Abstract
Microgravity conditions cause myocardial atrophy and dysfunction with limited therapeutics. Accumulating evidence demonstrates that the deficiency of nicotinamide adenine dinucleotide (NAD+) contributes to myocardial dysfunction under microgravity, making NAD+ boosting an appealing therapeutic approach. In this study, we sought to investigate whether β-nicotinamide mononucleotide (NMN), a precursor of NAD+, preserved cardiomyocytes size and myocardial function during microgravity. Simulated microgravity was induced by tail-suspension in C57BL/6 mice for 28 days. NMN (100 mg/kg body weight) was given every other day after the onset of tail-suspension. Tail-suspension reduced the NAD+ content in hearts and decreased the heart weight and cardiomyocytes size, and cardiac function. Administration of NMN attenuates myocardial atrophy and preserves myocardial function in tail-suspended mice. These cardioprotective effects of NAD+ repletion were associated with the reduction of oxidative stress and improvement of autophagic flux. These findings indicate that NMN may hold great potential as a therapeutic approach for myocardial abnormalities under microgravity conditions.
Introduction
Myocardial atrophy and cardiac dysfunction caused by microgravity environment1,2 are important factors leading to long-term orthostatic intolerance and decrease exercise capacity after astronauts return to the Earth. 3 However, the mechanism of myocardial atrophy caused by microgravity has not been fully elucidated, and the prevention and treatment strategies are extremely limited. Therefore, effective treatment countermeasures are required for attenuating myocardial dysfunction under microgravity conditions.
Nicotinamide adenine dinucleotide (NAD+) is a cofactor for enzymes involved in multiple metabolism pathways and a substrate for NAD+ dependent enzymes, including ploy-ADP-ribose polymerases, sirtuins, and CD38. 4 Therefore, NAD+ is indispensable for the maintenance of cellular redox balance, DNA repair, and other processes. 5 Stimulated microgravity elicits the reduction in myocardial NAD+ level and NAD+ boosting strategies have shown promise in alleviating tail-suspension-induced aging-related myocardial injury in mice. 6 Of particular interest, β-nicotinamide mononucleotide (shorten as NMN), a direct precursor of NAD+, is one of the intermediates in NAD+ biosynthesis and has been supported the safety in patients.7,8 A recent study reported that intraperitoneal injection of NMN prevented tail-suspension-related bone loss in rats. 9 Most recently, a new paper reported that oral administration of NMN attenuated aging-related myocardial dysfunction in response to tail-suspension in mice. 6 These findings strongly suggest that NMN may provide protective effects in cardiac abnormalities under microgravity conditions. However, the underlying mechanisms remain incompletely understood.
Accumulating studies indicate by which stimulated microgravity may lead to the progression of myocardial dysfunction is through oxidative stress.10–12 One of the main characteristic of stimulated microgravity is the accumulation of free radicals, eg, reactive oxygen species (ROS). 13 Increased oxidative stress has been shown in heart tissues from mice with microgravity stimulation.11,14,15 Recently study demonstrated a decrease in antioxidant proteins, including nuclear factor erythroid 2-related factor 2 (Nrf2) and superoxide dismutase 2 (SOD2),9,10 indicative of an increase in oxidative stress under stimulated microgravity conditions. Since the antioxidant system of cardiomyocyte is relatively weak, autophagy serves as an important defense mechanism against oxidative stress. 16 It is well known that oxidative stress mediates an autophagic flux blockage, suggesting that ROS and aberrant autophagy maybe inter-related. 17 Although both oxidative stress and aberrant autophagic flux are observed and contribute to myocardial atrophy in tail-suspended mice, 18 it remains elusive if ROS/perturbated autophagy are modulated by NMN under stimulated microgravity conditions. This study investigated whether boosting NAD+ with NMN could be a therapeutic approach to alleviate myocardial abnormalities stimulated by microgravity.
Methods
Animal Experiments
This investigation conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication, 8th Edition, 2011). This was a double-blind, randomized study, and all animal procedures were approved by the Laboratory Animal Research Committee of Soochow University, China.
The experimental mice were randomly divided into Sham group and tail-stimulated group. Stimulated microgravity was induced by tail-suspension on adult mice (male at 2 months) for 28 days as previously described. 11 Briefly, a segment of adhesive tape was affixed to both the tail of the mice and a swivel that was connected to a horizontal bar positioned at the upper part of the cage. Throughout the period of tail-suspension, the mice have free access to food and water. NMN (100 mg/kg/body weight) or saline were intraperitoneally injected into mice every other day. Four groups were included: (1) Sham + Saline (10 mice); (2) Sham + NMN (10 mice); (3) tail-suspension + saline (10 mice); (4) tail-suspension + NMN (10 mice); 28 days later, all mice were killed as described previously, 11 and serum and heart tissues were collected for further analysis.
For autophagic flux, bafilomycin A1 (1.5 mg/kg. i.p.) were injected into mice 2 h before killing.
Echocardiography
Mice were anesthetized with 1% inhalant isoflurane. Echocardiography was performed by using a Visual Sonics Vevo 2100 system equipped with a 40 MHz linear array transducer. M-mode and two-dimensional parasternal short-axis (40 μm × 80 μm) scans at the level of the papillary muscles were performed. Left ventricular (LV) end-systolic inner diameter, LV end-diastolic inner diameter, LV posterior wall thickness in end-diastole and end-systole, fractional shortening (FS%), and ejection fraction (EF%) were determined as we reported previously. 19
Histological Analysis
Mice were euthanized by cervical dislocation under isoflurane inhalation anesthesia. Heart tissues were collected, fixed, and sectioned as previously described. 20 For cell size measurement, several cross-sections of the whole heart (5 μm thick) were prepared and stained for membranes with wheat germ agglutinin (Thermo Fisher Scientific, USA) and nuclei with Hoechst 33258 (Thermo Fisher Scientific, USA). Every single cardiomyocyte was measured using ImageJ 1.48v software. At least a total of 200 cardiomyocytes were traced in each heart.
Western Blot Analysis
Fifty micrograms of protein from heart tissues were loaded onto SDS-polyacrylamide gels. After electrophoresis, the separate proteins were transferred onto PVDF membranes. After blocking in 5% nonfat milk for 1 h, the membranes were incubated with antibodies against LC3 A/B (1:1000 dilution; Cell Signaling Technology, USA), p62 (1:1000 dilution; Cell Signaling Technology, USA), Nrf2 (1:1000 dilution; Affinity Biosciences, China), Keap1 (1:1000 dilution; Proteintech, USA), and GAPDH (1:5000 dilution; Proteintech, USA), respectively. After washing steps, the membranes were incubated with secondary antibodies conjugated with horseradish peroxidase. The signals were developed via an enhancing version of chemiluminescence reaction. Quantitative analysis was carried out using ImageJ 1.48v software.
Real-Time Polymerase Chain Reaction
Total RNA of heart tissues was isolated using TRIzol reagent (Sigma Aldrich, USA) according to the manufacturer's instructions. RT-PCR was conducted to analyze the mRNA levels of Slc12a8 and GAPDH using Premix Ex Taq™ II (Takara, Japan) and the ABI Real-Time PCR system (Applied Biosystems, USA). The sequences of primers are as follows: Slc12a8, 5’-CACAGGGGCCATGATGACTT-3’ and 5’- CTTGAAGCAGGGCTCTGTCA-3’; GAPDH, 5’- AAAGGGCATCCTGGGCTACA-3’ and 5’-CAGTGTTGGGGGCTGAGTTG-3’.
Detection for NAD+
The content of NAD+ was detected by commercially available kits (Beyotime, China) according to the instructions.
Determination of Oxidative Stress Levels in Hearts
The method we determine oxidative stress levels was the same as our previous study. 15 The generation of ROS in heart tissue lysates was measured using the Amplex® Red hydrogen peroxide/peroxidase assay kit (Thermo Fisher Scientific, USA), according to the manufacturer's instructions. Briefly, frozen heart tissues were homogenized in an assay buffer. The homogenates (100 μg of protein) were incubated with a fluorescent probe Amplex® Red and hydrogen peroxide/peroxidase at 37 °C. The fluorescent product formed was quantified using a spectrofluorometer measured at 530/590 nm. Changes in fluorescence were expressed as arbitrary units.
Detection for Malondialdehyde
The lipid peroxidation in heart tissues was assessed by determining malondialdehyde (MDA) content using a commercially available kit (Nanjing Jiancheng Bioengineering Institute, China) according to the instructions.
Superoxide Dismutase Activity
SOD activity in heart tissues was measured using a total SOD assay kit (Beyotime, China) according to the manufacturer's instructions.
Statistical Analysis
Experimental data were analyzed using GraphPad Prism software (Version 8.0.1) and presented as means ± SD. Student t-test was used for two-group comparison. For multiple groups, differences were assessed by one-way-ANOVA or two-way-ANOVA, followed by Tukey's Honest Significant Different test for post-hoc analyses. A value of P < 0.05 was considered significant statistically.
Results
Tail-Suspension Treatment Induces NAD+ Reduction, Decrease Cardiomyocyte Size, Heart Mass, and Myocardial Function in Mouse Hearts
Tail-suspension for 28 days resulted in lower content of NAD+ in mouse heart tissues (Figure 1A). NMN has been widely used for NAD+ replenishment via its receptor Slc12a8. 21 As shown in supplementary figure S1, tail-suspension did not alter the mRNA expression level of Slc12a8. This result provides a rationale for the chosen of NMN as a therapeutic approach under microgravity conditions. After receiving NMN, the NAD+ content was increased in mouse heart tissues (Figure 1B). These results indicate NAD+ exhaustion in tail-suspended mouse hearts. Furthermore, this decrease of NAD+ negatively correlated with EF% (Figure 1C) and FS% (Figure 1D), heart mass (Figure 1E), cardiomyocytes size (Figure 1F), and Troponin I (Figure 1G) and CK-MB (Figure 1H) content in serum in mice with stimulated microgravity.

Tail-suspension induces NAD+ decrease, myocardial atrophy, and dysfunction in mice. (A, B) NAD+ content in heart tissues. (C, D) Myocardial systolic function in analyzed by echocardiography. (E) Heart weight/body weight (mg/g). (F) Quantification of cross-sectional areas of cardiomyocytes. (G, H) Troponin I and CK-MB levels in serum. Data are mean ± SD, N = 6 in each group. *P < 0.05 versus Sham + saline. NAD+, nicotinamide adenine dinucleotide.
Tail-Suspension Treatment Contributes to Oxidative Stress and Autophagic Flux Blockage in Mouse Heart Tissues
To further detect the effect of NAD+ loss on myocardial dysfunction, we detected the content of ROS and MDA in myocardial tissues from mice with different durations of tail-suspension. Tail-suspension increased the content of ROS (Figure 2A) and MDA (Figure 2B) in myocardium, which was consistent with the decline of NAD+ content. Since previous studies have implicated the impairment of autophagy in tail-suspended cardiac injury, 18 we evaluate LC3B, an indicator of autophagy, in sham and tail-suspended mice heart tissues. Tail-suspension treatment time dependently increased the protein level of LC3B in heart tissues (Figure 2C and 2E), suggesting the possibility of blockage of autophagic vacuole processing. To address this possibility, bafilomycin A1, an inhibitor of late stage of autophagy, was injected into mice. Administration with bafilomycin A1 increased the protein levels of LC3B in saline group, while LC3B protein levels were not further increased incubating with bafilomycin A1 in tail-suspended mice (Figure 2D and 2F). These findings suggest that the accumulation of LC3B in tail-suspended mouse hearts resulting from the inhibition of LC3B turnover, implicating the blockage of autophagic flux. These results demonstrate that the decrease of NAD+ content caused by tail-suspension was combined with cardiac autophagic flux blockage and oxidative stress in myocardium.

Oxidative stress and autophagy measurement. (A) ROS generation and (B) MDA content were analyzed in heart tissues. (C) Representative western blot images for the protein levels of LC3B and GAPDH. (E) Quantitation of LC3B over GAPDH ratio. (D) After 28 days with tail-suspension, mice were injected with Bafilomycin A1 (1.5 mg/kg, i.p.) or DMSO. 2 h later, western blot was performed to determine the protein levels of LC3B and GAPDH. (F) Quantitation of LC3B over GAPDH. Data are mean ± SD, N = 6 in each group. *P < 0.05 versus Sham + saline and †P < 0.05 versus Sham + saline. ROS, reactive oxygen species.
Prevention of Cardiac Injury and Dysfunction by NMN in Tail-Suspended Mice
To evaluate the in vivo effects of NMN, we injected NMN (100 mg/kg/body weight) or saline into mice. This does of NMN was chosen based on our and others recent studies and previous results which reported the safety and efficacy in increasing the content of NAD+ in multiple organ tissues in mice.7,22 After the onset of tail-suspension, mice were injected with NMN every other day or saline for 28 days. 28 days later, microgravity stimulation decreased EF% and FS% in tail-suspended mice (Figure 3A, B and supplementary table S1), indicating of cardiac dysfunction. Administration of NMN did not affect cardiac function in mice from saline group, but attenuate myocardial dysfunction in tail-suspended mice (Figure 3A, B and supplementary table S1). 28 days after tail-suspension, boosting NAD+ with NMN resulted in bigger heart weight (Figure 3C and supplementary table S2) and larger cardiomyocyte size (Figure 3D and 3E). As indicators of myocardial injury, the level of Troponin I and CK-MB in serum were significantly increased in tail-suspended mice compared with mice in sham group. Relative to saline treatment, administration of NMN results in lower level of Troponin I and CK-MB in serum of tail-suspended mice (Figure 3F and 3G). NMN treatment did not affect myocardial function, the ratio of heart/body weight, the cross-sectional areas of cardiomyocytes, and the level of Troponin I and CK-MB in sham animals (Figure 3A-G and supplementary table S1-2). There is no death in all groups. Taken together, these findings indicate that NMN protects the heart against microgravity induced by tail-suspension in mice.

Protective effects of NMN on heart injury in tail-suspended mice. (A, B) Myocardial systolic function determined by echocardiography. (C) Heart weight/body weight (mg/g). (D) Representative micropictures for cardiomyocyte cell membrane and nuclei staining with wheat germ agglutinin (red) and Hoechst 33258 (blue). (E) Quantification of cell size of cardiomyocytes. (F, G)Troponin I and CK-MB levels in serum. Data are mean ± SD, N = 6 in each group. *P < 0.05 versus Sham + saline and #P < 0.05 versus TS + saline. NMN, β-nicotinamide mononucleotide.
NMN Activates the Nrf2/Keap1 Anti-Oxidative Stress Pathway and Promotes Autophagy in Tail-Suspended Mouse Hearts
The inseparable relationships between NAD+, oxidative stress, and autophagy have been widely reported. 23 To explore whether NMN reduced microgravity-induced cardiac injury by improving autophagic flux and attenuating oxidative stress, NMN or saline were injected into mice separately. Consistently, the protein levels of LC3B were significantly increased in tail-suspension-stimulated mice, while NMN treatment normalized the protein levels of LC3B and p62 (Figure 4A-C). These results demonstrate that NMN protects autophagy in tail-suspended mouse hearts. We also analyzed the oxidative stress in tail-suspended mouse hearts. Compared to the tail-suspension group, the expression of anti-oxidative stress factor Nrf2 and SOD activity significantly increased with administration of NMN (Figure 4A, 4D and 4F). Meanwhile, the levels of Keap1, ROS generation, and MDA decreased (Figure. 4A, 4E, 4G and 4H). These results indicate that NMN activates the Nrf2/Keap1 antioxidant stress pathway and promoting autophagy.
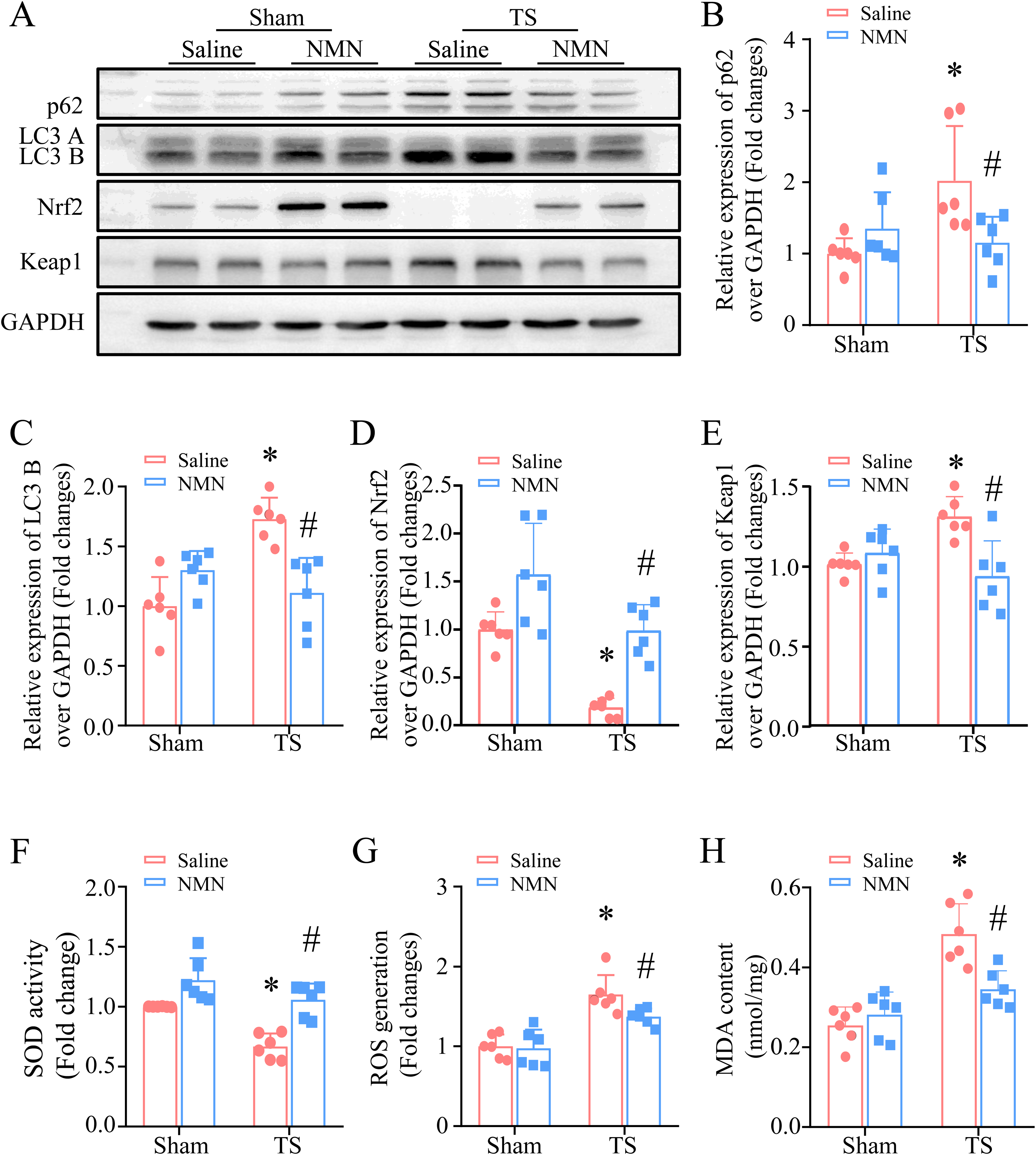
Effects of NMN on oxidative stress and autophagy in mice heart with tail-suspension. (A) During tail-suspension, mice were received with NMN or saline every other day for a total of 28 days. Autophagy was analyzed by the protein level of LC3B and p62. Representative Western blot images for LC3B, p62, Nrf2, Keap1 and GAPDH. (B, C, D, E) Quantitative analysis of the protein levels of p62, LC3B, Nrf2, and Keap1 relative to GAPDH. (F) SOD activity was determined in heart tissue lysates. (G, H) ROS production and MDA content were determined in heart tissues. Data are mean ± SD, N = 6 in each group. *P < 0.05 versus Sham + saline and #P < 0.05 versus TS + saline. NMN, β-nicotinamide mononucleotide; ROS, reactive oxygen species; Nrf2, nuclear factor erythroid 2-related factor 2; SOD, superoxide dismutase.
Discussion
The present study demonstrated that administration of NMN reduced oxidative stress and autophagy in the hearts, alleviated myocardial atrophy and improved cardiac function in mice after 28 days of tail-suspension, suggesting that boosting NAD+ with NMN may be a potential approach to attenuate myocardial abnormalities under microgravity conditions.
NAD+ deficiency was reported in serum from tail-suspended mice 6 and unloading rat bone. 9 To the best of our knowledge, this study demonstrates for the first time that tail-suspension induces NAD+ exhaustion in mouse heart tissues. Importantly, boosting NAD+ with NMN improved cardiomyocyte size and heart mass and preserved myocardial function in tail-suspended mice, underscoring a pivotal role of NAD+ in myocardial atrophy and abnormalities with stimulated microgravity. As further evidence in support of our findings, a recent study demonstrated that oral administration of NMN improved aging-related cardiac dysfunction caused by tail-suspension. 6 Thus, these findings strongly suggest that administration of NMN may present a potential therapeutic approach for myocardial disorders under stimulated microgravity conditions.
Studies from our and other labs demonstrated that oxidative stress was induced in tail-suspended mouse heart tissues and inhibition of ROS generation preserved cardiomyocyte size and heart weight and improved myocardial function in tail-suspended mice.10,24 These findings indicated an important role of oxidative stress in myocardial abnormalities under microgravity conditions. NAD+ is an important coenzyme for redox reactions; decreased NAD+ may negatively affect oxidative stress in microgravity-stimulated heart tissues. Indeed, accumulating evidence reported that boosting NAD+ with NMN abates oxidative stress in multiple cardiovascular diseases, including ischemia reperfusion, septic and hypertrophic cardiomyopathy and heart failure.19,25–27 Consistently, this study shows that the cardio-protective effects of NMN were also associated with the reduced oxidative stress. Oxidative stress represents an imbalance between endogenous antioxidant defense systems and ROS production systems. Nrf2 is a key transcription factor that regulate expression of antioxidant proteins that alleviating oxidative stress damage. Our findings showed that administration of NMN upregulate the protein expression levels of Nrf2. This upregulation of Nrf2 resulting in elevated levels of SOD and reduced ROS and MDA contents. These findings suggest that the protective effects of NMN may be mediated through antioxidant defense systems in myocardial abnormalities upon tail-suspension.
Another important finding is that tail-suspension-induced aberrant autophagy contributes to cardiac dysfunction, an event which is attenuated by NMN. Autophagy is a conserved process including autophagosome formation, maturation, fusion, and breakdown in lysosome, and the release of macromolecules back into the cytosol, allowing for the recycling of cellular building blocks. It has been demonstrated that tail-suspension induces aberrant autophagy in heart tissues whereas maintaining normal autophagy is critical for cardio-protection under microgravity conditions.18,28 In line with previous reports, the current study shows that aberrant autophagy is observed in heart tissues with stimulated microgravity. We also demonstrate that NMN restored autophagy in mouse heart tissues under microgravity condition. Further study to support this conclusion is that normal autophagy or mitophagy plays pivotal role in maintaining cardiac function under microgravity conditions.
The relationships between NAD+, oxidative stress, and autophagy are inseparable. Previous studies have demonstrated that NAD+ treatment enhanced autophagy triggered by oxidative stress. 29 Blocking autophagy with chloroquine or bafilomycin A1 inhibited the decease of ROS production by NAD+ administration.30,31 Moreover, the impairment of autophagy following Nrf2 downregulation indicates that oxidative stress also plays a pivotal role in the regulation of autophagy. 31 Our study has demonstrated that the interplay of oxidative stress and aberrant autophagy contributes to myocardial abnormalities under microgravity, which is prevented by NMN.
This study provides a new potential approach by which NMN attenuate myocardial abnormalities induced by stimulated microgravity and provide hope for translating these findings to human beings, especially for astronauts. NMN has demonstrated safety in human trials, supporting the potential as a therapeutic agent and its oral bioavailability to elevated NAD+ levels in humans are promising for non-invasive administration.7,8 While mice and humans exhibit significant differences in cardiovascular physiology, including heart rate, metabolic rate, and stress responses, which may affect the efficacy of NMN on myocardial function under microgravity conditions. Meanwhile, our study couldn’t account for the cumulative effects of space radiation or other environmental stressors in spaceflight. Additionally, the 28-day tail-suspension period in mice may not fully replicate the prolonged microgravity exposure experiences by astronauts during spaceflight. However, due to the difficulty of mimicking microgravity conditions in humans, tail-suspension is recognized as the most appropriate animal model and has been extensively employed because the observed response mimic observations made during exposure to microgravity.6,11,32 Different types of techniques, degree of tilt, and duration of head down contribute to some inconsistent observations in literature. Therefore, further studies are needed to investigate the efficacy of NMN in animals combined with other environmental stressors (eg, radiation) or astronauts during spaceflight.
In summary, we have demonstrated that administration of NMN attenuates oxidative stress and enhances autophagic flux thereby alleviating myocardial dysfunction under tail-suspension-stimulated microgravity conditions. Given that recent clinical trials have shown support for the safety in patients, this study favors future translational potential to attenuate myocardial dysfunction using NMN upon microgravity conditions.
Supplemental Material
sj-xlsx-1-cpt-10.1177_10742484251369609 - Supplemental material for Administration of β-Nicotinamide Mononucleotide Attenuates Myocardial Dysfunction in Tail-Suspended Mice
Supplemental material, sj-xlsx-1-cpt-10.1177_10742484251369609 for Administration of β-Nicotinamide Mononucleotide Attenuates Myocardial Dysfunction in Tail-Suspended Mice by Wenyu Gu, MSc, Dan Liu, PhD, Lei Wei, MSc, Ziying Yang, MSc, Shumin Jiang, MSc, ShiyuDai, PhD, Ting Cao, PhD, and Zhenya Shen, PhD in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
sj-pptx-2-cpt-10.1177_10742484251369609 - Supplemental material for Administration of β-Nicotinamide Mononucleotide Attenuates Myocardial Dysfunction in Tail-Suspended Mice
Supplemental material, sj-pptx-2-cpt-10.1177_10742484251369609 for Administration of β-Nicotinamide Mononucleotide Attenuates Myocardial Dysfunction in Tail-Suspended Mice by Wenyu Gu, MSc, Dan Liu, PhD, Lei Wei, MSc, Ziying Yang, MSc, Shumin Jiang, MSc, ShiyuDai, PhD, Ting Cao, PhD, and Zhenya Shen, PhD in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgements
Not applicable
Ethics Approval and Consent to Participate
Not applicable
Consent for Publication
All authors consent to the publication and reduction of this work.
Author Contributions
WG, DL, and LW conducted the experiments and analyzed the data; WG, DL, SD, TC, and ZS designed the experiments and wrote the paper; WG, DL, ZY, SJ, SD, and SZ discussed the data and revised the paper. All authors approved the final version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (92168203), Jiangsu Cardiovascular Medicine Innovation Center (CXZX202210), National Key R&D Program of China (2022YFA1104300), and Priority Academic Program Development of Jiangsu Higher Education Institutions.
Declaration of Competing Interest
All authors do not have any competing interests.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Statements and Declarations
Not applicable
Supplemental Material
Supplemental material for this article is available online.
List of Supplemental Materials
Supplementary Figure 1. Slc12a8 mRNA expression in heart tissues Supplementary Table S1. Echocardiographic analysis Supplementary Table S2. Body and heart weight Supplementary data for P-value and effect size.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
