Abstract
Background: Few studies have assessed the effect of prothrombotic blood abnormalities on the risk of deep vein thrombosis (DVT) with hormone replacement therapy (HRT). Methods: We studied postmenopausal women with suspected DVT in whom HRT use and prothrombotic blood abnormalities were sought. Cases had unprovoked DVT and controls had no DVT and without DVT risk factors. The risk of DVT was determined in women with and without prothrombotic abnormalities. Results: A total of 510 postmenopausal women with suspected DVT were assessed; 57 cases and 283 controls were identified. Compared to HRT, nonusers without the factor V Leiden mutation, the risk of DVT was increased in estrogen-progestin HRT users (odds ratio [OR], 3.2; 95% confidence interval [CI]: 1.2-8.6) and in nonusers with the factor V Leiden mutation (OR, 5.3; 1.9-15.4) and appears multiplied in users of estrogen-progestin HRT with the factor V Leiden mutation (OR, 17.1; 3.7-78). Compared to HRT, nonusers with normal factor VIII, the risk of DVT was increased in estrogen-progestin HRT users with normal factor VIII (OR, 2.8; 1.0-7.9) and in HRT nonusers with the highest factor VIII quartile (OR, 6.0; 2.1-17), and appears to be multiplied in women who are users of estrogen-progestin HRT with the highest factor VIII quartile (OR, 17.0; 3.6-80). Conclusions: In postmenopausal women who are estrogen-progestin HRT users, the presence of the factor V Leiden mutation or an elevated factor VIII level appears to have a multiplicative effect on their overall risk of DVT, increasing it 17-fold compared to women without these blood abnormalities who are HRT nonusers.
Introduction
Randomized trials and observational studies have consistently shown that users of oral hormone replacement therapy (HRT) have a 2- to-4-fold higher risk of developing deep vein thrombosis (DVT) compared to nonusers of oral HRT.1–6 However, few studies have assessed whether having a prothrombotic blood abnormality, such as the factor V Leiden mutation, modifies the risk of DVT with HRT use,7–10 and uncertainty remains about the impact on DVT risk in women with various prothrombotic blood abnormalities.11–13
This issue is relevant in at least 2 clinical scenarios. The first is in women with a family history of DVT in whom HRT use is being considered since prior knowledge that a prothrombotic abnormality increases the absolute risk of DVT to an unacceptable degree might justify thrombophilia screening to confirm or exclude inheritance of such abnormalities. The second scenario is in women with a known prothrombotic abnormality and no prior DVT in whom HRT is being considered. Prior knowledge that HRT will increase their absolute risk of DVT may deter the use of HRT in such women.
These scenarios are likely to be encountered in clinical practice when women reach menopause and are being considered for HRT to alleviate vasomotor and other climacteric-related symptoms. Despite the drawbacks of long-term HRT use,5,6 short-term use of HRT is recommended by gynecologic practice guidelines as first-line therapy to alleviate vasomotor flushing and other climacteric symptoms,14–17 which can last for a year or longer and be debilitating in up to 25% of all women.18,19 Furthermore, there are no known therapeutic alternatives to HRT that are as effective or more effective in alleviating climacteric-related symptoms.14–17
Against this background, we aimed to investigate in postmenopausal women if the presence of common inherited and acquired prothrombotic blood abnormalities modify the association between HRT use and the risk of developing DVT.
Patients and Methods
Study Design
This study was a nested case-control study, embedded within a larger cross-sectional study that assessed the risk of DVT with HRT use. 4 In the parent study, postmenopausal women with suspected lower limb DVT were identified from outpatient clinics, emergency departments, or in-hospital settings from 12 centers in Canada, Italy, and the Netherlands. Patients were separated into cases with unprovoked (or idiopathic) DVT who had no known antecedent DVT risk factors, and controls without DVT who, similarly, had no known DVT risk factors. In this substudy, patients were recruited from 6 clinical centers (3 in Canada, 2 in Italy, 1 in the Netherlands). All patients provided written informed consent for study participation and the study was approved by the Ethics Review Boards of participating institutions.
Patients
Consecutive postmenopausal women with suspected lower limb DVT were eligible for this study. For this substudy, we excluded patients (both cases and controls) with antecedent risk factors for DVT, other than possible HRT use. Postmenopausal status was defined by 1 of the following criteria20,21: spontaneous menopause, with no menses for at least 6 months; surgical menopause due to bilateral oopherectomy; or the presence of menopausal symptoms such as vasomotor flushing for at least 6 months. Patients were excluded if they had 1 or more of the following characteristics: suspected pulmonary embolism; amenorrhea due to primary or secondary ovarian failure; cognitive impairment or language barrier. Patients with suspected pulmonary embolism were excluded because the accuracy of diagnostic testing is suboptimal as up to 20% of patients will not have pulmonary embolism reliably diagnosed or excluded. 22
Eligible and consenting patients had a clinical assessment in which presenting features, risk factors for DVT, and factors that might affect HRT use were documented. Hormone replacement therapy use status was ascertained by direct patient questioning and was aided by showing patients photographs of current HRT preparations. After the clinical assessment, patients underwent objective diagnostic testing for DVT, with test interpretation by observers who had no prior knowledge of patients’ HRT use status. All patients had standardized blood testing to assess for the presence of prothrombotic blood abnormalities.
Determination of HRT Use Status
Hormone replacement therapy was defined as an oral or transdermal formulation consisting of an estrogen with or without a progestin. Patients were classified as HRT current-users or HRT nonusers, with the latter group consisting of previous- and never-users. Current-users were HRT users who had been receiving HRT for at least 1 month prior to the study assessment. Previous-users were patients who had received HRT prior to 1 month of the study assessment but not within the month before the assessment. Never-users were patients who had never received HRT. In current-users, the HRT type was classified as estrogen-progestin or estrogen-only, and as oral or transdermal.
Determination of DVT Status
Diagnostic testing for DVT was based on clinical center-specific diagnostic algorithms, but all the patients’ required objective confirmation or exclusion of DVT based on validated criteria. Deep vein thrombosis was confirmed by a noncompressible vein on ultrasound
23
or a constant intraluminal filling defect on venography.
24
Patients with confirmed DVT received conventional anticoagulant therapy at the discretion of the treating physician. Deep vein thrombosis was excluded by 1 or more of the following: normal venography
25
; normal ultrasound and normal SimpliRED
Identification of Cases and Controls
Cases and controls were identified centrally by the Study Coordinating and Methods Centre based on prespecified criteria. Cases were defined as patients with unprovoked (or idiopathic) DVT, in whom DVT occurred in the absence of the following DVT risk factors: recent (within 1 month) surgery, immobility, or trauma; thrombophilia; previous venous thromboembolism; active cancer (treated within 6 months or palliative). 30 Controls were defined as patients in whom DVT was excluded, and who did not have any of the aforementioned DVT risk factors. This was done to ensure comparability for predisposition to DVT in both case and controls.
Prothrombotic Blood Abnormalities Assessed
A priori, we aimed to investigate the effect of the following prothrombotic blood abnormalities on the association between HRT use and DVT: factor V Leiden mutation; prothrombin gene mutation; antiphospholipid antibodies (APLA) (anticardiolipin antibodies, lupus anticoagulant); elevated homocysteine; elevated factor VIII; elevated factor XI. Deficiencies in protein C, protein S, and antithrombin were not assessed because they are uncommon, occurring collectively in less than 5% of patients with DVT. 31
Laboratory Analysis
Blood was drawn from each patient into a 5-mL glass tube containing EDTA or 3.2% citrate (Becton Dickinson Vacutainer Systems, Franklin Lakes, New Jersey). Plasma was separated by centrifugation for 15 minutes (1700g). To ensure platelet-free plasma, plasma was removed, placed in a plastic tube, and centrifuged for 15 minutes (1700g). Plasma and the buffy coat from the EDTA tube were stored at −70°C. All blood samples were processed locally and were subsequently shipped to a central laboratory (Hemostasis Reference Laboratory Inc, Hamilton, Ontario, Canada) for analysis.
The factor V Leiden mutation was assayed using a sequence-specific polymerase chain reaction followed by digestion with MnI-1 restriction enzymes. The prothrombin gene mutation was assayed as previously described. 32 Homocysteine was assayed by the fluorescence polarization immunoassay on an IMX analyzer (Abbott Diagnostics, Mississauga, Ontario). Factors VIII and XI were assayed by a 1-stage clotting assay. Antiphosholipid antibodies were assayed by clotting assays (lupus anticoagulant) and by solid phase immunoassays (anticardiolipin antibodies). A high sensitivity partial thromboplastin time reagent, (PTT LA, Diagnostica Stago, Asnières, France), a dilute Russell viper venom test (American Diagnostic, Greenwich, Connecticut), and a dilute prothrombin time test were used to screen for antiphospholipid antibodies. Results greater than the normal range were confirmed using a hexagonal phospholipid assay (Staclot LA, Diagnostica Stago, Asnières, France) or a dilute Russell viper venom confirmatory test (American Diagnostics). Anticardiolipin immunoglobulin G (IgG) and IgM were assayed by an enzyme-linked-immunosorbent assay (ELISA) assay (Corogenix, Denver, Colorado).
Statistical Analysis
To assess whether prothrombotic blood abnormalities modify the association between HRT use and DVT risk, we used multivariable regression analysis to determine the risk of DVT in patients with an abnormality and in patients without an abnormality in 3 groups: HRT nonusers; estrogen-progestin HRT users; and estrogen-only HRT users. Reported risk estimates were based on multivariable modelling and were expressed as odds ratios (ORs), with corresponding 95% confidence intervals (CIs). All P values were 2-sided. All analyses were done with SAS V8.1 software (SAS Institute Inc, Cary, North Carolina).
A priori, we identified factors that might confound the effect of prothrombotic blood abnormalities on the association between HRT and DVT. These factors included in the regression models were as follows: clinical center; patient age (deciles); education level (primary, secondary, post-secondary); body mass index (≤19.9, 20-29.9, or ≥30 kg/m2); smoking status (yes/no); previous hysterectomy (yes/no); bilateral oopherectomy (yes/no); vertebral or long-bone fracture (yes/no); osteoporosis (yes/no); breast cancer (yes/no); and clinical likelihood for DVT (low, moderate, high). Variables in the final multivariable models were selected based on their level of statistical significance, defined as P < .10 (2-sided) in the univariate model. To determine whether the risk estimates were consistent across clinical centers, interaction terms between the HRT variable and clinical center were added to the model and assessed using the likelihood ratio test.
Results
Description of Patient Population
The patient characteristics are shown in Table 1 . There were 510 women with suspected DVT who were potentially eligible for this substudy of whom 112 (22%) had DVT confirmed and 398 had DVT excluded. From this group, we identified 57 cases (out of 112) with unprovoked DVT and no antecedent DVT risk factors (other than possible HRT use) and 283 controls (out of 398) without DVT and also had no antecedent DVT risk factors (other than possible HRT use) for the regression analyses.
Patient Characteristics
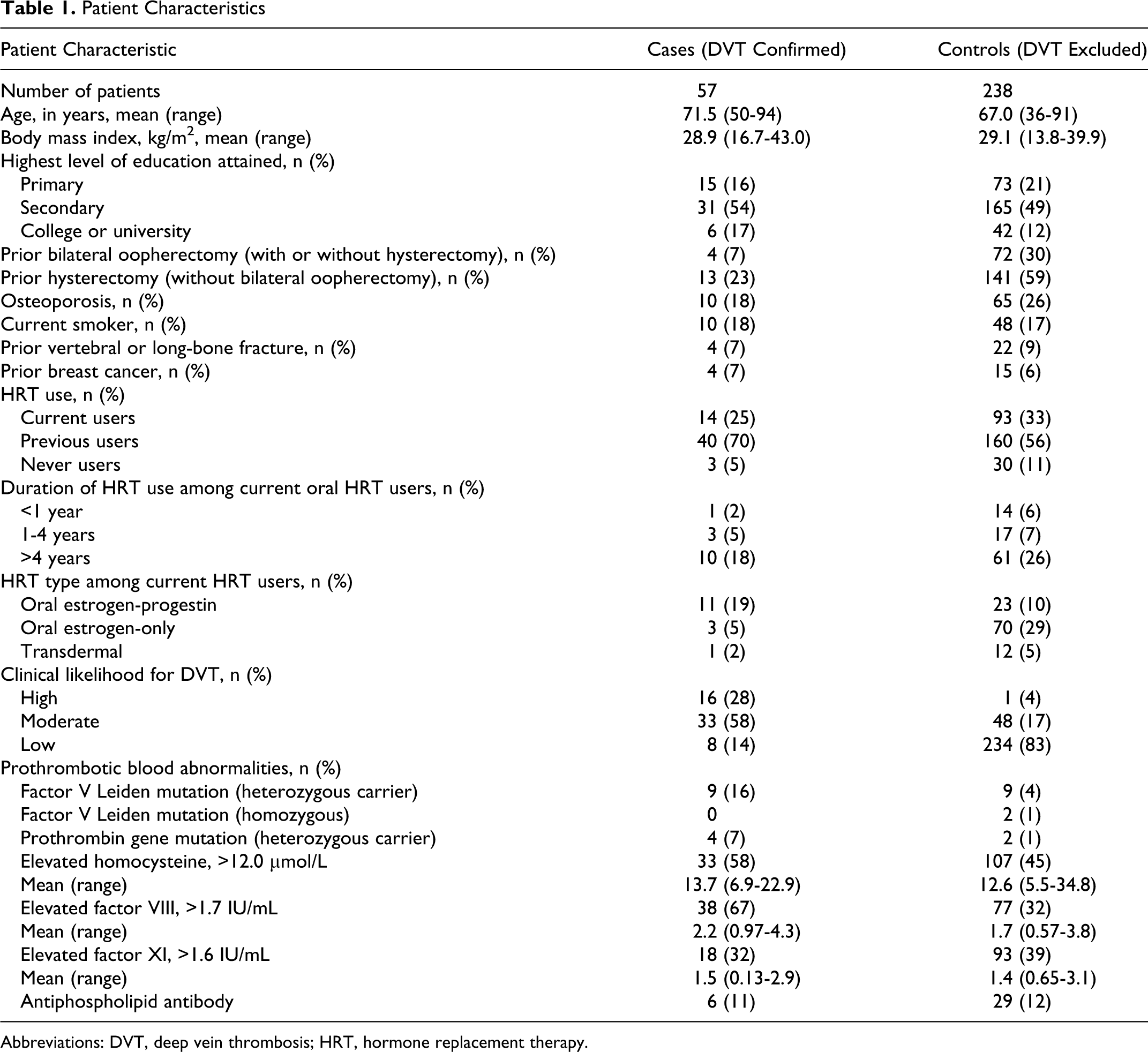
Abbreviations: DVT, deep vein thrombosis; HRT, hormone replacement therapy.
Multivariable Regression Analyses
The ability to perform statistically meaningful regression analyses (to assess effects on association between HRT and DVT) was limited to prothrombotic blood abnormalities in which there was both a significant association with DVT (irrespective of HRT use) and a sufficient number of patients with these abnormalities. As shown below, we were able to perform separate regression analyses only in patients with the factor V Leiden mutation and those with elevated factor VIII. This was not possible for other prothrombotic abnormalities in which there was no significant association with DVT (APLA, elevated homocysteine or elevated factor XI) or there were too few patients with these abnormalities (prothrombin gene mutation).
Effect of prothrombotic blood abnormalities on risk of DVT
After controlling for prespecified potential confounders, we found an increased risk of DVT in patients who had the factor V Leiden mutation (OR, 4.4; 95% CI: 1.7-11.3), the prothrombin gene mutation (OR, 10.2; 1.8-57), or an elevated factor VIII >1.7 IU/mL (OR, 3.6; 2.2-5.9). There was no significant increased risk of DVT in patients with an elevated factor XI >1.6 IU/mL (OR, 1.5; 0.6-3.2), an elevated homocysteine >12.0 μmol/L (OR, 1.0; 0.9-1.1), or with APLA (OR, 1.0; 0.4-2.6).
Effect of factor V Leiden mutation on risk of DVT With HRT Use and HRT Type
As shown in Table 2 , compared to HRT nonusers without the factor V Leiden mutation (reference group), the risk of DVT was increased in users of estrogen-progestin HRT without the factor V Leiden mutation (OR, 3.2; 1.2-8.6) and in HRT nonusers with the factor V Leiden mutation (OR, 5.3; 1.9-15.4). The risk of DVT in women who were both users of estrogen-progestin HRT and had the factor V Leiden mutation appeared to be multiplicative (OR, 17.1; 3.7-78.0). There was no significant increased risk of DVT in estrogen-only HRT users without the factor V Leiden mutation (OR, 0.3; 0.08-1.2) and in estrogen-only HRT users with the factor V Leiden mutation (OR, 1.7; 0.3-9.0).
Effect of Factor V Leiden Mutation on Risk of DVT According to HRT Type

Abbreviations: DVT, deep vein thrombosis; HRT, hormone replacement therapy.
Effect of elevated factor VIII on risk of DVT With HRT Use and HRT Type
As shown in Table 3 , patients were separated into factor VIII level quartiles (0.5-1.3 IU/mL, 1.4-1.7 IU/mL, 1.8-2.0 IU/mL, >2.1 IU/mL) to permit a comparable patient distribution of patients across groups, with HRT nonusers in the normal or near-normal quartile (0.5-1.3 IU/mL) as the reference group. Compared to HRT nonusers who had a normal factor VIII level, the risk of DVT was increased in estrogen-progestin HRT users with a normal factor VIII level (OR, 2.8; 1.0-7.9) and in HRT nonusers with the highest factor VIII level (OR, 6.0; 2.1-17.0), and appears to be multiplied in women who are both users of estrogen-progestin HRT and had the highest factor VIII level (OR, 17.0; 3.6-80.0). There was no significant increased risk of DVT in estrogen-only HRT users without an elevated factor VIII level (OR, 0.3; 0.1-1.3) or in estrogen-only HRT users with the highest factor VIII level (OR, 2.0; 0.3-11.0).
Effect of Elevated Factor VIII on Risk of DVT According to HRT Type
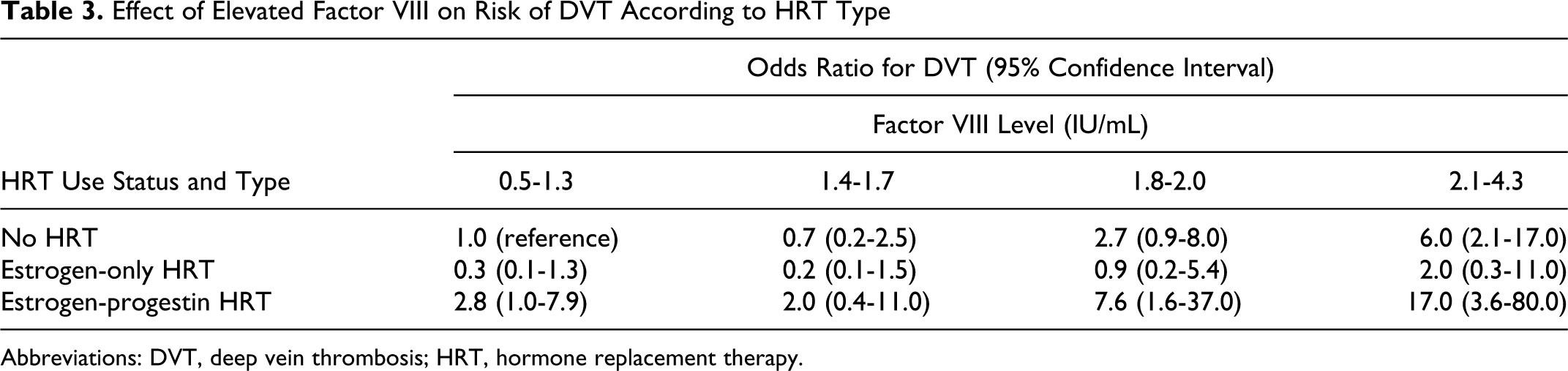
Abbreviations: DVT, deep vein thrombosis; HRT, hormone replacement therapy.
Discussion
The principal finding from this study is that in postmenopausal women who are users of oral estrogen-progestin HRT, the presence of the factor V Leiden mutation or an elevated factor VIII level appears to have a multiplicative effect on the overall risk of DVT, increasing it approximately 17-fold compared to women without these prothrombotic blood abnormalities who do not use HRT and approximately 6-fold compared to women without these blood abnormalities who use HRT.
Our findings are likely to be valid based on the following considerations. First, cases with DVT and controls without DVT who had testing for prothrombotic blood abnormalities were derived from the same population of postmenopausal women with suspected DVT, thereby ensuring that all patients were subjected to the same referral process and minimizing the likelihood for differential HRT use in cases and controls. Second, it is likely that data about HRT use status and diagnostic classification (DVT confirmed or excluded) were reliable because these were collected prospectively, during a face-to-face patient interview, and DVT was diagnosed by objective criteria and diagnostic testing was performed without prior knowledge of HRT use. Furthermore, HRT use status was determined prior to and independent of diagnostic testing for DVT, thereby minimizing the likelihood for diagnostic suspicion bias. Third, laboratory testing was done in a central laboratory using standard commercial assays, thereby minimizing the likelihood of unreliable measurements due to different assay methods.
Our finding that patients with an elevated factor VIII were at increased risk of DVT with HRT use warrants further comment. Since factor VIII is an acute-phase reactant, which may increase in response to an acute infectious or inflammatory disease, it is possible that the higher factor VIII levels observed in patients with DVT than those without DVT may have been caused by the acute DVT rather than being a preexisting factor that contributed to its pathogenesis. Given our cross-sectional study design, we were unable to repeat factor VIII measurements at a time remote from the acute DVT when, presumably, elevated factor VIII levels related to acute disease would resolve. 33 However, patients in our comparator group without DVT were not “healthy controls” and also had conditions such as popliteal bursitis or superficial phlebitis that would have likely also triggered an acute-phase reactant response and elevated factor VIII as in cases with acute DVT. Furthermore, the apparent graded response between factor VIII levels and risk of DVT suggests a causal link.34,35 Overall, the significance of elevated factor VIII as a putative risk factor for DVT requires further study, especially to ascertain if there is a factor VIII cut-off level (or serial levels) that best discriminates between an increased risk of DVT and other conditions that may also increase factor VIII. 36
There are additional findings that are, at best, hypothesis-generating and require further study. An elevated homocysteine or an elevated factor XI level did not appear to confer an increased risk of DVT irrespective of whether HRT was used or the type of HRT used. However, the effect of these purported prothrombotic blood abnormalities on the risk of DVT is questionable.35,37 Although the prothrombin gene mutation in itself conferred a more than 10-fold increased risk of DVT, there were too few patients with this abnormality to assess its effect on the association between HRT and DVT. Furthermore, the lack of an association between the presence of APLA and DVT is surprising and, possibly, is due to chance.38,39 Alternatively, such an association may have been undetected since there may have been falsely abnormal APLA laboratory tests in patients with suspected DVT in whom DVT was excluded, which would mask a true association between APLA and acute DVT. 40 The lack of an association between hyperhomocsyteinemia and thrombosis has been reported elsewhere.37,41 Finally, although the risk of DVT in women with the factor V mutation or an elevated factor VIII level who were estrogen-only HRT users appeared less than in estrogen-progestin HRT users, this finding should be interpreted with caution and may be due to chance.
Our findings complement the findings of other studies that have assessed the effect of prothrombotic blood abnormalities on the association between HRT use and DVT. Compared to the present study, there was similar increased risk of DVT in HRT users with the factor V Leiden mutation compared to HRT nonusers without this abnormality in the Oxford Regional Health Case-Control Study (OR, 15.5; 3.1-77) and the combined analysis of the HERS-ERA Study data (OR, 14.1; 2.7-72).10,12 In the women’s health initiative (WHI) Study, there was also an increased risk of DVT in HRT users with the factor V mutation compared to HRT nonusers without this abnormality, although the magnitude of this risk appeared lower than in the present study. 13 In the Oxford study, the prothrombin gene mutation increased the risk of DVT in HRT users compared to nonusers (OR, 11.0; 2.7-44). Two other observational studies found that users of HRT with either the factor V Leiden or prothrombin gene mutations had a 9-fold (OR, 9.1; 4.5-18.2) to 25-fold (OR, 25; 0.9-50) increased risk of VTE compared to HRT nonusers without a prothrombotic blood abnormality.42,43 These studies, however, did not assess the effects of other prothrombotic abnormalities on the risk of DVT with HRT use.
Taken together, the findings from the present and previous studies support the premise that prothrombotic blood abnormalities further increase the risk of DVT with HRT use. This finding is similar to the observed effect of prothrombotic blood abnormalities to further increase the risk of DVT with oral contraceptive use.44–46
The clinical implications of this study should be considered within the context of the totality of evidence relating to the risk of DVT in women with a prothrombotic blood abnormality who are exposed to HRT. Our findings, coupled with those of others, suggest that in women with an asymptomatic prothrombotic blood abnormality who develop climacteric symptoms, consideration should be given to nonhormonal treatments because of the markedly increased risk of DVT in such patients, particularly if they are carriers of the factor V Leiden mutation. Moreover, in women with a family history of DVT who develop climacteric symptoms, screening for prothrombotic blood abnormalities should be considered prior to initiation of HRT since decisions about HRT use may be predicated on the presence of and type of prothrombotic abnormality detected in addition to the individual family history.
To summarize, in postmenopausal women who are users of estrogen-progestin HRT, the presence of the factor V Leiden mutation or an elevated factor VIII level appears to have a multiplicative effect on their overall risk of DVT, increasing it 17-fold compared to women without these blood abnormalities who are HRT nonusers.
Footnotes
Acknowledgment
The authors would like to express their gratitude to the research nurses and other staff who enrolled patients at each clinical site.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Grant from the Canadian Institutes of Health Research.
