Abstract
Clinical trials of anticoagulants often exclude special populations. We assessed the proportion of special populations in real-world orthopedic surgery and the incidence of venous thromboembolism (VTE)-related outcomes. Data on patients with hip (n = 11 483) or knee replacement (n = 19 390) were extracted from IMS’ PharMetrics Patient-Centric Database. There was high prevalence of patients aged ≥75 years (20.3%), CYP3A4-inhibitor use (21.5%), and chronic warfarin use (9.5%). Venous thromboembolism events were increased with each increasing year of age (hip: odds ratio [OR] 1.02, 95% confidence interval [CI] = 1.01-1.03; knee: OR 1.01, 95%CI = 1.00-1.02) and chronic warfarin use (hip: OR 1.56, 95%CI = 1.13-2.17; knee: OR 1.33, 95%CI = 1.03-1.72); in hip patients with renal insufficiency (OR1.61, 95%CI=1.11-2.36); and in knee patients with atrial fibrillation (OR 1.41, 95%CI = 1.06-1.88). Major bleeding was higher in hip patients with hepatic impairment (OR 21.99, 95%CI = 2.04-236.62), each increasing year of age (OR 1.08, 95%CI = 1.01-1.15), and chronic warfarin use (OR 7.11, 95%CI = 1.16-43.46). Special populations are prevalent in real-world orthopedic surgery, which may impact VTE-related outcomes.
Introduction
Patients undergoing major orthopedic surgery are at particularly high risk of venous thromboembolism (VTE). Without effective thromboprophylaxis, the incidence of hospital-acquired deep-vein thrombosis (DVT) is approximately 40% to 60% following total hip replacement (THR) and approximately 40% to 85% following total knee replacement (TKR). 1 Furthermore, the incidence of pulmonary embolism (PE) following THR or TKR is 0.9% to 28% and 1.5% to 10%, respectively, with fatal PE occurring in 0.1% to 2.0% of cases. 1 The American College of Chest Physicians recommends that patients undergoing major orthopedic procedures receive thromboprophylaxis with a low-molecular-weight heparin (LMWH), fondaparinux, or adjusted-dose vitamin K antagonist for ≥10-days postsurgery. Given the persistent risk of VTE for several weeks after surgery, extended-duration thromboprophylaxis is recommended for up to 35 days after THR or hip fracture surgery and suggested after TKR. 1
In addition to established prophylactic agents, new oral anticoagulants are in the advanced stages of clinical development for VTE prevention. For example, dabigatran etexilate and rivaroxaban have recently been licensed for use as thromboprophylaxis in major orthopedic surgery in Europe and Canada. Dabigatran etexilate is an oral pro-drug that is converted to the direct thrombin inhibitor, dabigatran. 2 Rivaroxaban is a direct inhibitor of factor Xa 3 and is currently under review by the Food and Drug Administration (FDA).
However, caution with prophylactic agents needs to be exercised in several special patient populations and in those taking specific other medications (Table 1 ).4–10 Cautions or contraindications are described on the prescribing information of some anticoagulants for patients with renal insufficiency, hepatic impairment, concomitant use of antiplatelets, or use of a CYP3A4 inhibitor.5–7 Furthermore, some patient populations are of additional concern, for example, patients with acute coronary syndrome (ACS) because of the potential for rebound events. 4 Oral warfarin use is particularly associated with drug interactions, primarily as a result of cytochrome P450 (CYP) metabolism, which can affect the maintenance of therapeutic levels of anticoagulation. 10
Selection of Subpopulations

Abbreviations: ACS, acute coronary syndromes; CYP, cytochrome P450; VTE, venous thromboembolism.
These special patient groups are often excluded from clinical trials, and few studies have evaluated the proportion of special patient groups present in everyday surgical practice and assessed the potential impact on VTE-related clinical outcomes. The objective of this analysis was to evaluate the proportion of real-world THR and TKR patients with a history of ACS, renal insufficiency, hepatic impairment, atrial fibrillation (AF), or patients aged ≥75 years. The proportion of real-world THR and TKR patients prescribed concomitant medications was also assessed, with regard to CYP3A4 inhibitors, chronic warfarin use, and antiplatelet drugs. In addition, the study evaluated VTE prophylaxis status and the incidence of VTE-related clinical outcomes (including DVT, PE, and bleeding) within each subpopulation.
Methods
Study Population
IMS’ PharMetrics Patient-Centric Database was used, which comprises commercial health plan information collected from over 94 different managed-care organizations, and includes the complete pharmacy and medical records of more than 58 million patients. The database includes both inpatient and outpatient diagnoses (International Classification of Diseases, Ninth Revision [ICD-9-CM] codes) and procedures (Current Procedural Terminology, Fourth Edition [CPT-4] codes and Healthcare Common Procedure Coding System) and prescription records (National Drug Classification codes). As such, the reliability of the data relies on the coding accuracy of the physicians in the hospitals. Both health care plan paid and charged amounts are available for all services rendered, along with dates of service. Additional information includes patient demographics (year of birth, gender, geographic region), plan type (health maintenance organization, preferred provider organization), payer type (commercial, self-insured), provider specialty, and plan enrollment. This database has been used in many real-world health outcomes research studies. 11
In this study, patients aged ≥18 years with THR or TKR as their primary CPT-4 code were included. An 18-month period was examined for each patient, beginning 12 months prior to, and ending 6 months after the index surgical event, with the index surgical event having to have occurred in 2007. Therefore, records from January 2006 to June 2008 were extracted. In the event of multiple hip or knee surgeries during the study period, only the first surgery was included. Patients with hospital admission for both THR and TKR were allocated to the THR cohort due to a higher degree of morbidity within the cohort.
The primary objective was to evaluate the proportion of special populations among patients with orthopedic surgery, as well as the VTE prophylaxis status within each subpopulation and the incidence of VTE-related clinical outcomes, including DVT, PE, major bleeding, minor bleeding, and thrombocytopenia. Using CPT-4, the ICD-9-CM, and National Drug Classification codes, the proportion of patients with orthopedic surgery having one of the following factors was assessed: history of ACS (intermediate coronary syndrome and acute myocardial infarction); renal insufficiency (acute renal failure, chronic kidney disease, and renal failure); hepatic impairment (chronic liver disease, hepatitis, or cirrhosis); AF or atrial flutter; and age ≥75 years. In addition, the concomitant use of specific drugs was assessed, namely: chronic warfarin use for any medical reason; antiplatelet drugs (clopidogrel, ticlopidine, and acetylsalicylic acid); and the prescription of a strong or moderate CYP3A4 inhibitor including antiviral HIV protease inhibitors (indinavir, nelfinavir, ritonavir), macrolide antibiotics (erythromycin, telithromycin, clarithromycin), azole antifungals (fluconazole, itraconazole, ketoconazole, posaconazole, voriconazole), nefazodone, quercetin, aprepitant, verapamil, amiodarone, chloramphenicol, ciprofloxacin, cyclosporine, diltiazem, imatinib, echinacea, ergotamine, metronidazole, norfloxacin, delavirdine, efavirenz, nevirapine, ethinyl estradiol, saquinavir, fluoxetine, and fluvoxamine. In-hospital VTE prophylaxis patterns were determined for each special population cohort, as well as for all THR patients and all TKR patients. The following pharmacological agents were studied: warfarin, unfractionated heparin (UFH), enoxaparin, dalteparin, tinzaparin, and fondaparinux.
The study determined VTE-related clinical outcomes among each of the subpopulations. The primary efficacy endpoint was the overall rate of hospital-acquired VTE as one of the secondary hospital discharge diagnoses. Venous thromboembolism events were classified based on the time of occurrence, namely, VTE events that occurred during the index surgery hospitalization and those that occurred within 180 days after the index surgical discharge as a postsurgical hospital readmission. The study also examined readmissions associated with ACS. The primary safety endpoint was the overall rate of adverse events (major bleeding, minor bleeding, and thrombocytopenia) as one of the secondary hospital discharge diagnoses.
Statistical Analyses
Descriptive statistics were used to measure and describe patient demographics and prophylaxis status, with P values provided by chi-square test when appropriate. The clinical outcomes were summarized and compared statistically across the special populations. A logistic regression analysis was utilized to determine the relationship between the clinical endpoints and the special populations. A binary response model (Yes vs No for the presence of a specified clinical condition or drug use) was used for the study analysis, and the Fisher scoring algorithm was employed to determine maximum likelihood estimations. The analysis also used full-rank parameterization of the classification variables. Odds ratio (OR) estimates with Wald 95% confidence intervals (CI) were reported for each regression parameter. P values were derived from the analysis of maximum likelihood estimates. A P value of .05 was used to determine the level of statistical significance. All statistical analysis was carried out using SAS (SAS Institute, Cary, North Carolina).
Results
This analysis of 30 873 patients included 11 483 patients with THR and 19 390 with TKR (Table 2 ). There were more female patients in both the THR (54.6%) and the TKR (62.6%) groups. Mean ages in the THR and TKR cohorts were similar at 65.2 years and 64.1 years, respectively. Median length of stay in hospital was 3 days in both cohorts; however, the proportion of patients who remained in hospital for ≥7 days was nearly twice as high for patients with THR (9.9%) than for TKR (5.5%).
Summary of Patient Demographics and Characteristics

Abbreviations: ACS, acute coronary syndrome; CYP, cytochrome P450; SD, standard deviation.
When the special populations were analyzed, a large proportion of patients were aged ≥75 years (26.1% of patients with THR and 16.9% of patients with TKR). Atrial flutter was recorded in 9.0% of patients with THR and 6.9% of patients with TKR. Renal insufficiency was noted in 5.2% of patients with THR and 4.0% of patients with TKR. Approximately 1.5% to 2% of patients analyzed had a history of ACS or hepatic impairment. About a fifth of patients used a medication that interacted with CYP3A4 (19.8% for THR and 22.5% for TKR) and 1 in 10 had chronic warfarin use (9.2% and 9.7%, respectively). Fewer individuals, 3.4% of patients with THR and 3.2% of patients with TKR, were prescribed antiplatelet drugs.
Prophylaxis Status
Overall, less than half of patients received a pharmacological agent for prophylaxis during their index surgery hospitalization (43.9% of patients with THR and 46.7% of patients with TKR; Table 3 ). Significantly lower rates of inpatient prophylaxis were observed in patients with THR and patients with TKR aged ≥75 years (26.8% and 34.6%, respectively) compared with the total cohort populations (both comparisons P < .0001; Table 3). Patients with renal insufficiency also had significantly lower rates of prophylaxis use compared with the overall population for THR (30.9%; P < .0001) as well as TKR (42.0%; P < .01). Significantly higher rates of prophylaxis were observed in patients with chronic warfarin use (80.6% for THR patients and 83.8% for TKR patients) and in those who received a CYP3A4 inhibitor (58.2% and 61.7%, respectively) compared with the total cohort populations (all comparisons P < .0001).
Patients Receiving Venous Thromboembolism Prophylaxis
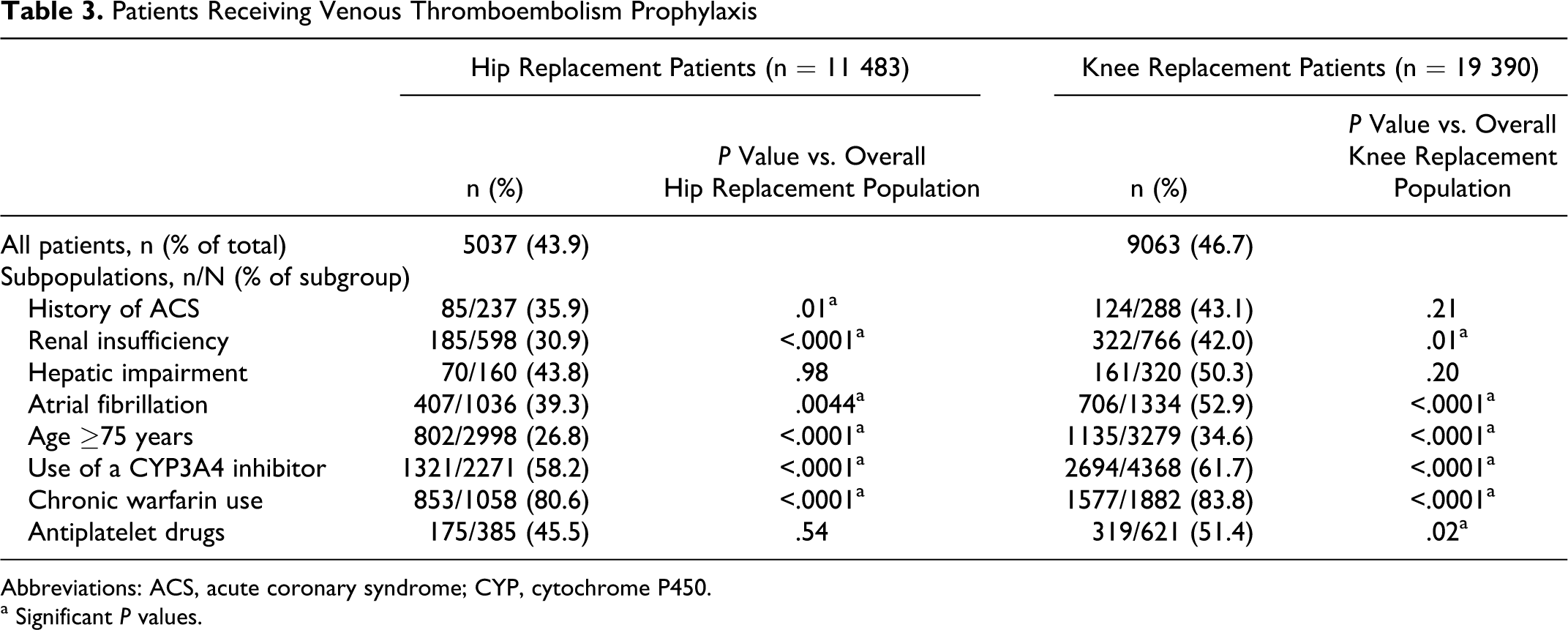
Abbreviations: ACS, acute coronary syndrome; CYP, cytochrome P450.
a Significant P values.
Overall, the 2 most commonly prescribed anticoagulants in patients with THR and TKR were warfarin (27.3% and 30.3% of the total, respectively) and enoxaparin (15.2% and 15.5% of the total, respectively), with few patients receiving fondaparinux (both 1.7%), dalteparin (1.4% and 1.2%), UFH (0.03% and 0.06%), or tinzaparin (0.01% and 0%). A similar pattern of type of anticoagulant prescribed was found for each of the different special patient populations.
Clinical Outcomes
Unadjusted clinical event rates in each of the special populations are presented for THR (Table 4 ) and TKR (Table 5 ). The relationship between the clinical endpoints and the special populations was determined through a logistic regression analysis, using a binary response model (Yes vs No for the presence of a specified clinical condition or drug use; Tables 6 and 7 ).
Unadjusted Clinical Event Rates in Subpopulations of Hip Replacement Patients and Those Using Specified Drugs
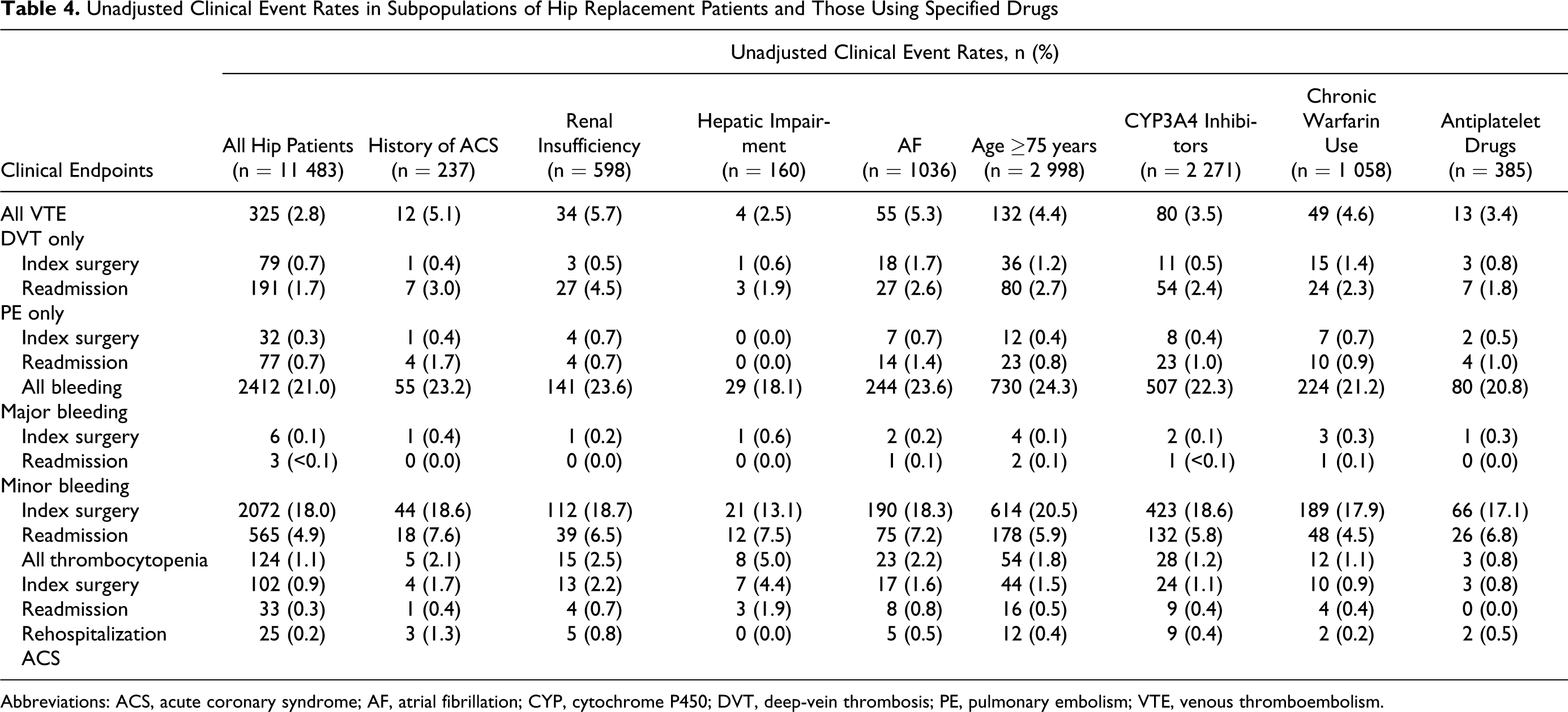
Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; CYP, cytochrome P450; DVT, deep-vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Unadjusted Clinical Event Rates in Subpopulations of Knee Replacement Patients and Those Using Specified Drugs
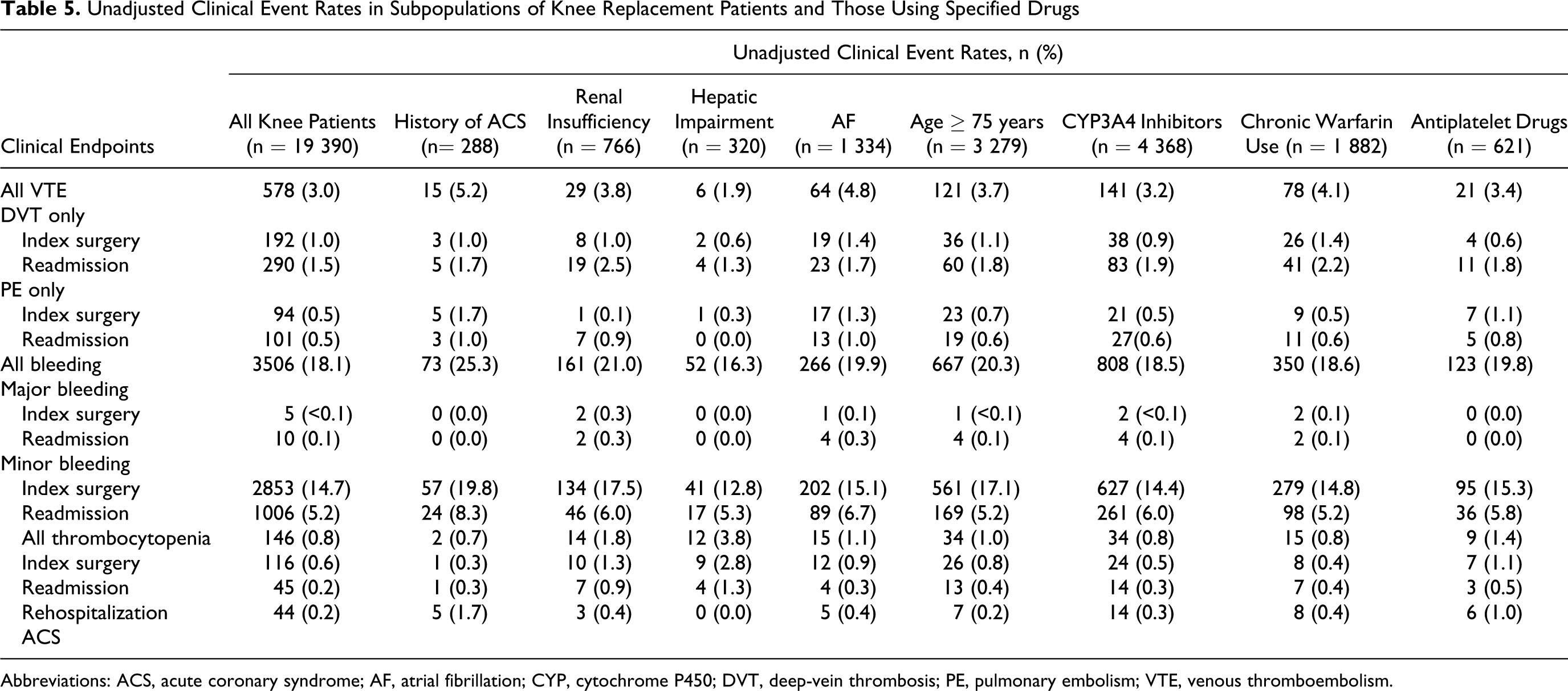
Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; CYP, cytochrome P450; DVT, deep-vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Clinical Endpoints in Subpopulations of Hip Replacement Patients and Those Using Specified Drugs a
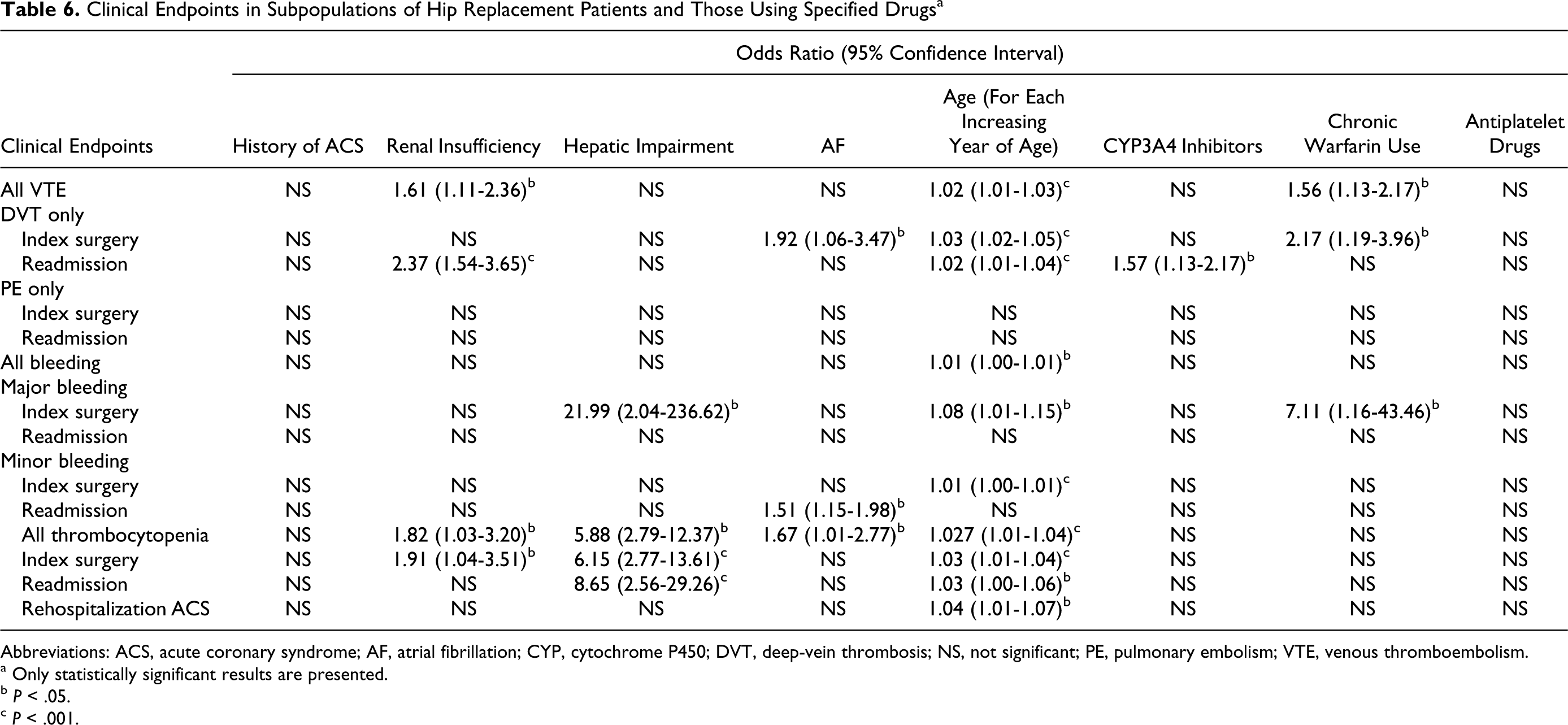
Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; CYP, cytochrome P450; DVT, deep-vein thrombosis; NS, not significant; PE, pulmonary embolism; VTE, venous thromboembolism.
a Only statistically significant results are presented.
b P < .05.
c P < .001.
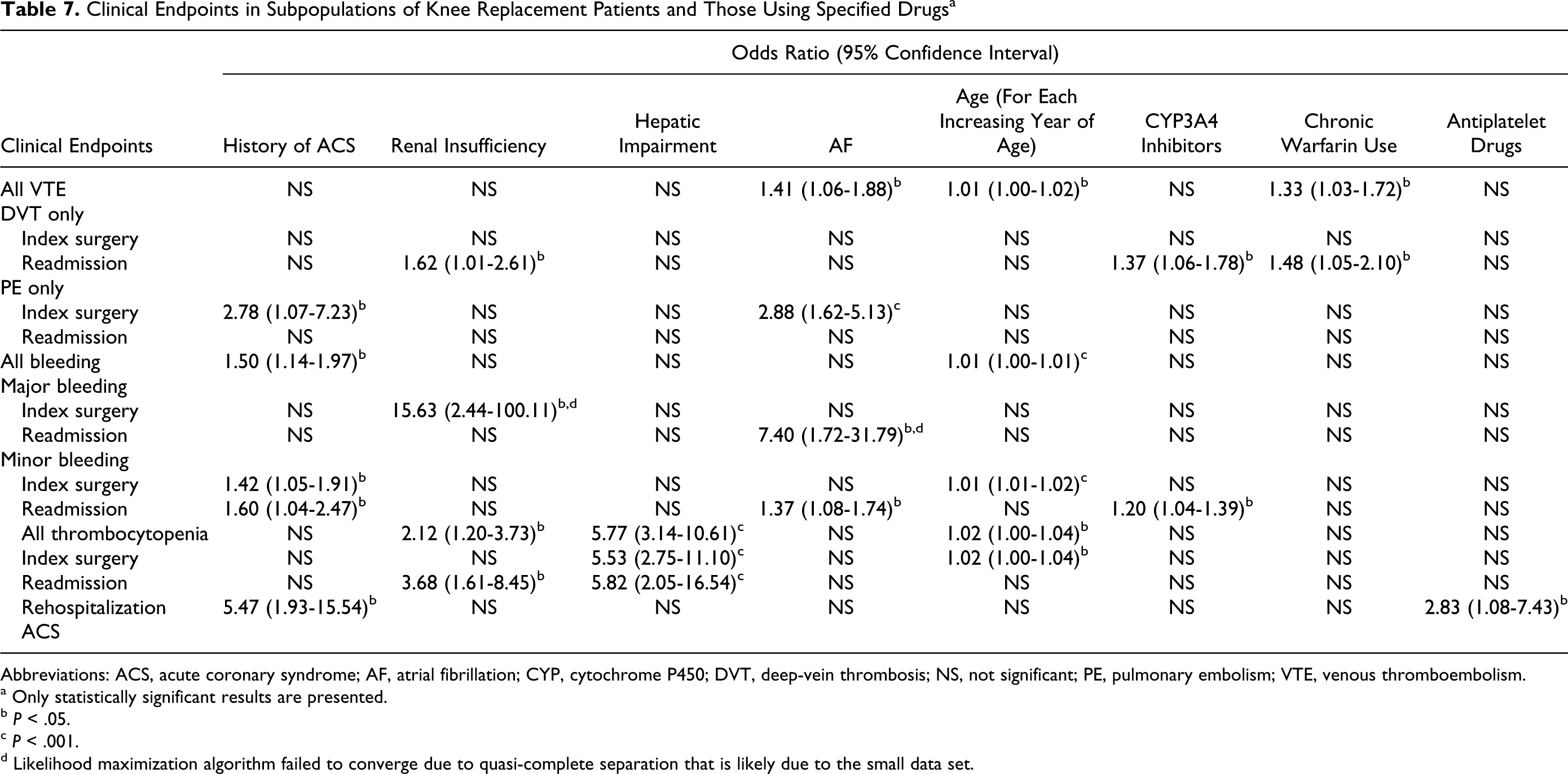
Clinical Endpoints in Subpopulations of Knee Replacement Patients and Those Using Specified Drugs a
Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; CYP, cytochrome P450; DVT, deep-vein thrombosis; NS, not significant; PE, pulmonary embolism; VTE, venous thromboembolism.
a Only statistically significant results are presented.
b P < .05.
c P < .001.
d Likelihood maximization algorithm failed to converge due to quasi-complete separation that is likely due to the small data set.
The risk of developing VTE was significantly higher in patients with THR having renal insufficiency (OR 1.61, 95% CI = 1.11-2.36), on chronic warfarin use (OR 1.56, 95% CI = 1.13-2.17), and with each increasing year of age (OR 1.02, 95% CI = 1.01-1.03; Table 6). For patients with TKR, the risk of developing VTE was significantly higher in those with AF (OR 1.41, 95% CI = 1.06-1.88), on chronic warfarin use (OR 1.33, 95% CI = 1.03-1.72), and with each increasing year of age (OR 1.01, 95% CI = 1.00-1.02; Table 7).
The likelihood of major bleeding was higher in patients with THR having hepatic impairment (OR 21.99, 95% CI = 2.04-236.62), each increasing year of age (OR 1.08, 95% CI = 1.01-1.15), and chronic warfarin use (OR 7.11, 95% CI = 1.16-43.46). In patients with TKR, an increase in the odds of major bleeding during the index surgery hospitalization was observed in patients with renal insufficiency (OR 15.63, 95% CI = 2.44-100.11), but the wide confidence interval indicated that the validity of this observation is questionable, perhaps due to the small data set.
Clinical outcomes that were significantly affected in each of the special populations are presented for patients with THR (Table 6) and TKR (Table 7). A history of ACS had no significant impact on outcomes in patients with THR (Table 6). In contrast, prior ACS significantly increased the odds of patients with TKR having PE during the index surgery hospitalization by nearly 3-fold (OR 2.78, 95% CI = 1.07-7.23, P < .05) and increased postoperative readmission ACS by over 5-fold (OR 5.47, 95% CI = 1.93-15.54, P < .05) compared with the overall TKR population (Table 7). A history of ACS also increased the likelihood of a bleeding event (OR 1.50, 95% CI = 1.14-1.97, P < .05) and minor bleeding during the index surgical admission (OR 1.42, 95% CI = 1.05-1.91, P < .05) and upon readmission (OR 1.60, 95% CI = 1.04-2.47, P < .05) in patients with TKR (Table 7).
The risk of developing VTE was significantly higher in patients with THR having renal insufficiency (OR 1.61, 95% CI = 1.11-2.36, P < .05) but not in patients with TKR (Tables 6 and 7). In both patients with THR and TKR surgery, renal insufficiency also increased the likelihood of readmission for DVT (OR 2.37, 95% CI = 1.54-3.65, P < .001 and OR 1.62, 95% CI = 1.01-2.61, P < .05, respectively) and increased the odds of total thrombocytopenic events (OR 1.82, 95% CI = 1.03-3.20, P < .05 and OR 2.12, 95% CI = 1.20-3.73, P < .05, respectively). The validity of the large increase in the odds of major bleeding during the index surgery hospitalization observed in patients with TKR is questionable, perhaps due to the small data set.
Hepatic impairment was significantly associated with an almost 6-fold increased odds of the incidence of thrombocytopenia in both patients with THR (OR 5.88, 95% CI = 2.79-12.37, P < .001) and TKR (OR 5.77, 95% CI = 3.14-10.61, P < .001; Tables 6 and 7). The increase in likelihood of thrombocytopenia was observed for both the index surgery hospitalization and readmissions. More than a 20-fold increased likelihood of major bleeding during the index surgery hospitalization was also observed in patients with THR having hepatic impairment (OR 21.99, 95% CI = 2.04-236.62, P < .05).
Atrial flutter was associated with a higher risk of DVT during the surgical hospitalization in patients with THR (OR 1.92, 95% CI = 1.06-3.47, P < .05). In patients with TKR, AF was associated with a higher risk of VTE (OR 1.41, 95% CI = 1.06-1.88, P < .05) and PE during index surgery hospitalization (OR 2.88, 95% CI = 1.62-5.13, P < .001; Tables 6 and 7). Increased likelihood of minor bleeding on readmission was also observed in patients with AF in both cohorts.
Advancing age was associated with a large number of VTE-related outcomes. Each increasing year of age was linked with a significantly higher risk of VTE, all bleeding, and minor bleeding and thrombocytopenia during the index surgery hospitalization in both cohorts (Tables 6 and 7). In addition, DVT was significantly increased with advancing age in patients with THR during the index surgery hospitalization (OR 1.03, 95% CI = 1.02-1.05, P < .001) and upon readmission (OR 1.02, 95% CI = 1.01-1.04, P < .001; Table 6). In this cohort, there was also increased likelihood with older age of major bleeding during the index surgery hospitalization (OR 1.08, 95% CI = 1.01-1.15, P < .05) and increased postoperative readmission ACS (OR 1.04, 95% CI = 1.01-1.07, P < .05).
Regarding the use of specific concomitant drugs, CYP3A4 inhibitor utilization increased the likelihood of readmission for DVT in both patients with THR (OR 1.57, 95% CI = 1.13-2.17, P < .05) and TKR (OR 1.37, 95% CI = 1.06-1.78, P < .05; Tables 6 and 7). In addition, use of CYP3A4 inhibitors was associated with a higher risk of minor bleeding during readmission in patients with TKR (OR 1.20, 95% CI = 1.04-1.39, P < .05). The risk of developing VTE was significantly higher with chronic warfarin use in patients with THR (OR 1.56, 95% CI = 1.13-2.17, P < .05) and TKR (OR 1.33, 95% CI = 1.03-1.72, P < .05). Chronic warfarin use was also associated with more than 7-fold increased likelihood of major bleeding during the index THR hospitalization (OR 7.11, 95% CI = 1.16-43.46, P < .05). Use of antiplatelet drugs did not have a substantial impact on VTE-related outcomes, with the exception of rehospitalization for ACS, which was increased by nearly 3-fold in patients with TKR (OR 2.83, 95% CI = 1.08-7.43, P < .05).
Discussion
The results of this real-world study of more than 30 000 patients demonstrate that special populations are prevalent among “everyday” patients who undergo THR and TKR. These patient groups are often excluded from clinical trials so it is important to estimate their prevalence in everyday surgical patients and evaluate their impact on efficacy and safety outcomes. The pharmacological VTE prophylaxis rates in this real-world study were low among the total populations studied, despite the fact that patients undergoing major orthopedic surgery are at particularly high risk of VTE. Only 43.9% to 46.7% of patients following THR or TKR received pharmacological prophylaxis. These findings are consistent with other studies that have demonstrated low prophylaxis rates in surgical patients in general,12–14 as well as in patients with orthopedic surgery.15–17
The present study included 4.4% of patients with renal insufficiency, a condition particularly linked with the need for anticoagulant dose considerations. Many anticoagulants are predominantly eliminated by the kidneys. The use of therapeutic doses of anticoagulants in patients with renal impairment may lead to inefficient clearance and bioaccumulation, resulting in excessive anticoagulation and a subsequent increased risk of bleeding complications. 18 In the present study, renal insufficiency was associated with an increased risk of rehospitalization for DVT and thrombocytopenia in both patients with THR and TKR. Patients with renal insufficiency were also one of the subgroups with the lowest prophylaxis rate, perhaps reflecting the reluctance of surgeons to prescribe prophylaxis due to safety concerns. Enoxaparin can be prescribed to patients with renal insufficiency provided FDA-approved dose adjustments are used in patients with severe renal impairment (CrCl <30 mL/min). 5 Fondaparinux is contraindicated in patients with severe renal impairment as is the new oral anticoagulant, dabigatran. 6 Rivaroxaban may be used with caution in patients with CrCl of 15 mL/min to 29 mL/min but is not recommended in patients with CrCl <15 mL/min. 7
Hepatic impairment was noted in 1.6% of patients in the present study. Hepatic impairment consistently increased the odds of thrombocytopenia in both cohorts and also resulted in a greatly increased likelihood of major bleeding in patients with THR. The first new oral anticoagulant, ximelagatran, was withdrawn from market due to increased risk of liver toxicity. 19 Other new anticoagulants have so far not been associated with increased liver toxicity 20 ; however, any effects of new agents on liver function may be exacerbated in the presence of established hepatic insufficiency. The use of LMWHs may lead to elevations in liver transaminase levels, but increases are generally transient, asymptomatic, and are not associated with long-term adverse sequelae.5,21 The manufacturer recommends that caution be exercised when enoxaparin is used in patients with hepatic impairment. 5 Rivaroxaban can be used with caution in patients with cirrhosis having moderate hepatic impairment (Child Pugh score B) if it is not associated with coagulopathy. 7 Dabigatran is not recommended in patients with elevated liver enzymes (>2 × the upper limit of normal) as these patients were excluded from clinical trials with dabigatran. 6
A fifth of patients in the current study were aged ≥75 years. Pharmacological prophylaxis rates were particularly low among patients ≥75 years, with only 27% and 35% of these patients receiving pharmacological prophylaxis after THR and TKR, respectively. Advancing age is associated with increased risk of VTE, such that 18- to 28-fold increases in VTE risk were observed in patients aged ≥70 years compared with those aged 20 years to 29 years. 8 Elderly patients also have a higher incidence of age-related renal disease and less lean body mass. 9 Elderly patients are at increased risk of bleeding with all anticoagulants, independent of their reliance on renal elimination. 9 In the present study, increasing age had a small, but significant impact on VTE, bleeding, and thrombocytopenia in both cohorts. Rehospitalization for ACS was also increased in elderly patients with THR.
The use of medications that inhibit CYP3A4 was observed in approximately 20% of patients, which may be of concern to patients receiving oral anticoagulants metabolized via CYP3A4. CYP3A4 is 1 of 2 CYP450 isoforms that metabolize the R enantiomer of warfarin 10 ; CYP3A4 also metabolizes rivaroxaban. 7 Due to increased exposure, it is not recommended that rivaroxaban be coadministered with drugs that strongly inhibit both CYP3A4 and P-glycoprotein efflux transporter, including systemic azole-antimycotics (eg, ketoconazole, itraconazole, voriconazole, and posaconazole) or HIV protease inhibitors (e.g. ritonavir). Caution should be exercised when rivaroxaban is taken with strong CYP3A4 inducers (eg, rifampicin, phenytoin, carbamazepine, phenobarbital, or St John’s Wort) as this may lead to reduced rivaroxaban plasma concentrations. In the present study, use of CYP3A4 inhibitors was associated with an increased risk of rehospitalization for DVT in patients with THR and TKR, despite this subgroup having one of the highest prophylaxis rates. The most often prescribed anticoagulant was warfarin, which—as mentioned—is metabolized by CYP3A4. Hypothetically, it is possible that the use of CYP3A4 inhibitors decreased the effectiveness of warfarin, 22 which may have led to an increased risk of DVT if patients were not being monitored properly and decreases in international normalized ratio were not detected early enough for appropriate adjustment of warfarin doses.
Use of antiplatelet drugs was recorded in 3.3% of patients of the study population. Furthermore, patients with TKR taking antiplatelet drugs had higher prophylaxis rates than the overall population. In general, the use of antiplatelet drugs and anticoagulants is cautioned due to the possible increased risk of bleeding. According to instructions from the manufacturer, it is not recommended that dabigatran be used concomitantly with clopidogrel and ticlopidine. 6 Rivaroxaban should be used with care in patients concomitantly treated with acetylsalicylic acid and platelet aggregation inhibitors. 7 Use of enoxaparin concomitant with platelet inhibitors, including acetylsalicylic acid and salicylates, is recommended to be avoided whenever possible, and to be monitored if coadministration is essential. 5
In the present study, use of antiplatelets was associated with an increased risk of rehospitalization for ACS in patients with TKR.
The present study highlights the extent to which special patient populations are prevalent among real-world orthopedic surgery patients and their potential impact on efficacy and safety outcomes. These findings should be taken into account when providing orthopedic surgery patients with prophylactic regimens. Although new oral anticoagulants have been evaluated in clinical trials as an alternative in VTE prevention in orthopedic surgery, clinical trial results should be viewed with caution until they are replicated in routine care settings across a broad spectrum of patients with orthopedic surgery. 20
Regarding limitations, although the database contains information from a large number of different managed-care organizations across diverse geographical locations in the United States, it is possible that it may not be representative of the US population as a whole. This study assessed VTE incidence after hospital discharge from the index surgery as readmission for VTE. This does not include VTE occurring postdischarge and being treated in the outpatient setting, leading to an underestimation of VTE rates. In addition, asymptomatic DVT was not included as VTE was assessed as a hospital diagnosis for VTE and imaging techniques are not routinely applied to prospectively screen for DVT in clinical practice. Patients with contraindications to pharmacological prophylaxis were not excluded from this study. If these patients received mechanical prophylaxis as recommended, this study may have underestimated the thromboprophylaxis rate. The status of using mechanical prophylaxis could not be reliably recorded in the database used in this study.
To conclude, the present study highlights that special patient populations are prevalent in the real-world orthopedic setting and this may impact on the efficacy and safety of prophylaxis regimens. Studies on the effects of different patient characteristics on clinical outcomes are warranted, particularly with new anticoagulants where few real-world data exist.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The authors received editorial/writing support in the preparation of this manuscript from Hester van Lier, PhD, of Excerpta Medica, funded by sanofi-aventis US, Inc. The authors are fully responsible for all content and editorial decisions. HCO Research received research funding from sanofi-aventis US, Inc for this study. Steven Deitelzweig: honoraria, research funding, and speakers bureau for sanofi-aventis, Bristol-Myers Squibb, and Scios; speakers bureau for Pfizer. Jay Lin: employee at Bruce Wong & Associates Inc, which received funding to carry out this work from sanofi-aventis US, Inc. Grace Lin: employee at HCO Research, which received funding to carry out this work from sanofi-aventis US, Inc.
