Abstract
Guidelines for the diagnosis and management of heparin-induced thrombocytopenia (HIT) vary between hospitals. Recent guidelines recommend initiating alternative anticoagulant therapy in patients with suspected HIT while awaiting laboratory test results confirming the presence of heparin–PF-4 antibodies (PF-4). This retrospective chart review was designed to assess the current state of management of patients with thrombocytopenia suspected to be due to HIT at 26 US hospitals. Most hospitals (25 of 26; 96.2%) had guidelines in place for the management of suspected HIT, with 7 (26.9%) having a “halt heparin, test, and await results” (ie, “test and wait”) policy. One-third of hospitals had a wait time for obtaining PF-4 antibodies of 3 days or more. Hospital guidelines for the management of HIT may actually discourage the use of optimal HIT management strategies.
Introduction
Heparins are used ubiquitously for the treatment and prevention of thromboses. 1 –3 It is estimated that 12 million people, or one-third of hospitalized patients in the United States, are exposed to heparins each year. 4,5 However, heparins have several limitations, including patient-to-patient variability in anticoagulation response, an inability to inhibit clot-bound thrombin, and risk of associated thrombocytopenia. 6 –9
Thrombocytopenia is common, occurring in up to 36% of hospitalized patients receiving heparin. 10 It is associated with poor short-term outcomes, including the risk of thrombosis and death. 3,10 Heparin-induced thrombocytopenia (HIT) is an antibody-mediated response to heparin. 11 Serologically confirmed HIT occurs in an estimated 1% to 5% of patients exposed to heparin (variations in frequency depend on patient population and type of heparin used) and may evolve into symptomatic thrombosis, which can lead to loss of extremities or death. 3,10 –12
Heparin-induced thrombocytopenia should be suspected when thrombocytopenia (platelet count of <150 000/mm3 or a fall from baseline of 30%-50%) occurs in patients within 5 to 14 days of heparin initiation or whenever thrombosis occurs in patients receiving heparin or low-molecular-weight heparin (LMWH) therapy. 11,13 Delayed-onset HIT, which begins several days following the discontinuation of heparin therapy, may also occur in approximately 3% to 5% of patients. 11,14,15
Despite its serious nature, thrombocytopenia may be overlooked in patients receiving heparin therapy and can result in an unacceptable risk of adverse outcomes. 10,16 –18 The Complications After Thrombocytopenia Caused by Heparin (CATCH) registry reported data from a prospective, observational study of 3536 patients in 48 US hospitals from March 2003 to May 2004. 10,16 Patients were grouped into 3 strata: the prolonged heparin stratum consisted of patients treated with heparin or LMWH for at least 96 hours; the cardiac care unit (CCU) stratum comprised patients in CCU who developed thrombocytopenia while on heparin; and the HIT serologic studies stratum consisted of all patients who had a HIT assay performed during the study period. 10 Of the 2420 patients 16 in the prolonged heparin stratum, 881 (36.4%) developed thrombocytopenia. 10 Only 89 (10.1%) of these patients were suspected of having HIT, with suspicion of HIT being documented a median of 1 day (range, 0-4) after thrombocytopenia developed. In 20.9% of these patients, a thromboembolic complication (TEC) had occurred prior to documented suspicion of HIT. More than half of the 881 patients with thrombocytopenia (56.0%) had a high probability of HIT, based on the 4T (Thrombocytopenia, Timing, Thrombosis, and Absence of OTher Features) scoring system. 10,19
In patients included in the CATCH registry, appropriate treatment was often delayed or not provided, even when HIT was suspected. For example, among thrombocytopenic patients in the prolonged heparin and CCU strata (n = 881 and n = 1090, respectively), those with a high probability and a documented suspicion of HIT had a median time to initiation of serologic testing of 3 days. 10 A hematology consult was obtained in about half of these patients (41.2% and 52.5% in the prolonged heparin and CCU strata, respectively). 10 Patients in the prolonged heparin stratum continued to receive heparin for a median of 70 hours after HIT was suspected; those in the CCU stratum continued to receive heparin for a median duration of 32 hours. 10 The 2012 American College of Chest Physicians Evidence-Based Clinical Practice Guidelines recommend the use of alternate nonheparin anticoagulants when HIT is strongly suspected or confirmed. 11 In the CATCH registry, a direct thrombin inhibitor (DTI) was initiated in only 19.1% of patients with suspected HIT in the prolonged heparin stratum and in 21.7% of those in the CCU stratum. Even among patients with the highest risk of HIT, DTIs were administered to only 29.4% and 35.6% of patients in the prolonged heparin and CCU strata, respectively. 10 Overall, about 23% of patients with thrombocytopenia in the prolonged heparin stratum and about 36% in the CCU stratum died or experienced a TEC during their hospital stay. 10
These data suggest that the incidence of thrombocytopenia during treatment with unfractionated heparin or LMWH is high, occurring in as many as 1 in 3 patients. 10 While current clinical guidelines recommend that clinicians halt heparin and begin alternate anticoagulation when HIT is suspected, the guidelines do not provide specific guidance on when alternative treatments should be initiated. 11 It appears that diagnosis and treatment gaps clearly exist in the setting of HIT. In addition to the recent findings from the CATCH registry, results from a review of thromboembolic episodes observed during prospective studies of HIT between 1980 and 2005 suggested that the prevalence of clinically confirmed thrombosis in patients with HIT who did not receive alternate anticoagulation was 30% to 50%, depending on patient characteristics. 5
Based on observations from the CATCH registry, it appears that clinicians may be reluctant to initiate alternate anticoagulant therapy in at-risk patients with suspected HIT. 10 Reasons for this apparent reluctance are unclear. To further our understanding of “real-world” management of HIT and the utilization of DTIs, we conducted a study to (1) ascertain how HIT is diagnosed and treated in HIT-aware hospitals and (2) gain an understanding of how DTIs are used, with the specific goal of identifying usage and restriction patterns in US hospitals.
Methodology
The registry assessed utilization of argatroban (GlaxoSmithKline; Research Triangle Park, North Carolina) as a surrogate for DTIs. Argatroban was chosen because it is the DTI with the broadest indication and is accepted as a standard of care in the setting of HIT. 11,20
The registry comprised 26 US hospitals within the Top 200 strata of argatroban usage in 2008, identified from a third-party data source (Wolters Kluwer; Bridgewater, New Jersey). Participants were asked to provide information regarding the hospital formulary status of DTIs, institutional guidelines or protocols for the management of patients with suspected HIT, heparin antibody monitoring/testing, and coagulation testing. Additionally, participants were asked to retrieve patient data files for 10 or more consecutive patients who were most recently treated with argatroban at the participating institution. All hospital and patient identifications were blinded for data analysis.
Results
Hospital Demographics
Participants comprised pharmacy directors or pharmacists from 26 US hospitals and data were provided for 400 patients. Hospital affiliation types were divided between academic hospitals (n = 8; 30.8%), community teaching hospitals (n = 13; 50.0%), and community nonteaching hospitals (n = 5; 19.2%). Larger hospitals (≥500 beds) accounted for 57.7% (n = 15) of the sample; hospitals with ≤499 beds accounted for 42.3% (n = 11). The formulary status of nonheparin anticoagulants at the participating hospitals is shown in Table 1.
Formulary Status of Nonheparin Anticoagulants at Participating Hospitals.a
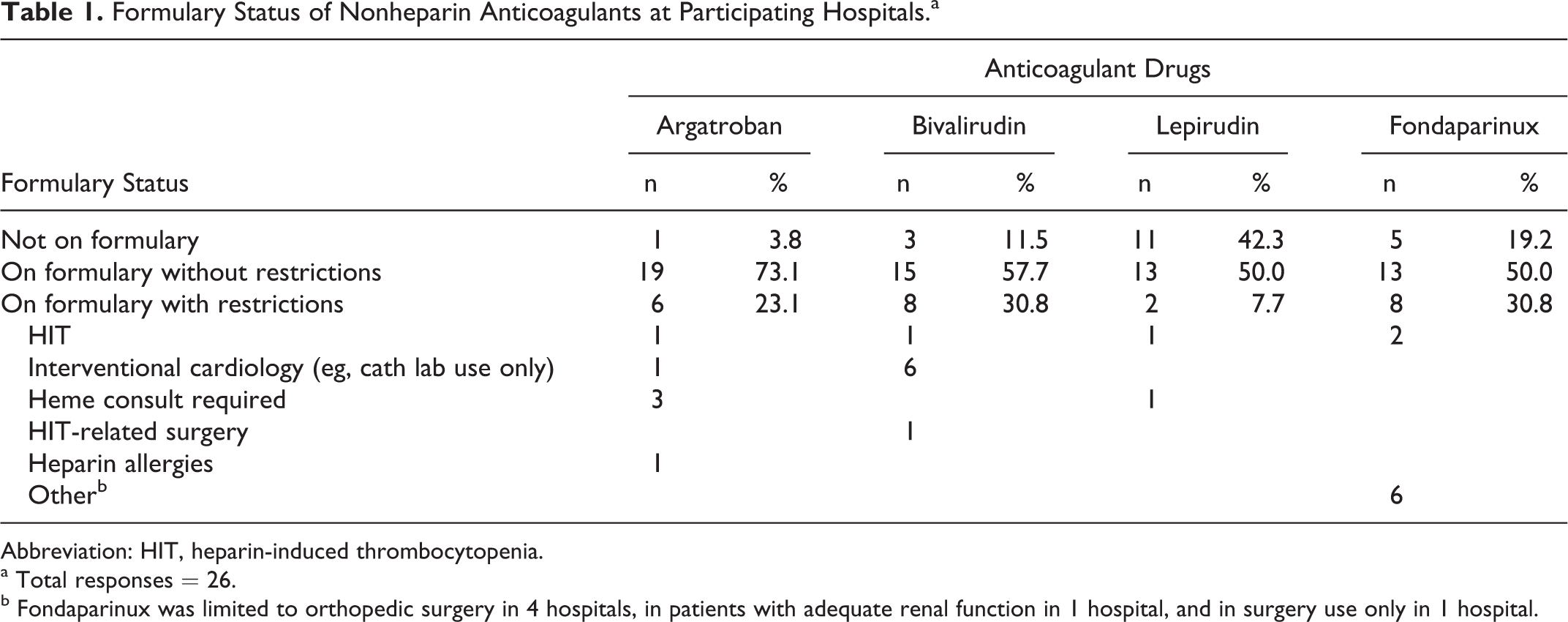
Abbreviation: HIT, heparin-induced thrombocytopenia.
a Total responses = 26.
b Fondaparinux was limited to orthopedic surgery in 4 hospitals, in patients with adequate renal function in 1 hospital, and in surgery use only in 1 hospital.
Hospital Guidelines
The majority of participating hospitals (25 of 26; 96.2%) had guidelines in place for the management of suspected HIT. Two-thirds of participating hospitals (17 of 26; 65.4%) had policies consistent with current national guidelines recommending halting heparin, testing for heparin antibodies, and initiating therapy prior to antibody test results (“test and treat” policy). However, 7 sites (26.9%) had guidelines which recommended halting heparin, testing for heparin antibodies, and awaiting antibody test results (test and wait policy); and 1 hospital (3.8%) had guidelines in place recommending halting heparin, testing for heparin antibodies, and initiating therapy based on the probability of HIT (using the 4T score). 19 Smaller hospitals (fewer than 500 beds) were more likely to have guidelines in place recommending to begin treatment while awaiting test results (eg, test and treat policy). Figure 1 summarizes these results by hospital size. Nonacademic hospitals were more likely to have a test and wait policy.
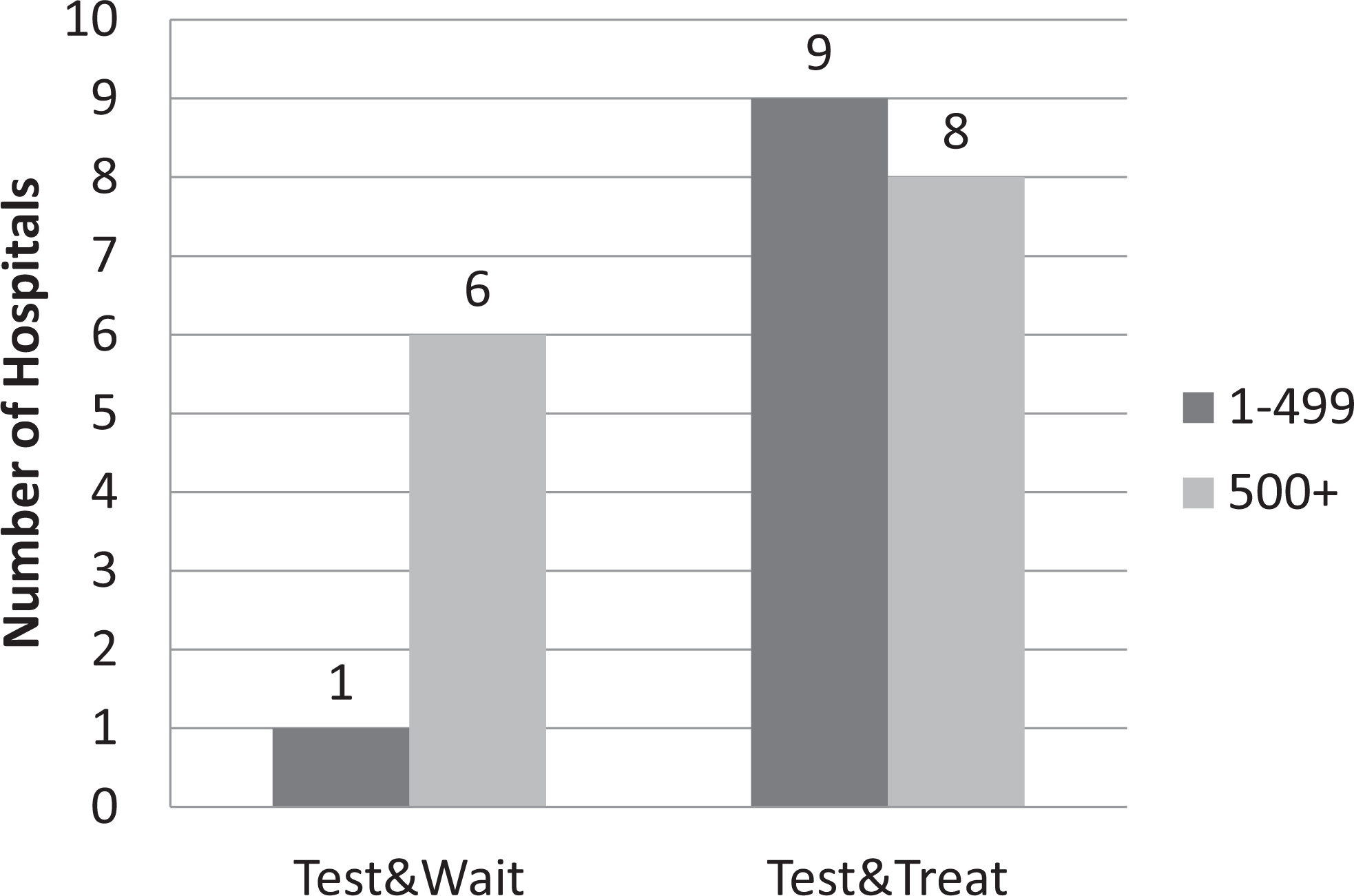
Smaller hospitals more likely to begin an alternative anticoagulant while awaiting test results.
Laboratory Testing
The most commonly used serologic test for HIT was the heparin–platelet factor-4 (PF-4) enzyme-linked immunoabsorption assay (ELISA). Survey responders reported the average hospital turnaround times for antibody testing. The average amount of time required to get test results varied from hospital to hospital. Of the 26 participating hospitals, 8 (30.8%) typically required less than 1 day to get antibody test results, while 10 (38.5%) required 1 to 2 days, and 8 (30.8%) required at least 3 days. In one-third (5 of 15) of the largest hospitals (≥500 beds), the typical wait for antibody test results was 3 or more days. Seven hospitals had a test and wait policy recommending halting heparin and waiting for the return of antibody test results prior to initiating alternate therapy; the typical wait time for test results was less than 1 day in 4 (57.1%) of 7, 1 to 2 days in 2 (28.6%) of 7, and 3 or more days in 1 (14.3%) of 7 hospitals. Only 1 hospital’s guidelines specifically recommended confirming ELISA results with functional testing (eg, serotonin release assay [SRA]).
Laboratory Testing Performed
Of the 400 patient cases reviewed, full information was available for 391. Of these, 280 (71.6%) patients had a heparin antibody test performed during the reported hospital stay and 111 (28.4%) did not. Of the 280 patients tested, 65 (23.2%) and 26 (23.4%) of the 111 who were not tested were in hospitals with a test and wait policy. One hundred ninety-six (70.0%) patients given an antibody test and 82 (73.9%) of those not given an antibody test were in hospitals with a test and treat policy. Among the 280 patients administered an antibody test, positive antibody test results were reported in 172 (61.4%) patients and negative results were reported in 108 (38.6%) patients. Participants’ responses regarding antibody test results, hospital length of stay, and details on argatroban use are summarized in Table 2.
Reason for Argatroban Use, Argatroban Use Levels, and Length of Hospital Stay, by Antibody Test Status.
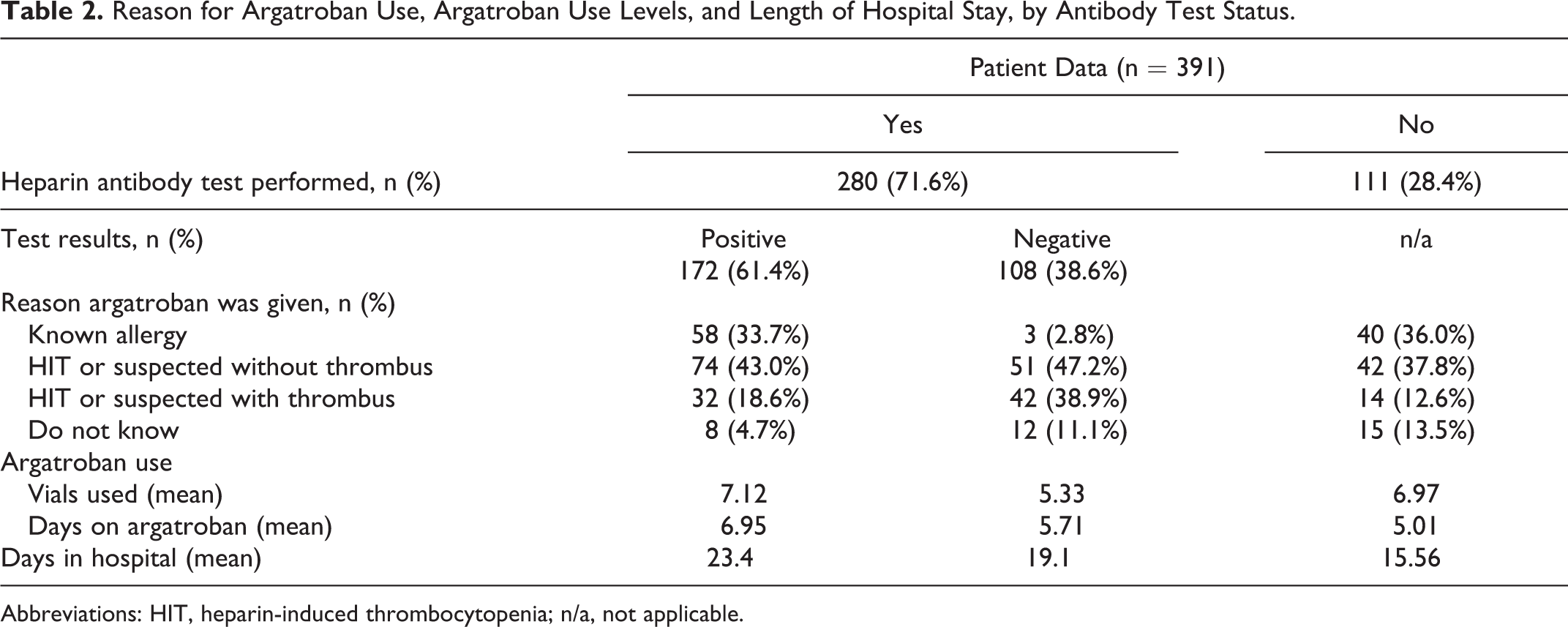
Abbreviations: HIT, heparin-induced thrombocytopenia; n/a, not applicable.
Hospital Guidelines for Test-Positive Patients
Most participating hospitals (24 of 26, 92.3%) had guidelines in place for the management of patients with positive heparin antibody test results; 2 hospitals (7.7%) had no guidelines in place. Regarding the management of patients with positive heparin antibody tests, 8 (30.8%) hospitals had guidelines recommending the initiation of alternate treatment (ie, a “treat” policy); 10 (38.5%) had guidelines recommending the initiation of alternate treatment while waiting for confirmation of antibody test results (ie, a “treat and confirm” policy). Additionally, 5 (19.2%) hospitals had guidelines recommending halting anticoagulant treatment until the diagnosis was confirmed by hematology consult (ie, a “confirm then treat” policy), and 1 (3.8%) had guidelines recommending the initiation of alternate treatment while waiting for heparin antibody test to be confirmed via platelet SRA test.
Recommended Anticoagulants
Argatroban and lepirudin were most frequently listed as recommended anticoagulants in hospital guidelines for the management of patients with HIT. Some of the participating hospitals had more than 1 recommended anticoagulant listed in their guidelines; thus, 36 responses were obtained from the 26 participants. Argatroban was listed as a recommended treatment in 22 (84.6%) hospital guidelines, 6 hospitals (23.1%) listed lepirudin as a recommended treatment, and 5 hospitals (19.2%) listed fondaparinux as a recommended treatment.
Discussion
This study, which collected data from 400 patients in 26 US hospitals, was undertaken to gain useful information on identification and treatment patterns of HIT. Previously, data compiled from more than 2400 patients in the CATCH registry showed that suspicion of and evaluation for HIT were infrequent and delayed, even among high-risk patients. In addition, suspicion of HIT often did not arise until after a TEC had occurred. 10 In general, the results of this study are consistent with the apparent treatment inertia observed in CATCH. Data from this study suggest that apparent treatment gaps may exist in a large proportion of the participating HIT-aware hospitals, even when a suspicion of HIT is documented. Furthermore, these gaps appear to be associated with institutionally defined clinical guidelines for the management of patients with or at risk for HIT. Based on these results, the HIT management guidelines established in 7 of the 26 participating hospitals were not consistent with current, evidence-based clinical practice guidelines. 11 That is, in 27% of the participating hospitals, institutional policies dictated that when HIT was suspected, alternate anticoagulant treatment was to be withheld until the results of antibody testing were available. Interestingly, this test and wait policy was not isolated to smaller or nonteaching hospitals. In fact, this policy was held by 2 (25%) of 8 academic, 2 (15%) of 13 community teaching, and 3 (60%) of 5 community nonteaching hospitals. Overall, 6 of these 7 institutions were large, 500+ bed hospitals. Antibody test results were delayed by at least 1 day in 3 (43%) of the 7 hospitals with a wait-and-see policy, and, in 1 of the 7, antibody test results were typically delayed at least 3 days. Thus, it appears that 3 (43%) of the 7 hospitals have institutionally defined policies in place that leave potentially at-risk patients without anticoagulant treatment for 1 day or more, and for as long as 3 days depending on delays in obtaining antibody test results.
We did not determine whether the individual hospital guidelines were followed with respect to argatroban administration. Notably, in 111 (28.4%) argatroban-treated patients no laboratory testing was performed despite the fact that nearly all local guidelines recommend testing. Furthermore, in 108 patients (27.6%) in whom antibody testing was negative, argatroban appeared to be continued despite these negative results.
Specific reasons for the apparent shortfall in HIT management practices observed in this study are unclear. Potentially, a lack of awareness of serious risks and consequences associated with thrombocytopenia, safety and/or efficacy concerns about currently available medications, and perceived high treatment costs among clinicians may be contributing factors.
It is possible that restrictions on drug use and HIT management protocols in participating hospitals were put in place to help manage treatment costs. The data compiled in this study suggest that these policies may not result in reduced costs. For example, among the 111 patients who did not have a heparin antibody test performed, the majority (74%) were in hospitals with a “test and treat” policy. Similarly, among the 280 patients who did receive antibody testing, 70% were in hospitals with a “test and treat” policy. Notably, on average, patients who did not receive antibody testing had shorter hospital stays and received argatroban for a shorter length of time compared with patients who had heparin antibody testing performed. Thus it appears that moving quickly to address HIT or suspected HIT (via a “halt heparin, treat with alternate, then confirm” policy) may have positive implications for lowering drug usage/costs and hospital lengths of stay, even when serologic confirmation of HIT is not obtained.
Several limitations of this study should be noted. The results described are purely observational in nature; no statistical analysis was performed, and no control or reference groups were included. We did not determine adherence to local guidelines for the use of ELISA testing in patients receiving argatroban. In addition, only HIT-aware hospitals with high rates of DTI (argatroban) use participated. Given this limitation, it could be argued that greater treatment gaps might be observed if the study had included hospitals with lower rates of DTI utilization. Finally, the number of participants included in this study (26 hospitals) is relatively small. Nevertheless, results appear to support learnings gathered from a patient population of more than 2400 in the CATCH registry.
Conclusions
In patients with HIT or suspected HIT, treatment with alternate anticoagulant therapies may be delayed by 1 day or more in a large proportion of the participating DTI-aware hospitals. At some institutions, directives included in hospital guidelines for the management of HIT may actually discourage health care providers from following optimal HIT management strategies. Institutions should review current HIT management policies to determine whether these policies improve the institution’s performance in managing patients with suspected HIT.
Footnotes
Acknowledgment
The author thanks Michael Morren, RPh, of Peloton Advantage, LLC, for writing and editorial support. The author gratefully acknowledges the participating hospitals and thanks HRA Research (Parsippany, New Jersey) for compiling and analyzing the data provided by registry participants.
Authors’ Note
The opinions expressed in the current article are those of the author. The author received no fee for involvement in the design or execution of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Canyon Pharmaceuticals, Inc sponsored the survey. The author received no honorarium or other form of financial support related to the development of this manuscript.
