Abstract
The von Willebrand factor (vWF) aptamer, ARC1779 that blocks the binding of vWF A1-domain to platelet glycoprotein 1b (GPIb) at high shear, may deliver a site-specific antithrombotic effect. We investigated the efficiency of ARC1779 on platelet function in patients with coronary artery disease (CAD) on double antiplatelet therapy. Blood from patients taking aspirin and clopidogrel and from normal volunteers was treated ex vivo with ARC1779 or abciximab, either prior to perfusion (pretherapy) or 10 minutes following the initiation of perfusion (posttherapy) on damaged arteries. Under pre- but not posttherapy, platelet adhesion was significantly reduced by ARC1779 at 83 and 250 nmol/L and by abciximab (100 nmol/L) versus placebo (4.8, 3.8, and 2.9 vs 7.3 platelets × 106/cm2,
Introduction
Antithrombotic therapy has been shown to reduce the risk of death and myocardial infarction (MI) from 20% to 4% - 8% in acute coronary syndromes (ACS). 1 However, it is commonly complicated by major bleeding, which is strongly associated with an increased risk of subsequent mortality. 2 Therefore, contemporary drug development programs seek to identify molecular targets delivering reduced bleeding risk with preserved antithrombotic efficacy.
On vascular injury, von Willebrand factor (vWF) promptly interacts with the newly exposed subendothelial components, setting in motion the processes of platelet adhesion and aggregation.3,4 The A1 domain of vWF reversibly links platelets to the subendothelial matrix via an interaction with platelet glycoprotein 1b (GPIbα). Following initial platelet capture from flowing blood at the site of vascular injury, platelet GPVI stimulation and GPIIb/IIIa activation ensues, leading to irreversible platelet adhesion, spreading, aggregation, and thrombus formation. 4 High shear stress is necessary and sufficient for vWF to adopt the proper conformation to support platelet adhesion and aggregation. Therefore, in the absence of high shear force, vWF is not sufficiently activated and its contribution to thrombogenesis is greatly reduced.5,6
ARC1779, an aptamer that binds to the A1 domain of activated vWF, inhibits binding of vWF to the GPIb receptor on platelets, inhibiting all of the vWF-mediated activation pathways and potentially blocking pathological thrombosis.7,8 We showed that ARC1779 inhibited botrocetin- and shear force-induced platelet aggregation. It also reduced adhesion of platelets to collagen-coated matrices, formation of platelet thrombi on denuded porcine arteries, and formation of occlusive thrombi in cynomolgus monkeys. 9 In a phase I study of 47 healthy participants, ARC1779 demonstrated a favorable early safety profile with no bleeding complications and only minor adverse events. 7 More recently, it has been shown that ARC1779 potently and specifically inhibits vWF activity and vWF-dependent platelet function, even in the setting of acute MI where vWF activity is increased. 10 Actually, ARC1779 is being evaluated in a phase II clinical study on cerebral microembolism in patients undergoing carotid endarterectomy (CEA). However, the pharmacodynamic effect of ARC1779 in combination with other commonly used antiplatelet agents is unknown. We, therefore, sought to investigate the efficiency of ARC1779 on platelet activation, adhesion, and aggregation in the presence of 2 antiplatelet agents (aspirin and clopidogrel) used routinely in clinical practice in patients with coronary artery disease (CAD), as well as in unstable carotid disease.
Materials and Methods
Design of the Study and Protocol
The ethics committee of the Montreal Heart Institute approved the study, and all participants provided written informed consent. This is a single-center, randomized, open-label study in patients undergoing percutaneous coronary intervention (PCI) at the Montreal Heart Institute. Patients were eligible for the study if they were between 18 and 75 years of age, had documented stable CAD defined as prior MI (>90 days) and/or at least 1 lesion >50% in a major epicardial artery on angiography, and were receiving maintenance treatment with dual aspirin 81 mg and clopidogrel 75 mg daily at least 7 days prior to study entry. We excluded patients with a recent episode of ACS (<90 days), stroke within 3 months, malignancies, active bleeding or bleeding diathesis, oral anticoagulation with a coumarin derivate or prothrombin time >1.5 times control, platelet count <100 000/mm3, serum creatinine >180 mmol/L, or liver disease resulting in bilirubin level >2 mg/dL. Healthy volunteers who were not consuming prescription or over-the-counter medications for at least 10 days before blood sampling were selected as the control group.
Blood from patients with CAD treated with ASA (aspirin) + plavix (clopidogrel) were compared to those obtained from normal volunteers. Patients were randomly assigned to pretherapy or posttherapy ex vivo with ARC1779 (Archemix corporation, Cambridge, Massachusetts) or abciximab (ReoPro, Eli Lilly, Toronto, Canada). Treatments for pre-therapy were incubated 5 minutes before the onset of perfusion, whereas those for posttherapy were administered 10 minutes after the beginning of the perfusion. Based on previously published data,7,9 we compared escalating doses of ARC1779 (25, 83, and 250 nmol/L), with abciximab (100 nmol/L) and with saline, the vehicle of ARC1779. Synthesis, structure, and pharmacology of ARC1779 were reported previously.7,9
Platelet Adhesion
Platelet isolation and labeling, as well as the perfusion experiments were conducted as described previously.9,11 Briefly, a 60-mL sample of venous blood from each participant was anticoagulated with 6 mL of H-D-Phe-Pro-Arg-chloromethylketon (PPACK) in saline (50 nmol/L final concentration; Calbiochem, QC, Canada), and a 30-mL with anticoagulant citrate dextrose (ACD; Baxter, Mississauga, Canada). The ACD blood was used to isolate and radiolabel platelets with In111 and resuspended in the remaining 60 mL of the PPACK blood. Fresh porcine aortas (Aggromex, St-Blaise, Canada) were isolated and denuded by lifting and peeling off the intima to expose the subjacent media. The segments were placed into Badimon perfusion chambers with a 1 mm internal diameter × 10 mm long. The chambers were placed in parallel in a thermostatically controlled water bath at 37°C, thus permitting simultaneous parallel, pairwise perfusion over arterial tissues of treated or untreated blood at a high shear (6974/s). Blood (10 mL) was recirculated over the arterial segments for 15 minutes in the flow chambers. A known GPIIb/IIIa antagonist (100 nmol/L of abciximab or ReoPro) and a placebo (physiological saline) were used as controls, and ARC1779 was tested at 25, 83, and 250 nmol/L. The arterial segments were then fixed in formalin 1% and placed in polystyrene tubes for gamma counting to quantify platelet adhesion, as described previously. 12 After compilation of gamma counting, the arterial segments were prepared and observed by scanning electron microscopy.
Platelet Aggregation
Blood from each treatment was collected and diluted 1:1 in physiological saline. Platelet aggregation was measured at 37°C in a whole blood lumi-aggregometer (Chrono-log Corporation, Havertown, Pennsylvania) after the addition of adenosine diphosphate (ADP) 10 µmol/L (Chrono-log), ristocetin 1 mg/mL (Helena Laboratories, Beaumont, Texas), arachidonic acid (AA) 500 µmol/L (Chrono-log), thrombin receptor-activating peptide 1 (TRAP-1 5 µmol/L; Peptides International, Louisville, Kentucky), and collagen (2 ug/mL, Chrono-log). Aggregation was recorded for 10 minutes and expressed in ohms (Ω).
Platelet Activation
The effects of ARC1779 on platelet activation was evaluated in healthy volunteers, where 50 µL of blood were fixed in 950 µL of phosphate buffered saline (PBS)-paraformaldehyde 1% for 30 minutes before being centrifuged at 7000
Statistical Analysis
Data from the characteristics of the study population were expressed as mean and standard error of mean (SEM) for continuous variables and as total number of patients and percentages for categorical variables. Continuous data were analyzed using a nonparametric 1-way analysis of variance (ANOVA) followed by Dunnett test and expressed as mean (SEM), whereas categorical data were analyzed using the chi-square test. All
Results
Study Population
The characteristics of the patient population and the healthy controls are summarized in Table 1. The mean age of the CAD patient group was 57.2 ± 1.5 years (81.5% male) and the mean age of the healthy volunteer group was 40 ± 5.3 years (20% male). The mean duration of combined aspirin and clopidogrel treatment prior to study entry was 10 ± 5 months.
Baseline Characteristics of the Study Population
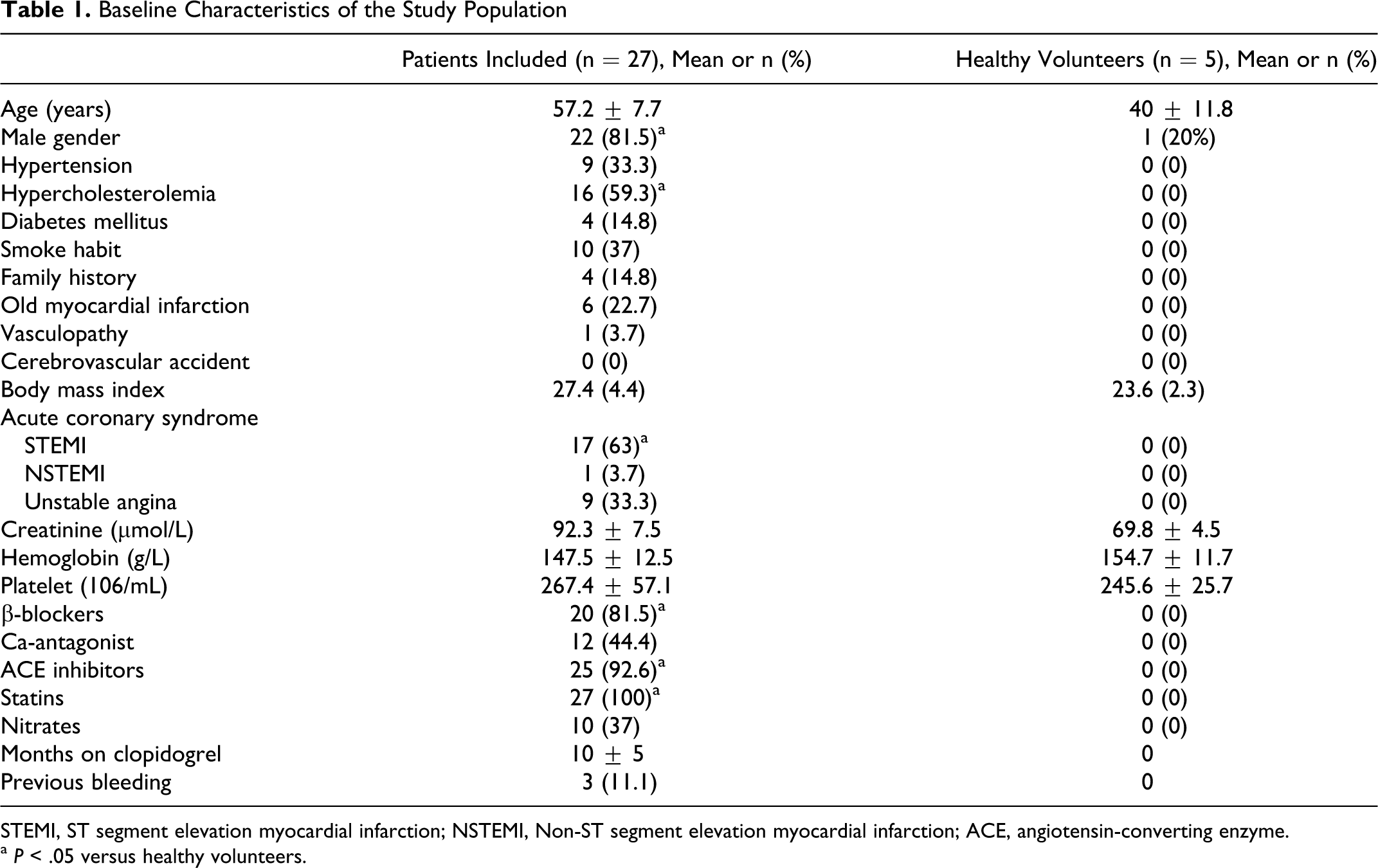
STEMI, ST segment elevation myocardial infarction; NSTEMI, Non-ST segment elevation myocardial infarction; ACE, angiotensin-converting enzyme.
a
Platelet Adhesion
Pretherapy was used with the blood of 5 healthy volunteers and 17 patients with CAD, whereas blood from the remaining 10 patients with CAD was used to assess posttherapy. As shown in Figure 1, platelet adhesion was highest when the arterial samples were perfused with blood from healthy volunteers treated with placebo. Platelet adhesion in this group averaged 81.9 ± 23.6 × 106 platelets/cm2. Administration of abciximab before perfusion almost completely inhibited platelet adhesion to 2.2 × 106 platelets/cm2. In the healthy controls, ARC1779 at 25 and 83 nmol/L had no significant effect on platelet adhesion, whereas it significantly reduced platelet adhesion by 89% to 8.8 × 106 platelets/cm2 at 250 nmol/L.
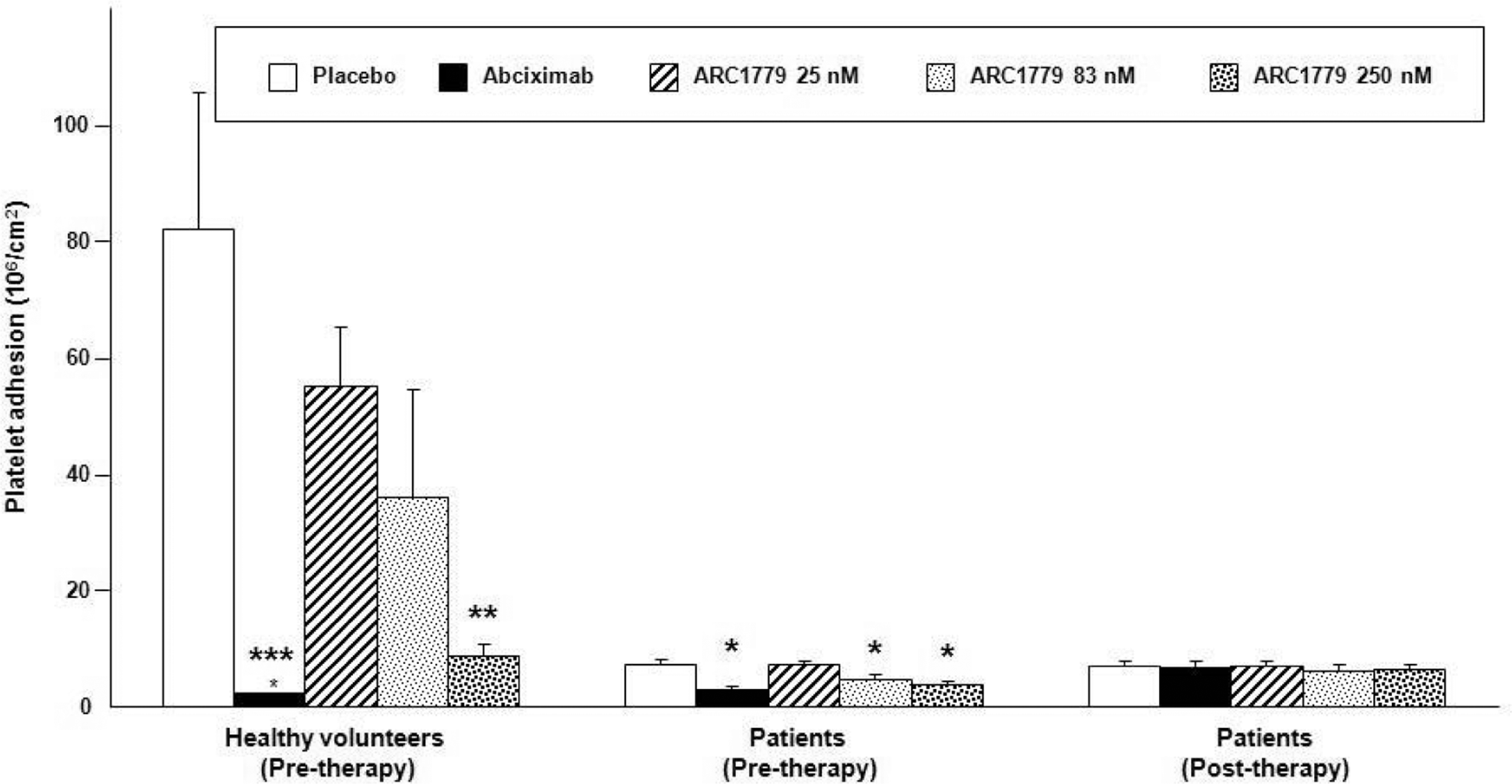
Effects of ARC1779 and abciximab on platelet adhesion in healthy control and in patients on aspirin and clopidogrel. 111In-labeled human platelets in whole blood were circulated over denuded (injured) porcine arterial segments in perfusion flow chambers at a shear rate of 6974/s for 15 minutes at 37°C. In 17 patients and in 5 normal volunteers, the drugs (abciximab and ARC1779) were preincubated with the blood for 5 minutes before each perfusion and defined as pretherapy. In 10 patients, the drugs were added 10 minutes after the onset of blood perfusion and defined as posttherapy. (Mean ± SEM, *
In patients pretreated with clopidogrel and aspirin and randomized to pretherapy, a significant reduction in adhesion was observed in perfusions with placebo compared to that observed in healthy volunteers, 7.3 ± 1.0 × 106 platelets/cm2 versus 81.9 ± 23.6 × 106 platelets/cm2. Treatment of patient blood with ARC1779 at 83 and 250 nmol/L or with abciximab further reduced platelet adhesion to 4.8, 3.8, and 2.9 × 106 platelets/cm2, respectively (Figure 1).
In patients treated with dual antiplatelet therapy in which the posttherapy protocol was followed, no significant difference was observed in platelet adhesion among the different therapies compared to the perfusion without ARC1779 (Figure 1).
The results of the adhesion assay were confirmed by scanning electron microscopy visualization of platelet adhesion in each of the treatment groups. As shown in Figure 2, arterial surfaces exposed to the blood of healthy volunteers with placebo, accumulated a heavy thrombotic matrix, which was markedly attenuated when blood from patients treated with aspirin and clopidogrel was used. Thrombus formation was further decreased by both abciximab and ARC1779, given as pretherapy, with an equal degree of thrombus reduction with both treatments.
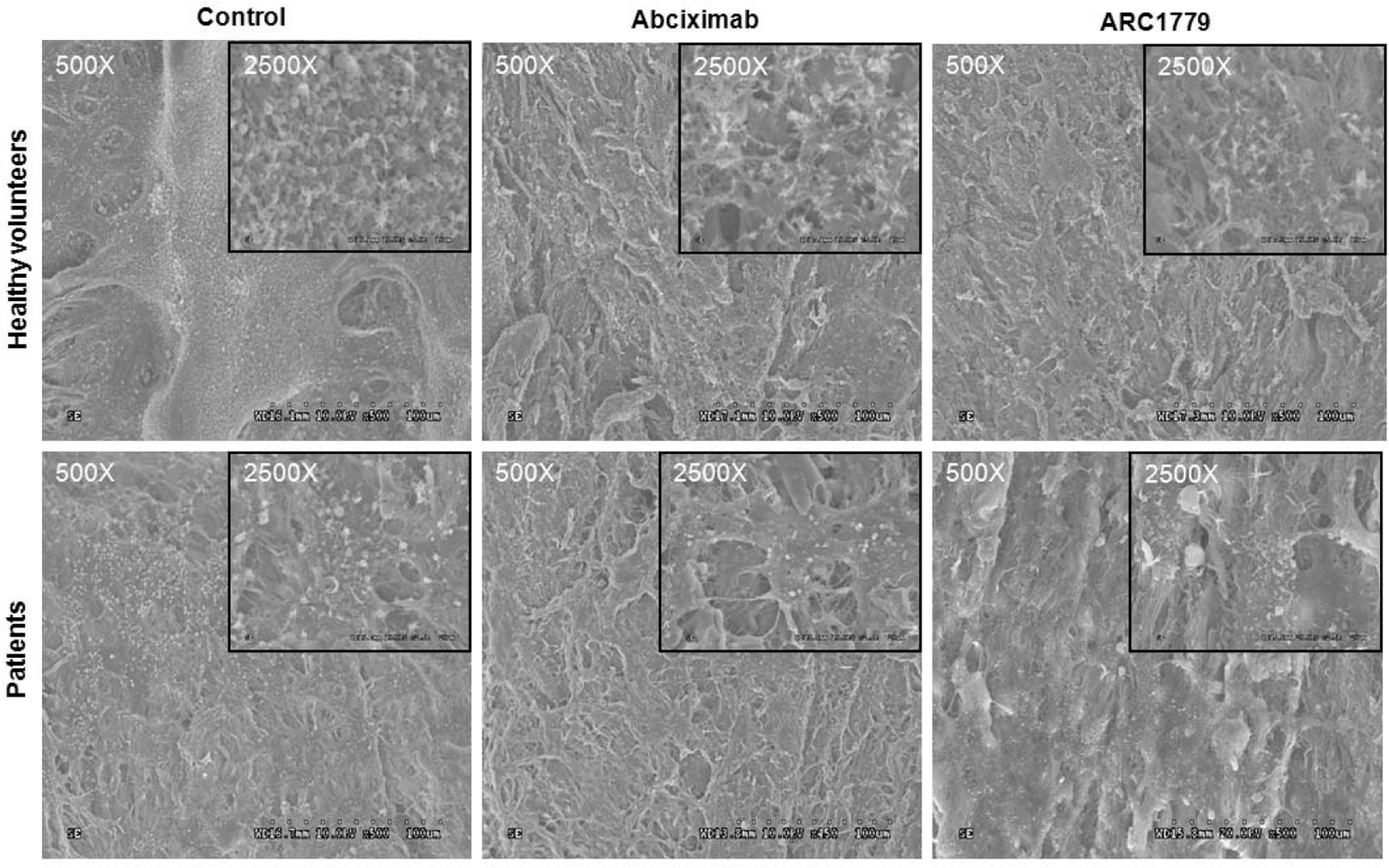
Representative scanning electron microscopy micrographs of the resulting platelet adhesion following exposure of whole blood to denuded (injured) porcine arterial segments in perfusion flow chambers at a shear rate of 6974/s for 15 minutes at 37°C. Both the effect of abciximab (100 nmol/L) and ARC1779 (250 nmol/L) are depicted in healthy volunteers and patients under pretherapy. Magnifications ×500, Insert ×2500.
Platelet Aggregation
The impact of the different treatments on whole blood platelet aggregation in response to different platelet agonists was evaluated and presented in Figure 3. As expected, the responses to ADP and AA were completely prevented in patients on double antiplatelet therapy with aspirin and clopidogrel. Moreover, abciximab abolished platelet aggregation in response to TRAP-1, ADP, and AA both in healthy volunteers and in patients. The response to collagen activation was similar to TRAP-1. In healthy volunteers, collagen-induced aggregation averaged 18 Ω that was reduced to 2 Ω with abciximab, but unaffected by ARC1779 (18 Ω). In patients, collagen-induced aggregation was reduced to 3 Ω, abolished by abciximab, and unaffected by ARC1779 (4 Ω). In contrast, ARC1779 had no significant effect on platelet aggregation in response to TRAP-1, ADP, AA, or collagen. In response to ristocetin, abciximab and ARC1779 were equally effective in reducing platelet aggregation by 50% only in healthy volunteers. In patients receiving clopidogrel and aspirin, a reduction of ristocetin-induced aggregation was observed in the control group and in the group treated with ARC1779 but not with abciximab.
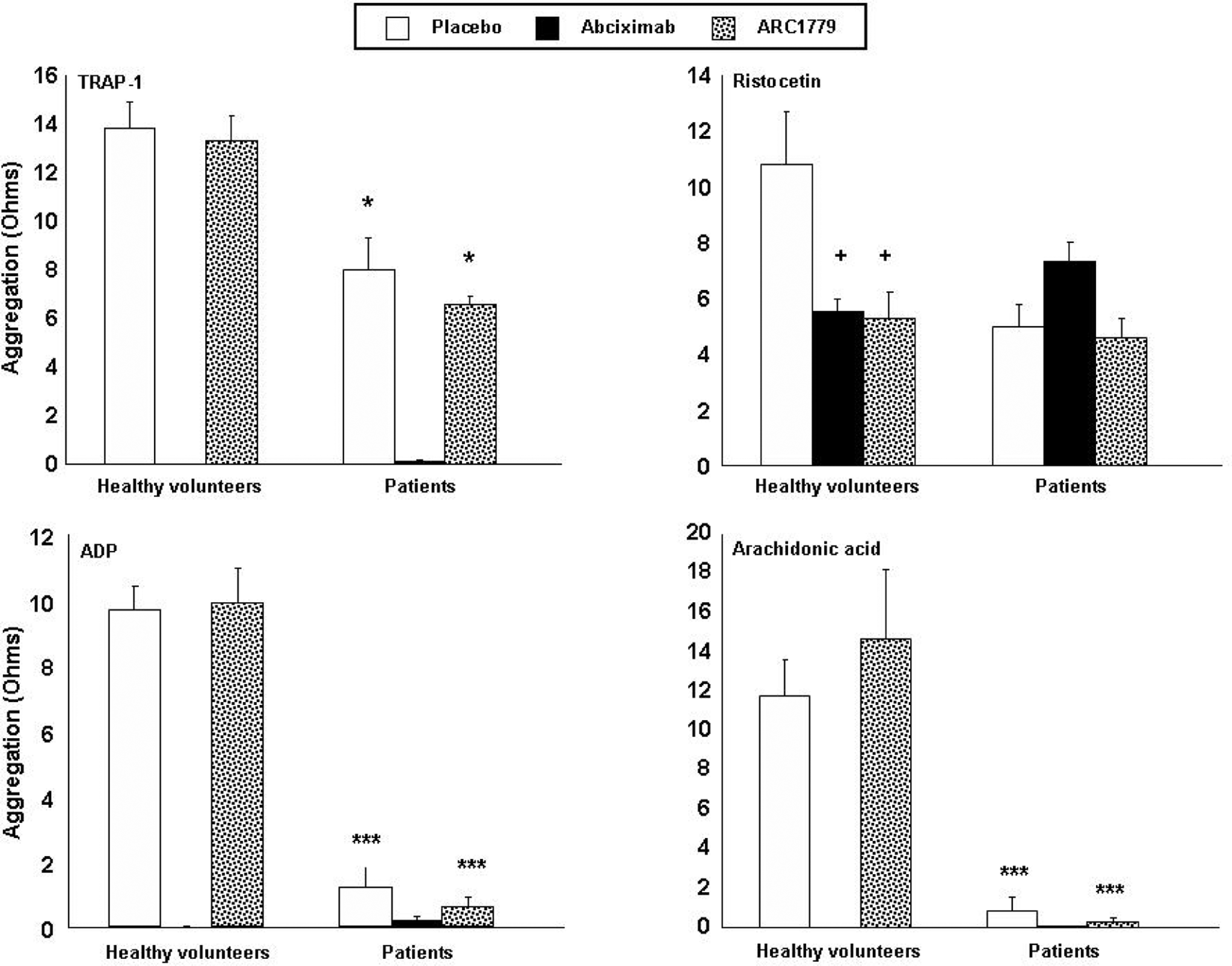
Effects of ARC1779 and abciximab on platelet aggregation in healthy control and in patients on aspirin and clopidogrel. Whole blood platelet aggregation in healthy volunteers and in patients in response to different agonists (TRAP-1 5 µmol/L, ristocetin 1 mg/mL, ADP 10 µmol/L, and arachidonic acid 500 µmol/L). Aggregation was monitored over 15 minutes in the presence of abciximab (100 nmol/L) or ARC1779 (250 nmol/L). Mean ± SEM, *
Platelet Activation
Platelet activation was assessed by the measurement of P- selectin (CD62P) translocation and vWF expression on the platelet surfaces, as well as by the determination of platelet−leukocyte aggregates, in the presence of ARC1779 or abciximab. This was done on blood samples from healthy volunteers after the perfusion experiments. As shown in Figure 4, neither abciximab nor ARC1779 has a significant effect on P-selectin or vWF expression. Although the binding of platelets to leukocytes increased after blood perfusion (control) in comparison with nonperfused blood (baseline), this increase was not significantly affected by ARC1779 or abciximab.
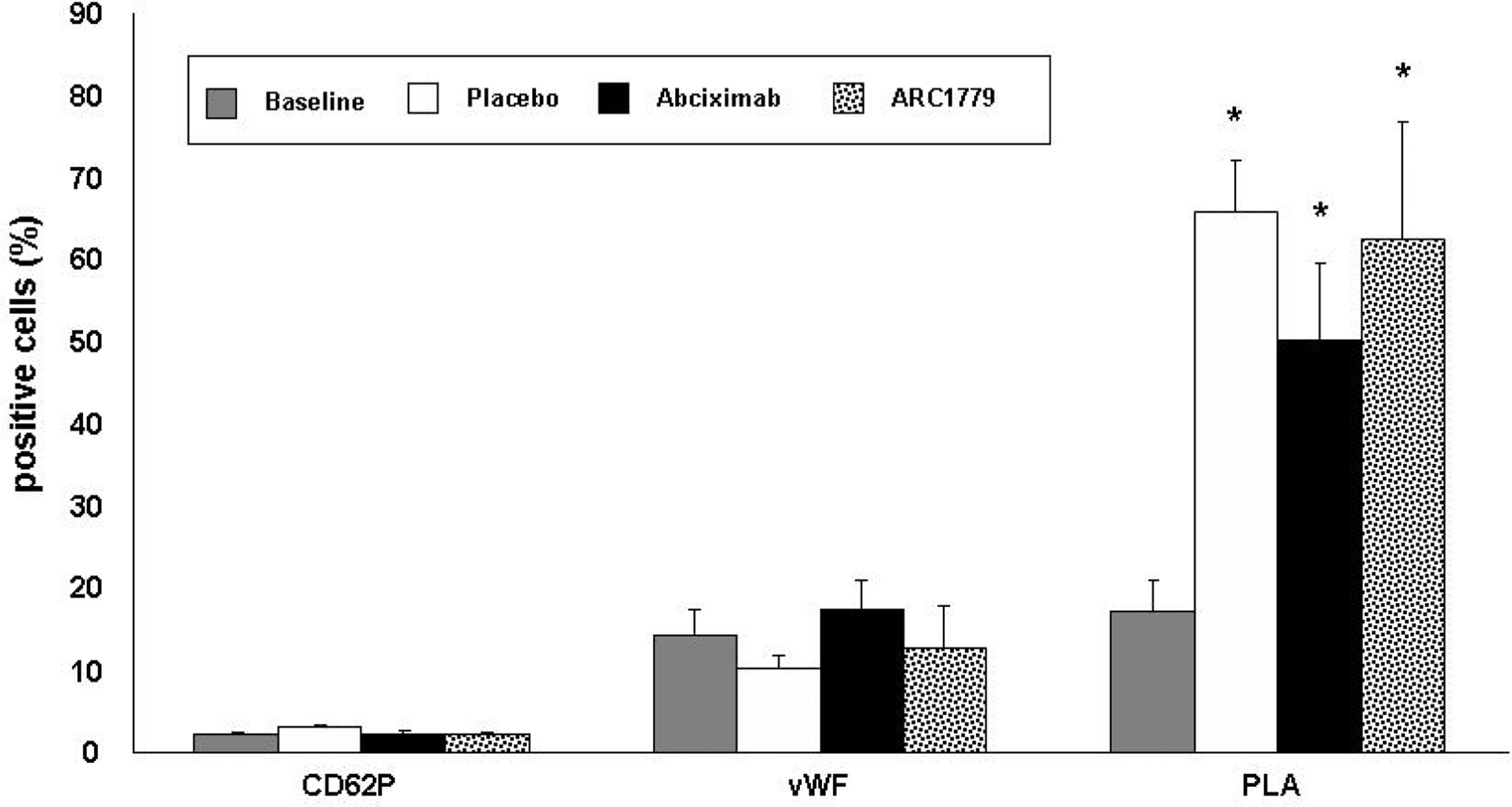
Effects of ARC1779 and abciximab on platelet activation. Whole blood platelet activation as assessed by P-selectin translocation (CD62P), vWF expression and platelet/leukocyte aggregate (PLA) formation following exposure of whole blood to denuded (injured) porcine arterial segments in perfusion flow chambers at a shear rate of 6974/s for 15 minutes at 37°C. Platelet activation was studied in healthy volunteers in the presence of abciximab (100 nmol/L) or ARC1779 (250 nmol/L). Mean ± SEM, *
Discussion
We investigated, in an ex-vivo model, the effects on platelet function of escalating concentrations of a vWF antagonist aptamer (ARC1779) in healthy volunteers and in CAD patients receiving aspirin and clopidogrel and compared it to placebo and abciximab administration. When preincubated with the blood before perfusion (pretherapy), we observed similar levels of reduction in platelet adhesion with the highest dose of ARC1779 (250 nmol/L) and abciximab (100 nmol/L), compared to the placebo group. These results were consistent in healthy volunteers, as reported previously,7,9 and in patients under dual antiplatelet therapy with aspirin and clopidogrel. In contrast, none of the treatments showed any benefit in the posttherapy settings, when treatments were delayed 10 minutes after the perfusion. In contrast to abciximab, ARC1779 did not significantly affect platelet aggregation in response to TRAP-1, AA, and ADP. In addition, ARC1779 was without any effect on P-selectin expression and platelet−leukocyte binding.
Despite their documented clinical benefits, contemporary antiplatelet agents are limited by interindividual treatment response variability,13–15 delayed onset of action,16–18 irreversibility, and increased bleeding complications. Thus, current research on antiplatelet therapy is mainly focused on the development of new agents capable of reducing bleeding complications while preserving their antithrombotic efficacy. The compatibility of novel antiplatelet therapies with aspirin and clopidogrel must be determined prior to their clinical investigation among patients with ACS and those undergoing PCI or CEA, as these 2 molecules are considered upstream therapy in such populations.19,20
In the current study, we analyzed the effect of ARC1779 alone and in combination with aspirin and clopidogrel. ARC1779 reduced significantly platelet adhesion, even in patients pretreated with aspirin and clopidogrel. However, no differences were observed on aggregation in healthy volunteers between control group and ARC1779, except when the platelet-agglutinating agent ristocetin was used. Compared to healthy volunteers, patients receiving clopidogrel and aspirin demonstrated a reduction in aggregation response to the different tested agonists. However, the addition of ARC1779 did not further attenuate platelet aggregation. Thus, the effect of ARC1779 is additive to aspirin and clopidogrel in preventing platelet adhesion but not in inhibiting platelet aggregation. We hypothesize that this observation may be explained by the different molecular targets of the 3 antiplatelet agents used in this study: the anti vWF aptamer binds to the A1 domain of vWF that interacts with the platelet GPIb receptor at high shear rate, whereas aspirin irreversibly inhibits the cyclooxygenase pathway leading to a reduction in thromboxane A2 (TxA2) formation from AA, and clopidogrel prevents ADP from interacting with the P2Y12 receptor. When the endothelial layer is disrupted, platelets adhere to newly exposed vWF and collagen via GPIb and GPVI, respectively. Thereafter, they become activated, change shape, and secrete secondary mediators such as ADP and TxA2, which in turn induce further platelet activation propagation and thrombus formation. 21 ARC1779 inhibits the initial adhesive contact between the platelet GPIb receptor and extracellular matrix-bound vWF at high shear, whereas aspirin and clopidogrel interferes with platelet aggregation, limiting the autocrine/ paracrine activation loop triggered by secreted TxA2 and ADP during platelet activation. 21 This property may be particularly useful in patients with high platelet reactivity despite treatment with clopidogrel or aspirin, given that ARC1779 offers an alternative pathway of platelet inhibition.
ARC1779 has a rapid onset of action, achieving maximum concentration levels in 7 to 30 minutes. 7 Hence, in situations where rapid antiplatelet action is needed, as in primary PCI or CEA, ARC1779 might be considered a promising therapy for future investigation. In this study, we simulated established administration protocols used in clinical practice with abciximab administered before PCI in the emergency room (upstream therapy) or periprocedurally in the cardiac catheterization laboratory (downstream therapy). Since contradictory results have been observed in recent studies on the administration of abciximab prior to PCI,22,23 we decided to study the behavior of ARC1779 in this setting. We observed that, in patients already pretreated with aspirin and clopidogrel, there was no significant decrease in platelet adhesion with ARC1779 or with abciximab administered as downstream therapy. In contrast, administration of the drug as upstream therapy had an additive effect on the dual antiplatelet therapy, reducing significantly platelet adhesion compared to placebo. This observation should be considered in future protocols of clinical studies with ARC1779.
von Willebrand factor is active only in the presence of high intravascular shear forces found in stenosed arteries or at the atherosclerotic plaque. High shear stress is necessary for vWF to adopt the proper conformation to support platelet adhesion and aggregation. Therefore, in the absence of high shear force, vWF is not sufficiently activated and its contribution to thrombogenesis is greatly reduced. Indeed, ARC1779 inhibits the initial adhesive contact between the platelet GPIb receptor and extracellular matrix-bound vWF at high shear, whereas aspirin and clopidogrel interferes with platelet aggregation. Thus, conventional antiplatelet therapies provide systemic effects that may contribute to bleeding events; whereas a site-specific drug like ARC1779 may help minimize bleeding. Thereby, a vWF antagonist may theoretically avoid hemorrhagic complications normally seen with conventional antiplatelet agents. In our ex-vivo perfusion chamber model, we studied the behavior of the ARC1779-vWF-platelet interaction in a high laminar shear environment (6974 s−1) emulating flow conditions in a moderately stenotic epicardial coronary artery. To assess possible systemic effects of ARC1779, FACS was performed and no differences were observed in platelet vWF and P-selectin expression, and in platelet−leukocyte aggregate formation compared to placebo group. This observation supports the concept of a localized effect of ARC1779, without systemic antithrombotic effects. Failure of systemic antiplatelet therapy to remain localized to the primary site of action, the coronary or carotid vasculature, may contribute to bleeding events, and a site-specific drug may help minimize bleeding. However, this potential benefit has to be confirmed in larger clinical studies powered for clinical bleeding end points.
Our study has several limitations. First, this was an ex vivo study which may not fully replicate the in vivo pharmacologic effects of ARC1779. Second, the relatively small sample size may affect the precision of our pharmacodynamic estimates. Third, CAD patients were on multiple co-medications and the impacts of any potential interactions with ARC1779 are unknown. Fourth, the mean age and the proportion of male gender were both higher in patients, as compared to healthy volunteers. Fifth, clinical end points were not studied, and the effects of ARC1779 on thrombotic and bleeding end points will require investigation in larger clinical studies.
Conclusions
In this preclinical ex vivo study in patients with CAD, we showed that an anti-vWF aptamer, ARC1779, has comparable antithrombotic efficacy to abciximab among patients receiving aspirin and clopidogrel but with lesser effects on platelet activation and aggregation. Because vWF functions predominantly in a high shear environment, ARC1779 may deliver a site-specific antithrombotic effect while minimizing bleeding risk. These important proof-of-concept data may constitute the framework for randomized clinical investigations of this novel antiplatelet therapy among coronary patients in the setting of PCI in ACS, or cerebral microembolism post CEA.
Footnotes
Drs Arzamendi and Dandachli contributed equally to this work. This study was previously presented at the following conferences:
Acknowledgment
We are grateful to M. Lyne Mongeon and Dr Maryam Tabrizian for their assistance in scanning electron microscopy.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: in part supported by Archemix corp., Cambridge, USA.
