Abstract
Anticoagulation therapy is central to the management of thromboembolic disorders, and the use of direct oral anticoagulants offers several advantages over standard therapy with parenteral heparins and vitamin K antagonists. In phase III clinical trials, the direct oral anticoagulants (given once or twice daily) all demonstrated favorable benefit–risk profiles compared with conventional standard therapy for the treatment and secondary prevention of venous thromboembolism and for stroke prevention in patients with nonvalvular atrial fibrillation. In clinical practice, many factors may influence overall clinical outcomes in patients receiving anticoagulant therapy, including adherence and persistence to the prescribed therapy, which becomes particularly important during long-term therapy. When choosing an anticoagulant for an individual patient, the pharmacological and clinical profile of the anticoagulant, its dosing regimen, and the patient’s clinical characteristics (eg, renal function and comorbidities) and preferences should be considered. This review examines the rationale for and clinical evidence of the selected dosing regimens of the direct oral anticoagulants for the treatment of venous thromboembolism and stroke prevention in nonvalvular atrial fibrillation. The potential influence of dosing strategies (eg, once- or twice-daily dosing) and other factors on patient adherence and therapy persistence are also discussed.
Keywords
Introduction
Thromboembolic disorders such as venous thromboembolism (VTE) and stroke arising from atrial fibrillation (AF) are major public health concerns worldwide. Atrial fibrillation, which is estimated to have an incidence as high as 28.3 per 1000 person-years, 1 is associated with a 5-fold increase in stroke and a 3-fold increase in congestive heart failure and can also lead to cognitive dysfunction and premature death. 2,3 The worldwide projected incidence rate of VTE (comprising deep vein thrombosis [DVT] and pulmonary embolism [PE]) was reported as 0.21 to 1.48 per 1000 person-years, with a prevalence of 0.20% to 0.66%. 4 Venous thromboembolism and its long-term complications (risk of recurrence, postthrombotic syndrome, and chronic thromboembolic pulmonary hypertension) 5 –7 are associated with significant morbidity and mortality. 8,9 Postthrombotic syndrome affects 20% to 50% of patients with DVT, and chronic thromboembolic pulmonary hypertension can occur in up to 9% of patients with PE within the first 2 years. 10 –12 Recurrence rates of VTE vary depending on patient characteristics, such as age and sex, and on the origin of the index event. 13 Thromboembolic disorders represent a rapidly increasing burden to patients, physicians, and health care systems, 14,15 in part owing to the rise in the proportion of elderly people in many populations, such as in the United States. 16,17
Anticoagulation therapy is central to the management of thromboembolic disorders, and standard of care comprises heparins and/or vitamin K antagonists (VKAs). 18 –20 However, VKA therapy is associated with several drawbacks: a slow onset of action, a narrow therapeutic window, a need for frequent coagulation monitoring and dose adjustment, multiple drug–drug and food–drug interactions, and an increased risk of bleeding. 21,22 Low-molecular weight heparins (LMWHs) are more predictable compared with VKAs; however, parenteral administration is a burden for some patients and health care systems. 21 The direct oral anticoagulants (OACs) have been developed to overcome the limitations of traditional anticoagulant therapy. As a result of successful phase III trials, apixaban, dabigatran, edoxaban, and rivaroxaban have been approved for the prevention of stroke in patients with nonvalvular AF and for the treatment and prevention of recurrent VTE (Table 1). 23 –36
Summary of Guidance for Use of Direct Oral Anticoagulants for the Treatment and Prevention of Recurrent Venous Thromboembolism and the Prevention of Stroke in Patients With Nonvalvular Atrial Fibrillation.
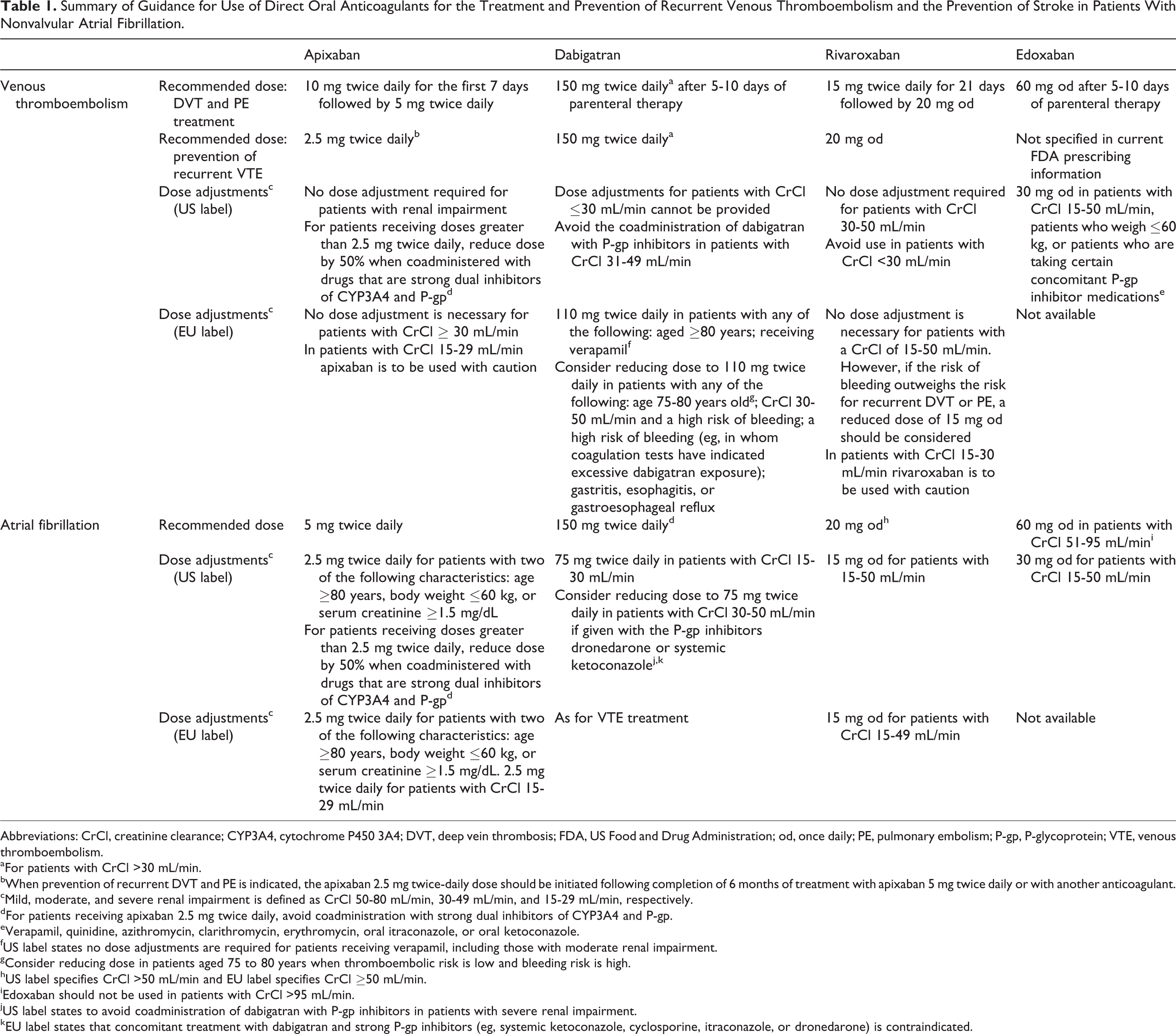
Abbreviations: CrCl, creatinine clearance; CYP3A4, cytochrome P450 3A4; DVT, deep vein thrombosis; FDA, US Food and Drug Administration; od, once daily; PE, pulmonary embolism; P-gp, P-glycoprotein; VTE, venous thromboembolism.
aFor patients with CrCl >30 mL/min.
bWhen prevention of recurrent DVT and PE is indicated, the apixaban 2.5 mg twice-daily dose should be initiated following completion of 6 months of treatment with apixaban 5 mg twice daily or with another anticoagulant.
cMild, moderate, and severe renal impairment is defined as CrCl 50-80 mL/min, 30-49 mL/min, and 15-29 mL/min, respectively.
dFor patients receiving apixaban 2.5 mg twice daily, avoid coadministration with strong dual inhibitors of CYP3A4 and P-gp.
eVerapamil, quinidine, azithromycin, clarithromycin, erythromycin, oral itraconazole, or oral ketoconazole.
fUS label states no dose adjustments are required for patients receiving verapamil, including those with moderate renal impairment.
gConsider reducing dose in patients aged 75 to 80 years when thromboembolic risk is low and bleeding risk is high.
hUS label specifies CrCl >50 mL/min and EU label specifies CrCl ≥50 mL/min.
iEdoxaban should not be used in patients with CrCl >95 mL/min.
jUS label states to avoid coadministration of dabigatran with P-gp inhibitors in patients with severe renal impairment.
kEU label states that concomitant treatment with dabigatran and strong P-gp inhibitors (eg, systemic ketoconazole, cyclosporine, itraconazole, or dronedarone) is contraindicated.
Compared with the VKAs, direct OACs have a rapid onset of action, predictable pharmacokinetics and pharmacodynamics that eliminate the need for routine coagulation monitoring, and minimal food and drug interactions. 37 These direct OACs are given either once daily (od) or twice daily in the approved indications. Effective anticoagulation is essential for the management of thromboembolic disorders, but therapeutic levels and clinical benefits are not achieved unless adherence remains high. This is particularly important during long-term therapy, such as stroke prevention in patients with AF. This article examines the rationale for and clinical evidence of the selected dosing regimens of the direct OACs for the treatment of VTE and stroke prevention in nonvalvular AF, and discusses the potential influence of dosing strategies on patient adherence and therapy persistence.
Acute and Extended Treatment of VTE
Current Anticoagulation Strategies and Understanding the Dosing Rationale of Direct OACs
Anticoagulation in patients with VTE aims to prevent thrombus extension and embolism and recurrent events, including potentially fatal PE. 6 The traditional strategy is a dual-drug approach, usually with parenteral heparin (eg, LMWH) overlapping with and followed by a VKA (eg, warfarin). 18 –20 Anticoagulation is usually recommended for at least 3 months after an initial venous thromboembolic event, but the optimal duration of extended treatment is unknown, and depends on the individual benefit–risk balance for the patient (eg, nature of the first event, risks of recurrence, and bleeding). 20 Extended anticoagulation (>3 months) is beneficial in patients with VTE, and the direct OACs have demonstrated noninferiority in the reduction of recurrent venous thromboembolic events versus standard therapy. 26,27,29,32,33,36,38,39
Phase II dose-finding studies of anticoagulants usually focus on safety outcomes to ascertain the risk of bleeding across a range of doses and regimens. The phase II Oral Direct Factor Xa inhibitor BAY 59-7939 in patients with acute symptomatic Deep Vein Thrombosis (ODIXa-DVT) study in patients with a proximal DVT compared the efficacy and safety of rivaroxaban 10, 20, or 30 mg twice daily, or 40 mg od with a standard regimen of enoxaparin overlapping with and followed by a VKA during a 12-week treatment period. 40 All rivaroxaban twice-daily regimens achieved a reduction in patients’ thrombotic burden at day 21 (without recurrent symptomatic VTE or VTE-related death) that was similar to levels observed with standard therapy. 40 The improvement in the thrombus burden after 21 days of treatment was not statistically significant with od versus twice-daily regimens; recurrent venous thromboembolic events and major bleeding events were uncommon in all treatment groups. 40 These results supported the idea that a twice-daily regimen may intensify the efficacy of the initial treatment.
Recurrence rates after discontinuing anticoagulant therapy can be high (7% of patients experience a recurrent episode within 12 months), and the risk of recurrence can persist. 26,41,42 Extended treatment with anticoagulants is often required for those at high risk of recurrence. A second phase II dose-ranging study of rivaroxaban investigated the potential of od administration for extended treatment. 43 Patients with an acute symptomatic DVT were treated with rivaroxaban od at doses of 20, 30, or 40 mg or standard therapy for 12 weeks. The incidence of the composite of symptomatic VTE and asymptomatic deterioration in thrombotic burden was similar for all doses, as were rates of bleeding. 43 Based on these results and those of the ODIXa-DVT study, the rivaroxaban regimen used in the phase III EINSTEIN program consisted of an initial intensified treatment with 15 mg twice daily for 21 days, followed by 20 mg od for continued treatment. 26,27
The Botticelli study compared the efficacy and safety of 3 dosing regimens of apixaban (5 mg and 10 mg twice daily, and 20 mg od for 84-91 days) with standard LMWH/VKA therapy. 44 Efficacy and safety profiles were similar across the dosing regimens and did not differ significantly from standard treatment. 44 As a result, the treatment regimen tested in the phase III AMPLIFY study of apixaban consisted of the elevated (10 mg twice daily) regimen for intensified initial treatment followed by 5 mg twice daily. 29
Phase III clinical studies confirmed the efficacy and safety of the selected doses of apixaban (Apixaban for the Initial Management of Pulmonary Embolism and Deep-Vein Thrombosis as First-Line Therapy [AMPLIFY] and Apixaban after the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis with First-Line Therapy–Extended Treatment [AMPLIFY-EXT]), 29,38 dabigatran (Efficacy and Safety of Dabigatran Compared to Warfarin for 6 Month Treatment of Acute Symptomatic Venous Thromboembolism [RE-COVER], Phase III Study Testing Efficacy & Safety of Oral Dabigatran Etexilate vs Warfarin for 6 m Treatment for Acute Symp Venous Thromboembolism (VTE) [RE-COVER II], Twice-daily Oral Direct Thrombin Inhibitor Dabigatran Etexilate in the Long Term Prevention of Recurrent Symptomatic VTE [RE-SONATE], and Secondary Prevention of Venous Thrombo Embolism (VTE) [RE-MEDY]), 32,33,39 edoxaban (Hokusai-VTE), 36 and rivaroxaban (EINSTEIN DVT, EINSTEIN PE, and EINSTEIN EXT) 26,27 for the treatment and/or secondary prevention of VTE. In all phase III clinical trials for the treatment of acute VTE, the direct OACs showed noninferior efficacy compared with standard therapy. Rates of major bleeding were significantly reduced with rivaroxaban and apixaban in EINSTEIN PE 27 and AMPLIFY, 29 respectively. Each of these direct OACs also demonstrated superior efficacy and an acceptable safety profile compared with placebo during extended treatment to prevent VTE recurrence. 26,38,39 It should be noted, however, the study designs and patient populations differed between these studies.
Stroke Prevention in Patients With Nonvalvular AF
Anticoagulants reduce the risk of stroke and systemic embolism in patients with AF. 45 Guidelines recommend oral anticoagulation in patients with a CHA2DS2-VASc score ≥2, but it can also be considered in patients with a lower risk of stroke (CHA2DS2-VASc [Congestive heart failure, Hypertension, Age ≥ 75 years (2 points), Diabetes mellitus, Stroke or transient ischemic attack (2 points), Vascular disease, Age 65–74, Sex category (female)] score of 1). 46 It should be noted, however, that the CHADS2 scoring system ([Congestive heart failure, Hypertension, Age ≥ 75 years, Diabetes mellitus, Stroke or transient ischemic attack (2 points)]; instead of CHA2DS2-VASc) was used in phase III stroke prevention trials with the direct OACs. 30,31,34,35
Current Anticoagulation Strategies and Understanding the Dosing Rationale of Direct OACs
Phase III clinical trials of stroke prevention in patients with AF used both twice daily (dabigatran and apixaban) and od (rivaroxaban and edoxaban) regimens. Prevention of Embolic and Thrombotic Events in Patients With Persistent Atrial Fibrillation (PETRO), a double-blind, dose-escalating phase II study, tested 3 dabigatran twice-daily regimens (50, 150, and 300 mg) in patients with nonvalvular AF. 47 The 150 and 300 mg twice-daily regimens were effective in preventing thromboembolic events, although they were associated with a higher rate of total bleeding events than the 50 mg twice-daily regimen. 47 The 150 mg twice-daily regimen was selected together with a lower dosing regimen of 110 mg twice daily in an attempt to reduce bleeding events in the phase III Randomized Evaluation of Long Term Anticoagulant Therapy (RE-LY) trial. 30 For apixaban and rivaroxaban, the final doses tested in phase III trials were inferred from VTE treatment data, and indication-specific phase II dose-finding studies were not conducted. 48 For rivaroxaban, population pharmacokinetic modeling and simulation were also undertaken to assist in dose selection. 49 The plasma concentration–time profile simulated in patients with AF and normal renal function was similar to the profile estimated using a rivaroxaban 20 mg od regimen in patients with DVT. Further simulations focusing on patients with AF and moderate renal impairment (creatinine clearance of 30–49 mL/min) showed that, owing to decreased renal function and increased rivaroxaban plasma concentrations, the rivaroxaban 15 mg od regimen should reach a similar plasma concentration–time profile to that observed with the 20 mg od dose in patients with AF and normal renal function. 49 As a result, the rivaroxaban 20 mg od regimen (or 15 mg od dose in patients with creatinine clearance of 30–49 mL/min) was used in the phase III Rivaroxaban Once daily, Oral, Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF) study. 31 For edoxaban, 30 and 60 mg od doses were tested in the Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation–Thrombolysis in Myocardial Infarction 48 (ENGAGE AF-TIMI 48) trial. 35
Phase III trials revealed that rivaroxaban 20 mg (or 15 mg) od (ROCKET AF), 31 dabigatran 110 mg twice daily (RE-LY), 30 and edoxaban 30 mg od (ENGAGE AF-TIMI 48) 35 were as effective as dose-adjusted warfarin, whereas dabigatran 150 mg twice daily, 30 apixaban 5 mg twice daily (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation [ARISTOTLE]), 34 and edoxaban 60 mg od 35 showed superior efficacy to warfarin in preventing stroke and systemic embolism in patients with nonvalvular AF (Table 2). All direct OACs were associated with a significantly reduced risk of intracranial bleeding, one of the most feared complications of warfarin therapy. 30,31,34,35 However, gastrointestinal bleeding was significantly increased by edoxaban (60 mg od), dabigatran (150 mg twice daily), and rivaroxaban use. 30,31,35 Direct comparisons between these trials cannot be made because of the different study designs and patient populations. For example, ROCKET AF (rivaroxaban) enrolled higher risk patients with a mean CHADS2 score of 3.5, compared with 2.1 in RE-LY (dabigatran) and ARISTOTLE (apixaban), and 2.8 in ENGAGE AF-TIMI 48 (edoxaban). The time in therapeutic range (TTR) of international normalized ratios of warfarin therapy in these studies also varied between 55% in ROCKET AF, 64% in RE-LY, 62% in ARISTOTLE, and 65% in ENGAGE AF-TIMI 48 (Table 2). Further analyses of ROCKET AF and RE-LY found that the treatment effects of rivaroxaban and dabigatran, compared with warfarin, were consistent regardless of the trial center TTR levels. 53 –55
Summary of Phase III Studies of Rivaroxaban, Apixaban, and Dabigatran Versus Dose-Adjusted Warfarin in Patients with Nonvalvular Atrial Fibrillation. 30,31,34,35,50 –52
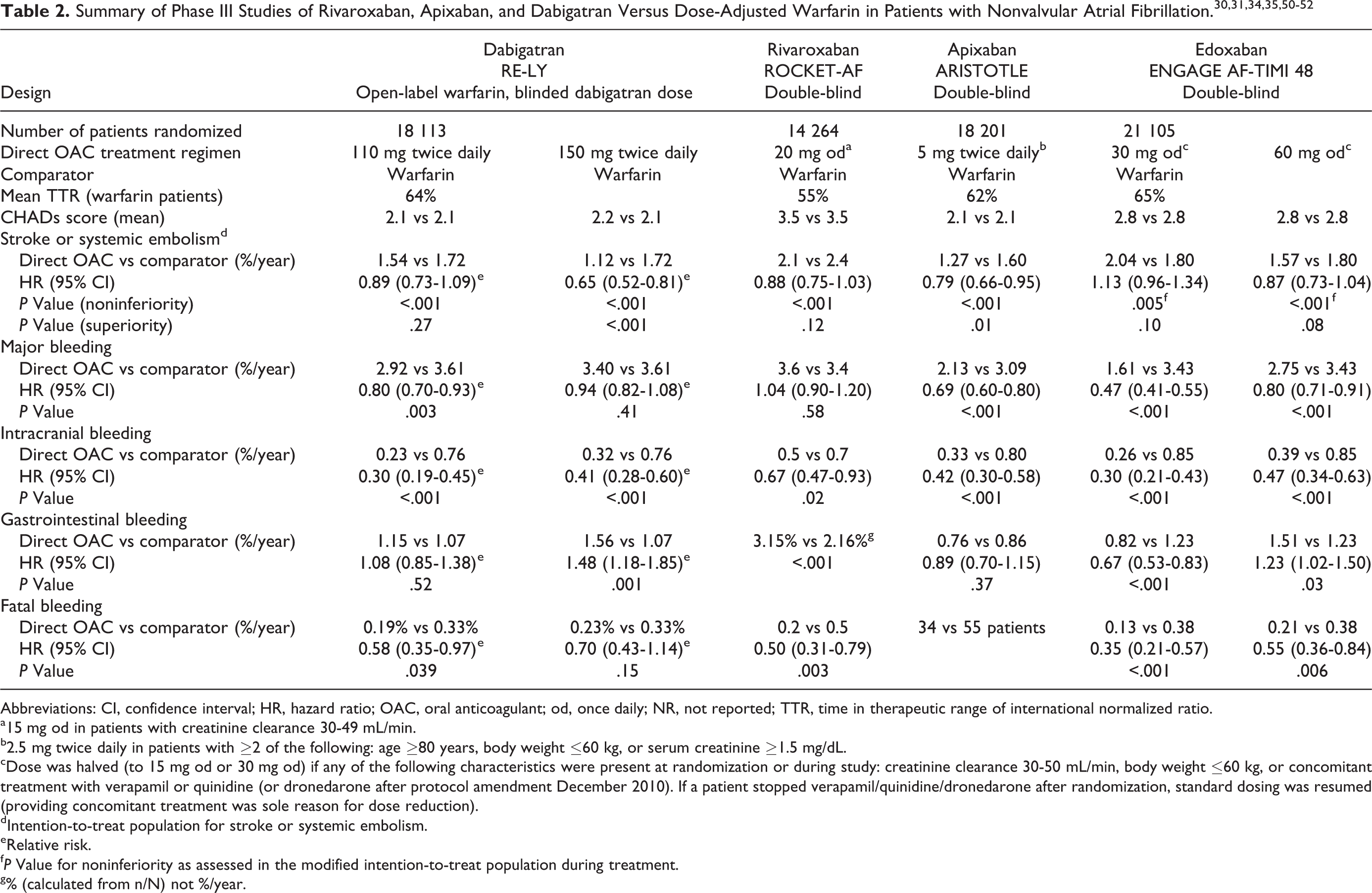
Abbreviations: CI, confidence interval; HR, hazard ratio; OAC, oral anticoagulant; od, once daily; NR, not reported; TTR, time in therapeutic range of international normalized ratio.
a15 mg od in patients with creatinine clearance 30-49 mL/min.
b2.5 mg twice daily in patients with ≥2 of the following: age ≥80 years, body weight ≤60 kg, or serum creatinine ≥1.5 mg/dL.
cDose was halved (to 15 mg od or 30 mg od) if any of the following characteristics were present at randomization or during study: creatinine clearance 30-50 mL/min, body weight ≤60 kg, or concomitant treatment with verapamil or quinidine (or dronedarone after protocol amendment December 2010). If a patient stopped verapamil/quinidine/dronedarone after randomization, standard dosing was resumed (providing concomitant treatment was sole reason for dose reduction).
dIntention-to-treat population for stroke or systemic embolism.
eRelative risk.
f
g% (calculated from n/N) not %/year.
Adherence and Persistence to Therapy in Clinical Practice
Adherence, or compliance, to medication is defined as the process by which patients take their medications as prescribed and includes 3 components: initiation (beginning with administration of the first dose), implementation (the extent to which the actual dose taken corresponds to the prescribed dosing regimen from the first to the last dose, and the intensity of drug use for the duration of therapy), and discontinuation (the end of therapy; administration of the last dose at the end of the treatment period or because of premature discontinuation). 56 Persistence describes the duration of time from initiation to discontinuation of therapy. 57 Typically, patients who have medication available >80% of the time (based on prescription refill data), or a medication possession ratio of 80% to 100%, are considered to have acceptable adherence for the treatment of cardiovascular conditions. 58
Adherence to medication in clinical practice has been estimated as being ≤80% 59 and can be affected by numerous factors (Table 3). 60 Depending on the type of treatment regimen, adherence may be harder to achieve. For example, a single-drug, od, oral regimen will be easier to adhere to than a complex regimen with 2 or more drugs, food and drug interactions, and different routes of administration (Table 4). Estimates of adherence to short-term therapy range between 70% and 80%, but long-term therapy is associated with much lower levels of approximately 40% to 50%. 60 In a study of adherence in patients prescribed warfarin, approximately 92% of patients had ≥1 missed or extra bottle opening, 36% of patients missed >20% of bottle openings, and 4% of patients had >10% extra bottle openings. 61 This poor adherence had a significant effect on anticoagulation control: patients who missed ≥20% of bottle openings were at increased risk of subtherapeutic coagulation (adjusted odds ratio, 2.10; 95% confidence interval [CI] 1.48-2.96), and those who had >10% of extra bottle openings were more likely to be over-anticoagulated (adjusted odds ratio, 1.73; 95% CI 1.09-2.74). 61 In routine clinical practice, warfarin therapy has been shown to be associated with poor treatment persistence. Recent analyses of real-world data from claims databases have shown that rivaroxaban is associated with better treatment persistence than warfarin in patients with AF. 62,63 Dabigatran has also been shown to have higher persistence than warfarin in patients with newly diagnosed AF. 64
Factors Influencing Adherence. 60

Dosing Strategies and Impact on Adherence.

Potential Impact of Dosing Strategies on Adherence and Persistence
Patients receiving medications for cardiovascular diseases showed higher adherence with od compared with more frequently scheduled dosing regimens. 65,66 A meta-analysis of dosing frequency and adherence specific to oral therapies demonstrated significantly higher adherence rates for od regimens. 67 Lower health care resource utilization costs were also associated with od regimens compared with multiple dosing over a range of acute and chronic diseases. 67
Nonadherence leads to poor clinical outcomes, higher rates of hospitalization, and increased financial burden. 60 In a UK analysis of hypertension drug regimens, hospital admission costs were indirectly linked to adherence when it was shown that serious cardiovascular outcomes occurred less frequently in patients taking a single-tablet regimen compared with patients receiving individual component therapy. 68 An inversely proportional correlation between dosing frequency and adherence to medication has been established for a diverse range of medical conditions, including patients with cardiovascular disease. 65,66,69 –73 A review of 51 studies of patients with chronic conditions showed that the “taking adherence” (number of bottle openings divided by the prescribed number of doses) was 93.0% for od dosing (95% CI 91.2-94.7) versus 85.6% for twice-daily dosing (95% CI 82.5-88.8). 73 In 29 chronic cardiovascular disease studies, od dosing regimens had higher adherence compared with twice-daily and 3-times-daily regimens by approximately 14.0% and 27.5%, respectively. 65 Significant improvements in adherence and a reduction in health care resource utilization and costs were also observed when reducing the frequency of oral therapies (from multiple dosing to od dosing) across acute and chronic disease conditions. 67
There are currently limited real-world data comparing adherence and use of the direct OACs with standard therapy. 74 Use of a direct OAC (either od or twice daily) is currently mentioned by the European Heart Rhythm Association guidelines as a plausible strategy to optimize adherence. 75 It has been reported that an od dosing regimen has been associated with higher adherence than a twice-daily regimen in patients with cardiovascular disease, 66 as well as in patients with AF receiving medication for diabetes or hypertension. 76 Several studies have reported real-life persistence with the direct OACs. Data from the Dresden new oral anticoagulant registry showed low rates of rivaroxaban discontinuation in the daily care of unselected patients with AF, with 90.9% of patients remaining on therapy after the first 6 months of treatment, and an overall discontinuation rate of 13.6/100 person-years. 77,78 In addition, data from this registry also demonstrated that 85% of patients receiving rivaroxaban for the treatment of VTE were still taking rivaroxaban at 6 months, indicating a high persistence rate. 79 A cross-sectional cohort study of patients receiving dabigatran (administered twice daily) in a real-life setting showed that 30% of patients had missed their medication and 12% had inadequate adherence. 80 Elsewhere, the use of dabigatran in patients with AF has been shown by real-world data to be well tolerated and effective compared with warfarin, and associated with rates of discontinuation ranging from 3.2% to 6.5% for 110 mg and 150 mg twice-daily doses, respectively, and 25.8/100 patient-years overall. 81,82 However, it should be noted that the reported persistence (or discontinuation) rate with dabigatran may not be directly related to its dosing regimen (ie, twice daily); in the RE-LY clinical trial, dyspepsia was the most frequently cited adverse event with dabigatran use (11.8% for 110 mg dose and 11.3% for 150 mg dose, compared with 5.8% for warfarin), which was thought to have contributed to its discontinuation rates (15% to 16% at 1 year). 30
When choosing between direct OACs, differences between the dosing regimens should be considered as one factor based on the physician’s understanding of the specific patient’s history of compliance with medications, as well as other factors such as the patient’s renal function, comorbidities, and preference, particularly for patients requiring long-term therapy. 48 The od dosing regimens may be associated with improved adherence in patients receiving chronic anticoagulation therapy for VTE (39% to 61% higher likelihood of adherence) or nonvalvular AF (26% higher likelihood of adherence) compared with twice-daily regimens. 76,83 Simple and convenient therapy offered by direct OACs may change patients’ attitudes toward anticoagulant use and improve adherence to treatment. This will assist elderly patients, who are more likely to have multiple comorbidities and take concomitant medicines, and those who require long-term anticoagulant treatment. 84
Treatment satisfaction is also an important patient-reported outcome to consider in clinical studies. Low treatment satisfaction could lead to lower adherence and therapy persistence. Data from subanalyses of the open-label EINSTEIN DVT and EINSTEIN PE studies showed that rivaroxaban was associated with clinically significant improvements in patient-reported benefits and burden compared with standard therapy. 85,86 This reported benefit was consistent over 12 months in patients with DVT, suggesting that adherence was maintained. 85
It is important to emphasize that pharmacological data do not predict clinical outcomes. 87 The od dosing regimens selected for rivaroxaban were based on data from phase II and phase III clinical studies and the expectation that od regimens would provide a simple and convenient administration, potentially improving adherence. Although all the direct OACs have relatively short half-lives (apixaban ∼12 hours, edoxaban 10-14 hours, dabigatran 12-14 hours, and rivaroxaban 5-13 hours), dabigatran and apixaban are given twice daily for the treatment of VTE and for stroke prevention in AF, whereas rivaroxaban and edoxaban are given od. 23 –25,88 The inhibitory constants (Ki) for factor Xa (apixaban: 0.08 nmol/L, edoxaban: 0.56 nmol/L, and rivaroxaban: 0.4 nmol/L) and thrombin (dabigatran: 4.5 nmol/L) are low. 89 –92 For example, the plasma levels of rivaroxaban at 24 hours after dosing (ie, Ctrough levels prior to the next dose) are estimated to be well above the Ki value (ie, concentrations required to cause 50% inhibition of factor Xa). 93 There is currently no clinical evidence to support improved outcomes with twice-daily dosing relative to od dosing in a situation of a missed dose, and further studies and data from routine clinical practice would be needed to provide evidence and insights. Nevertheless, all direct OACs have demonstrated efficacy and safety in clinical trials regardless of whether dosed od or twice daily. 26,27,29 –36,38,39
Conclusion
Several direct OACs are being incorporated into routine clinical practice and present simplified and effective therapeutic options for the management of several thromboembolic disorders. These agents are given either od or twice daily, with specific regimens based on data from dose-finding studies and/or pharmacokinetic modeling, and subsequently confirmed in large, randomized phase III trials. Because of their relatively short half-lives (eg, compared with VKAs), strict adherence to the respective dosing schedule is essential to maintain effective anticoagulation. Adherence and persistence may differ in real-world versus strictly controlled clinical trial settings. Evidence suggests that od dosing regimens have better adherence levels compared with twice-daily dosing regimens. However, there are many factors that can influence adherence to therapy and treatment persistence in routine clinical practice. When choosing a direct OAC for a patient, the dosing regimen should be considered alongside other aspects affecting the suitability of one agent over another, including efficacy and risk of bleeding, and the patient’s renal function and comorbidities. 94 –96 Patient education and regular follow-ups are also important to ensure good adherence and optimal clinical outcomes.
Footnotes
Acknowledgments
The authors would like to acknowledge Emma Marlow, who provided editorial support with funding from Bayer HealthCare Pharmaceuticals and Janssen Scientific Affairs, LLC.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article; Professor Alpesh Amin is a consultant and speaker for Pfizer, Bristol-Myers Squibb, and DSI. Professor Joel C. Marrs has no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial support was funded by Bayer HealthCare Pharmaceuticals and Janssen Scientific Affairs, LLC.
