Abstract
Thromboembolic disease is a common complication of hip fracture in the elderly. Anticoagulants represent a standard of care in preventing postoperative thrombotic complications following surgical fixation. We asked whether levels of antibody to heparin–platelet factor 4 (PF4) complex were differentially present in unfractionated heparin (UFH) versus Enoxaparin, following hip fracture and whether one particular subtype of antibodies was more prevalent. Plasma samples from elderly patients sustaining a hip fracture treated with either enoxaparin or UFH were collected pre- and postoperatively and analyzed using enzyme-linked immunosorbent assay (ELISA) sandwich method for the prevalence of antiheparin-PF4 antibodies and later subtyped. The prevalence of antiheparin-PF4 antibodies was higher in the UFH group especially on postoperative day 7. Patients treated with UFH showed a greater prevalence of antiheparin-PF4 antibodies and a greater prevalence of immunoglobulin G (IgG) subtype. Heparin and enoxaparin are capable of generating heparin-induced thrombocytopenia (HIT) antibodies in elderly patients undergoing orthopedic surgery but perhaps not to the same extent. When comparing low-molecular-weight heparin (LMWH) with UFH, the incidence of new antiheparin-PF4 antibody production is higher in patients treated with UFH.
Keywords
Introduction
Hip fracture continues to present a major public heath concern carrying considerable morbidity and mortality. Orthopedic intervention in this population often leads to postoperative complication, including thromboembolism. Between 1986 and 2005, the annual mean number of hip fractures in patients over age 65 was 957.3 per 100 000. 1 The incidence of hip fractures worldwide in 1990 was 1.6 million, with a projected incidence in 2050 of 6.26 million. The average age of occurrence is 77 years for women and 72 years for men. With the growing aging population, reducing the risks following hip fracture remains of paramount concern. 1 In stratifying risk of venous thromboembolism, hip fracture repair presents the highest risk category with fatal pulmonary embolism (PE) in 4% as opposed to elective arthroplasty (0.3%-0.4%).
Without thromboprophylaxis, recent estimates suggest that nearly 60% of elderly patients undergoing orthopedic surgery may experience postsurgical thromboembolic complications in the form of deep venous thrombosis (DVT) orPE. 2 Of these, 5% go on to have fatal complications. Unfractionated heparin (UFH) and low-molecular-weight heparins (LMWHs) are often administered2–4 as the standard of care in prophylaxis to reduce the perioperative risk by approximately 40%. While they apparently reduce the thromboembolic risk, they increase bleeding complications and may induce thrombosis in certain cases and must be monitored closely. 5
Orthopedic surgical interventions often result in activation of hemostatic and inflammatory processes contributing to the pathophysiology associated with thrombotic and vascular complications observed in these patients. Antibodies to the heparin–platelet factor 4 (PF4) complex (HIT antibodies) have been observed in up to 30% of patients treated both UFH and LMWH. 6 These antibodies are involved in the HIT syndrome and have been shown to activate platelets and endothelial cells leading to thrombosis. 7 Nonfunctional HIT antibodies are also generated in these patients, which may contribute to adverse outcomes even in patients without the HIT syndrome for several weeks.8,9 Torigoshi et al demonstrate that the incidence of DVT was significantly higher in the seroconverted patients treated with UFH as compared with that of the nonseroconverted, with a higher titer in those that were symptomatic. 8
To date, no study has established the difference in presence of antibodies to heparin-PF4 complex between UFH and LMWH in hip fracture patients. While it has been demonstrated that the immunoglobulin G (IgG) subtype of HIT antibody is more pathologic, no study has examined its presence in orthopedic patients undergoing chemoprophylaxis for hip fracture.10,11 In this study, we evaluated the levels of antibodies to heparin-PF4 complex in patients with hip fracture undergoing prophylaxis with either UFH or LMWH and the differential level of Ig subtypes. Reducing thromboembolic complications following hip surgery is of utmost concern in orthopedic surgery. The mere presence of antiheparin-PF4 antibody even in the absence of heparin-induced thrombocytopenia (HIT) can increase thromboembolic complications.
Materials and Methods
Plasma samples were taken from an open prospective randomized control study, PK-532 (n 341), in which patients aged 18 and over presenting with acute fractures of the femoral neck and/or fractures of the upper third of the femur were included. Exclusion criteria were a high risk of bleeding contraindicating the use of LMWHs, active bleeding, severe liver or renal disease, contraindications for use of heparin or enoxaparin, breastfeeding, or pregnancy. The patient makeup consisted of female and male participants that were treated with either enoxaparin (40 mg once daily) or UFH (5000 IU twice daily). The frozen plasma samples were randomly selected for analysis from each group (N = 50 for both UFH and enoxaparin). The first thromboprophylactic doses were given immediately after patient inclusion in the study and continued for 7 ± 2 days after surgery or until full mobilization.
Blood samples were collected prior to surgery and at days 1, 3, 5, and 7 postoperatively and frozen at −70°C. The average age for the group receiving enoxaparin was 77.1 (±10.1) with 39% of these patients being male. In the UFH group, the average age was 75.5 (±12.7), with 51% of the patients being male. These samples were selected randomly and no prior preferences for sample selection with regard to gender were made. These samples were sent to Loyola University Chicago, Hemostasis & Thrombosis Laboratories from the Department of Orthopedics, Arhus University Hospital, Arhus; Copenhagen University Hospital, Hillerad; and Aalborg University Hosptial, Aalborg, Denmark.
Patient plasma samples chosen for this current study were selected at random. The goal of this study was to detect, quantify, and compare levels of antibodies to heparin-PF4. For this reason, 50 patients were analyzed in both the UFH and enoxaparin groups. Subset analysis was performed only for plasma samples with a positive antiheparin-PF4 antibody response. Investigators were blinded to the information at selection and the key was e-mailed from the collecting site principal investigator. From there, the samples were selected at random to reach equal numbers for the 2 groups. The samples were divided in their respective enoxaparin and UFH groups. No other information was available to investigators or technicians in the laboratory on the subdivision of these samples.
Frozen plasma samples were thawed in batches and analyzed for the prevalence of antiheparin-PF4 antibodies using a commercially available enzyme-linked immunosorbent assay (ELISA) method (GTI, Milwaukee, Wisconsin). A wash buffer, a substrate solution, human negative, and positive controls were prepared as directed by the kit. Samples were diluted with a calibrator diluent in a 1:200 ratio. Three hundred microliters of working wash solution was added to all wells for 10 minutes, then aspirated and decanted. Fifty microliters of the standard or sample solutions were placed in the appropriate antibody precoated microtiter wells and allowed to incubate for 40 minutes with the exception of blank wells at 37°C. After washing, 50 μL of the conjugate solution was added and allowed to incubate for 40 minutes. After a final wash, 100 μL of the substrate solution was added to the wells and allowed to incubate for 30 minutes at room temperature. Following this, a stop solution was added and the microtiter was measured at an absorbance of 405 with a reference filter of 490 nm with blank wells subtracted from overall optical density (OD). Test results of greater than 0.40 were regarded positive results.
Antibodies were also subtyped for heparin-dependent antibodies using an ELISA method-specific isotyping assay (Anira Columbus, Ohio). This method used different capture probes in the sandwich ELISA methods. After thawing of samples at room temperature, cell lysate, substrate, conjugate, positive IgG, IgA, IgM controls, and negative controls were prepared according to the kit instructions. Samples were diluted with calibrator diluent at 1:100 ratio. Fifty microliters of cell lysate was introduced to the micro ELISA plate wells. Two hundred microliters of positive control of IgG, IgA, IgM, negative controls, and diluted sample were added to the cell lysate with the exception of 2 blanks and incubated for 60 minutes at room temperature (18°C-25°C). Following successive washes, 200 μL immunoconjugate was added in the micro ELISA plate wells and incubated for 60 minutes at room temperature. After successive washing, 200 μL substrate was added into the wells and color allowed to develop for 5 minutes. Fifty microliters of stop sulfuric acid was then added at the same interval as the substrate and read after 10 minutes at 450 nm (A450) and blanks subtracted.
As a realistic negative control, HIT antibody levels were compared from an additional 26 plasma samples (12 males and 17 females) from normal human volunteers that were obtained along with a previously collected pool of normal human plasma (NHP) from healthy volunteers of age 20 to 55. Additionally, a patient not undergoing hip fracture with known levels of HIT antibodies was analyzed as a positive control and compared with the provided positive controls. Prior to analysis, these samples were thawed and mixed well at room temperature.
The data on each individual was analyzed in terms of cumulative mean ± SD and the limits. Individual groups were compared using analysis of variance (ANOVA), correlation coefficients were determined comparing various parameters.
Results
Among the hip fracture surgery patients in our study, the prevalence of antibodies heparin-PF4 was higher in the UFH group as measured by average OD (P < .05). Only values greater than the designated cutoff of .40 OD A405 was designated as positive for PF4 antibody. The HIT antibody titer was directly proportional to the absorbance values. The elevation in HIT antibodies was especially evident on postoperative day 7 (see Figure 1 ). Four patients were positive at baseline and remained positive throughout. Besides the 4 positive at baseline, 5 (total 9[18%]) of 50 patients treated with enoxaparin were ELISA positive by standardized cutoff of OD at any time point for antibodies to heparin-PF4 complex. This is compared with 16 ([32%] 4 positive at baseline and 12 additional positive) of 50 patients analyzed in the UFH group. Control normal human plasma demonstrated no antibodies to heparin-PF4 complex as judged by absorbance values. The positive control showed a high titer of antibodies that were not reached at any postoperative day even among positive patients. Normal plasma controls were negative with none having OD over 0.4 and an average OD of 0.29. The patient analyzed for positive control had an OD positive throughout at an average of 1.3 OD.
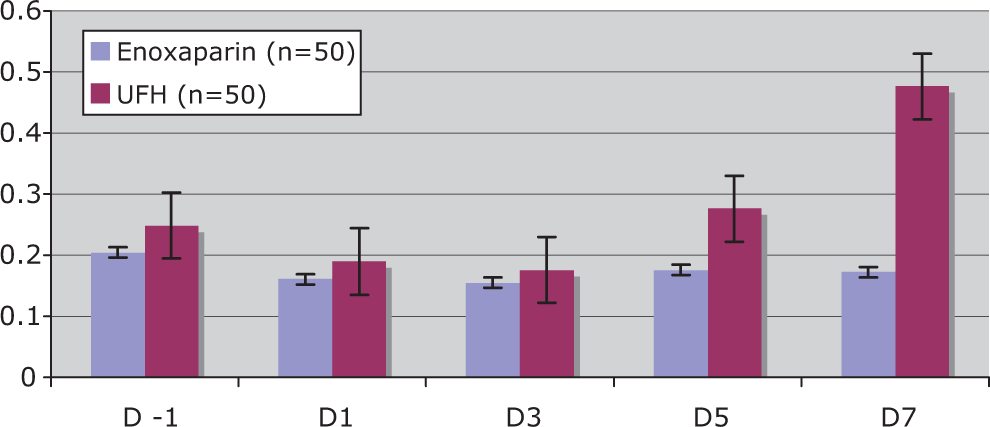
Heparin-induced thrombocytopenia antibodies in hip fracture. X-axis denotes the days at blood draw, and Y-axis denotes absorbance at 429 nm with a spectrophotometer.
Subtyping studies on positive samples revealed that the IgG subtype was more prominent in UFH-treated patients compared to those treated with enoxaparin (P < .05). Again these antibodies were especially prevalent on postoperative day 7 (see Figure 2). Of the total 25 patients that had a positive antiheparin-PF4 antibody response from the total 100 patient plasma samples analyzed in our study, 10 were positive for IgG subtype. In the UFH group, those samples that were positive for antiheparin-PF4 antibody exhibited a higher proportion of IgG subtype antibody (56%) in contrast to enoxaparin which had much lesser (11%) of IgG antibodies. Of the 25 patients, 7 were positive for IgA, with 4 also being positive for IgG. Eight were positive for IgM, with only 1 being positive for IgA and none positive for IgM. Each patient was analyzed only at time points in which they were positive by global GTI methods, as mentioned above. The positive control was positive for IgG but not for IgA or IgM, while negative controls run were not positive for IgG, IgA, or IgM.
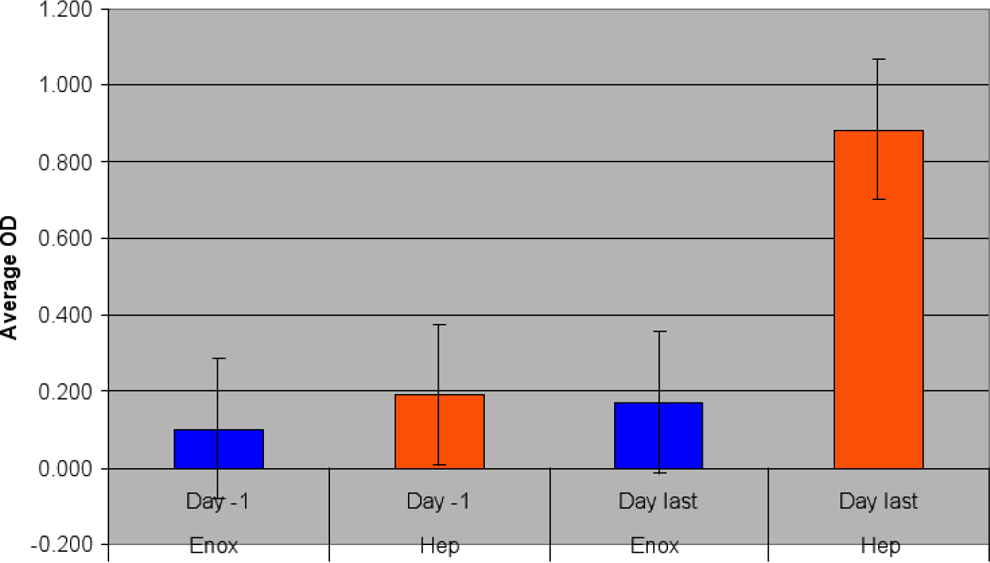
Hyphen Isotype IgG Average OD heparin vs. enox treated patients.
Discussion
The purpose of this study was to examine the difference in HIT antibody generation in hip fracture patients treated with LMWH and UFH in order to determine whether one drug may be more safe in avoiding thrombosis associated with these antibodies. The results demonstrate that LMWHs notably induced less of a production of antibodies to heparin-PF4 including dramatically less of the potentially harmful IgG subtype in hip fracture patients. In making decisions about prophylaxis in hip fracture, these factors must be considered.
A limitation of our study is that patients were not separated by age, severity of fracture, method of surgical reduction, or gender. Further subgroups may have revealed patterns of antibody production further. While the purpose of the study was to examine general drug effects in hip fracture patients, these factors may in fact play a role and this may limit the extent to which our findings can be applied in specific clinical situations. Also this study does not include whether patients were naive to heparins prior to inclusion in the study. Four patients were positive for antibodies to heparin-PF4 on day 1. As a result, this may obscure the extent to which individual drugs instigated the response.
As previously reported in HIT patients, the platelet count falls 4 or more days following the onset of heparin therapy. 9 This is consistent with the increasing prevalence of antibodies and peak on day 7 seen in our study especially in the UFH group (Figure 1). Our finding of 32% of hip fracture patients seroconverting to HIT-positive antibodies after prophylaxis with UFH is consistent with previous retrospective study demonstrating that 34% of patients seroconvert even when there is no evidence of thrombocytopenia. 8 Nearly 16% of these patients exhibited symptomatic DVT. Similar to previous reports in hip fracture patients, the antiheparin-PF4 titer was higher on day 7.
Furthermore, type II HIT is a prothrombotic with thrombosis occuring due to platelet activation from antibodies to heparin resulting in immunogenic complex formation of heparin and PF4.6,10,11 Laboratory analysis of LMWHs has revealed that they only induce antibodies to PF4 1.5% of the time and less commonly instigate a thrombotic response with IgG subtype. 12 Our analysis of LMWH in orthopedic patients showed a higher rate of antibody development than previously reported (18%), but still lower than that of UFH (32%). Moreover, the IgG subtype was consistent with the previous reports and was very low in the enoxaparin group.
Randomized controlled trials show a consistently lower rate of DVT for LMWHs when compared to UFH.3,13,14 This prevalence of increased HIT antibodies, coupled with higher IgG levels in UFH-treated hip fracture patients, may offer an explanation as to why this effect occurs. Our study evaluated the differential antibody response for relative levels of antiheparin-PF4 antibodies among the UFH and enoxaparin groups, but clinical outcomes in terms of safety and efficacy of drugs were not measured as our study had a small sample size; and correlation with clinical findings may not be accurately determined. There is a need for future study of newer anticoagulants such as newer anti-Xa and anti-IIa inhibitors to determine their interaction with antibodies to HIT and to see whether they offer an even lower rate of IgG antibodies, thereby leading to lower rates of thrombosis.
In summary, hip fracture patients undergoing surgery are regularly treated with different agents of prophylaxis to thromboembolic complications. These agents possess varying ability to cause a patient to seroconvert to HIT antibodies which may promote thrombosis for weeks following such seroconversion. This may contribute to increased DVT seen in patients treated with UFH versus enoxaparin. Additional research may clarify the prophylactic agent posing the least risk of HIT antibody IgG production to promote better outcomes in orthopedic surgery patients.
Clinical Implications
Orthopedic interventions have classically led to postoperative venous thromboembolic complications despite prophylaxis with anticoagulants. With the establishment of differential quantification of antibody production between these two commonly utilized anticoagulants, one may be able to help surgeons select which drug will be safer in patients with hip fracture and perhaps design agents to further decrease the risk of VTE following surgical intervention. Because it is established that HIT antibodies, specifically IgG subtype causes patient thrombotic events to increase, it is important to understand the potential risks of these events between drugs and perhaps monitor more closely the antibody levels in patients treated with UFH. Reducing the number of fatal and nonfatal thromboembolic complications within orthopedic surgery patients becomes even more crucial as the population continues to age and the number of hip fractures remains proportional.
Footnotes
Acknowledgment
This study was funded by the internal research funds of the Hemostasis and Thrombosis Research Laboratories, Loyola University Chicago and Cardiovascular Diseases and Educational research funds (CDER), Cypress. We are grateful to Dr Richard Gameli, Dean of Stritch school of Medicine and doctors Richard Kennedy and Linda Brubaker of the office of research services for their support and encouragement for the completion of this study.
Ethical Board Review statement: Each of the sites that recruited the patients in this study were individually reviewed and approved by the local IRB. This work was performed at Loyola University Chicago Stritch School of Medicine.
There is no conflict of interest for any of the authors in relation to any aspect of this study.
This study was funded by the internal research funds of the Hemostasis and Thrombosis Research Laboratories, Loyola University Chicago and Cardiovascular Diseases and Educational research funds (CDER), Cypress.
