Abstract
Total joint arthroplasty (TJA) improves the quality of life for patients with end-stage osteoarthritis but is associated with an increased risk of venous thromboembolism (VTE), thus pharmacologic thromboprophylaxis is recommended for most patients. Patients with congenital bleeding disorders may develop severe arthropathies due to repeated hemarthroses and derive similar benefit from TJA as the general population. No guidelines for pharmacologic thromboprophylaxis in this population exist, however, as the risks and benefits are not well defined. We undertook the current study to assess the safety and efficacy of pharmacologic VTE prophylaxis in patients with congenital bleeding disorders undergoing TJA. We retrospectively reviewed the medical records of patients with bleeding disorders who underwent TJA at our academic institution between 1987 and 2012. We identified 28 patients who underwent 38 TJA procedures. Low-molecular-weight heparin (LMWH) was administered in 29 procedures (76%) and was discontinued early in 3 procedures (2 patients) due to nonjoint bleeding. No symptomatic VTE was identified, and no joint or deep wound infections were seen. Twenty-two patients accounting for 31 procedures were contacted to discuss their experience with TJA. All reported decreased pain, and 97% reported improved function after the surgery. Impressively, 97% stated that they would choose to have the surgery again. These results confirm the benefit of TJA in patients with congenital bleeding disorders and end-stage arthropathies and suggest that LMWH thromboprophylaxis is safe. No patient in our cohort developed symptomatic VTE, whether or not thromboprophylaxis was administered, thus necessity of thromboprophylaxis remains an unanswered question.
Introduction
Joint pain and dysfunction due to advanced arthropathy may be a source of morbidity for people with bleeding disorders, especially in patients with hemophilia. Repeated bleeding into the joint (hemarthrosis) leads to hypertrophy of synovial tissue and results in recurrent bleeding due to the friability and hyperemia of the new synovium. Ultimately, this repetitive process leads to degenerative arthritis and end-stage arthropathy. 1 While prophylaxis with coagulation factor replacement therapy reduces the incidence and severity of arthropathy in children with hemophilia, 2 a need for joint replacement in adults still exists. 3
Joint replacement surgery improves function, relieves symptoms, and enhances quality of life for patients with hip and knee arthropathies. 4,5 While total joint arthroplasty (TJA) carries substantial bleeding risk, the procedure is also associated with increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE), collectively known as venous thromboembolism (VTE), and extended postoperative pharmacologic thromboprophylaxis is recommended for most patients. 6 Patients with bleeding disorders present a unique challenge with respect to postoperative thromboprophylaxis. On one hand, these patients are at risk of bleeding due to the nature of their hemostatic disorder but, on the other hand, surgery is generally performed only after normal hemostatic potential is restored with the use of coagulation factor concentrates. The net effect, presumably, is to render these patients at average risk of VTE and therefore candidates for perioperative thromboprophylaxis. However, whether or not pharmacologic thromboprophylaxis is beneficial for patients with bleeding disorders undergoing major orthopedic surgery is an unanswered question.
Dartmouth-Hitchcock Medical Center houses the only federally funded Hemophilia Treatment Center in New Hampshire and maintains an active joint replacement program for patients with bleeding disorders. Our clinical practice has been to ensure normal hemostasis using coagulation factor concentrates perioperatively, and since 2000, we have administered pharmacologic thromboprophylaxis concurrently to prevent VTE. As the usefulness of this practice is unknown, we undertook the current study to assess the safety and efficacy of perioperative pharmacologic VTE prophylaxis in patients with congenital bleeding disorders undergoing joint replacement surgery. A secondary goal of the study was to assess functional improvement and patient satisfaction with the procedure.
Materials and Methods
We performed a retrospective chart review, using a protocol approved by the Dartmouth College Committee for the Protection of Human Subjects, to identify patients with congenital bleeding disorders who underwent TJA between 1987 and 2012. All patients were treated with coagulation factor concentrates and other hemostatic agents appropriate to their disorders for at least 4 and up to 6 weeks postoperatively per institutional practice. Coagulation factor concentrates were administered perioperatively according to World Federation of Hemophilia guidelines. 7 Coagulation factor levels were measured in platelet-poor plasma in the hospital special coagulation laboratory using standard automated protocols. Although coagulation factor levels were not measured routinely beyond the inpatient period of the hospitalization, coagulation factor concentrate doses were targeted to achieve a peak factor level of 100% for the surgery, then 80% to 100% through the first postoperative day, 60% to 80% through day 3, 40% to 60% through day 14, and 30% to 50% through the remainder of the first 28 days. Thereafter, coagulation factor infusions were administered prior to physical therapy sessions and as needed. Coagulation factor assays were performed on a STA-R or STA-R Evolution automated coagulation analyzer (Diagnostica Stago, Inc, Parsippany, New Jersey) using clot-based assays for factors VIII, IX, and XI (STA PTT reagent and STA deficient plasmas, Diagnostica Stago) and an immunoturbidimetric assay (STA Liatest, Diagnostic Stago) for von Willebrand factor antigen levels. Low-molecular-weight heparin (LMWH) thromboprophylaxis, when used, was initiated either preoperatively or on postoperative day 1 with the intent to continue through postoperative day 28. One patient with factor XI deficiency and an inhibitor was treated with epsilon aminocaproic acid and recombinant factor VIIa infusions preoperatively. Postoperative hemostasis in this patient was supported with recombinant factor VIIa infusions at 4- to 12-hour intervals during the first postoperative day and with oral tranexamic acid concurrently and for 2 weeks postoperatively. Low-molecular-weight heparin was administered for 2 weeks rather than the full 28 days for this patient. We collected demographic information, thromboprophylaxis data, range of motion (ROM) measurements, and reported pain before and after surgery, and we assessed early (<30 days) and late complications (bleeding, infection, symptomatic thrombosis). We used a 2-sided Student t test to evaluate differences in ROM pre- and postoperatively. Finally, we contacted patients to assess retrospectively their satisfaction with the surgery by soliciting answers to 3 yes or no questions: (1) Do you have less pain in the affected joints after compared to before surgery; (2) do you have improved function of the affected joint(s) after compared to before surgery; and (3) if you could do it over, would you have the surgery again? Data are presented descriptively using median values and ranges where appropriate.
Results
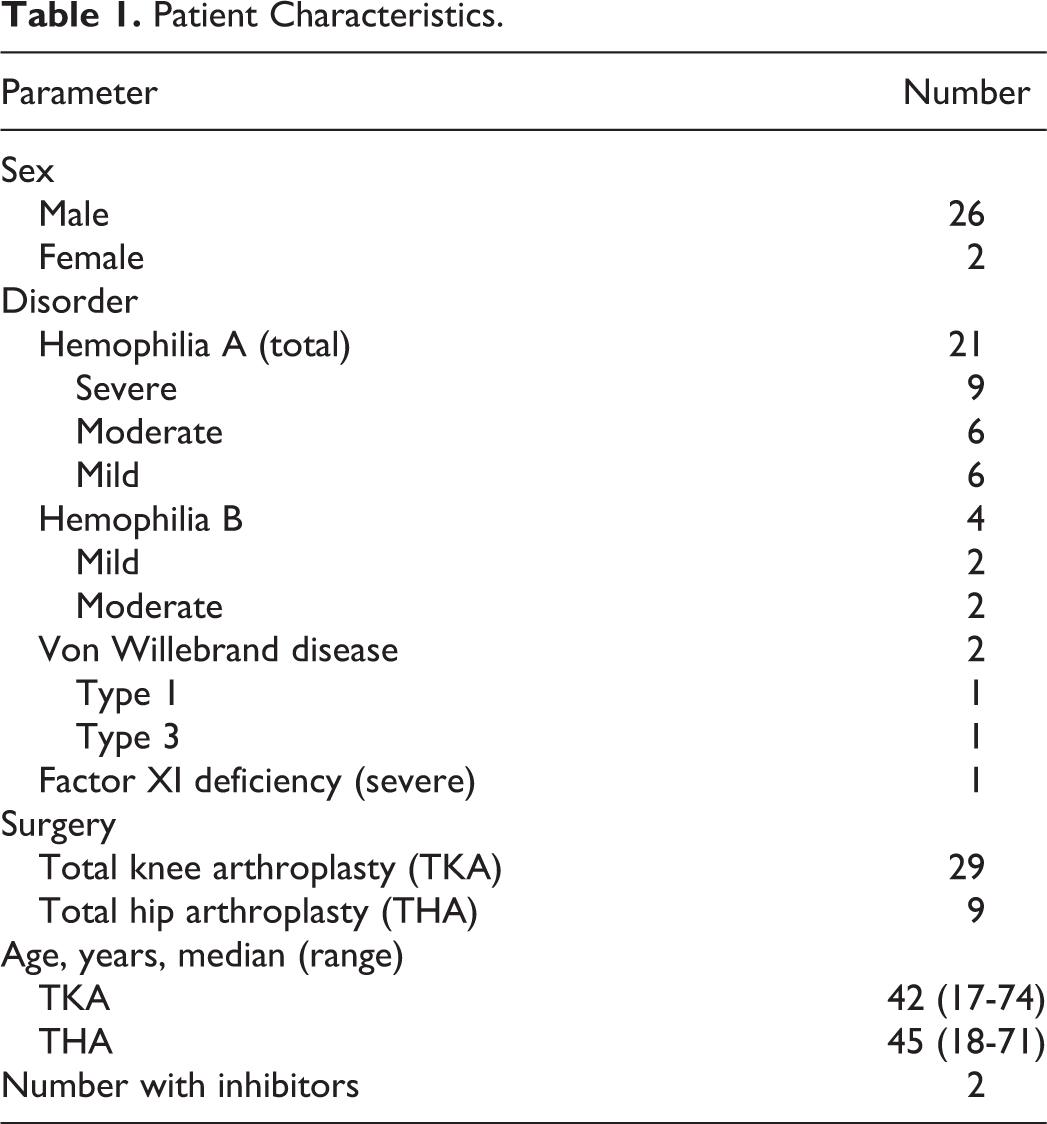
We identified 38 procedures involving 29 knees (total knee arthroplasty [TKA]) and 9 hips (total hip arthroplasty [THA]) in 28 patients with congenital bleeding disorders. Patient characteristics are summarized in Table 1. Males with hemophilia A constituted the majority of TJA cases, and inhibitors were present in 1 patient with hemophilia A (1.5 BU) and 1 patient with factor XI deficiency (0.5 BU).
Patient Characteristics.
We documented the use of at least 1 dose of prophylactic LMWH postoperatively in 29 (76%) of 38 procedures. Seven procedures, all performed in 2000 or earlier, were conducted without the use of pharmacologic thromboprophylaxis. The medical records were incomplete in 2 procedures, and we could not determine whether or not thromboprophylaxis was used in these cases. Enoxaparin was used in all cases in which thromboprophylaxis was administered. Details of anticoagulant dosing and duration are summarized in Table 2. Thirteen of 29 procedures were associated with a full 4-week thromboprophylaxis course and 3 with a 2- or 3-week course. Thromboprophylaxis was administered during the inpatient hospitalization with the intent to administer for 4 weeks for 13 procedures; however, adherence to the outpatient regimen could not be confirmed.
Anticoagulant Dose and Duration.

Abbreviations: q 24 hours, every 24 hours; q 12 hours, every 12 hours.
The main bleeding and thrombosis outcomes are summarized in Table 3. Thromboprophylaxis was permanently discontinued in 3 procedures in 2 patients due to bleeding. One patient with severe hemophilia A undergoing a TKA developed hematuria after removal of a foley catheter; however, the hematuria was self-limited, and blood product support was not required. One patient with mild hemophilia A (factor VIII level 10%) underwent bilateral TKA (2 simultaneous procedures) with enoxaparin initiated the evening before surgery and continued at a dose of 30 mg every 12 hours postoperatively. He developed hypotension and anemia on the second postoperative day that was eventually attributed to operative blood loss as there was no evidence for postoperative bleeding. He improved with fluid resuscitation and required a packed red blood cell transfusion but did not require reoperation due to bleeding. Low-molecular-weight heparin injections were, however, not restarted.
Bleeding and Thrombosis Complications.

There were no symptomatic VTE in the entire population, despite no pharmacologic thromboprophylaxis having been administered to one-quarter of the patients. Early complications (<30 days) included 5 cases of wound site erythema, without documented bleeding or infection, and 2 hemarthroses in patients who did not receive pharmacologic thromboprophylaxis. Late complications (>30 days) occurred in 3 patients. These included 2 patients with aseptic loosening in the prosthetic knees leading to TKA revisions at 9 years and 11 years after the surgery. The latter of these patients developed septic arthritis associated with the revision, requiring surgical debridement and a repeat TKA 1 year later. The third patient developed a progressive flexion contracture and required a TKA revision at 2 years after the initial surgery.
Improvement in ROM was documented in all 9 patients with THA and in 17 (59%) of 29 patients with TKA by 18 months postoperatively (Table 4). We contacted 22 (78%) of 28 study participants accounting for 31 (82%) of 38 procedures to inquire about their satisfaction with the surgery. Nearly all patients in both the TKA and the THA groups reported decreased pain and improved function of the affected joints after surgery and most expressed satisfaction with the decision to undertake the surgery (Table 5).
Range of Motion Outcomes Between 12 and 18 Postoperative Months.
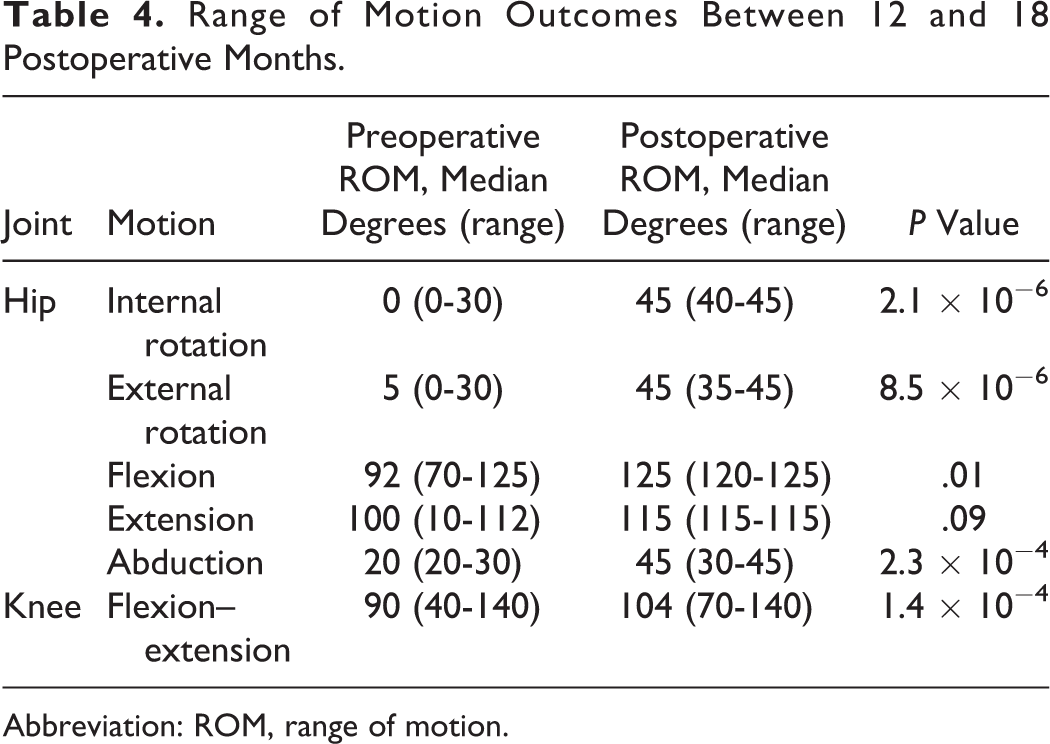
Abbreviation: ROM, range of motion.
Patient Satisfaction.
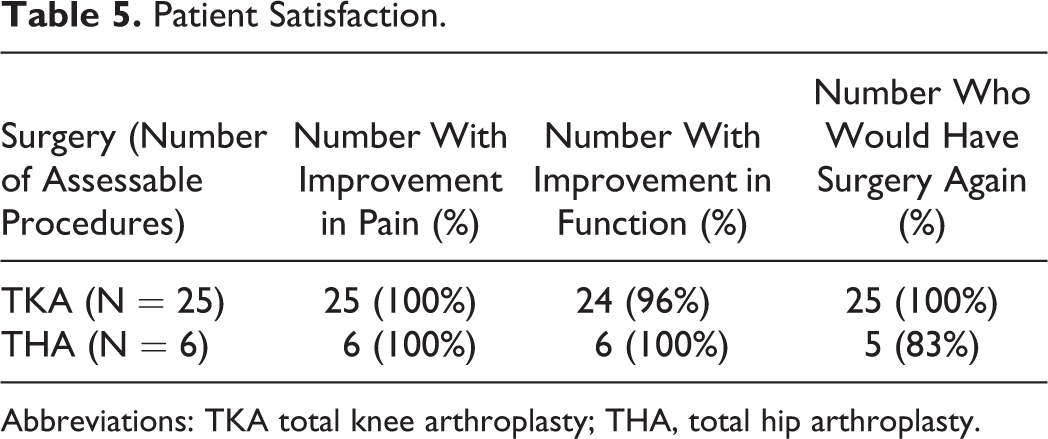
Abbreviation: TKA total knee arthroplasty; THA, total hip arthroplasty.
Discussion
Major orthopedic surgery to address severe hemophilic arthropathies is being performed worldwide with excellent outcomes and substantial improvement in quality of life. 8 –10 Total joint arthroplasty in the general population is associated with an increased risk of VTE that is mitigated with the use of postoperative pharmacologic thromboprophylaxis, which is now standard for most patients who are not perceived to be at increased risk of bleeding. 6 Data on VTE risk in patients with congenital bleeding disorders undergoing TJA are limited, however, and evidence-based guidelines for postoperative VTE prevention do not exist. Our data suggest that administering LMWH postoperatively to patients with bleeding disorders in whom hemostasis is supported with coagulation factor concentrates is generally safe, though 2 patients in our cohort developed bleeding that may have been abetted by anticoagulation. The first patient developed hematuria associated with the use of a foley catheter, but this episode was self-limited and managed without the use of blood product transfusions. The second patient sustained operative blood loss to an extent that a postoperative red blood cell transfusion was required. Of note, he received an enoxaparin injection the evening before surgery and twice daily dosing, a practice that is no longer generally in use at our institution.
We were unable to show that perioperative thromboprophylaxis is beneficial in this population. Notably, there were no symptomatic VTE episodes in our cohort, despite 24% of procedures being associated with no pharmacologic thromboprophylaxis. The validity of our findings, however, is limited by the small size and retrospective nature of our study. Nevertheless, similar findings have been reported by other groups and have been reviewed, 11 with an estimated rate of symptomatic VTE in patients with hemophilia calculated at 0.5% (95% confidence interval 0.1-0.9). More recent descriptions support previous reports of a low risk of VTE in hemophiliac joint replacement surgery. For example, Raza et al reported on 23 patients with hemophilia who underwent elective THA or TJA. 12 Only 2 patients in their cohort received LMWH prophylaxis, and 1 of these patients developed a major bleeding complication. No symptomatic VTE was observed. A Japanese group reported no symptomatic or ultrasonographically detected VTE in 33 patients with hemophilia undergoing 38 TKA procedures, 13 and a Chinese group identified no symptomatic VTE in 32 patients undergoing 52 joint arthroplasties without the use of pharmacologic thromboprophylaxis. 14 Finally, Siboni and colleagues reviewed their experience with orthopedic surgery in a heterogeneous population of 22 patients with rare bleeding disorders undergoing 35 orthopedic surgery procedures. 15 Thromboprophylaxis was used in only 1 case, and, though there were no episodes of DVT or PE, 2 patients who did not receive thromboprophylaxis developed superficial thrombophlebitis.
The incidence of clinically silent VTE in TJA in the general population is reported at up to 40% to 60% and of symptomatic VTE up to 1% to 10% in the absence of thromboprophylaxis. 6 Although this has not been rigorously studied, contemporary case series tend to indicate that VTE occurs much less frequently in patients with hemophilia after major orthopedic surgery, despite restoration of normal hemostasis (by laboratory parameters) and omission of pharmacologic thromboprophylaxis. The incidence is not zero however, and subclinical VTE has been reported in up to 10% of patients with hemophilia undergoing major orthopedic surgery when screened with Doppler ultrasound. 16 Although we identified no cases of symptomatic VTE in our patients, we cannot exclude asymptomatic VTE in our retrospectively reviewed population, as screening for asymptomatic VTE in orthopedic surgery patients is not part of the standard of care in our institution.
The explanation for the lower incidence of both symptomatic and asymptomatic VTE is not obvious but may reflect the younger age at which patients with bleeding disorders tend to undergo joint replacement surgery compared to the population with osteoarthritis as the underlying cause of joint disease. In turn, younger patients tend to have a lower prevalence of comorbid conditions associated with increased risk of VTE (eg, heart failure). Alternatively, coagulation factor replacement therapy, especially using episodic rather than continuous infusion protocols, may not entirely restore hemostasis to “normal.” While peak coagulation factor levels may be in the hemostatic range, there may be substantial periods during the day when low factor levels predominate and which may be protective against VTE.
The American College of Chest Physicians (ACCP) guideline for thromboprophylaxis after major orthopedic surgery suggests using intermittent pneumatic compression devices rather than anticoagulation for patients at increased bleeding risk. 6 While patients with congenital bleeding disorders may fit this designation, the use of coagulation factor concentrates perioperatively is expected to alleviate much of the excess bleeding risk. Nevertheless, because of the apparent small incidence of postoperative VTE in this population, it seems rational to administer pharmacologic thromboprophylaxis only if additional risk factors are present (eg, advanced age, obesity, cancer, or other concurrent medical conditions). The Padua Prediction Score risk assessment model has been validated for VTE risk stratification of medical inpatients and might plausibly be adapted for the hemophiliac population. 17 A cumulative risk score of ≥4 corresponds to a high risk of VTE (11%), whereas a low score is associated with a low VTE risk (0.3%). Alternatively, though a specific instrument for orthopedic surgery is not available, the Caprini score for VTE risk assessment was developed in general surgery patients 18 and may be a useful tool in patients with hemophilia undergoing orthopedic surgery. Any risk assessment tool, however, will have to take into account type of bleeding disorder and coagulation factor concentrate used. For example, compared to patients with congenital factor VIII deficiency, patients with other types of bleeding disorders may have a higher risk of VTE, and the type of coagulation factor concentrate administered may play a role in VTE risk. While there seems to be a very low risk of VTE in patients with factor VIII deficiency, the risk seems to be higher in patients with factor IX deficiency. 11,19 This excess VTE risk in patients with factor IX deficiency may have more to do with coagulation factor replacement products than with the nature of the disorder itself, however, and may not be as much of an issue with recombinant and highly purified factor IX products that have largely replaced intermediate purity products and prothrombin complex concentrates. 20 –22 Patients with inhibitors may be at higher risk when treated with bypass agents, though recent data suggest this increase in risk may be minimal. 23,24 Finally, patients with von Willebrand disease may be at higher risk of VTE than patients with factor VIII deficiency. This may be related to high levels of factor VIII that are often achieved after infusion of intermediate purity plasma-derived products, thus care should be taken to avoid over replacement. 25,26
Presently, the optimal thromboprophylaxis strategy for patients with bleeding disorders undergoing major orthopedic surgery is not defined. A survey of US Hemophilia Treatment Centers in 2005 identified a marked variation in management of this problem, 27 thus the question is ripe for prospective investigation. In the meantime, based on our study and others, young patients having factor VIII deficiency without medical comorbidities or obesity may be candidates for mechanical thromboprophylaxis and early ambulation without anticoagulants or a course of anticoagulation limited to the inpatient period after TJA. Moreover, for patients for whom pharmacologic thromboprophylaxis is selected, a duration of 14 days rather than the extended period recommended by the ACCP for patients without bleeding disorders 6 may be appropriate, corresponding to the time when a stable wound has been established and the daily coagulation factor concentrate dose is lowered. 28
We saw no early deep wound or joint infections in our population. This is likely attributable to both surgical and supportive care factors, but we also note a low incidence of early hemarthoses, and it is likely that the two outcomes are related. Bleeding into a prosthetic joint, including microbleeds, early on seems to be associated with an increased risk of infection, 14,29 –31 and we observed our use of aggressive coagulation factor support perioperatively likely contributed to the low incidence of bleeding complications. The absence of deep infections in our series further suggests the safety of prophylactic LMWH in our population, as we might have expected a higher infection rate if LMWH was contributing to microbleeding in the operated joints.
Major orthopedic surgery carries substantial risk in patients with bleeding disorders and must only be undertaken with careful attention to the risk–benefit calculation. The results of this study suggest that TJA for hemophilic arthropathies in our population was highly beneficial. Nearly all patients in our cohort expressed subjective improvement in pain and function that, for the most part, corresponded to increased ROM. Most importantly, all but one stated a willingness to undergo the surgery again. This parameter is a measure of meaningful improvement that has been validated as reflecting a clinically important difference in health-related quality of life after TJA. 32
Our study has several important limitations. The retrospective design forced us to depend on the accuracy and completeness of existing medical records and did not allow us to capture complete information for all patients. For example, the use of intermittent compression devices for thromboprophylaxis is a potential confounder, and their implementation was difficult to ascertain from the records. In many cases, we were also not able to evaluate other risk factors for VTE in these patients. Additionally, there is no control group, thus we do not have a good baseline estimate of postoperative VTE or bleeding risk in our population and instead rely upon estimates from the existing medical literature, which may not be generalizable. Finally, our study population is not uniform and time period is long, making it likely that surgical and supportive care protocols are heterogeneous and evolved over time. For example, pharmacologic thromboprophylaxis was not documented in patients in this study between 1987 and 2000. The routine use of enoxaparin thereafter reflects Food and Drug Administration approval and availability of the medication in the United States as well as evolving changes in the standard of care for thromboprophylaxis in major orthopedic surgery. Despite the shortcomings of our study, our data suggest that the use of LMWH after TJA is safe in patients with congenital bleeding disorders who receive concurrent hemostatic support postoperatively.
Conclusion
While there are risks associated with TJA in patients with congenital bleeding disorders, our data suggest that they are outweighed by the benefits, with the majority of patients experiencing decreased pain and improved function. The advances in coagulation factor concentrate manufacturing and the virtual elimination of infectious disease transmission as well as improvements in management of existing chronic infections have allowed patients with congenital bleeding disorders to enjoy unprecedented longevity. Accordingly, we are seeing an increase in comorbidities associated with aging in this population. Moreover, we are seeing an increase in obesity in patients with bleeding disorders that is commensurate with the increase observed in the general population nationally. As both advanced age with its concomitant comorbidities and obesity are significant risk factors for VTE, defining the optimal thromboprophylaxis regimen in this population is more important than ever. Moreover, the role of newer, target-specific oral anticoagulants remains to be clarified. We have demonstrated that pharmacologic thromboprophylaxis appears generally safe in this population; whether it is necessary, however, is unknown and should be the subject of prospective trials using risk-adapted protocols.
Footnotes
Acknowledgments
The authors would to gratefully acknowledge Anna Briggs, Hemophilia Center Program Assistant, for her invaluable assistance with data collection for this study.
Authors’ Note
DLO, BMH, YG, and LM designed and performed the research. PB contributed essential research expertise. DLO, BMH, and YG analyzed the data. DLO, BMH, YG, and PB wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
