Abstract
The aim of this study is to investigate the effects of exogenous
Keywords
Introduction
Platelet activation and aggregation play an important role in the development of cardiovascular diseases (CAD). The interaction between plasma lipoproteins and platelets can affect platelet activation.
1
As it is known, low-density lipoprotein (LDL) is an atherogenic lipoprotein and elavated plasma LDL levels promote thrombotic events or atheriosclerotic lesion progression.
2
Low-density lipoprotein enhances expression of glycoprotein IIb/IIIa (GpIIb/IIIa), P-selectin, and fibrinogen binding as well as increasing platelet aggregation and trombaxane A2 release. Increased reactive oxygen species in various pathological conditions such as CAD and diabetes give rise to oxidative processes like lipid peroxidation in LDL, which causes oxidative modification of LDL. Oxidized LDL (ox-LDL) is more effective on platelet activation than native LDL. It has been reported that ox-LDL increases platelet aggregation without an agonist.
3
Oxidized-LDL stimulates platelet activation primarily by diminishing nitric oxide sythase (NOS) expression and release of nitric oxide (NO) in endothelial cells and platelets.4,5 Thus, it can promote atherosclerotic lesion formation on the surfaces of arteries. At the same time, it not only increases foam cell formation but also increases cell proliferation in smooth muscles and platelet adhesion.6–8 High-density lipoprotein (HDL) has anti-atherogenic effects on the platelet function. High-density lipoprotein reduces total body cholesterol by a mechanism known as reverse cholesterol transport and thereby reduces the risk of developing CAD. However, HDL has additional antithrombotic actions. It also promotes the production of the atheroprotective signalling molecule NO.
9
Several studies showed that ox-LDL reduces NOS activity but HDL reverses ox-LDL’s effects on neutrophils.
10
Extracellular
Nitric oxide, previously known as an endothelium-derived relaxing factor, is a biological mediator and is produced from
Platelets themselves synthesize NO.
17
Investigations on NO production in platelets began in 1990 with a study of Randomski et al
18
describing the presence of
Expression of P-selectin on the platelet surface membrane is a specific and useful index of platelet activation. 22 Platelets and endothelial cells possess P-selectin. P-selectin is an adhesion molecule of selectin family and plays an important proinflamatory role in interactions of leukocytes, platelets, and vascular endothelium. P-selectin is normally stored in the α-granule membranes of resting platelets. P-selectin is rapidly translocated to the plasma membrane and is expressed on the platelet surface after activation by several mediators such as thrombin, histamine, and protein kinase C.22,23
The aim of this study was to investigate the effects of exogenous
Materials and Methods
Participants
All participants were healthy adult donors. Exclusion criteria were medical history of renal, liver, and cardiovascular disease; acute-chronic infectious diseases; thrombotic disease; alcohol consumption; smoking; use of antiplatelet, anticoagulant, or lipid-lowering drugs for at least 10 days. Inclusion criteria: normolipidemic, age between 20 and 35 years, and no medical history of platelet function disorders. All participants gave written informed consent to study participation. The study protocol was approved by the Ethics Committee at the Medical Faculty of Marmara University.
Chemicals
CD62-FITC was from Beckman Coulter (Marseille, France); QIFI kit was from DAKO (Copenhagen, Denmark); TITAN GEL Lipoprotein Kit was from Helena Laboratories (Sunderland, UK; Cat No: 3045). Paraformaldehyde (PFA), phosphate buffered saline (PBS), thiobarbituric acid (TBA), apyrase, adenosine diphosphate (ADP),
Isolation of Lipoproteins and Modification of Native LDL
For the purification of lipoproteins, peripheral venous blood of normolipemic healthy volunteers (N = 9, F:M = 5:4) was collected into vacutainer tubes containing EDTA (1 mg/mL) after 12 hours fast and was incubated at room temperature for 20 minutes. Blood samples were centrifuged at 150
Oxidation of native LDL (nLDL) by copper ions was performed according to a commonly used protocol. Before oxidation, nLDL was dialyzed overnight at 4°C against phosphate buffer saline (PBS, 8 mmol/L NaH2PO4, 5 mmol/L KCl, 125 mmol/L NaCl) to remove EDTA. Oxidation of nLDL (1 mg/mL) was carried out with 10 μmol/L CuSO4 in PBS for 48 hours at 37°C. Oxidation of nLDL was terminated by refrigeration. After dialysis, lipid peroxidation (LPO) levels were measured by the TBA method and expressed as malondialdehyde (MDA) equivalent. 26
The electrophoretic mobility of isolated lipoprotein fractions and ox-LDL on agarose gels (Titan gel lipoprotein system, Helena Laboratories) was visualized by staining with Fat Red 7B.
Platelet Isolation
Peripheral blood from the participants (N = 8, F:M = 4:4) was collected into 10-mL tubes containing 1 mL of 130 mmol/L trisodium citrate for a final ratio of 9:1 and 1 U/mL apyrase was added to prevent activation during isolation. Venous blood samples were obtained with a 21-gauge butterfly needle from participants. The blood samples were centrifuged at 200
Determination of NO Levels
The levels of NO in platelets were measured by using the method of Chen et al,
27
with minor modifications, indicated as nitrite plus nitrate (NOx) which is commonly used to monitor NO concentration. Buffer or 1 mmol/L
Quantitation of Platelet P-selectin Receptor Numbers
Flow cytometric analysis of P-selectin expressed on platelets was performed by a previously described method.
22
Washed platelets were incubated with
Following the incubation for total 1 hour, FITC-labeled CD62-P (anti-P-selectin) was added to each tube. The tubes were incubated for 15 minutes in the dark. For fixation, PFA (%1 vol/vol) was added and then the samples were diluted with PBS. CD62-P fluorescence intensity was measured by flow cytometer.
For the estimation of unspecific and background fluorescence, an unactivated sample was stained with an isotype FITC-conjugated immonuglobulin G (IgG) control. The analysis of all samples were carried out on FACS Calibur flow cytometer (Becton Dickinson, San Jose, California, USA). Daily quality control and fluorescence standardization were checked with CaliBrite beads (Becton Dickinson). A total of 50 000 cells were counted in each tube. The results represent the means of duplicate samples. Platelet discrimination was made according to scatter properties of cells at FS/SS (Forward Scatter/Side Scatter) scattergram with logaritmic scale. The negative control cursor was set to 2% of the fluorescence of control cells on histograms. The results were expressed as percentage of positive cells and number of receptors per cell.
Calculation of Specific Antibody Binding Capacity per Platelet
Before the flow cytometric analysis of the platelet samples stained with specific CD62-P antibodies, calibration beads, which are used as reference beads for calculation of binding capacity of CD62-P were run. The calibration beads were prepared according to the kit’s procedure (QIFI kit, DAKO, Denmark) and the same voltage and gain values were used for the measurement of samples. The calibration beads were covered with known numbers of IgG molecules in increasing numbers (3100 to 623 000). Mean fluorescence intensity (MFI) values of the calibration beads were measured by flow cytometer. Mean fluorescence intensity values for each of the platelet samples were also determined. Histograms of the geometric mean fluorescence intensity of 10 000 events were recorded and used to plot a log-log graph of the mean fluorescence intensity versus the number of antibodies attached to each bead. The number of platelet- bound CD62P antibody was estimated from this graph on the basis of the geometric mean fluorescence intensity of the sample. After subtraction of nonspecific binding, the number of specifically bound antibody molecules was taken as the number of bound sites for CD62P/platelet.
Statistics
Data were expressed as mean SD. The results were analyzed by the nonparametric Mann-Whitney U-test. Comparisons of multiple groups were made using the friedman test (SPSS 11.0 Chicago, Illinois).
Results
Analysis of Isolated Lipoproteins
High-density lipoprotein and nLDL were isolated from the plasma of 9 different persons by discontinuous density gradient ultracentrifugation. The composition of the lipoprotein fractions was assessed by examining the TG, CHO, protein, Apo A-I and Apo B contents (Table 1). Electrophoretic mobilities and protein, CHO, and apoprotein contents of LDL and HDL that we isolated were compatible with the expected criteria (Figure 1). Our findings have shown that lipoprotein isolation was succesfully performed. Table 2 shows oxidation levels of lipoproteins. We observed that lipid peroxidation levels of ox-LDL were found significantly higher than the nLDL (
The Composition of the Lipoprotein Fractions

Abbreviations: CHO, cholesterol; TG, triglyceride; Apo A-I, apoprotein A-I; Apo B, apoprotein B; LDL, low-density lipoprotein; nLDL, native LDL; HDL, high-density lipoprotein.
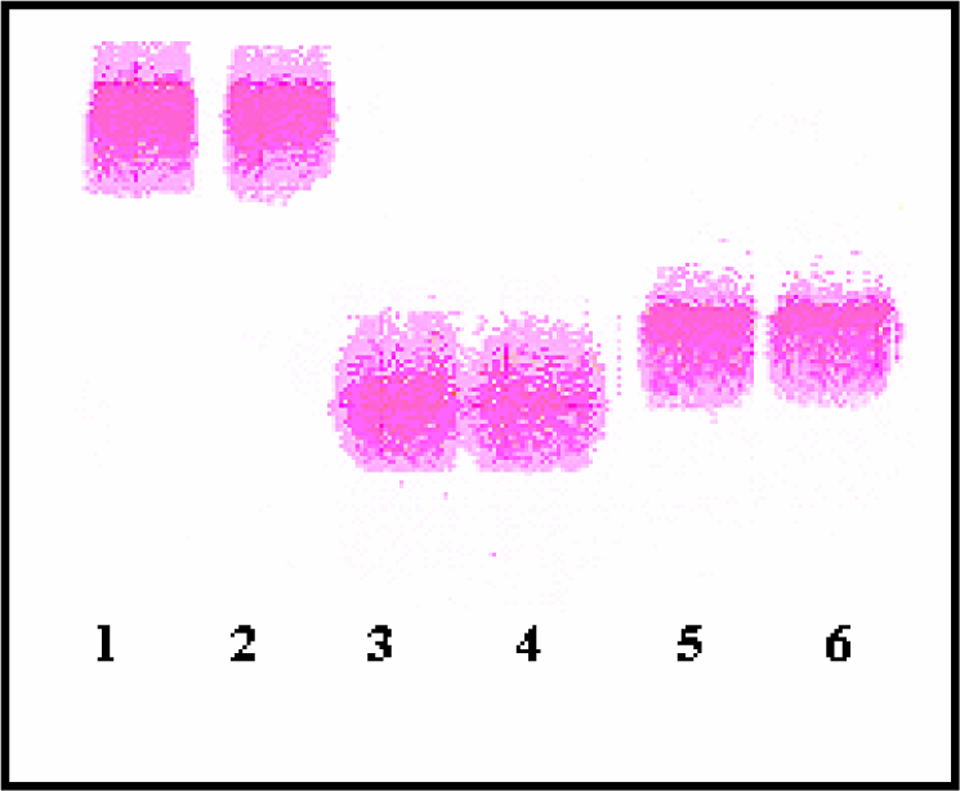
Lipoprotein electrophoresis (1-2: LDL, 3-4 ox-LDL, 5-6: HDL)
Lipid Peroxidation (LPO) Levels of Lipoproteins
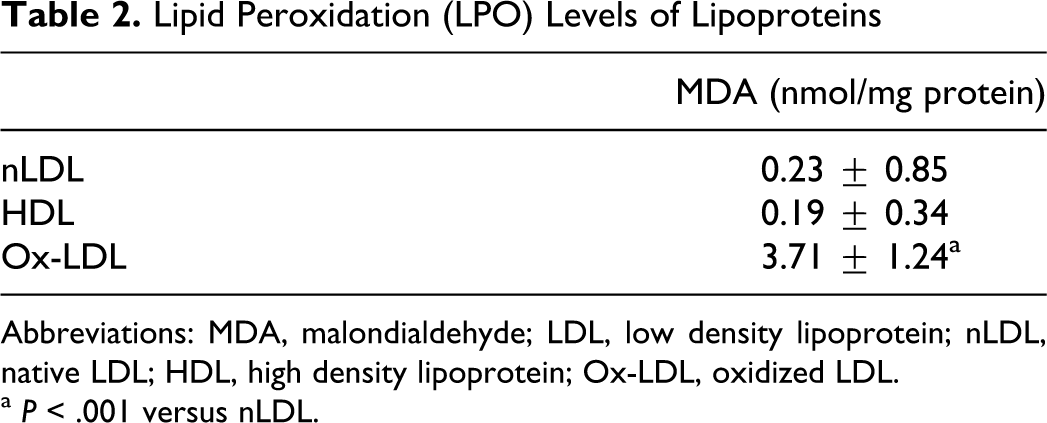
Abbreviations: MDA, malondialdehyde; LDL, low density lipoprotein; nLDL, native LDL; HDL, high density lipoprotein; Ox-LDL, oxidized LDL.
a
Platelet NO Levels
To see the effects of exogenous
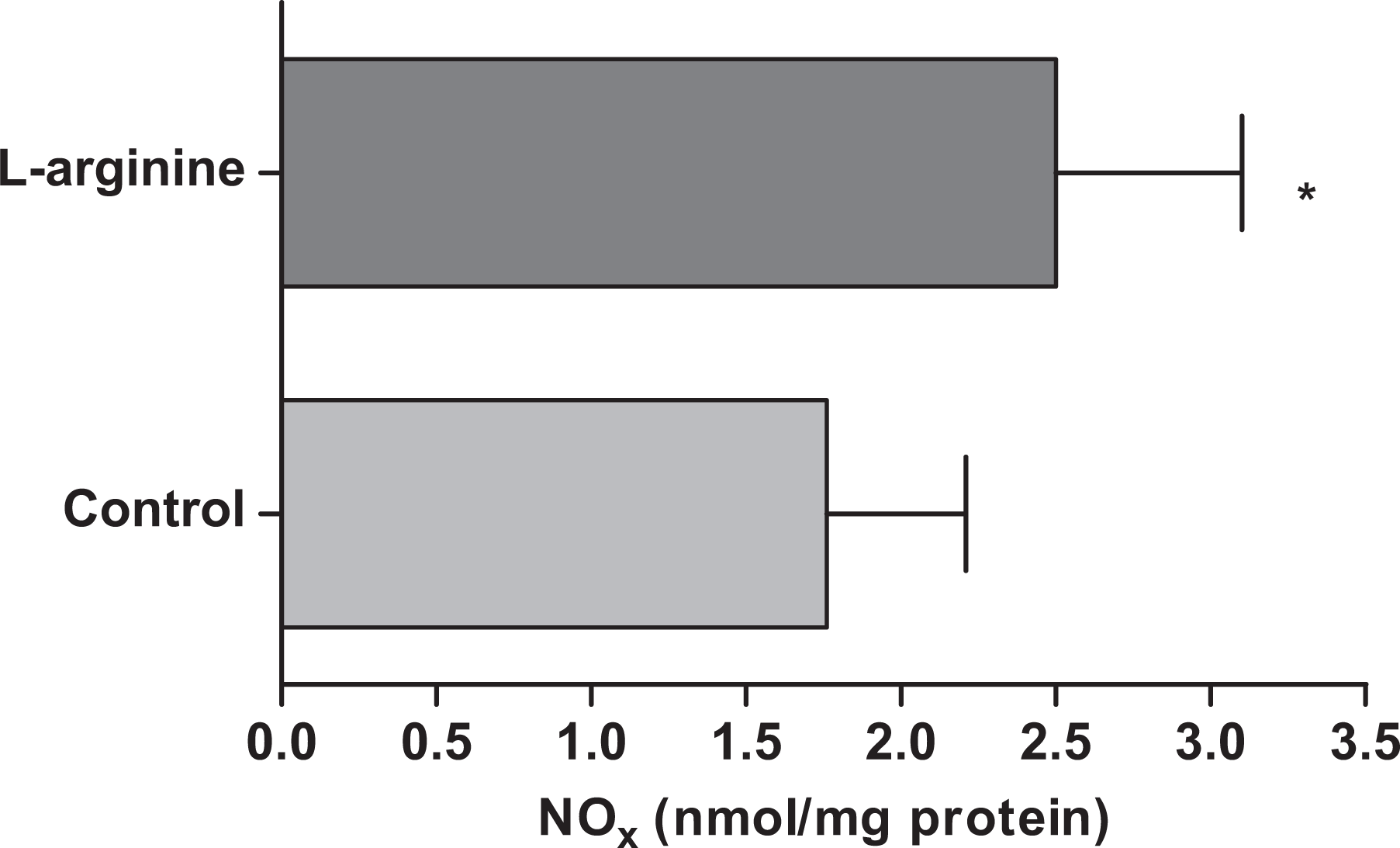
Effect of
Flow Cytometric Analysis of Platelet Receptor numbers on l-Arginine-Treated Platelets
Histograms of control group and platelets treated with
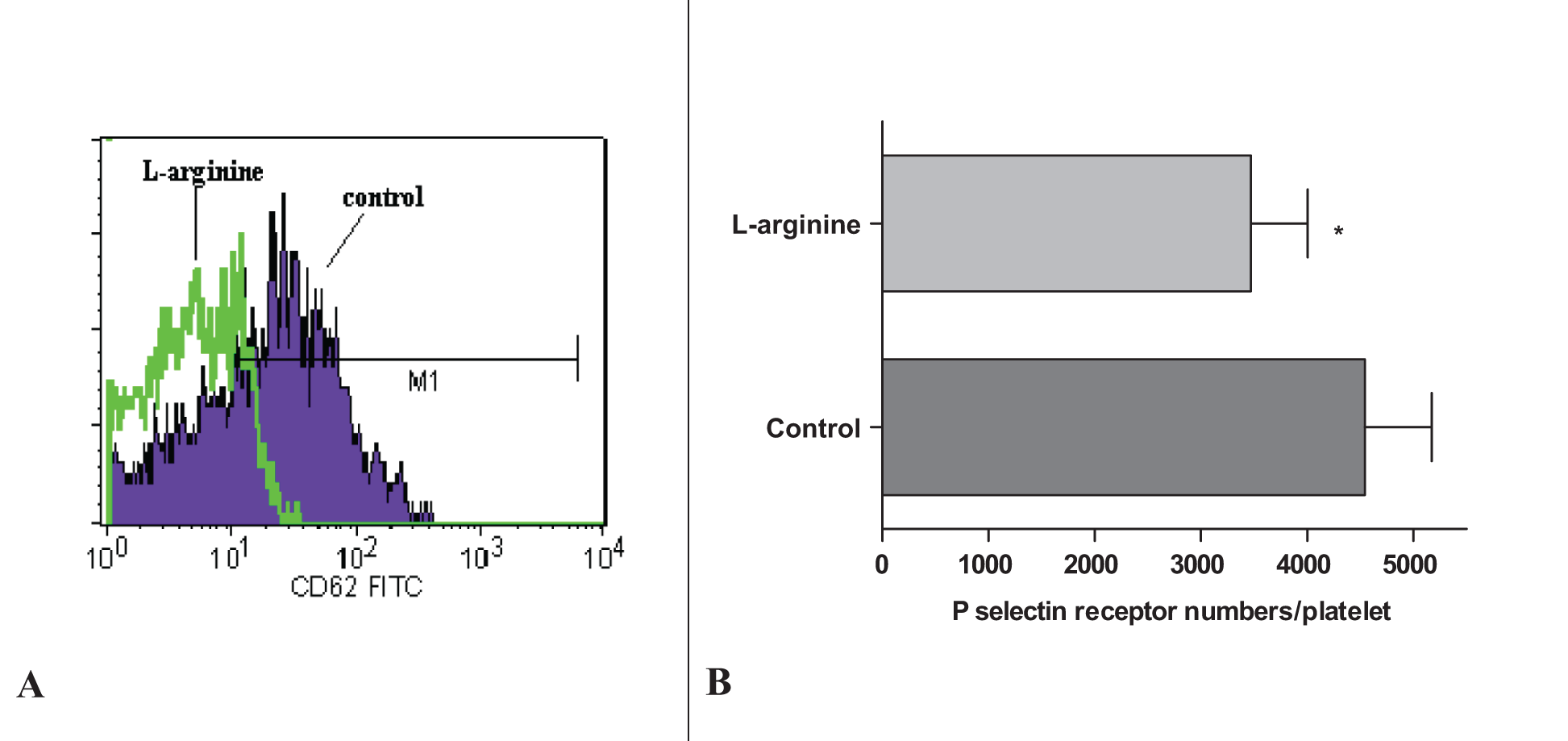
A, Histogram of platelet sample. CD62-P-positive platelets are in the area marked as M1. B, P-selectin receptor numbers on
The Effects of Lipoproteins on Platelet P-Selectin Numbers in the Absence or Presence of l-Arginine
As shown in Figure 4, nLDL and primarly ox-LDL promoted platelet P-selectin numbers (control: 4543±629, nLDL: 6160 ± 881, ox-LDL: 7051 ± 800,
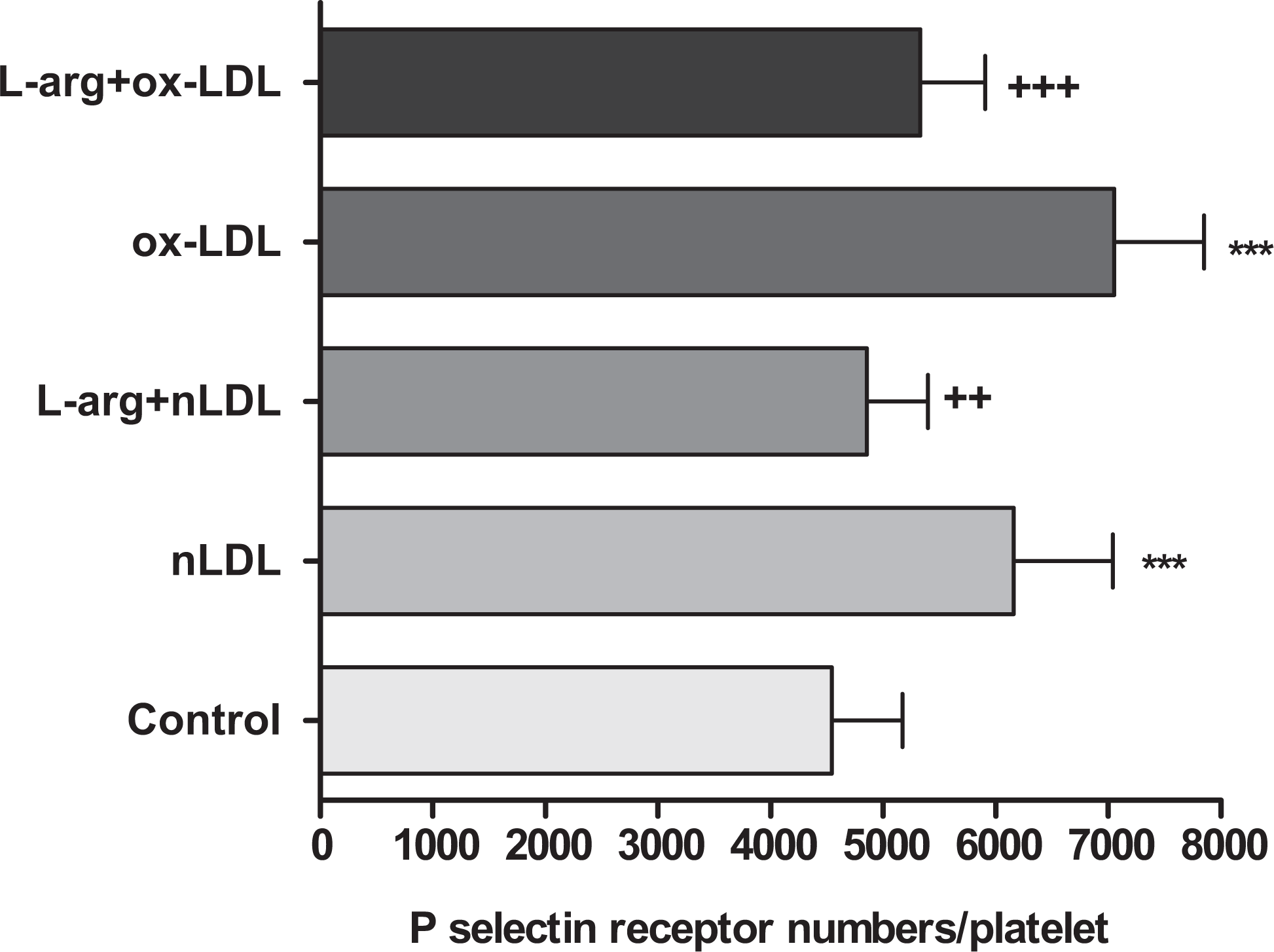
The effects of lipoproteins on platelet P-selectin receptor numbers in the absence or presence of
The Effects of HDL on P-Selectin Receptor Numbers in the Absence or Presence of l-arginine and ox-LDL
When the platelets were incubated with only HDL, some reduction was observed in the numbers of P-selectin; however, this reduction was not found significant compared to the control group (control: 4543 ± 629, HDL: 3894 ± 526,
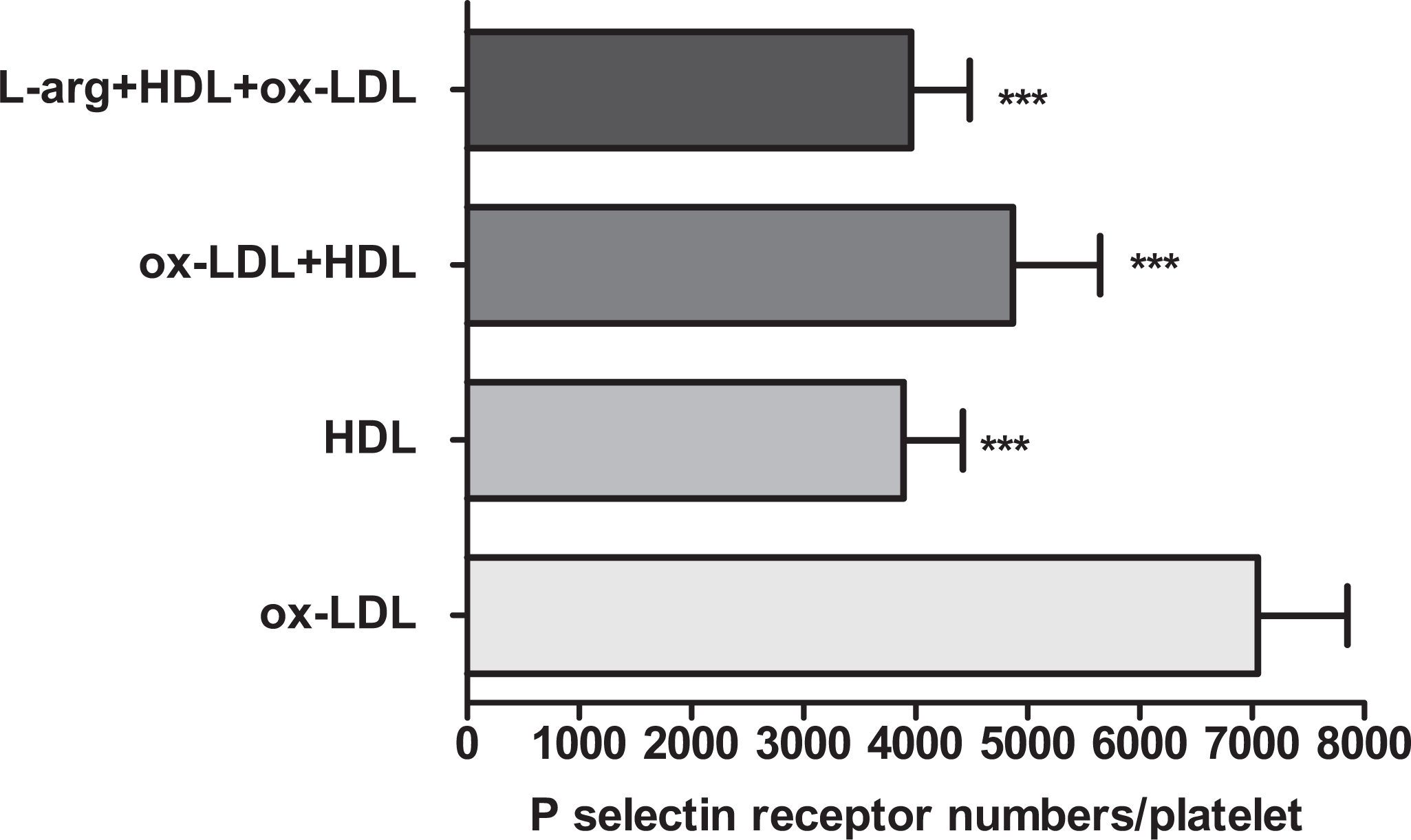
The effects of HDL on P-selectin receptor numbers of ox-LDL-treated platelets. Washed platelets were incubated with tested compounds and then the cells were labeled with anti-CD62 P-FITC. Platelet P-selectin receptor numbers were determined by flow cytometry (***
Discussion
Platelet-derived NO plays a role as a natural antithrombotic mechanism and inhibits additional platelet recruitment to a growing thrombus.
28
Malinski et al
29
reported that activated platelets release approximately 10-11 mol NO/min per 108 platelets. In our study, we first investigated the effects of exogenous addition of
In the current study, the platelet P-selectin expression was flow cytometrically measured to observe the effect of
Hyperactivity of human blood platelets is accompanied by an enhanced risk of atherosclerosis and arterial thrombosis. 34 Lipoproteins, especially modified lipoproteins such as ox-LDL, can also affect platelet functions. There are several studies in the literature that separately investigated the relationship between plasma lipoproteins with platelet activation and platelet NO production with platelet activation via P-selectin expression.35–37 However, there is not a comprehensive study that has investigated the effects of endogenous NO production on the relationship of nLDL, HDL, and ox-LDL with platelet activation.
In the in vitro studies carried out with lipoproteins, it was observed that the dose-dependent incubation of LDL with platelets can result in shape changes, aggregation, and secretion in the platelets. 38 Activation of LDL receptors (apoER2′ or GpIIb/IIIa) in platelets initiates different signal ways such as phosphorilation of p38MAPK, activation of cytosolic phospholipase A2, and formation of thrombaxane A2, a potent platelet-activating agent. Ox-LDL is much more effective than natural LDL in platelet signal routes via several receptors (GpIIb/IIIa, LOX-1, CD36, apoER2, SR-A).3,39,40
The effect of ox-LDL on platelet NO production proceeds in 2 ways, namely the inhibition of NOS and uptake of
High-density lipoprotein has a cardiovascular protective role with its endothelial and antithrombotic effects.
9
In our previous study, we concluded that HDL has a protective role by reducing the platelet activation and oxidative stress triggering effects of LDL/ox-LDL.
41
In the current study, we indicated that HDL alone also inhibited the platelet activation increase depending on ox-LDL. The effect of HDL was further fortified by the application of
As a result, it can be concluded that platelet NOS activity is sensitive to
The limitation of the study is the low number of samples. Nevertheless, the small sample size of the study allows the finding of significant clinical differences.
Footnotes
This study was presented at the 2007, AACC Annual Meeting, San Diego, California, July 15-19, 2007.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Supported in part by Marmara University Research Unit Grants SAG 070/060904.
