Abstract
Background:
Cardiomyocyte apoptosis by coronary microembolization (CME) contributes to myocardial dysfunction, in which mitochondrial pathway and death receptor pathway are activated. Lectin-like oxidized low-density lipoprotein receptor 1 (LOX-1) is a membrane protein involved in apoptosis. The study aimed to explore the role of LOX-1 in the activation of these 2 major apoptotic pathways.
Methods:
Twenty Bama miniature swine were randomized into 4 groups (n = 5 per group). The groups were Sham, CME, LOX-1 small-interfering RNA (siRNA), and control siRNA. Microspheres were injected into the left anterior descending artery of swine to establish CME model. Twelve hours after operation, cardiac function, serum c-troponin I level, microinfarct, and apoptotic index were examined. The levels of LOX-1, Bcl-2, Bax, cytochrome c as well as cleaved caspase 9, -8, and -3 were detected.
Results:
Myocardial dysfunction, enhanced serum c-troponin I, microinfarct, and apoptosis were induced following CME. Moreover, CME induced increased expression of LOX-1, Bax, cytochrome c, cleaved caspase 9, -8, and -3 as well as decreased Bcl-2 expression levels. The LOX-1 siRNA reversed these effects by CME except cleaved caspase 8 expression, while the control siRNA had no effect.
Conclusion:
Coronary microembolization induces cardiomyocyte apoptosis via the LOX-1-dependent mitochondrial pathway and caspase 8-dependent pathway.
Keywords
Introduction
Coronary microembolization (CME) resulting from the embolization of atherosclerotic and thrombotic debris into the microvasculature is a severe and frequent complication during percutaneous coronary intervention (PCI), thrombosis, and surgical revascularization. 1 It could induce malignant arrhythmias, contractile dysfunction, reduced coronary reserve, and irreversible myocardial damage including apoptosis and necrosis. 2 Furthermore, CME is the most likely culprit of absence of reflow and represents a strong independent predictor of poor prognosis. Apoptosis occurs following CME and partially contributes to CME-induced myocardial injury. 3 Apoptosis inhibition may attenuate the progress of chronic cardiac dysfunction following CME and improve the prognosis. Activation of mitochondrial apoptotic pathway and death receptor pathway has been considered as 2 major pathways in apoptosis. 4,5 Bcl-2, Bax, cytochrome c, and caspase 9 are markers of the mitochondrial pathway. Caspase 8 activation is a key process of the death receptor pathway. Lectin-like oxidized low-density lipoprotein receptor 1 (LOX-1) is a newly identified lectin-like oxidized LOX for several ligands including tumor necrosis factor α (TNF-α). 6 It expresses itself in atherosclerotic plaques and has been demonstrated to be critical in several cardiovascular diseases. 7 –9 However, whether or not it is involved in the activation of mitochondrial pathway and death receptor pathway following CME is currently unknown. Therefore, we sought to explore the role of LOX-1 in CME-induced apoptosis in a swine model of CME established by injecting microspheres into the left anterior descending (LAD) artery.
Materials and Methods
Animal Modeling and Grouping
The protocol was approved by the Animal Care and Use Committee of Guangxi Medical University, China. The investigation adhered to the Guide for the Care and Use of Laboratory Animals published by the US National Institute of Health (NIH Publication NO. 85-23, revised 1996). Twenty Bama miniature swine (25-30 kg) of either sex were purchased from the Animal Center, Agricultural College of Guangxi University (Nanning, China). Swine were randomized into 4 groups (n = 5 per group): Sham, CME, LOX-1 small interfering RNA (siRNA), and control siRNA. The CME model was induced by injecting plastic microspheres (diameter 42 μm) into the LAD artery. The initial anesthesia was induced gradually by intramuscular injection of the combination of ketamine (20 mg/kg) and atropine (2 mg) and maintained with diazepam injected via left or right ear vein. A 6F radial arterial sheath (Cordis, Fremont, CA) was placed in the right femoral artery. Swine were anticoagulated with 200 IU/kg heparin injected intravenously after a sheath placement, then 100 IU/kg/h to maintain heparinization. A 6F JL 4.0 guiding catheter was utilized for coronary angiography. A 1.8F infusion catheter (Cordis Inc) was then placed in the LAD artery with the tip located between the second and third diagonal branches. The swine in the CME group received a total dosage of 100 000 microspheres with the diameter of 42 μm (Dynospheres; Dyno Particles; Lillestrom, Norway) suspended in a 10 mL normal saline within 40 minutes, while animals in the sham-operated group received the same dosage of normal saline instead. Swine in the LOX-1 siRNA and control siRNA groups were subjected to LOX-1 siRNA and control siRNA, respectively, via intracoronary artery, 24 hours prior to CME.
The Transfection of LOX-1 siRNA
The LOX-1 messenger RNA sequence was found in GenBank (accession: NM_ 213805) and synthesized by GenePharma company (Shanghai, China). The sense strand and the antisense strand of the LOX-1 siRNA were 5′-GGGAACUCACAGAAAUGAUTT-3′ and 5′-AUCAUUUCUGUGAGUUCCCTT-3′, respectively. The control siRNA was purchased from the same company. A total mass of 300 μg LOX-1 siRNA or control siRNA was diluted with 600 μL transfection reagent (Entranster—in vivo; Engreen, Beijing, China) mixed thoroughly by pipetting the solution up and down. Then the mixture was injected into the LAD artery via the 1.8F infusion catheter, followed by 2 mL normal saline injection.
Cardiac Function Monitoring
Twelve hours following operation, swine were anesthetized and positioned in a right lateral decubitus position. This process was performed by an experienced specialist using a GE VIVID 7 system with a 1.5 to 4.3 MHZ transducer. In brief, the transducer was placed on the left anterior chest wall to measure the left ventricular ejection fraction (LVEF), left ventricular end-diastolic dimension (LVEDD), left ventricular end-systolic dimension (LVESD), fractional shortening (FS), and cardiac output (CO).
Serum c-Troponin I Measurement
EDTA-anticoagulated blood sample (1.0 mL) was obtained from the femoral artery of each swine 12 hours after operation, prior to euthanasia, and centrifuged at 4000 rpm for 15 minutes. Serum samples were stored at −80°C for detection by electrochemistry method according to manufacturer’s instruction (Roche Inc, Switzerland).
Tissue Sampling
After cardiac function detection, 10% potassium chloride (10 mL) was used to euthanize the swine via ear vein injection. Hearts were isolated and cleaned with normal saline immediately. The anterior wall of the left ventricle dominated by LAD artery was isolated. Parts of myocardial tissue were immediately frozen in liquid nitrogen and stored at −80°C for Western blot analysis. The others were fixed with 4% paraformaldehyde for 12 hours, embedded in paraffin, and serially sectioned into slices of 4 μm thickness for hematoxylin and eosin (HE) staining, hematoxylin basic fuchsin picric acid (HBFP) staining, and terminal deoxynucleotidyl transferase-mediated dUTP Nick End-Labeling (TUNEL) assay.
Microinfarct Size Measurement
The HBFP staining is an important method used to detect early myocardial ischemia or infarct region. The normal myocardial tissue was stained yellow or brown while the ischemia or necrotic myocardial tissue red. A DMR-Q550 pathological image analyzer (Leica, Germany) was used to observe HBFP-stained sections. Microinfarct area was counted in 5 randomly selected visual fields (magnification, × 100) in each section using QWin analysis software (Leica, Germany), and 5 sections per swine were examined. The area of microinfarction was determined by planimetry and expressed as percentage of the total analyzed area.
TUNEL Assay
Apoptotic cardiomyocytes were detected using TUNEL assay kit (Roche, USA). The TUNEL-positive signal was located in nuclei, and apoptotic nuclei were stained yellow–brown while the normal light blue. Meanwhile, morphological features of apoptosis (small, condensed nuclei, cell shrinkage, and nuclear fragmentation) were taken into consideration. In each section, 10 random high-powered fields (magnification, ×400) were observed to count TUNEL-positive nuclei, and apoptotic index (%) was calculated as the percentage ratio of TUNEL-positive cell nuclei to the total nuclei.
Western Blot Analysis
Total protein was extracted using lysis buffer (containing Phenylmethanesulfonyl fluoride [PMSF]). The bicinchoninic acid method was used to determine protein concentration, and 30 μg protein sample was separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes (Millipore, Bedford, Massachusetts), which were blocked with 5% nonfat milk dissolved in 0.05% Tris Buffered Saline with Tween (TBST) at room temperature for 1 hour followed by incubation overnight at 4°C with following primary antibodies: LOX-1 (1:1000, abcam, Shanghai), Bcl-2 (1:500, BIOSS, USA), Bax (1:1000, abcam), cytochrome c (1:1000, Cell Signaling Technology, USA), caspase 9 (1:1000, abcam), caspase 8 (1:1000, Cell Signaling Technology), and caspase 3 antibody (1:1000, abcam). The membranes were washed and then incubated with the fluorescent secondary antibody (1:5000, LICOR, USA) at room temperature for 2 hours. The Odyssey 2-color infrared fluorescence imaging system (LICOR) was utilized to scan the membranes. Band density was determined by Quantity One software (Version 4.6.2, Bio-Rad, USA).
Statistical Analysis
Statistical analyses were performed using SPSS 19.0 statistical software (IBM Corporation, Chicago, IL). Quantitative data were expressed as mean ± standard deviation (SD). Analysis of variance was used for comparison among multiple groups, followed by Student-Newman-Keuls test for the further conduction of post hoc analysis. Pearson correlation analysis was performed for linear correlation. Statistical significance was presented as P < .05.
Results
In this study, there was no statistically significant difference (P > .05) among groups in regard to heart rate or body weight (see Table 1)
Physiological data of Swine among Groups.

Abbreviations: CME, coronary microembolization; Pre, preprocedure; Post, postprocedure; LOX, low-density lipoprotein receptor; siRNA, small interfering RNA.
Cardiac Dysfunction Following CME
The myocardial dysfunction was induced following CME, characterized by increased LVEDD and LVESD as well as decreased LVEF, FS, and CO (P < .05) as shown in Table 2. The LOX-1 siRNA improved myocardial function following CME (P < .05), while the control siRNA had no significant effect (P > .05).
Effects of LOX-1 siRNA on Cardiac function Following CME.
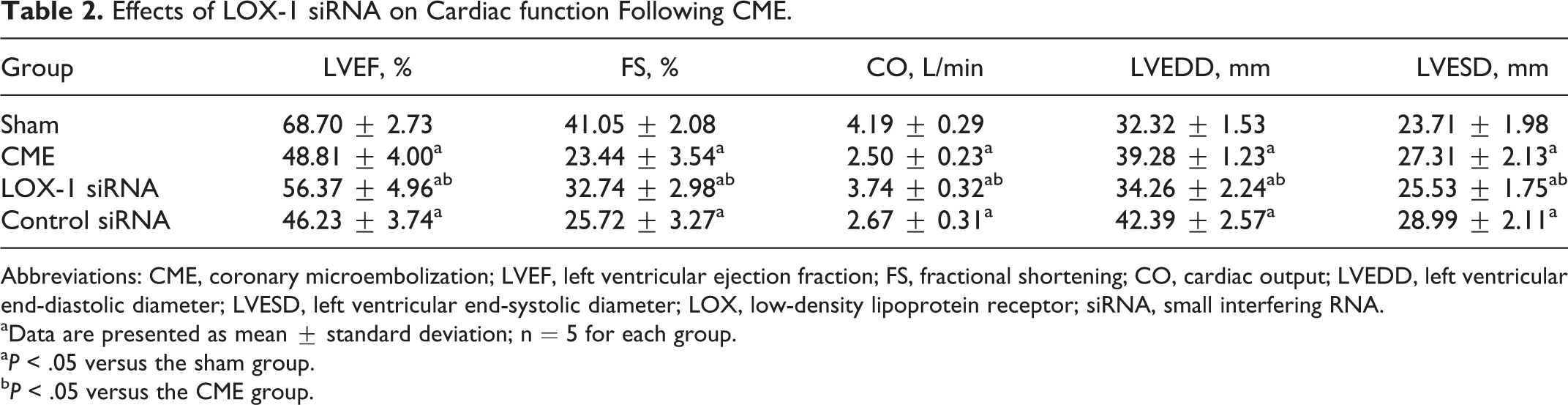
Abbreviations: CME, coronary microembolization; LVEF, left ventricular ejection fraction; FS, fractional shortening; CO, cardiac output; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; LOX, low-density lipoprotein receptor; siRNA, small interfering RNA.
aData are presented as mean ± standard deviation; n = 5 for each group.
aP < .05 versus the sham group.
bP < .05 versus the CME group.
Elevated Serum c-troponin I Level Following CME
The serum c-troponin I level was significantly enhanced following CME (P < .05) as shown in Table 3. Pretreatment with LOX-1 siRNA attenuated significant serum c-troponin I level (P < .05). The control siRNA had no significant effect on serum c-troponin I level (P > .05).
Effect of LOX-1 siRNA on Serum c-Troponin I Level After CME.a
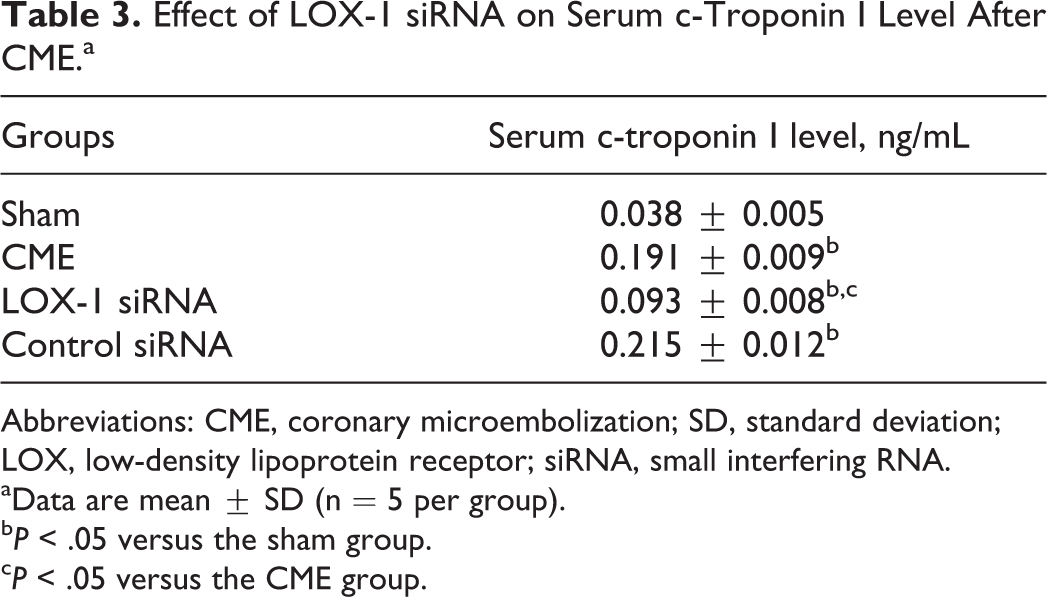
Abbreviations: CME, coronary microembolization; SD, standard deviation; LOX, low-density lipoprotein receptor; siRNA, small interfering RNA.
aData are mean ± SD (n = 5 per group).
bP < .05 versus the sham group.
cP < .05 versus the CME group.
Microinfarct Following CME
No infarct was observed in the sham group. As shown in Figure 1, HE staining showed multiple focal microinfarct around microspheres. Additionally, cardiomyocytes surrounding microinfarct were edematous with inflammatory cell infiltrates. As shown in Figure 2, HBFP staining revealed nontransmural multiple focal microinfarct in hearts of swine subjected to CME, mainly in the subendocardium, and the anterior wall of the left ventricle. No microinfarct was found in the sham group, while it was significantly evident in the CME group (P < .05). LOX-1 siRNA significantly reduced the microinfarct area (P < .05). The control siRNA had no significant effect (P > .05).

Pathohistological examination by HE staining at 12 hours following coronary microembolization (magnification, ×200; bar = 50 μm). The HE staining showed multiple focal microinfarct around microspheres with inflammatory cells infiltration. The arrow points to microspheres. HE indicates hematoxylin and eosin.

Microinfarct measurement with HBFP staining (magnification, ×100; bar = 25 μm). The normal tissue was stained yellow, while the infarct red. A, HBFP staining showed no microinfarct in the sham group (a). Infarct was evident following coronary microembolization (b). The increase was inhibited by LOX-1 siRNA (c), while the control siRNA showed no effect (d). B, The bar graph shows infarct size. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. HBFP indicates hematoxylin basic fuchsin picric acid; LOX, low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.
Cardiomyocyte Apoptosis Following CME
The apoptotic index was significantly enhanced in the CME group (P < .05) as shown in Figure 3. The LOX-1 siRNA pretreatment significantly decreased apoptotic index (P < .05). The control siRNA had no significant effect (P > .05).

Apoptosis detected by TUNEL staining (magnification, × 400; bar = 100 μm). A, TUNEL-positive signal was located in nuclei, and apoptotic nuclei were stained yellow-brown while the normal cell nuclei light blue. The arrow indicates TUNEL-positive nucleus. a, sham; b, CME; c, LOX-1 siRNA; d, Control siRNA. B, The bar graph shows apoptotic index. Apoptosis was evident in the CME group. LOX-1 siRNA decreased apoptotic index. The control siRNA showed no effect. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. TUNEL indicates terminal deoxynucleotidyl transferase-mediated dUTP Nick End-Labeling; CME, coronary microembolization; LOX, low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.
Expression of LOX-1 protein Following CME
The LOX-1 protein expression detected by Western blot was significantly increased in the CME group (P < .05) as shown in Figure 4. Pretreatment with LOX-1 siRNA significantly inhibited its upregulation (P < .05). The control siRNA had no effect on LOX-1 expression (P > .05).

LOX-1 upregulation. A, Western blot showed that LOX-1 protein expression was increased by coronary microembolization (CME). LOX-1 siRNA inhibited its increase despite CME. The control siRNA had no effect. Representative blots shown. B, Semiquantitation of bands of LOX-1. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. LOX indicates low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.
Expression of Apoptotic Related Proteins Following CME
The Bcl-2 expression was reduced, and the expression of other proteins (Bax, cytochrome c and cleaved caspase-9, cleaved caspase-8, and cleaved caspase-3) was increased in the CME group compared with those in the sham group as shown in Figures 5 to 7. LOX-1 siRNA reversed the expression of Bcl-2, Bax, cytochrome c, cleaved caspase-9, and cleaved caspase 3 following CME but not cleaved caspase-8. The control siRNA showed no effect.
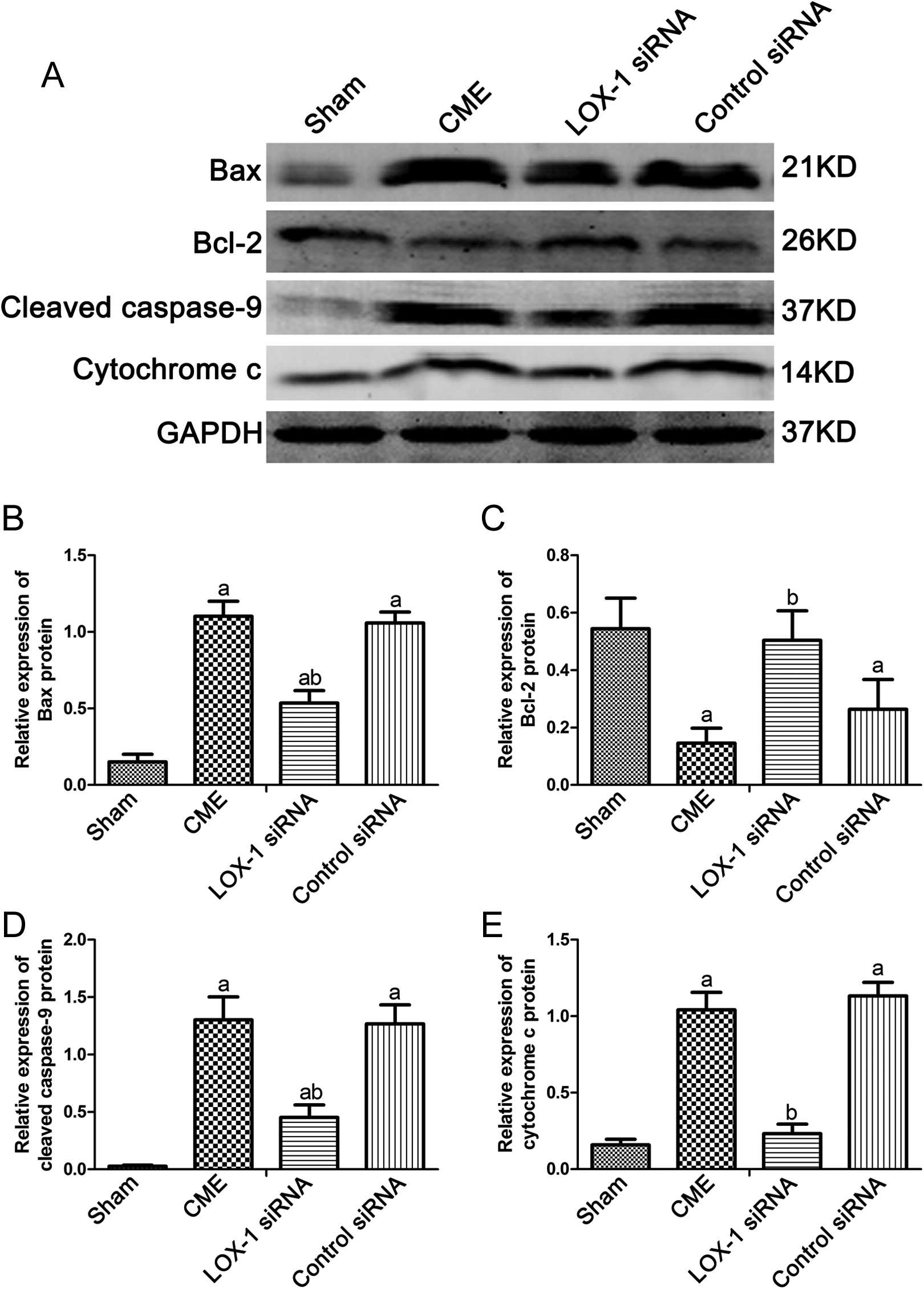
Mitochondria apoptosis-related protein activation. A, Bcl-2 expression was decreased, and the expression of Bax, cytochrome c, and cleaved caspase-9 was increased following coronary microembolization (CME). LOX-1 siRNA reversed the expression of CME on the expression of these proteins. The control siRNA had no effect. Representative blots are shown. B, Semiquantitation of bands of Bcl-2, Bax, cytochrome c, and cleaved caspase-9. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. LOX indicates low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.

Caspase-8 activation. A, Cleaved caspase-8 expression was enhanced following coronary microembolization (CME). Both LOX-1 siRNA and control siRNA showed no effect on cleaved caspase-8 expression. Representative blots are shown. B, Semiquantitation of bands of cleaved caspase-8. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. LOX indicates low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.

Caspase-3 activation. A, Cleaved caspase-3 expression was enhanced following coronary microembolization (CME). LOX-1 siRNA inhibited its increase, while the control siRNA showed no effect. Representative blots shown. B, Semiquantitation of bands of cleaved caspase-3. Data are mean ± SD (n = 5 per group). aP < .05 versus the sham group; bP < .05 versus the CME group. LOX indicates low-density lipoprotein receptor; siRNA, small interfering RNA; SD, standard deviation.
Linear Correlation Analysis
The level of LOX-1 positively correlated with apoptotic index (r = .864; P < .01), whereas it negatively correlated with LVEF (r = −.871; P < .01). This is shown in Figure 8.

Linear correlation analysis. The level of LOX-1 protein was positively correlated with apoptotic index (A) and negatively correlated with LVEF (B). LOX indicates low-density lipoprotein receptor; LVEF, left ventricular ejection fraction.
Discussion
The present study was designed to explore the role of LOX-1 in CME-induced apoptosis in a swine model of CME showing that (1) myocardial dysfunction, microinfarct, enhanced serum c-troponin I level, and apoptosis were induced following CME. (2) Mitochondrial apoptosis-related proteins (Bcl-2, Bax, cytochrome c, and caspase-9), caspase-8, and caspase-3 were all activated in hearts following CME. In addition to that LOX-1 protein expression was markedly enhanced. (3) LOX-1 siRNA reversed these effects by CME but not caspase 8, while the control siRNA showed no effect.
Coronary microembolization and its sequelae are frequent clinical events in patients undergoing PCI. Furthermore, CME seems to be inevitable during PCI but differs in its severity. The mild CME does not cause clinical symptom, while the serious CME may induce no reflow and even sudden death. Bose et al concluded that patients with large necrotic core may be more susceptible. 10 Periprocedural myocardial injury is closely related to microembolization. A model resembling those patients who had embolism in their coronary microvasculature during PCI is critical for understanding the pathophysiology of CME. An ideal animal model that could totally simulate the CME phenomenon seen in clinics does not exist; however, the cardiovascular system of the swine closely resembles that in humans. Some researchers caused CME by injecting 3000 microspheres (42 μm) per mL/min showing perfusion–contraction mismatch with reduced contractile function and unchanged or even elevated blood flow. 11,12 Chen et al induced CME in mini-pigs by injecting a mean dosage of 120 000 microspheres into LAD artery, finding myocardial dysfunction, microinfarcts, elevated inflammatory cytokines, and apoptosis. 3 Li et al established CME model in rats by injecting homologous thrombotic material finding inflammation and left ventricular dysfunction. 13 These results are partially consistent with those in clinical scenarios. These models could partially simulate the CME phenomenon in clinics but differ in its severity. Our experimental approach simulated the occurrence of CME during PCI through injecting a dosage of 100 000 microspheres of 42 μm diameter into the LAD artery finding that myocardial dysfunction, microinfarct with inflammation, elevated serum c-troponin I, and apoptosis were induced in a swine model. These events are consistent with sequelae of patients subjected to CME in clinical settings. Therefore, this swine model of CME could partially simulate the status of patients subjected to CME in clinical scenarios.
LOX-1, Apoptosis-Related Proteins, and Apoptosis Following CME
Cardiomyocytes loss by apoptosis is involved in several cardiovascular diseases including heart failure, ischemia–reperfusion injury, and acute myocardial infarction (MI). 14,15 Decreased apoptosis by caspase inhibition has been determined to improve myocardial function and therefore might improve clinical outcomes. 16 –18 Recent reports determined that apoptosis plays an important role in CME-induced myocardial impairment, despite earlier reports of almost negligible effect. 3,19 This was confirmed in the present study by showing that CME induced increased apoptotic index and cardiac dysfunction. The mitochondria has been the primary concern since it was described as the powerhouse of cells to generate adenosine triphosphate. However, emerging studies have proposed its role in the process of apoptosis. 20,21 It is well known that the involvement of mitochondria is dependent on actions of Bcl-2 and Bax to promote the release of apoptotic proteins such as cytochrome c. Then the released cytochrome c induces the activation of caspase 9 and -3, and therefore apoptosis occurs. 4 Our previous research revealed that caspase 9, caspase 8, and caspase 3 were activated in the process of apoptosis following CME, which means mitochondria pathway and death receptor pathway were activated following CME. 22,23 Moreover, in the present study, we showed that the expression of Bax and cytochrome c was enhanced, and Bcl-2 expression was decreased following CME.
The LOX-1 is a newly determined membrane receptor for several ligands. It was reported to be highly expressed in atherosclerotic lesion and has been proposed in several cardiovascular diseases including acute coronary syndrome. Li et al showed that LOX-1 upregulation during myocardial ischemia–reperfusion injury was associated with apoptosis and cardiac dysfunction. 24 Inhibition of LOX-1 by LOX-1 antibody improved myocardial function and reduced apoptotic index. It means that LOX-1 contributes to apoptosis during ischemia–reperfusion injury. Recently, Hong et al showed that LOX-1-mediated endoplasmic reticulum stress pathway contributes to ox-LDL-induced apoptosis on endothelial cells. 25 However, its role in the regulation of apoptosis by CME remains to be elucidated. In the present study, we showed that effects of CME on mitochondria apoptotic proteins were markedly blocked by an LOX-1 siRNA, however it did not lead to any significant changes on cleaved caspase-8 expression. Moreover, we showed that the relative expression of LOX-1 was positively correlated with apoptotic index. These results indicate that CME induces apoptosis by activating the LOX-1-mediated mitochondria pathway and caspase-8-dependent pathway. And signal transduction of cardiomyocyte apoptosis resulting from CME is shown in Figure 9.
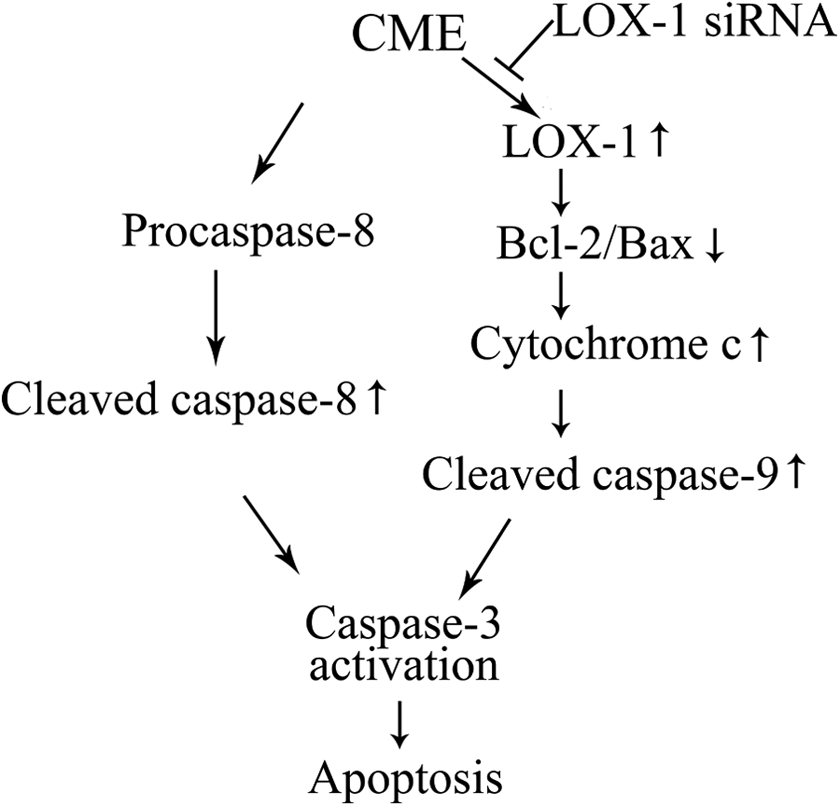
Signal transduction of CME-induced cardiomyocyte apoptosis. CME indicates coronary microembolization; LOX-1, lectin-like oxidized low-density lipoprotein receptor 1.
LOX-1 and Myocardial Injury Following CME
Coronary microembolization resulting from the washout of atherothrombotic debris into coronary microcirculation could induce microinfarct and therefore enhanced serum markers of myocardial impairment, such as troponin I. Final infarct size is proposed as a strong predictor of myocardial function recovery and long-term clinical outcome following infarction. Markers of myocardial necrosis in serum samples including troponin I is generally measured to estimate infarct area. 26 In this study, we showed that CME induced cardiac dysfunction, increased microinfarct size, and serum c-troponin I level. Therapeutic strategies targeting to reducing infarct area might therefore improve myocardial function in return. 27 Thus, LOX-1 siRNA reduced microinfarct area and serum c-troponin I level and improved myocardial function. These effects may partially be involved in apoptosis inhibition attributed to LOX-1 siRNA. Tumor necrosis factor α has been determined as a casual factor for myocardial dysfunction following CME. 28 Furthermore, TNF-α is in vitro stimuli for LOX-1 upregulation. 29 This means TNF-α may induce cardiac dysfunction following CME via the LOX-1 signaling pathway, which needs further research.
The level of soluble form of LOX-1 (sLOX-1) is believed to reflect LOX-1 expression of atherosclerotic plaque. Elevated sLOX-1 level of circulating blood is a strongly predictor of atherosclerotic progression. Recently, Kobayashi et al revealed that plasma sLOX-1 could diagnose the early stage of ST-elevation MI. 30 Coronary microembolization is thought to be a major cause of PCI-related periprocedural MI. Troponin and CK-MB are well-known biomarker of myocardial injury. Balin et al revealed sLOX-1 levels were significantly in positive correlation with those of troponin and CK-MB. 31 This means sLOX-1 may be an early and novel biomarker of MI. This will be further confirmed in our future research.
Conclusion
Our results indicate that CME induces cardiomyocyte apoptosis in a swine model via the activation of LOX-1-dependent mitochondrial pathway and caspase-8-dependent pathway. LOX-1 might be a potential target for cardioprotection.
Footnotes
Author Contributions
Lang, L contributed to conception, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Tao, L contributed to design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript; You, Z contributed to acquisition, analysis, and interpretation; You, W contributed to acquisition, analysis, and interpretation; Qiang, S contributed to analysis; LI, T contributed to acquisition; Chun, L contributed to analysis and critically revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Nature Science Foundation of China (NO. 81260042).
