Abstract
Background:
Oxidative stress may play an important role in the development of myocarditis. We investigated the effects of
Methods and Results:
A rat model of porcine myosin-induced EAM was used. After the immunization with myosin, NAC (20 mg/kg/d) or saline was injected intraperitoneally on days 1 to 21. Additional myosin-immunized rats treated with NAC were orally given 25 mg/kg/d of NG-nitro-
Conclusion:
Treatment with NAC ameliorated myocardial injury via NO system in a rat model of myocarditis.
Introduction
Myocarditis is an inflammatory disease of the myocardium commonly associated with cardiotropic infections and has been linked to the development of autoimmunity. Myosin-induced myocarditis in rats is a murine model of experimental autoimmune myocarditis (EAM) used to investigate the pathophysiology in acute as well as chronic cardiac dysfunction and dilated cardiomyopathy (DCM). 1,2 Myocyte mitosis and the presence of cardiac precursor cells in adult hearts have been recently reported, and the death of large numbers of cardiomyocytes is thought to result in the development of heart failure. 1,2 Accordingly, restoring lost myocardium would be desirable for the treatment of myocarditis and DCM. 3
Recent reports indicate that
Nitric oxide (NO) is a gaseous free-radical molecule that has been shown to be a mediator of vital physiological functions.
10,11
Recent evidence has suggested that tissue regeneration is at least in part related to the NO system.
12,13
Many cell types are capable of producing NO through the enzymatic conversion of
Acute myocarditis is associated with loss of cardiomyocytes as mentioned earlier. To treat it, replacement by a therapeutic regime aiming at tissue regeneration might be needed. In the current study, we examined the effects of NAC treatment on rats with EAM in relation to tissue regeneration and the NO system.
Methods
Immunization
Experimental autoimmune myocarditis was created in 6-week-old Lewis rats by subcutaneous injection of 0.2 mL of porcine cardiac myosin (10 mg/dL; Sigma), in the foot pads on days 1 and 8, as described previously. 2 Unimmunized rats were prepared. The rats were observed daily at the end of the experiment. The reason why we use young (6-week-old) rats is that the incidence of myocarditis is high around 5- to 6-week-old rats compared with old rats.
The protocols were approved by the Animal Care Committee of Kyoto University. The investigation conforms to the
Medical Experiment
A total of 30 rats were divided to group 1 (control; no treatment, n = 15) or group 2 (NAC treatment, n = 15). Additional immunized rats treated with NAC were orally (via drinking water) given 25 mg/kg/d of NG-nitro-
Additional groups of unimmunized rats were treated for 21 days with saline (control; n = 4), NAC (20 mg/kg/d; n = 4), NAC (20 mg/kg/d) plus
Histological Examination
Heart weight and body weight were recorded. Portions of the mid-ventricular level were fixed with formalin, embedded in paraffin, sectioned at 4-μm thickness, and stained with hematoxylin and eosin and with Mallory-azan and the remaining portions were processed to immunohistochemical staining. The sections were evaluated in a blinded manner for the characterization of infiltration, myocardial necrosis, and fibrosis as reported previously. 2,13,14
Immunohistochemical Study
Macrophages (MΦ+), CD4+-T cells, CD8+-T cells, and c-kit+ cells were stained using the indirect immunoperoxidase method as previously described by us. 15 For quantitation of MΦ+, CD4+, CD8+, and c-kit+ cells, the sections were examined by 1 examiner blindly. For negative controls, sections were prepared.
In Situ Detection of Superoxide Production in Hearts
Unfixed frozen cross-sections of the specimens were stained with dihydroethidium (Molecular Probe, Oregon) according to the previously validated method. 15 -17 For quantitation of ethidium fluorescence from hearts, fluorescence (intensity × area) was measured as described previously. 15
Intercellular Adhesion Molecule 1 Expression
Expression of intercellular adhesion molecule 1 (ICAM-1) was examined as described previously. 7 Protein samples were probed with the anti-ICAM-1 antibody (M-19; Santa Cruz Biotechnology, Santa Cruz, California). The β-actin samples were probed as internal controls for loading. Resulting bands were quantified as optical density × band area by the image analysis system.
Echocardiographic and Hemodynamic Studies
Echocardiography was performed before myosin immunization and on day 21 postmyosin injection. Left ventricular (LV) end-diastolic dimension, LV end-systolic dimension, interventricular septal thickness, and posterior wall thickness were measured. Ejection fraction (EF) of the left ventricle and LV mass were determined. 15 Blood pressure was measured serially by the tail cuff method in all the groups throughout the study.
Statistical Analysis
Data were expressed as mean ± standard deviation. Comparison of the parameters was performed by 1-way analysis of variance, followed by Fisher protected least-significant difference test.
Results
Attenuation of Myocarditis by NAC Treatment
The incidence of myocarditis was 100% in immunized rats (Figures 1
-3). Myocardial necrosis and fibrosis scores were significantly less severe in the NAC-treated group and NAC plus
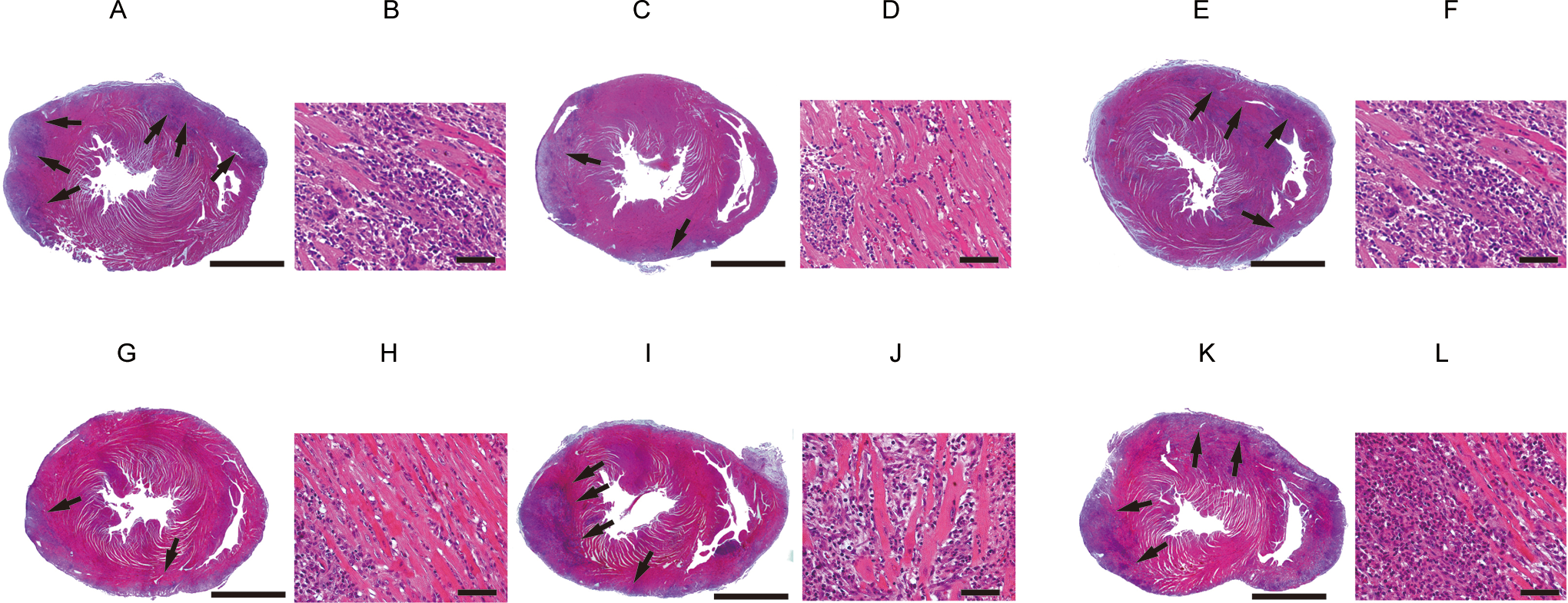
Effects of NAC treatment on myocardial necrosis with infiltrations in myocarditis. Representative myocardial sections show markedly reduced inflammation with tissue necrosis by NAC treatment (C and D) and NAC plus

Effects of NAC treatment on myocardial fibrosis in myocarditis. Representative myocardial sections show markedly reduced myocardial fibrosis by NAC treatment (B) and NAC plus

Effects of NAC treatment on superoxide production and myocardial MΦ+ expressions in myocarditis. Representative in situ superoxide production (white arrows) and myocardial sections immunohistochemically stained for MΦ+(brown cells, arrows) demonstrate the reduced expression of the positive cells in NAC group (C and D) and NAC plus
Cardiac Pathology, Immunohistochemistry, and DHE Staining.a

Abbreviations: DHE, dihydroethidium; MΦ, macrophage; ND, not detected;
aMean ± SD.
bData were obtained by the number of positively stained cells by all the counterstained cells (each trial n = 5).
cFor quantitation of ethidium fluorescence, fluorescence(intensity × area) was measured using a high-power image (each trial n = 5).
d
e
Most of the stained cells on day 21 were MΦ+ and CD4+. CD8+cells were relatively few. The percentages of MΦ+ cells in NAC-treated and NAC plus
Presence of Myocardial c-kit+cells by NAC Treatment
It is generally accepted that myocardial c-kit+ cells were rare in normal hearts. 3,18 In this study, c-kit+ cells could be demonstrated only in the NAC group but not in the other groups (Figure 4).

Myocardial c-kit+cells by NAC treatment in NAC-treated group (left) and control group (right). Myocardial c-kit+cells (brown cells, arrows) were demonstrated in NAC-treated rat. Inset boxes show magnified examples. Bar indicates 100 μm. c-kit+ stain in NAC-treated group (A). Negative stain in control group (B). NAC indicates
In Situ Superoxide Production
Ethidium fluorescence was noted from the myocardial lesions of EAM, and NAC treatment significantly suppressed the staining. That is, superoxide production was significantly decreased by the treatment of NAC or NAC plus
Western Blotting
Expression of ICAM-1 was increased in rats with myocarditis compared to normal rats (data not shown). Decreased expression of ICAM-1 in NAC-treated rats (immunized) compared with the control rats (immunized) was confirmed by Western blot analysis (Figure 5).

Western blot analysis of expression of intercellular adhesion molecule 1 (ICAM-1). A, Decreased ICAM-1 expression in NAC-treated rats (immunized) was confirmed by Western blot analysis. B, Densimetric analysis of relative protein levels. In rats treated with NAC, ICAM-1 expression was decreased but was increased by NAC plus
Cardiac Function
After 3 weeks, EF slightly decreased in rats with myocarditis compared to that of control (unimmunized) rats, indicating the presence of cardiac dysfunction in this animal model (Table 2). The significant improvement in cardiac function compared to the untreated (immunized) EAM rats was observed in the EAM rats treated with NAC but not in those with treated with NAC plus
Echocardiographic Data.a
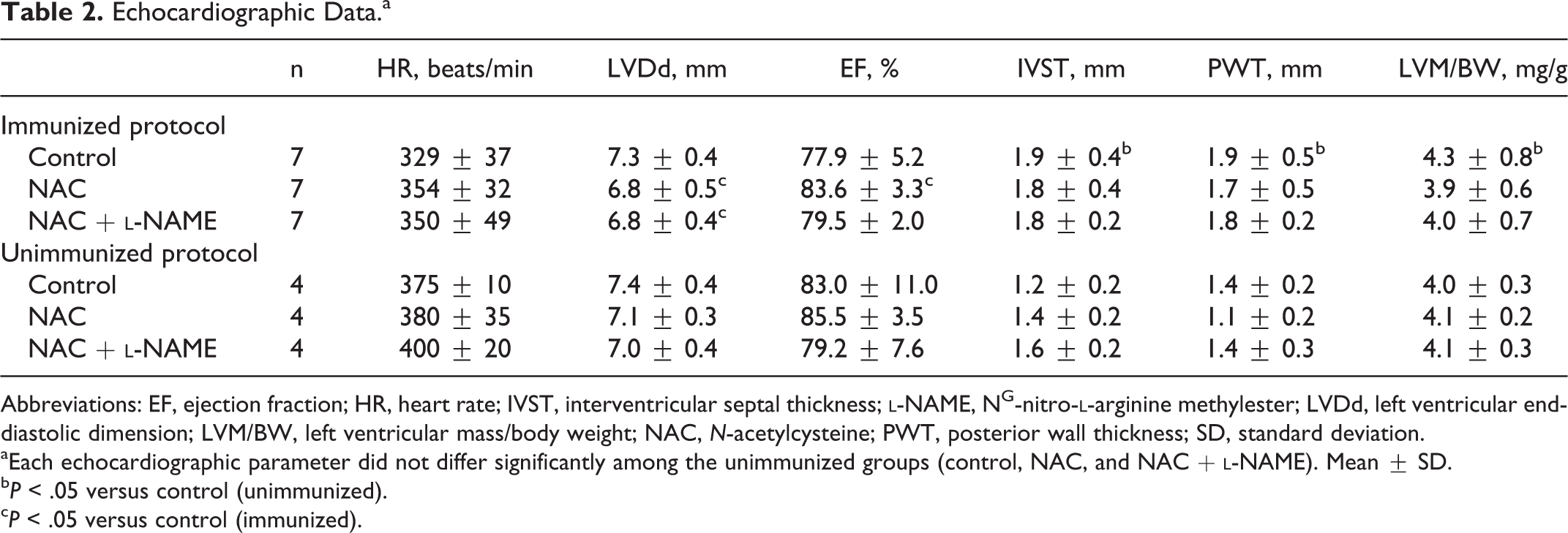
Abbreviations: EF, ejection fraction; HR, heart rate; IVST, interventricular septal thickness;
aEach echocardiographic parameter did not differ significantly among the unimmunized groups (control, NAC, and NAC +
b
c
Discussion
We showed here that the treatment with NAC against EAM ameliorated pathological findings, including myocardial necrosis and fibrosis associated with macrophage infiltrations, and suppressed superoxide production and ICAM-1 expression in the diseased myocardium. In addition, the observed beneficial effects of NAC upon myocarditis were reversed by the inhibition of NOS; that is, the effects of NAC seem to be via NO. Myocardial c-kit+cells demonstrated only in NAC-treated group may contribute, at least in part, to the improvement in myocardial lesions in this group.
There is increasing evidence to support the critical role of both free radicals and oxidative stress in the development of heart failure. 19 -21 Both clinical and experimental evidence support the hypothesis that oxidative stress is linked to hypertension, heart failure, and atherosclerosis.
The antiatherosclerotic effects of NAC in animal models, which were via NO system and antioxidative effects in part, were already reported.
7,22,23
Multipotent progenitor cells have been implicated as part of the normal cardiac repair mechanism in various adult pathophysiological states, including vascular ischemia, atherosclerosis, and pulmonary hypertension. 3,18,24 -26 Recent studies have demonstrated that tissue regeneration is associated with the induction of NOS. 9 -11 The cell-based therapies associated with the protective effects of NO have been reported. 27,28
Most recently, Niwano et al reported the potential usefulness of NAC for the suppression of ventricular structural and electrical remodeling by its antioxidative action in an EAM model. 29 In addition, it was also reported that chronic treatment with NAC is effective in improving cardiac function in Syrian cardiomyopathic hearts. 30 Indeed, echocardiographic data of the current study revealed that the increased LV mass produced by myocardial inflammation was suppressed by NAC treatment, although it did not reach a significant difference.
The effects of NAC are pleiotropic. It was already reported that NAC treatment normalizes tumor necrosis factor α via the inhibition of nuclear factor-κB activation in hypertensive rats, resulting in the protection of the myocardium. 31 Indeed, NAC treatment prevented expression of ICAM-1 in cardiac tissue in the current study. The ICAM-1 has been positioned as a critical mediator of inflammatory cell infiltrations in animals during inflammation with ventricular remodeling. 31,32 It is also well established that the recruitment of inflammatory cells to the inflammatory sites requires the binding to ICAM-1. Treatment of NAC reduced the expression of ICAM-1 and, therefore, improved cardiac remodeling in this study (Figure 6).
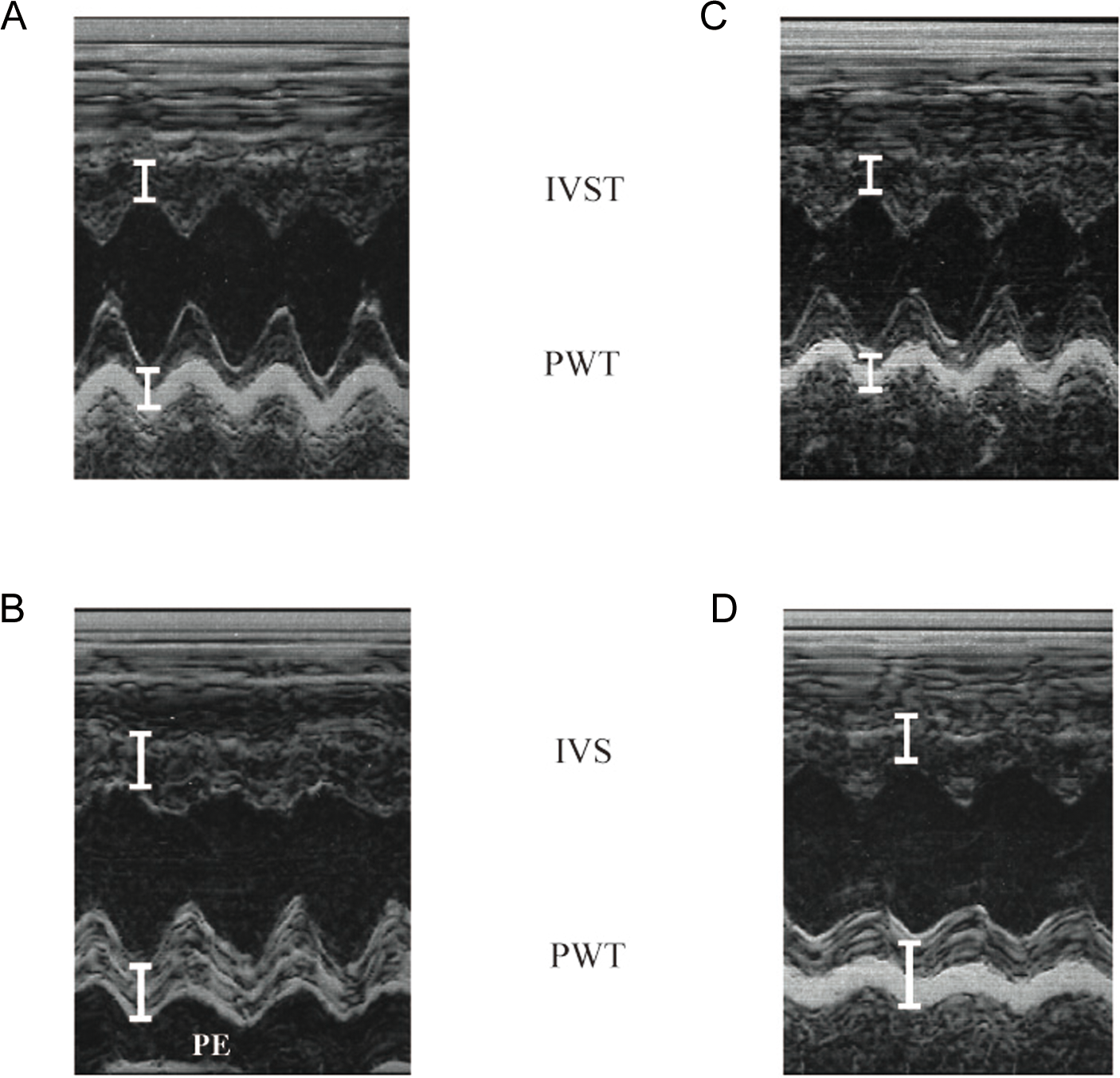
Echocardiographic findings. Echocardiography was performed on day 21. The parameters were measured as an average of 3 beats. Ejection fraction (EF) slightly decreased in the control (immunized) rat with EAM (B) compared to that of the control (unimmunized) rat (A). Pericardial effusion (PE) was observed in the rat with EAM (B). The significant improvement in cardiac function compared to the control rat with EAM (B) was observed in the EAM rat treated with NAC (C) but not in the EAM rat treated with NAC plus
There are some limitations in this study. First, although homing of bone marrow cells into the heart by NAC administration was demonstrated by myocardial c-kit+ cells, actual mobilization of bone marrow cells by NAC treatment and the precise mechanisms of NAC for myocardial c-kit+ cells expression were not demonstrated. Second, the pharmacokinetics of NAC may be different between rats and humans, as reported previously. 7,24,31 Different doses and routes of administration might be preferable. That is, further studies would be needed to determine an optimal dose–effect relationship. In the current study, however, we chose an intraperitoneal injection of 20 mg/kg/d of NAC as suggested by the previous report. 7 Finally, NO is a multiple signaling molecule involved in numerous processes, including host defense mechanism, molecular cell infiltration, and vascular smooth muscle homeostasis. 14,32,33 Significant contribution of NO in the development of EAM was already reported. 34,35 Ideally, multiple aspects of NO have to be considered in the current study. Our preliminary data have confirmed that the incidence of myocarditis is getting lower in the older rats than in the young rats. Accordingly, 6-week-old (young) rats were used in this experiment. It is known that heart rates in the young rats are higher than that in the old rats. In the current study, the so-called hyperkinetic state by relative tachycardia may contribute to the hemodynamic state of the young rats compared to the older rats; the hemodynamic changes produced by myocarditis may be hardly detected by echocardiography in the young rats.
In conclusion, NAC therapy suppressed acute EAM by the modulation of myocardial superoxide production via NO system associated with the myocardial regeneration at least in part.
Footnotes
Author Contribution
K. Shimada contributed to conception and design and analysis. H. Uzui contributed to analysis and critically revised the article. J.D. Lee gave final approval. T. Ueda critically revised the article. C. Kishimoto drafted the article and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by research grants from All Japan Coffee Associations, the Japanese Ministry of Education, Science, and Culture (18590772 and 23591040), The Univers Foundation, and The Shimizu Foundation for the Promotion of Immunology Research.
