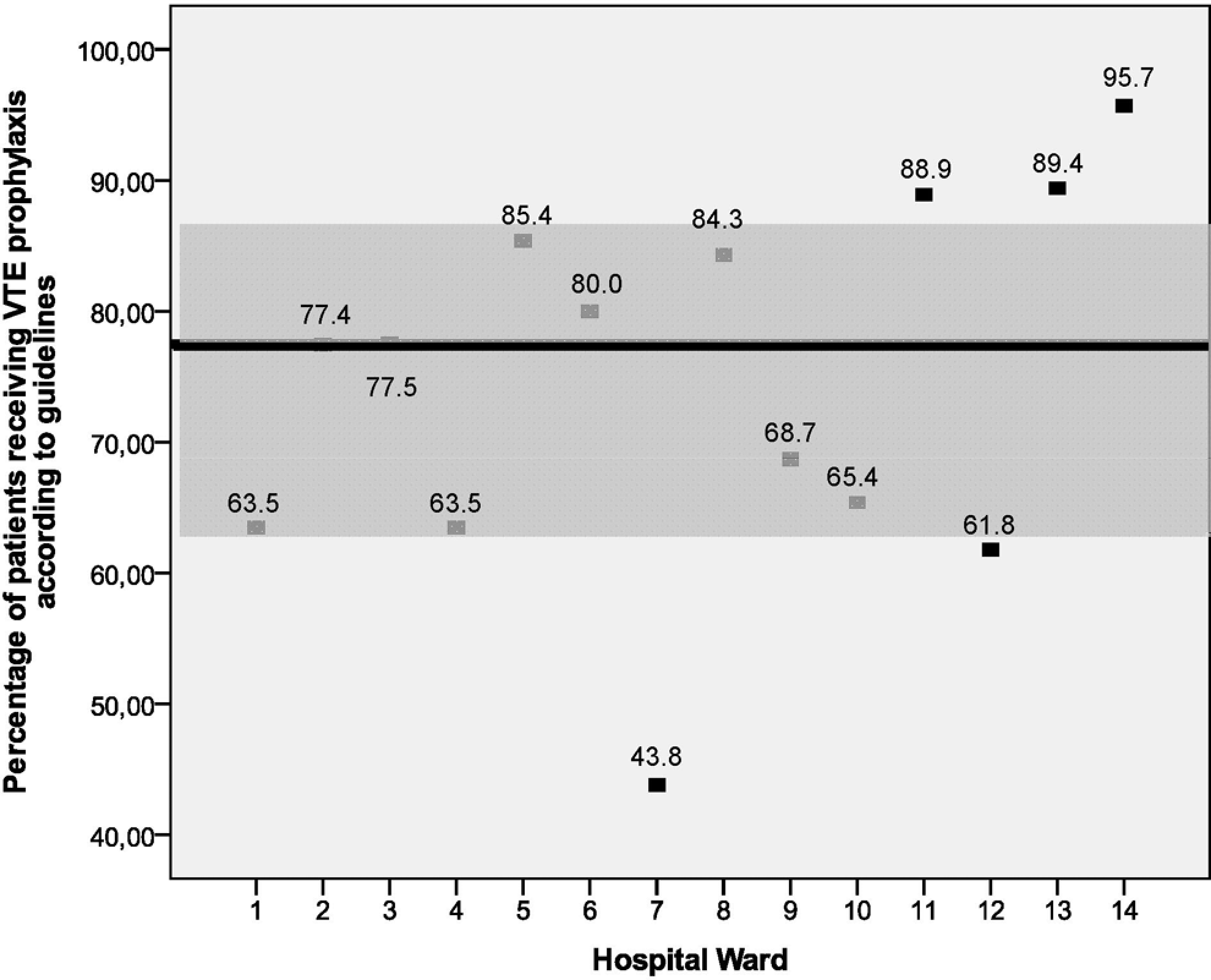
Compilance with antithrombotic guidelines for each hospital ward (black line: median of all hospital wards; gray range: interquartile range of all hospital wards; black holes: median of each hospital wards; in %).
Pharmaceutical Interventions in Patients of Noncompliance
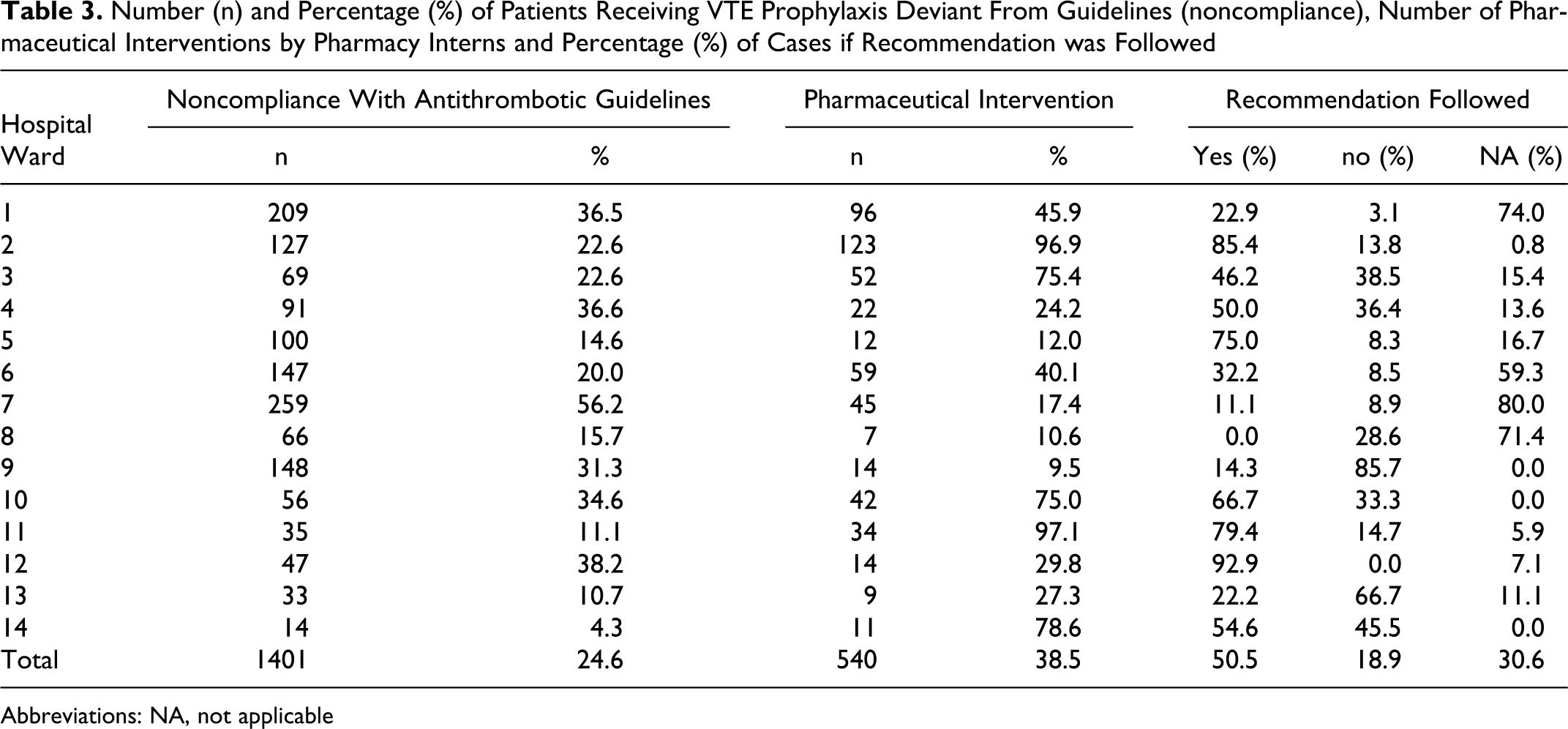
In patients of noncompliance with treatment guidelines, the PIs were supposed to carry out a pharmaceutical intervention to the attending physician. The VTE prophylaxis or heparin-bridging regimen was noncompliant with the guideline in 1401 (24.6%) patients. In 540 (38.5%) of these patients, a pharmaceutical intervention by the PI took place. In 50.5% of the intervention, the recommendations for a modified regime were followed, whereas the recommendations were not followed in 102 (18.9%) patients. No data were available for 165 (30.6%) patients. Detailed results for all hospital wards are given in Table 3 .
Number (n) and Percentage (%) of Patients Receiving VTE Prophylaxis Deviant From Guidelines (noncompliance), Number of Pharmaceutical Interventions by Pharmacy Interns and Percentage (%) of Cases if Recommendation was Followed
Abbreviations: NA, not applicable
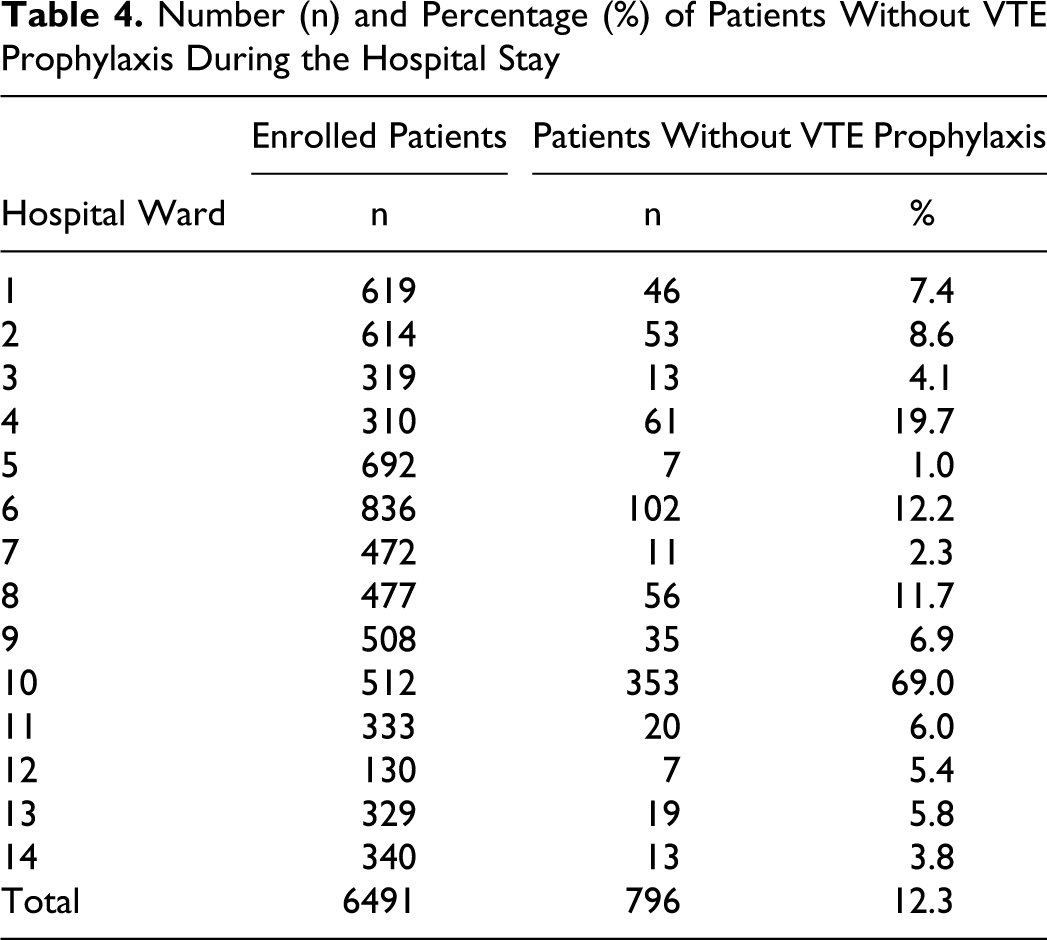
Patients Without VTE Prophylaxis
Of the 6491 enrolled patients, 769 did not receive VTE prophylaxis or heparin-bridging regimen (see Table 4 ).
Number (n) and Percentage (%) of Patients Without VTE Prophylaxis During the Hospital Stay
Thromboembolic and Bleeding Events
Thirty-six thromboembolic (0.55%) and 3 bleeding events (0.05%) were recorded during the hospital stay. Thromboembolic events were ischemic strokes (n = 9), DVT (n = 8), PE (n = 7), and others (n = 16), and 85% of them occurred in patients with high-thromboembolic risk. 38.5% (n = 15) thromboembolic and bleeding events occurred in patients that did not receive LMWH or UFH, according to the implemented guidelines.
Discussion
To our knowledge, this is the first study investigating the compliance with antithrombotic guidelines in surgery patients involving PIs.
The results of our study show that PIs under the supervision of a clinical pharmacist can make a valuable contribution toward a quality-assured antithrombotic management. Pharmacy interns rated the patients’ thromboembolic risk on the basis of diagnosis, duration of the surgery, comorbidities, and individual risk factors of the patient. Furthermore, PIs documented antithrombotic prophylaxis or bridging regimens utilizing medical records. Accordingly, they evaluated VTE prophylaxis and heparin-bridging regimens with antithrombotic guidelines to monitor compliance with the guidelines in daily clinical practice. Pharmacy interns made interventions recommending a change in drug or modification in dosage. Recommendations, however, were not always followed. This might be due to the lack of advanced clinical experience, knowledge, and personal authority of these young, most often female graduates.
Compliance with antithrombotic guidelines ranged from 43.8% up to 95.7% on the participating wards. The high compliance rate in some hospital wards show that the physicians are familiar with the guidelines, aware of the thromboembolic risk, and profit from a reminder system. However, there are many wards with rather low compliance. Most common reasons for inadequate prophylaxis were for example, lack of prophylaxis, incorrect type or dosage of prophylaxis. This may be caused by lack of awareness, less familiarity with guidelines, disagreement with guidelines, forgetfulness, overestimation of bleeding risk, underestimation of thromboembolic risk, and lack of a sufficient reminder system. We should mention that the treatment according to the guidelines is not suitable for all patients; in some cases the determination of treatment by the attending physician was based on a specific condition or circumstances of the patient, so that a drift from the guideline may be necessary. There is a significant gap between the availability of evidence-based guidelines and their application, resulting in a wide variation in VTE prescribing patterns. 4,10 Published 2,3,10–12 data have shown variable compliance rates with antithrombotic guidelines from 13.3% up to 89%.
To increase the compliance rate, several strategies are feasible, for example education (pocket cards, meetings), system support (clinical decision-support tools, risk assessment tools, computer reminder system), and process support (working groups to identify barriers). 4
The results of our study are beneficial for both the attending clinical pharmacist and the head physician of each hospital ward and clinic, respectively, to identify the weaknesses of the current antithrombotic guidelines and their clinical examination.
Both, frequency of pharmaceutical interventions by PIs in cases of noncompliance and the frequency of recommendations being followed by the attending physician, differ between the participating wards. The percentage of pharmaceutical interventions varied between 9.5% to 97.1% and the percentage of followed recommendations ranged from 0.0% to 92.9%. These results indicate that PIs can contribute in improving the compliance rate with antithrombotic guidelines, but the extent may depend on their knowledge and their acceptance in the medical team. Furthermore, pharmaceutical interventions by PIs are feasible in patients who are following “standard cases,” but interventions seem less feasible, for example, in patients with an extensive medical history, renal failure, a high risk of thromboembolic and bleeding. Therefore, support from clinical pharmacists with advanced experience in clinical pharmacy practice is desirable.
The number of patients without VTE prophylaxis varied highly between wards with different thromboembolic risk.
The number of thromboembolic and bleeding events was very low in our study. A number of recent studies have reported thromboembolic events
13–15
up to 2.5% and bleeding events
1,13,16
up to 6.3%. The low rate of thromboembloic events may be caused by the lack of a systematic evaluation of thromboembolic events such as venous ultrasound scanning,
Involving PIs in monitoring and improving compliance to antithrombotic guidelines is feasible and PIs may play an important role in the quality control of antithrombotic management. Considering this and the observation that compliance with antithrombotic guideline was often not sufficient, it might be an option to involve more PIs in this field in the future, since they are highly qualified and their salary costs are still low. Another advantage would be that involving PIs in a multidisciplinary team on a hospital ward gives them the opportunity to gain valuable clinical experience.
Due to the fact that antithrombotic management was only a part of the study the burden of work for the PIs was very high. Therefore, some issues were not evaluated although it would have been interesting, for example compliance with antithrombotic guidelines in patients not receiving any prophylaxis.
Limitations
Data were gathered from different hospital wards with deviant preconditions; some hospital wards and their medical teams have never been consulted by a clinical pharmacist on the ward before; therefore, the physicians were not familiar with receiving recommendations by a clinical pharmacist. In other cases, the ward and the physicians were used to get advice and recommendations by the clinical pharmacist. In those cases, PIs were much easily integrated into the medical team and the rate of followed recommendations was much higher.
Furthermore, there was no data available on the number of patients wearing elastic stockings for VTE prophylaxis, which can influence the number of thromboembolic events. In addition, there were no data available for evaluating the proportion of patients not receiving VTE prophylaxes during the hospital stay regarding justification or compliance with the guideline. Referring to this we have a gap in our process of evaluating VTE prophylaxis. However, we mentioned that the evaluation of the compliance with the antithrombotic guidelines was only one aspect of our project.
In conclusion, many patients are not receiving appropriate VTE prophylaxis or heparin-bridging regimens on hospital wards despite the fact that evidence-based antithrombotic guidelines are available. Pharmacy interns may play an important role in the antithrombotic management to monitor the status of the wards’ compliance with existing guidelines. However, a variety of limitations have to be considered in achieving a quality-assured process eventually.
Footnotes
Acknowledgments
We thank all participating pharmacies, Dom-Apotheke, Essen; Asklepios Kliniken Hamburg GmbH; Waisenhaus-Apotheke, Halle; Universitätsklinikum Heidelberg; Klinikum Fulda gAG; Klinikum rechts der Isar der TU München; and Dr.-Horst-Schmidt-Kliniken GmbH, Wiesbaden (Germany) and all pharmacy interns for their valuable contributions. We thank Christiane Sauerwein for generating the database and her contribution to study analyses, and Susanne vom Scheidt, both ZAPP (ABDA, Berlin) for her support in developing and running the study.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financed by the Federal Union of German Associations of Pharmacists (ADBA), Berlin, and the foundation “Dr. August und Dr. Anni Lesmüller-Stiftung”, Munich, Germany.
