Abstract
The primary objective was to evaluate the response rate of rituximab therapy for children with chronic immune thrombocytopenic purpura (ITP) and Evans syndrome (ES) and immune reconstitution of these children after rituximab therapy. Eleven patients with chronic ITP and 2 with ES between 6 and 18 years of age and platelet count less than 20 × 109/L received rituximab. Overall response (OR) was defined as an increase in platelet count above 50 × 109/L. The mean age of 13 children (9 girls, 4 boys) was 11.2 ± 3.8 years (6-18). One of the patients with ES had been splenectomized; others were not. The patients mean follow-up time was 10.3 ± 9.3 months after rituximab therapy. Two patients achieved complete response, 4 patients achieved partial response, and OR rate was 46% (6 of 13) after therapy. Seven patients have no response. In conclusion, rituximab may be considered prior to splenectomy in children with chronic ITP and ES with an acceptable toxicity profile.
Introduction
Immune thrombocytopenic purpura (ITP) is an immune-mediated disease characterized by transient or persistent decrease in platelet count and increased risk of bleeding. 1 The term “chronic ITP” is used for patients with ITP lasting for more than 12 months. 2 First-line treatment options including steroids, intravenous immunglobulin (IVIG), and anti-D immunoglobulin can increase platelet counts in more than 60% to 80% of patients. 3 Since pediatric patients with ITP have a risk of postsplenectomy sepsis, rituximab is a treatment option, increasingly preferred before splenectomy. 4
Rituximab, humanized chimeric monoclonal antibody, is directed against the CD20 antigen expressed on B lymphocytes. The mechanism of action relies on the basis of rapidly depletion of B lymphocytes by complement-mediated lysis, antibody-dependent cytotoxicity, induction of apoptosis, and inhibition of B-cell proliferation. 5 In English literature, there is limited number of studies about rituximab therapy and immune reconstitution after the therapy in children with chronic or refractory ITP. These studies have shown variable response rates to rituximab therapy ranging from 20% to 69%. 6 –9
In our center, rituximab therapy is preferred before splenectomy for children with chronic ITP and Evans syndrome. Thus, we decided to evaluate the response rate of rituximab therapy for children with chronic ITP and Evans syndrome, and immune reconstitution of these children after rituximab therapy followed-up in our center.
Materials and Methods
Eleven patients with chronic ITP and 2 patients with Evans syndrome treated with rituximab therapy between September 2008 and December 2011 at Ankara Children’s Hematology and Oncology Hospital were evaluated in this study. Patients of 6 to 18 years of age with the diagnosis of chronic ITP following for more than 12 months had used more than 1 first-line treatment options (steroid, IVIG, and anti-D immunoglobulin) for ITP, and having platelet count less than 20 × 109/L were eligible for rituximab therapy. Patients in the study group were refractory to all treatment options prior to rituximab therapy. All had previously received one or more courses of IVIG or methylprednisolone or anti-D immunoglobulin treatments. One patient with Evans syndrome had been splenectomized and had received immunosuppressive medications of cyclosporine A and azathiopurine.
Rituximab (anti-CD20, F. Hoffmann-La Roche, Basel, Switzerland) was given by intravenous infusion at a dosage of 375 mg/m2 for 4 consecutive weeks. All patients were premedicated with paracetamol and diphenhydramine before administration of rituximab. All infusions were administered at hospital. Side effects during infusion were recorded and treated symptomatically. Patients were followed regularly with complete blood counts, peripheral blood smears (absolute numbers of T and B lymphocytes), peripheral lymphocyte subsets (by flow cytometry), and serum immunoglobulin levels. Every patient’s bleeding score was assessed by Buchanan and Adix scoring system at treatment and during follow-up period. 10
Before the treatment, written informed consent was obtained from all patients and/or parents.
Definitions
Overall response (OR) was defined as an increase in platelet count above 50 × 109/L at least for 7 days. Response was classified as complete response (CR) if the platelet count was above 150 × 109/L, partial response (PR) if the platelet count was between 50 × 109/L and 150 × 109/L, minimal response (MR) if the platelet count was between 20 × 109/L and 50 × 109/L, and no response (NR) if there was no change from the baseline platelet count. Relapse was defined as a decrease in the platelet count to below 50 × 109/L again.
Statistics
Analysis of data was primarily descriptive, using standard deviations, ranges, mean, and median values. Wilcoxon rank sum test was used to asses any association between demographic and clinical characteristics of nonresponder and responder patients. Baseline and second, third, fourth weeks, and third month lymphocyte subsets and immunoglobulin levels were compared using Wilcoxon signed rank test. All analyses were performed using SPSS version 15.0 for Windows (SPSS Inc, Chicago, Illinois). P value ≤.05 is considered as statistically significant.
Results
Eleven patients with chronic ITP and 2 patients with Evans syndrome were evaluated in this study. The mean age of 13 patients (9 females and 4 males) was 11.2 ± 3.8 years (median: 10; 6-18 years). Patients’ clinical characteristics are presented in Table 1. Their follow-up period was 50.0
Patients’ Clinical and Demographic Characteristics.

Abbreviations: ITP, immune thrombocytopenic purpura; IVIG, intravenous immunoglobulin; OR, overall response; CR, complete response; PR, partial response; NR, no response.
After the rituximab therapy, 2 patients achieved CR, 4 patients achieved PR, and OR rate was 46%. One of the complete responders was a child with Evans syndrome who had a relapse 7 months after rituximab therapy, and he needed a second course of rituximab therapy. He achieved second CR with 3 infusions of rituximab and still followed up in CR for 6 months. Of the 4 partial responders, the first one had a relapse 5 months after rituximab therapy and required additional therapy. For the second one with Evans syndrome, the autoimmune antibodies (antiphospholipid, anticardiolipin, and anti-double-stranded DNA) were determined during the follow-up period. She relapsed at 13th month after rituximab therapy and underwent splenectomy. The third patient had synovitis in the right hip during rituximab therapy and she received antibiotics for synovitis. The third and fourth patient’s platelet count increased at sixth month and they have still been in PR for 8 months. One of the 7 nonresponders underwent splenectomy after 9 months from rituximab therapy.
The patients’ mean follow-up period was 10.3 ± 9.3 months (median: 8.5, range: 3-36 months) after rituximab therapy. Median time to response was 5.5 weeks (mean: 10.6 ± 10.4; range: 2-24 weeks) among all responders. Median time to response was 2.5 weeks (range: 2-3 weeks) in complete responders, whereas it was 15.5 weeks (range: 4-24 weeks) in partial responders (P ≤ .05). Response duration was median 6 months (range: 3-30 months) among all responders. Response duration was median 7 months (range: 3-24 months) in complete responders and 6 months (range: 5-30 months) in partial responders (P > .05).
The patients’ bleeding score was median 1 (range: 1-3). Five of the patients with moderate (grade 3) bleeding required supportive therapy. One of the patients with chronic ITP had recurrent otitis media and bronchiectasis which developed secondary to hypogammaglobulinemia after rituximab therapy and required 3 months of IVIG replacement therapy.
Comparison of responders and nonresponders to rituximab therapy has been shown in Table 2. There were no significant differences between responders and nonresponders with regard to age, gender, and platelet number before the therapy, duration of disease until the therapy, number, and response to previous treatments (Table 2).
Comparison of Responders and Nonresponders to Rituximab Therapy.
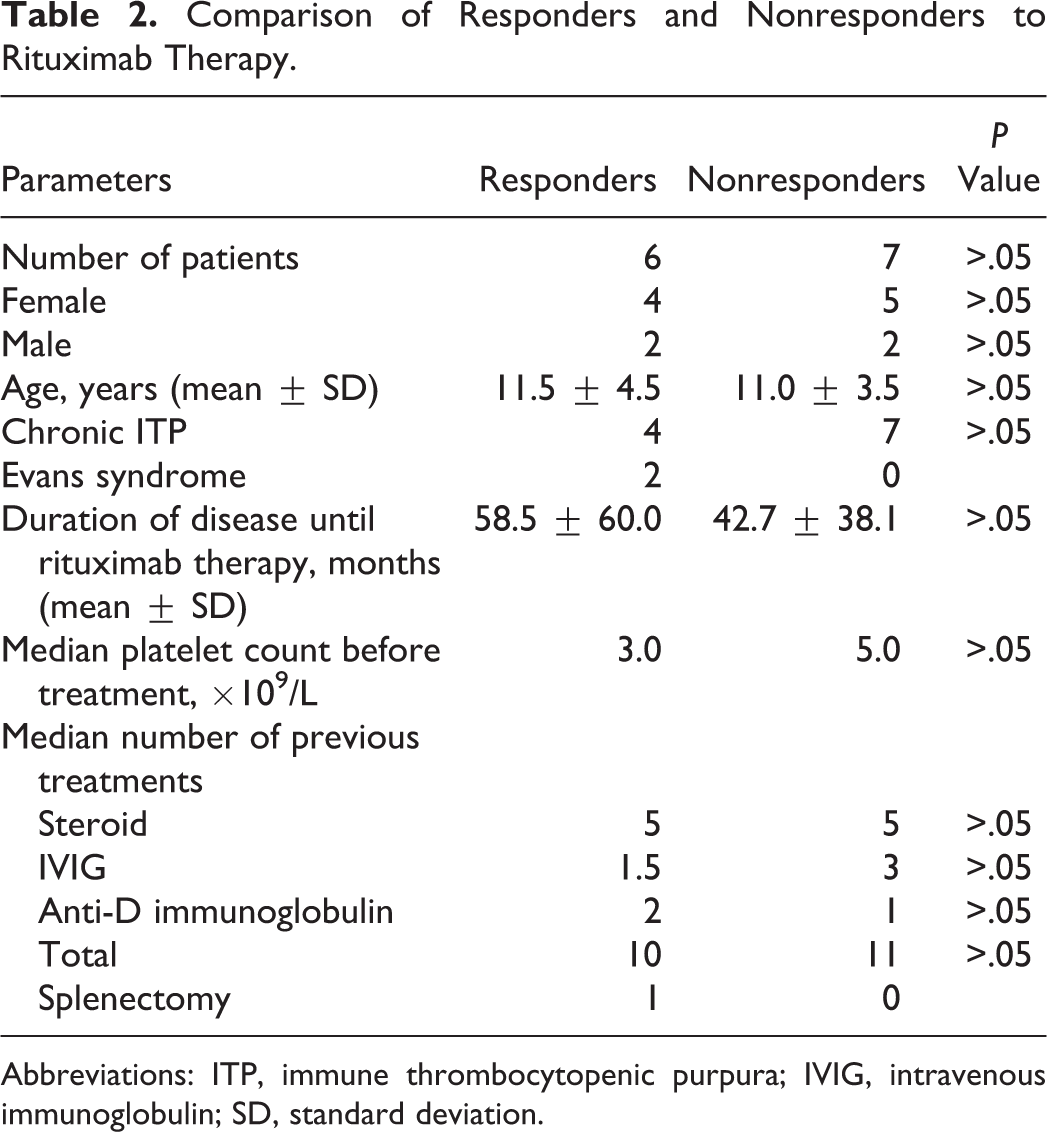
Abbreviations: ITP, immune thrombocytopenic purpura; IVIG, intravenous immunoglobulin; SD, standard deviation.
The patients’ peripheral lymphocyte subsets and serum immunoglobulin levels were presented in Table 3. CD19+ B lymphocytes decreased dramatically from 21.2% to 0.04% after the first infusion of rituximab therapy (P < .05), whereas B lymphocytes increased to 8% after 3 months of therapy (P > .05). No significant change was observed in other lymphocyte subsets. In all patients, the serum immunoglobulin (Ig) G and/or IgM levels decreased after 3 months of rituximab therapy. However, the decrease was statistically significant only for IgG levels at 3 months after the therapy (P < .05). At the same time, it is remarkable that the mean IgG level was still above 400 mg/dL after 3 months of the therapy.
Patients’ Peripheral Lymphocyte Subsets and Serum Immunoglobulin Levels.
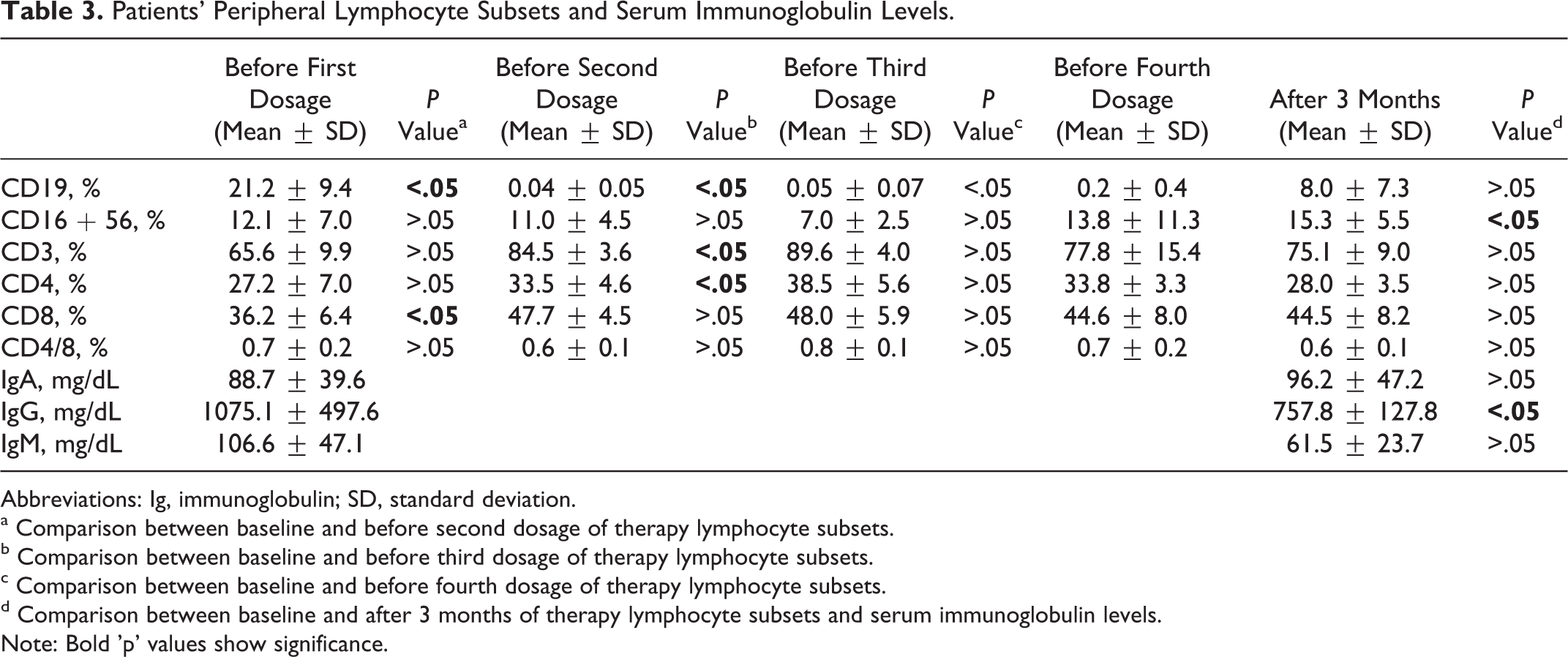
Abbreviations: Ig, immunoglobulin; SD, standard deviation.
a Comparison between baseline and before second dosage of therapy lymphocyte subsets.
b Comparison between baseline and before third dosage of therapy lymphocyte subsets.
c Comparison between baseline and before fourth dosage of therapy lymphocyte subsets.
d Comparison between baseline and after 3 months of therapy lymphocyte subsets and serum immunoglobulin levels.
Note: Bold 'p' values show significance.
Discussion
Physician management of patients with chronic ITP is different. Neunert et al designed a study about physician attitudes regarding management of children with severe chronic ITP. In all, 33% of physicians stated that they would recommend splenectomy for a child with chronic ITP. Of those who did not recommend splenectomy, 67% reported that they would treat such a child with rituximab. If initial drug therapy failed, 47% would proceed with splenectomy. 11 In our center, rituximab therapy is preferred before splenectomy for children with chronic ITP and Evans syndrome. Thus, we planned to report our experience about rituximab therapy in our patients with chronic ITP and Evans syndrome.
In this study, among 13 children with chronic ITP and Evans syndrome treated with rituximab therapy; 2 achieved CR, 4 achieved PR, and OR rate was 46%. Additionally, no difference was detected between responders and nonresponders in respect to age, gender, duration of the disease until rituximab therapy, previous treatments, and platelet count before rituximab therapy. In a study performed by Bennett et al, platelet count of >50 000/mm3 was achieved in 31% of the patients. They also reported that attainment of the primary outcome was not associated with age, prior pharmacologic responses, prior splenectomy, ITP duration, screening platelet count, refractoriness, or IgM reduction. 6
Rituximab is a murine–human protein conjugate that can cause allergic reactions. Reversible serum sickness symptoms were reported previously in 12% of the patients. 6,12 In our series, rituximab was well tolerated and caused transient tachycardia and hypotension in only 1 patient (7.7%) during infusion despite the premedication. This mild infusion reaction was treated by additional diphenhydramine and methylprednisolone infusion. No delayed infusion reactions or serum sickness were observed in our patients group.
In a previous study performed in children with chronic ITP, the median time to response in children with ITP was 3.5 weeks and the majority of the patients responded within 8 weeks after 4 doses of therapy.6 In another study, the authors have suggested that some patients may achieve a quite delayed response time (as long as 1 year after therapy). 13 In our study, median time to response was 2.5 weeks (2-3 weeks) in complete responders, whereas it was 15.5 weeks (4-24 weeks) in partial responders (P ≤ .05). Thus, we thought that the pediatric hematologist should wait for a reasonable time period of at least 6 months after rituximab therapy before another treatment is planned.
There are some studies about the mechanism of rituximab therapy in patients with chronic ITP. In the previous studies, the authors showed that regulatory T cells have reduced their suppressive capacity and the number of these cells has decreased in patients with ITP with active disease. As a result, autoreactive T cells involved in the production of platelet glycoprotein IIb-IIIa autoantibodies have increased in number and produced autoantibodies in patients with ITP. 14 Regulatory T cells preserve their suppressive features as normal controls in patients with ITP responded to rituximab therapy. 15
Recently, North American Chronic ITP registry has showed that there is a correlation between response to steroids and response to rituximab in patients with chronic ITP. Published response rates of rituximab therapy vary widely and this might be resulted from wide range of steroid refractoriness of the study population in the studies. 6 –9 It was speculated that overlapping biology of glucocorticoids and rituximab can explain the situation. However, many patients with steroid refractory ITP still respond to rituximab therapy and this could not be explained exactly. 16 Recently, it was reported that the combination of low-dose rituximab and steroid therapies showed higher response rates. This combination has increased the number of regulatory T cells to higher levels and this increase continues for a long time. 17 In our study, 6 patients responded to rituximab therapy and 5 of them had also steroid response previously.
In our study, response duration was 3 to 30 months. Wang et al also reported similar response duration of 4 to 30 months. 12 In our study, 3 of 13 patients relapsed. One of the relapsed children with Evans syndrome received second course of rituximab therapy after 7-month duration of remission and CR was achieved again. Thus, durable response was maintained in 30.8% (4 of 13) of patients. Since early relapses may be seen in patients with ITP treated with rituximab therapy, retreatment with rituximab therapy may offer good results. 18 Among 2 other relapsed patients, 1 with Evans syndrome underwent splenectomy after 30 months and the other with chronic ITP received steroid therapy after 5 months of the first rituximab therapy. If the third patient relapses again, second course of rituximab therapy is planned for him. He has still been in PR, now. Thus, if relapse of the chronic ITP is observed in the patient, second course of rituximab therapy may be tried. However, if there is NR to second course of rituximab therapy, then splenectomy should be recommended.
In our study, we have noted a dramatically decrease in B cells after rituximab therapy. In all patients, serum IgG and/or IgM levels decreased to a level below the normal range after rituximab therapy and the decrease was statistically significant for only IgG levels after 3 months of therapy. However, the mean IgG level was still higher than 400 mg/dL and all our patients except one did not need IVIG replacement after rituximab therapy. One of our patients had chronic otitis media and bronchiectasis related to hypogammaglobulinemia and needed only 3 months IVIG replacement therapy. He had no further serious infection and he is still in CR. Rao et al suggested that B-cell recovery took 6 to 12 months irrespective of dose and baseline immunoglobulin levels. 19 However, we cannot determine immunoglobulin levels at 6th and 12th month of rituximab therapy in our study. In previous studies, infections remained infrequent during and after rituximab therapy and occurred mostly in patients with an underlying predisposition to infections. It is recommended to perform vaccinations before administration of rituximab or after B-cell recovery. 20 Vaccinations were performed for our patients at least 2 weeks before rituximab therapy.
In conclusion, rituximab therapy with an acceptable safety profile should be considered prior to splenectomy or other potential interventions with higher toxicity in children with chronic ITP and Evans syndrome. Furthermore, retreatment with rituximab after relapse may enable additional responses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
