Abstract
Background:
The patent protection of low-molecular-weight heparins (LMWHs) expired, so the definition of criteria for the biological similarity between LMWH copies and the original product is a real need.
Aim:
The present in vitro study compared copies and branded enoxaparin using the specific anti-Xa activity and the calibrated automated thrombogram assay.
Methods:
Samples of platelet-poor plasma (PPP) and platelet-rich plasma (PRP) from 15 healthy volunteers were spiked with branded enoxaparin (Lovenox) or its copies (Cutenox, Dilutol, Enoxa, Fibrinox, Loparin, Lupenox, Novex, Noxprin, and Versa). The specific anti-Xa activity was measured in PPP, and thrombin generation was assessed in PPP and PRP in the presence of tissue factor or pancreatic cancer cells BXPC3.
Results:
The anti-Xa activity of enoxaparin copies ranged from 0.072 to 0.088 IU/μg, being lower as compared to the branded enoxaparin (0.095 anti-Xa IU/μg). The potency of each copy to inhibit thrombin generation varied in the 3 experimental systems. The presence of platelets or pancreatic cancer cells BXPC3 in human plasma induced significant modifications in the inhibitory efficiency of enoxaparin copies on thrombin generation, which distinguished them from the branded product.
Conclusion:
Enoxaparin copies showed significant variability regarding their inhibitory potency on thrombin generation. Platelets and cancer cells significantly increased the variability of the antithrombotic efficiency of the copies as compared to the branded enoxaparin. The present study underlines the need for the elaboration of additional functional criteria to evaluate the global antithrombotic capacity of enoxaparin copies in order to evaluate their potential sameness with the branded drug.
Background
Low-molecular-weight heparins (LMWHs) are cornerstone drugs in the primary prevention and treatment of venous thromboembolism and are widely used in patients with cancer. 1 –3 The smaller molecular size of polysaccharide chains of LMWHs, compared with unfractionated heparin (UFH), involves a more predictable pharmacological action, better subcutaneous bioavailability, and sustained antithrombotic activity. 4 Low-molecular-weight heparins are multitargeted anticoagulant drugs exerting their antithrombotic effect primarily via the binding of the pentasaccharide domain on antithrombin (AT). Due to the presence of the pentasaccharide domain in about 30% of the polysaccharide chains, LMWHs downregulate thrombin generation by accelerating the AT-dependent inhibition of factor Xa (FXa) and to a lesser extent by the inhibition of thrombin. 5
The LMWHs available in the market vary in their physicochemical properties, the anti-Xa/anti-IIa ratio, and their inhibitory effect on thrombin generation.
5
–7
The pharmacokinetic/pharmacodynamic properties of LMWHs are also different.
8
Like UFH, they are comprised of repeating disaccharide units of uronic acid (β-
The patent protection of the LMWH enoxaparin expired, so the extent and nature of the studies required to obtain a market authorization for LMWH copies is an issue of debate. In North America, the food and drug administration (FDA) classified LMWHs as semisynthetic drugs and their copies as generics, whereas in Europe, the European Medicines Agency (EMA) views them as biological medicines and consequently their copies as biosimilars. 12 Beyond the pharmacological definitions, the complex structure and activity of LMWHs make the production of a copy LMWH more challenging than that of a generic homogenous small molecule drug.
Several copies of enoxaparin are currently available worldwide. These agents may considerably differ from the branded product as well as from each other. 13 –16 Some copies of enoxaparin have similar pharmacokinetic properties, in terms of anti-Xa activity as compared to the original one. 17 –19 To the best of our knowledge, the comparisons of enoxaparin copies with the branded product have been done with assays either sensitive for the measurement of the specific anti-Xa activity of LMWHs or specific for the structural characterization of the oligosaccharide components. There are no available data—at least in the published literature—comparing branded and copies of LMWHs on the basis of their global effect on blood coagulation process. As a consequence, the studies published to date are inconclusive regarding the global functional antithrombotic similarity and sameness of enoxaparin copies compared with the branded product. The actual situation, which is characterized by the increasing number of enoxaparin copies available worldwide, makes the definition of criteria for the biological similarity between LMWH copies and the original product a real need.
Aiming to provide experimental evidence in order to respond to this need, the present study, conducted in vitro, develops an analytical approach for the comparison of branded enoxaparin and its copies by combining a standard pharmacological assay (the measurement of the anti-Xa specific activity) and a global coagulation assay, such as the calibrated automated thrombogram performed in human platelet-rich plasma (PRP) or platelet-poor plasma (PPP) as well as in an experimental model of cancer-induced hypercoagulability. The study aims to develop a rationale for the determination of the criteria for the similarity and sameness of branded and copies of LMWHs.
Materials and Methods
Enoxaparin Preparations
The reference branded enoxaparin sodium was Lovenox (Sanofi, France). The studied copies of enoxaparin were Cutenox (Gland Chemical/BioChimico, Brazil), Dilutol (Lazar laboratories, Argentina), Enoxa (Medis Laboratory, Tunisia), Fibrinox (Sandoz laboratories, Switzerland), Loparin (GlandPharma Limited, India), Lupenox (Lupin Ltd., India), Novex (Sothema, Morocco), Noxprin (Ajanta Ph, India), and Versa (Eurofarma, Brazil). All copies of enoxaparin were commercially available and were purchased from the market. The initial concentrations of branded enoxaparin and its copies are expressed as equivalent gravimetric units (μg/mL).
Cancer Cell Cultures
Adhesive pancreatic cancer cell lines BXPC3 from human pancreatic cancer and normal human plasma were used for thrombin generation experiments. The human pancreatic primary adenocarcinoma cell line BXPC3 was from American Type Culture Collection (Rockville, Maryland). Cells were expanded in Roswell Park Memorial Institute medium (RPMI)-1640 medium (Invitrogen, Cergy Pontoise, France) at 37°C in 100% humidified atmosphere with 5% CO2. Culture medium was supplemented with 10% (v/v) of inactivated fetal calf serum (Invitrogen), 5 mmol/L glutamine, and 50 U/mL penicillin–streptomycin (Sigma-Aldrich, Saint Quentin Fallavier, France). Adhesive cultures were developed on 75 cm2 culture flasks. At 50% confluence, the cells were incubated with trypsin for 5 minutes at 37°C, suspended in RPMI, thereafter washed twice in phosphate-buffered saline (PBS) and once in isotonic saline (NaCl) solution, subsequently, they were suspended in saline. Cells were used in the experiments only if the apoptotic cell number was lower than 2% of the whole cell count in the working suspension.
Human Plasma
Blood samples were obtained from 15 healthy volunteers, who were members of the laboratory staff and who had not taken any medication during the last 10 days prior to inclusion in the study. Samples were obtained by atraumatic antecubital venipuncture with the Vacutainer system. Blood was drawn in siliconized tubes (Becton Dickinson, Meylan, France) containing buffered 0.13 mol/L trisodium citrate (9 parts blood to 1 part citrate solution; 3.8% v/v). Citrated whole blood was centrifuged for 10 minutes at 150g at room temperature and the supernatant PRP was removed. The platelet count in PRP was adjusted to 1.5·105 L−1 by dilution with autologous PPP, which was obtained after further centrifugation of the remaining blood for 15 minutes at 2000g. Previous studies from our group showed that at this platelet count, the platelet-dependent enhancement of thrombin generation is in a plateau. 20 The PRP from 15 donors was spiked with increasing concentrations of branded enoxaparin or the studied copies. The PPP was prepared after double centrifugation of citrated whole blood for 20 minutes at 2000g at room temperature. A pooled PPP was prepared by mixing PPP from the healthy donors. The same pooled PPP was used in all experiments in order to avoid interindividual variability of thrombin generation assay. Pooled PPP was frozen at −80°C until assessment.
The concentration of the anti-Xa activity in plasma spiked with the enoxaparin was measured with the chromogenic assay Rotachrome Heparin from Diagnostica Stago (Asniéres, France). The concentrations of the studied compounds in plasma are expressed on a gravimetric basis or as anti-Xa IU/mL as measured in PPP.
Thrombin Generation Assay
Thrombin generation was studied in vitro using the Calibrated Automated Thrombogram and the respective software (Thrombinoscope b.v., Maastricht, the Netherlands). 21 Thrombin generation was studied in PPP and PRP in the absence or presence of pancreatic cancer cells BXPC3. The PRP experiments were performed with fresh material, whereas the PPP experiments were performed with frozen/thawed plasma.
Thrombin generation in PRP
In each well of a microplate, 80 μL of PRP supplemented with saline (control) or with the indicated concentrations of the studied compounds was mixed with 20 μL of diluted (1:1000 final dilution) recombinant human thromboplastin Innovin (Siemens, Paris, France), yielding 5 pmol/L TF in plasma as described elsewhere. 20
Thrombin generation in PPP
In separate experiments, 80 μL of PPP supplemented with saline (control) or with the indicated concentrations of the studied compounds was mixed with 20 μL of PPP-reagent 5 pmol/L Tissue factor (TF) (Thrombinoscope b.v., Diagnostica Stago).
Thrombin generation induced by cancer cells
To study the effect of the branded and copies of enoxaparin on thrombin generation triggered in the presence of the pancreatic cancer cells BXPC3, we used a validated experimental system described elsewhere. 22 Briefly, BXPC3 cells were expanded in the wells of a microtiter plate suitable for thrombin generation assessment as follows: BXPC3 cells which were expended in cell culture flasks were removed and suspended in a culture medium to obtain a concentration of 50 cells/µL. Then, 100 µL of this suspension was placed in each well of the microtiter plate (5000 cells/well) and incubated for 48 hours at 37°C in 100% humidified atmosphere with 5% CO2. During this period, cells adhered at the wells’ bottom. At 50% confluence, the plates were washed 3 times with PBS and once with saline. Then, 80 µL of normal PPP, spiked with the studied compounds or with saline (control), were added to each well. Thrombin generation was assessed as described earlier. In control experiments, the wells were filled with culture medium without cells. Each experiment was repeated 5 times. Saline (20 μL) was used in the control experiments. In preliminary experiments, we verified that the culture medium (solution of RPMI, glutamine, penicillin–streptomycin, and fetal calf serum) did not influence the thrombin generation process of normal PPP.
Thrombin generation was initiated by adding 20 µL of triggering solution containing CaCl2 (16.7 mmol/L final concentration) and fluorogenic substrate (Z-Gly-Gly-Arg-AMC, 417 µmol/L final concentration). Thrombin generation curves were corrected for substrate consumption and inner filter fluorescence effects using a Thrombin Calibrator (Thrombinoscope b.v.). At each cycle of the test, a plasma sample (80 μL) spiked with thrombin calibrator (20 μL) was run in parallel with each plasma sample spiked with the studied compound. Thrombin generation was assessed with the calibrated automated thrombogram assay (Thrombinoscope b.v). A plate reader fluorometer (Fluoroskan Ascent, ThermoLabsystems, Helsinki, Finland) and the appropriate software (Thrombinoscope b.v) were used. Among thrombogram parameters, we analyzed the mean rate index (MRI) of the propagation phase of thrombin generation (calculated by the formula: peak/(ttPeak − lagtime) and expressed in nmol/L/min). Interindividual and intra-assay variability of thrombin generation assay were assessed in healthy individuals and has been published elsewhere. 20 For practical reasons, the concentration–response curves from the experiments done in PPP, PRP, and PPP + BXPC3 cells in the presence of the studied agents and BXPC3 cells are not presented in the figures.
Statistical Analysis
The antithrombotic efficiency of the studied compounds was compared on the basis of the Inhibitory Concentration 50% (IC50) on the MRI. The IC50 was calculated by extrapolation from the concentration–response curve constructed for each studied compound in each experimental system. The nonparametric paired Mann-Whitney U test was applied to compare the inhibition of thrombin generation induced by each of the enoxaparin copies with that induced by the branded enoxaparin. To compare the studied copies versus the branded enoxaparin, we calculated the ratio (%) of the IC50 for the MRI for each agent according to the formula (1 − IC50branded/IC50copy)%. Results are shown as mean ± standard deviation (SD). The level of significance was set at P <.05. Statistical analysis was performed with SPSS software for Windows (release 10.0.1; SPSS Inc., Chicago, Illinois).
Results
Specific Anti-Xa Activity of Branded and Copies of Enoxaparin
The copies of enoxaparin, similarly to the branded product, when added at increasing gravimetrically equivalent concentrations in normal PPP induced a concentration-dependent increase of the anti-Xa activity in plasma (Figure 1). The specific anti-Xa activity per gravimetric unit of the copies and the branded enoxaparin calculated by this series of experiments is shown in Table 1. All copies of enoxaparin except Versa and Novex showed higher variability in their anti-Xa activity per gravimetric unit as compared to the branded enoxaparin.
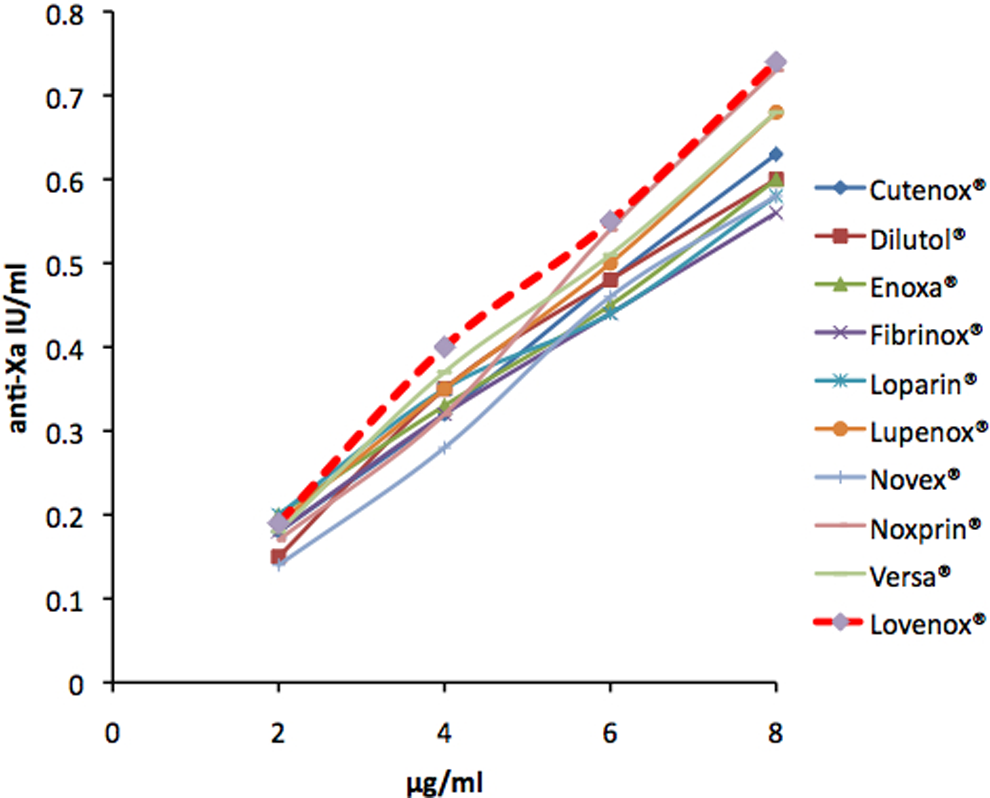
The concentration of the specific anti-Xa activity measured in normal platelet-poor plasma (PPP) spiked with increasing concentrations of copies and branded enoxaparin. Values are mean ± SD of 5 experiments.
The Specific Anti-Xa Activity per Microgram of Branded and Copies of Enoxaparin.a
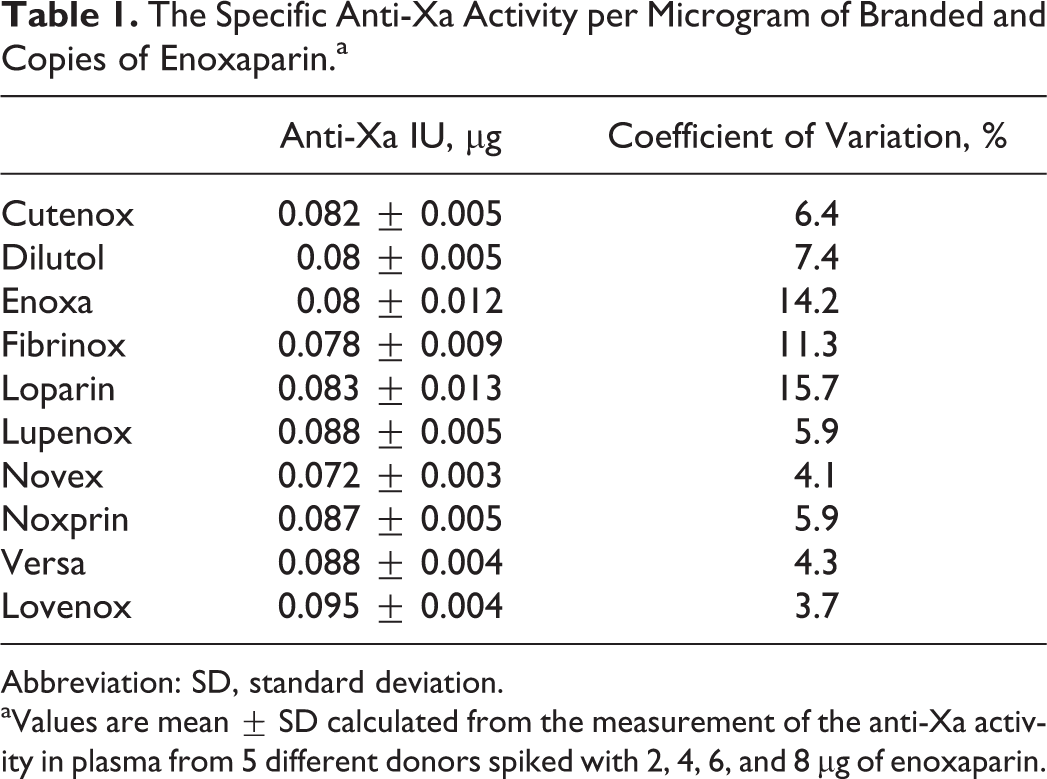
Abbreviation: SD, standard deviation.
aValues are mean ± SD calculated from the measurement of the anti-Xa activity in plasma from 5 different donors spiked with 2, 4, 6, and 8 μg of enoxaparin.
The variability of the anti-Xa activity in normal plasma spiked with increasing concentrations of the copies of enoxaparin was about 10% and was significantly higher than the interassay variability with branded enoxaparin with a coefficient of variation of about 3%.
Inhibitory Potency of Branded and Copies of Enoxaparin on Thrombin Generation
Copies and branded enoxaparin inhibited thrombin generation in a concentration-dependent manner in normal PPP, PRP, and PPP + BXPC3 cells. The comparison of the IC50 for the MRI for each one of the studied compounds in the different experimental systems revealed a significant variability of their antithrombotic potency (Tables 2 and 3).
Comparison on a Gravimetric Basis of the Inhibitory Potential of Branded and Copies of Enoxaparin on Thrombin Generation Triggered in PPP, PRP, and PPP in the Presence of BXPC3 Cells (PPP + BXPC3).a
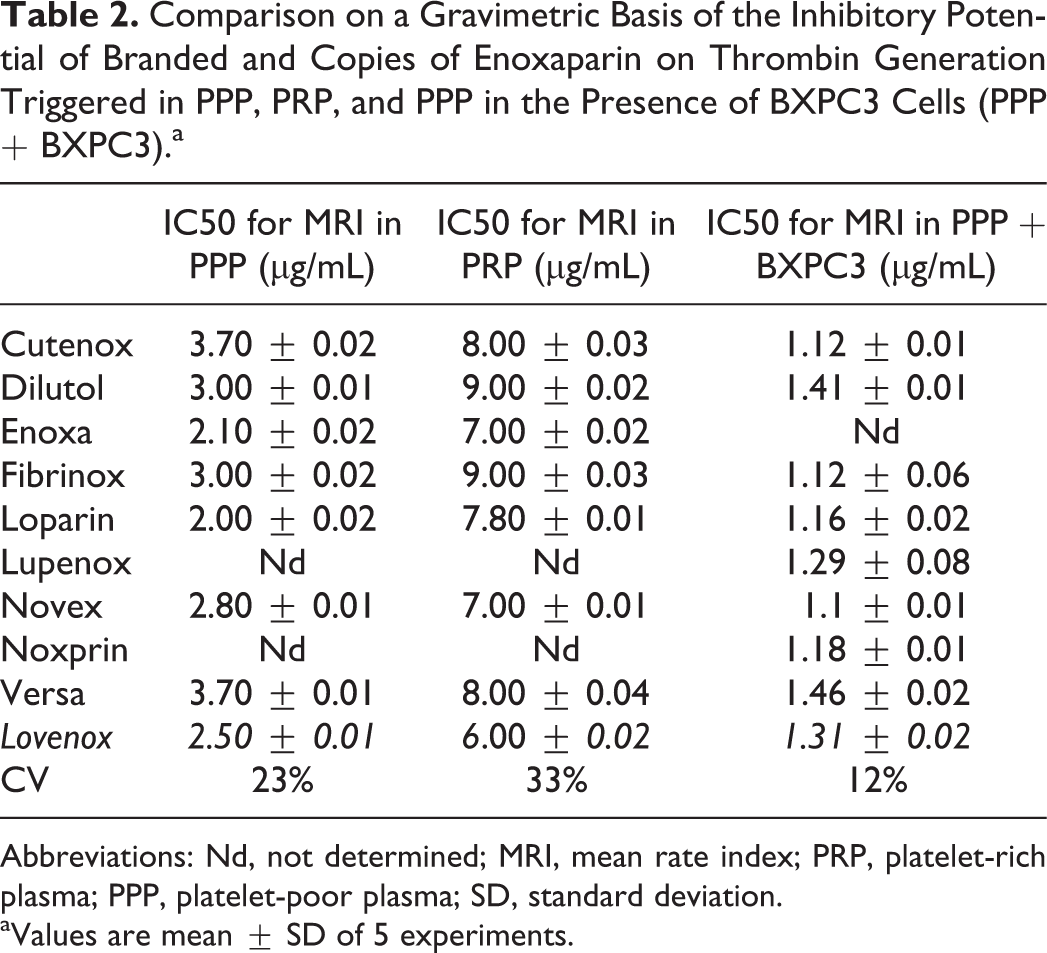
Abbreviations: Nd, not determined; MRI, mean rate index; PRP, platelet-rich plasma; PPP, platelet-poor plasma; SD, standard deviation.
aValues are mean ± SD of 5 experiments.
Comparison on the Equivalence of the Anti-Xa Activity of the Inhibitory Potential of Branded and Copies of Enoxaparin on Thrombin Generation Triggered in PPP, PRP, and PPP in the Presence of BXPC3 Cells.a
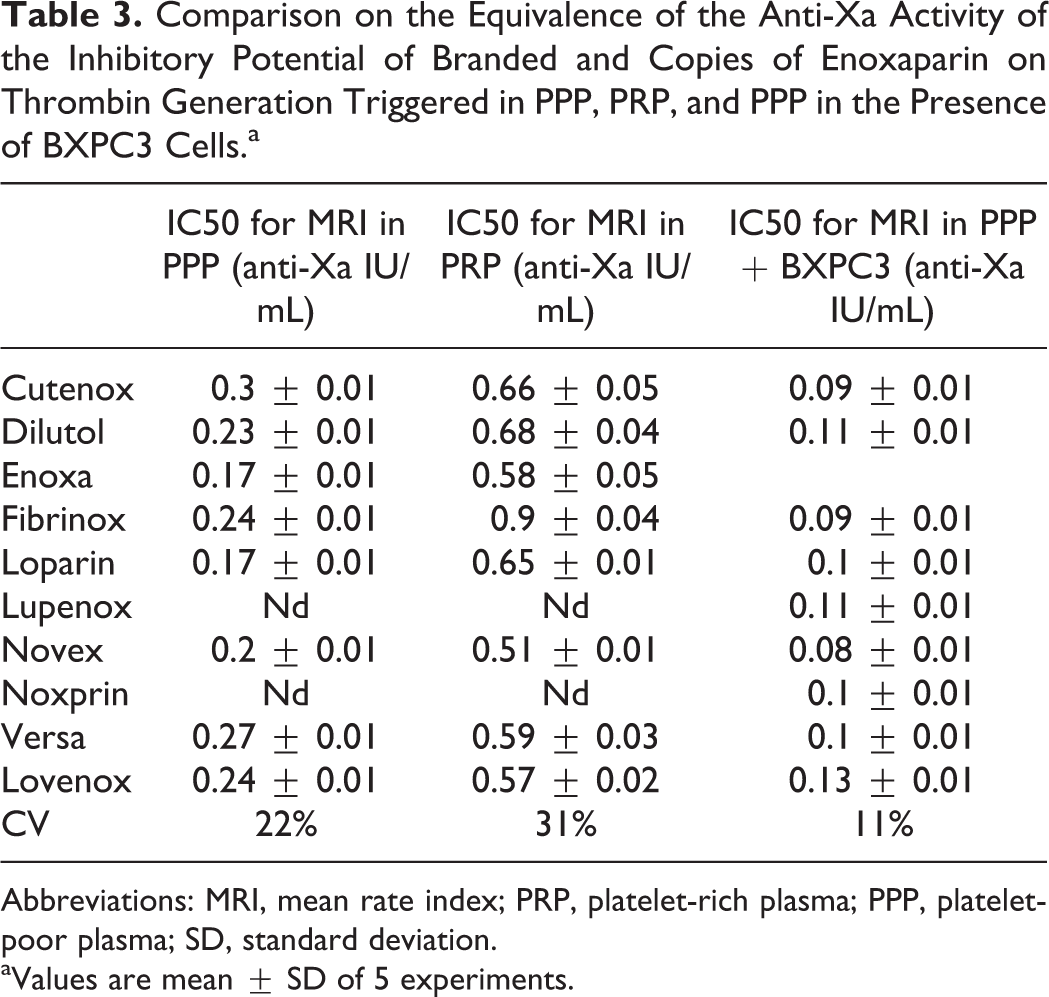
Abbreviations: MRI, mean rate index; PRP, platelet-rich plasma; PPP, platelet-poor plasma; SD, standard deviation.
aValues are mean ± SD of 5 experiments.
Comparison on Gravimetric Basis
When copies of enoxaparin were evaluated for sameness on a gravimetric basis, the coefficient of variation of the IC50 for the MRI was 23% in PPP, 33% in PRP, and 12% in PPP + BXPC3 cells. In each of the 3 experimental systems, the copies of enoxaparin showed significantly higher coefficient of variation of the IC50 for the MRI as compared to that observed for the branded enoxaparin when it was assessed in 10 different experimental cycles (2%).
Classification of enoxaparin copies according to the IC50 for the MRI in PPP showed that Loparin and Enoxa were more potent than Lovenox (20% and 16%, respectively). Novex was 12% less potent than Lovenox. Fibrinox and Dilutol were 20% less potent than Lovenox. Cutenox and Versa were 48% less potent than Lovenox.
The comparison of the IC50 for the MRI in PRP showed that all copies of enoxaparin were less potent than Lovenox. According to their potency, the copies of enoxaparin were classified as follows: Enoxa and Novex (−17%), Loparin (−30%), Cutenox and Versa (−33%), and Dilutol and Fibrinox (−50%).
The comparison of the IC50 for the MRI in PPP + BXPC3 cells showed a higher potency than Lovenox for Novex (16%), Fibrinox (15%), Cutenox (15%), Loparin (11%), and Noxprin (10%). Lupenox had similar potency as Lovenox. Dilutol and Versa were less potent than Lovenox (8% and 11%, respectively). These data are summarized in Figure 2.
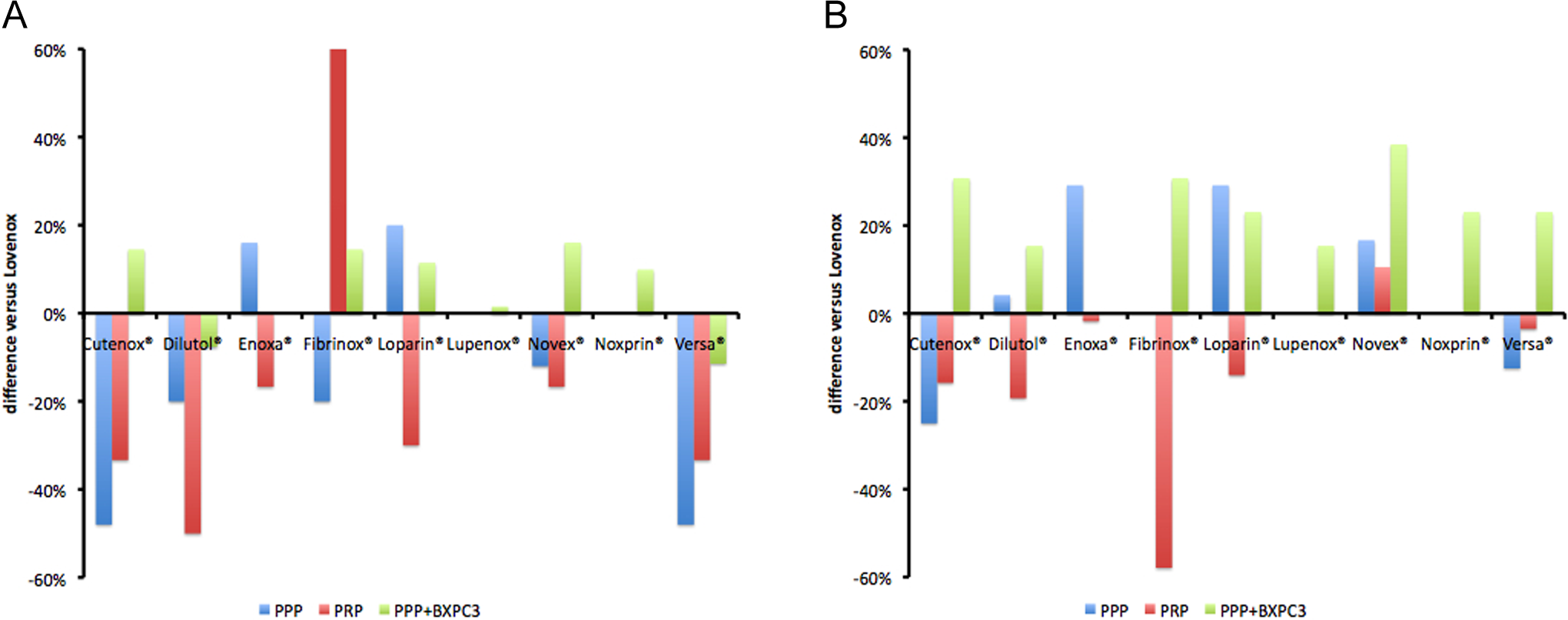
Relative potency of enoxaparin copies to inhibit thrombin generation in 3 different experimental systems. The inhibitory potency of each one of the enoxaparin copies was compared to that of branded enoxaparin. The comparison was done on a gravimetric basis (Frame A) and on the basis of equivalent anti-Xa activity (Frame B).
Comparison on the Basis of Equivalent Anti-Xa Activity
When copies of enoxaparin were assessed for sameness to branded enoxaparin on the basis of equivalent anti-Xa activity, the coefficient of variation of the IC50 for the MRI was 22% in PPP, 31% in PRP, and 11% in PPP + BXPC3 cells (Table 3). The coefficient of variation of the IC50 for the MRI in the presence of enoxaparin copies in each of the 3 experimental systems was significantly higher as compared to the intra-assay variability for each studied concentration of each preparation of enoxaparin which was 2%.
The comparison of the IC50 for the MRI in PPP showed that Enoxa, Loparin, and Novex were significantly more potent than Lovenox (29%, 29%, and 17%, respectively). Dilutol and Fibrinox showed a similar inhibitory potency as Lovenox. Versa and Cutenox were less potent as compared to Lovenox (−13% and −25%, respectively).
The comparison of the IC50 for the MRI in PRP showed that Novex was 11% more potent than Lovenox. Enoxa and Versa showed similar inhibitory potency as compared to Lovenox. Fibrinox, Dilutol, Cutenox, and Loparin were less potent as compared to Lovenox (−58%, −19%, −16%, and −14%, respectively).
The comparison of the IC50 for the MRI in PPP + BXPC3 cells showed that all copies of enoxaparin were more potent than Lovenox. These data are depicted in Figure 2.
Discussion
A critical question for the evaluation of the bioequivalence among copies and branded enoxaparin is what scientific data are needed to allow a conclusion of active ingredient sameness. The FDA developed a scientific approach to determining active ingredient sameness of the enoxaparin copies to the branded one. 23 The length of heparin chains with high affinity for AT determines their capacity to inhibit the other serine proteases than FXa, predominantly thrombin. 24 The degree of sulfation of heparin chains is an additional structural determinant for nonspecific binding with plasma proteins derived from platelets or other cells (ie, platelet factor 4 (PF4), fibronectin, etc). 25 The depolymerization process also determines the structural features of LMWHs. The ratio of the anti-Xa/anti-IIa activity and the anti-Xa activity per gravimetric unit of a LMWH preparation are considered as major determinants for their pharmacological characterization and have been retained as the most appropriate criteria for the characterization of the biochemical properties of enoxaparin. 26 These criteria have also been used by health authorities for the evaluation of the pharmacological potency of the enoxaparin copies and the comparison to the branded one when the sameness of these products was examined.
Theoretically, enoxaparin copies stemming from the same source material and produced by the same procedure of depolymerization as the branded one should manifest the same inhibitory potency on thrombin generation process. The studied preparations of enoxaparin copies are available in the market as being the same to the branded one according the actual criteria endorsed by the health authorities in the country in which the drug is approved. Based on this assumption, the present study evaluated the potency of the branded enoxaparin and some of the copies available in the world market to inhibit thrombin generation in normal human plasma triggered after TF pathway activation. Recent data published from our group showed that platelets or cancer cells modify the potency of the various branded LMWHs to inhibit thrombin generation in a variable and unpredictable manner. 27 If the branded and copies of enoxaparin are same, their exposition to platelets and cancer cells should induce the same magnitude of modifications on their inhibitory potency on thrombin generation. To control this hypothesis, we examined the effectiveness of copies and branded enoxaparin to inhibit thrombin generation in 3 different experimental systems. Thrombin generation was triggered in PPP by a reagent containing a physiologically relevant TF concentration (5 pmol/L) and an optimum concentration of procoagulant phospholipids (4 μmol/L). Thrombin generation in PRP was triggered by a physiologically relevant concentration of TF (5 nmol/L) in the presence of platelets. Platelets, which are activated by thrombin while it is generated, release proteins such as PF4 that interact with heparin chains and partially neutralize their antithrombotic potency. Finally, in the third experimental system, thrombin generation in PPP was triggered by the pancreatic cancer cells BXPC3, which express abundant amounts of TF. The BXPC3 cells do not express any detectable procoagulant phospholipids and consequently, the formation of prothrombinase and intrinsic tenase is suboptimal. 28
The specific anti-Xa activity per gravimetric unit is one of the principal criteria for the evaluation of the sameness between the copies and the branded LMWHs. The data presented herein demonstrate that the specific inhibitory strength of enoxaparin copies against FXa ranged from 0.072 to 0.088 anti-Xa IU/μg and it was lower as compared to that of the branded enoxaparin (0.095 anti-Xa IU/μg). Interestingly, for some of the enoxaparin copies the specific anti-Xa activity is not the same as that of the branded product. The influence of the donor variability on the specific antithrombotic activity measured in plasma in which branded LMWHs were added in vitro has been previously described. 29 Consequently, our data suggest that subcutaneous injection to patients of some copies of enoxaparin may not produce the same anti-Xa activity in plasma as that which would be produced after the injection of the same dose of the branded enoxaparin. Having the opportunity to examine 9 different copies of enoxaparin, our study clearly demonstrates that this variability concerns some but not all the copies.
The copies as well as the branded enoxaparin induced a concentration-dependent inhibition of thrombin generation in the 3 studied experimental systems. According to the criteria for the sameness of enoxaparin copies, the studied products should show close IC50 values for thrombin generation. Analysis of the IC50 values for the MRI (similar to the other parameters of thrombogram; data not shown) revealed significant differences among the copies of enoxaparin and between each of them and the branded enoxaparin.
The comparison on a gravimetric basis of the copies and the branded enoxaparin revealed a significant variability of their potency to inhibit thrombin generation. The control experiments showed that the coefficient of variation of the IC50 of the branded enoxaparin on thrombin generation was about 2%. However, this was not the case when the coefficients of variation of the IC50 for the MRI were calculated for the copies of enoxaparin. These values ranged from 12% to 33% depending on the experimental system. Interestingly, the effectiveness of each of the enoxaparin copies to inhibit thrombin generation as compared to the branded product varied in the 3 experimental systems. For example, Cutenox was less potent than Lovenox when they were compared in PPP, but it showed a significantly higher potency than Lovenox when they were compared in PPP in the presence of pancreatic cancer cells BXPC3. Enoxa was 20% more potent than Lovenox in PPP, but it was 17% less potent than Lovenox in PRP.
The variability of the inhibitory potency of enoxaparin copies on thrombin generation was significantly higher in PRP as compared to PPP. In addition, when assessed in PRP, all copies of enoxaparin were significantly less potent than the branded one. Thus, we conclude that enoxaparin copies are more vulnerable to platelet-induced inhibition as compared to the branded product. The variability of the IC50 values of enoxaparin copies was lower in PPP enriched with BXPC3 cells as compared to those observed in PPP. As mentioned earlier, the main difference between the 2 experimental systems refers (i) to the concentration of the procoagulant phospholipids, which favors the formation of the enzymatic complexes (prothrombinase and intrinsic tenase), and (ii) to the presence of the cellular membranes of cancer cells that might bind nonspecifically heparin chains. Consequently, the comparison of the 2 experimental systems demonstrated that the formation of prothrombinase and intrinsic tenase is a determinant factor for the variability of the inhibitory potency of enoxaparin copies on thrombin generation. Taking into consideration that according to the manufacturers the studied copies of enoxaparin had similar mean molecular weight (within specifications range) and the same specific anti-Xa and anti-IIa activity per gravimetric unit, the data presented herein allow us to conclude that interactions with cancer cells and platelets are responsible for this variability. The procedure for the evaluation of the sameness of enoxaparin copies, which is proposed in the present study, allows the enoxaparin copies to be distinguished not only from the branded enoxaparin but also among them. The findings of the present study argue in favor of the statement that some of the studied enoxaparin copies, although they respond to the pharmacological criteria sameness established by the FDA and EMA, are not the same to the branded drug.
The in vitro evaluation of the inhibitory potency on thrombin generation, using the calibrated automated thrombogram, could be an additional method for the assessment of the sameness of the LMWHs and their copies. In vitro thrombin generation assay triggered by low concentrations of TF in the presence of platelets or cancer cells is informative for the global antithrobotic potency of LMWHs and their copies in more physiologically relevant conditions as compared to those in the prothrombin time or in the activated partial thromboplastin time or the measurement of the specific anti-Xa activity. However, thrombogram assay presents several limitations since the activation of FXIII by thrombin and consequently its effect on the stabilization of fibrin monomers cannot be assessed. In addition, the present experimental procedure underestimates the effect of thrombin on the thrombomodulin-dependent activation of PC and the consequent downregulation of thrombin generation. Similarly, the interference of thrombin generation with the fibrinolysis via the activation of TAFI is not assessed. These thrombin-dependent physiological processes are potentially influenced by LMWHs and consequently might reveal additional differences between the copies and the original products. The present study provides evidence that the introduction of a functional criterion in the procedure of the evaluation of the sameness of the copies and the branded LMWH, this assay is of value. However, this assay should not be used as a sole functional criterion for the assessment of the bioequivalence of the LMWH copies. This new approach for the evaluation of functional bioequivalence needs to be enriched with additional global assays of blood coagulation, fibrin formation, and fibrinolysis. Finally, the functional approach for the evaluation of the bioequivalence of LMWHs and their copies should be completed by in vivo experimental modes, which provide the best estimation for the in vivo thrombin generation. 30
In conclusion, the findings of the present study underline the need for the elaboration of functional criteria, which evaluate the global antithrombotic capacity of LMWH copies in order to evaluate their potential sameness with the branded drug.
Footnotes
Acknowledgments
This study was supported by unrestricted grant from Sanofi. The authors acknowledge Mme Hayat Mokrani for her skillful technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
