Abstract
Objective
To assess the effect of sodium-glucose cotransporter 2 inhibitors (SGLT2-I) on cardiac remodeling and prognosis in type 2 diabetes mellitus (T2DM) patients presenting with acute myocardial infarction (AMI).
Methods
In this single-center retrospective active-comparator study, consecutive diabetic AMI patients undergoing percutaneous coronary intervention (PCI) between 2021 and 2023 were enrolled. Patients were divided into SGLT2-I users and non-SGLT2-I users based on discharge medications. The primary endpoint was the left ventricular remodeling index (LVRI), defined as the relative change in LV end-diastolic volume after six months. The secondary outcomes included major adverse cardiovascular events (MACE), comprising all-cause mortality, hospitalization for heart failure, nonfatal MI, and nonfatal stroke.
Results
The study comprised 423 T2DM AMI patients(with or without ST-segment elevation), with 239 SGLT2-I users and 184 non-SGLT2-I users. At six months, LVRI was significantly lower in the SGLT2-I users compared to the non-SGLT2-I users (3.49 ± 19.71 vs 7.06 ± 15.15, P = .042). The non-SGLT2-I users exhibited a higher prevalence of positive LVR (LVRI > 0%) (64.67% vs 50.63%, P = .004) and pathological LVR (LVRI > 20%) (19.57% vs 12.13%, P = .036). Multivariate logistic regression indicated that SGLT2-I was associated with a reduced risk of LVR (OR 0.6; 95%CI 0.38-0.97; P = .035). During a mean follow-up of 25 ± 8 months, Kaplan-Meier analysis demonstrated a lower rate of MACE-free survival in the non-SGLT2-I users (P = .005).
Conclusions
SGLT2-I protects against LVR and lowers the risk of adverse cardiovascular outcomes in T2DM AMI patients.
Introduction
Sodium-glucose cotransporter 2 inhibitors (SGLT2-I) were originally created to manage hyperglycemia in patients with type 2 diabetes mellitus (T2DM) by preventing glucose reabsorption in the kidney's proximal tubules. 1 Subsequent large-scale clinical trials have demonstrated that SGLT2-I can reduce the occurrence of cardiovascular and renal complications in patients with T2DM, 2 as well as decrease the rates of hospitalization for heart failure (HF) and cardiovascular mortality in individuals with chronic HF.3,4 The cardioprotective effects of SGLT2-I may be attributed to their ability to improve cardiac energy metabolism and systolic function, suppress the sympathetic nervous system, prevent ischemia/reperfusion injury, and mitigate adverse cardiac remodeling.5–8
In patients with acute myocardial infarction (AMI), left ventricular remodeling (LVR) can occur despite successful percutaneous coronary intervention (PCI), ultimately leading to the progression of HF. 9 The EMMY trial conducted the first assessment of the effects of SGLT2-I in AMI patients, demonstrating that SGLT2-I effectively improves postinfarction cardiac structural and functional parameters. 10 However, it should be noted that the EMMY trial had a limited sample size of diabetic patients, which may impact the ability to determine the true efficacy and safety of SGLT2-I therapy in diabetic patients with AMI. Recent clinical studies have demonstrated that T2DM AMI patients treated with SGLT2-I experience a significant reduction in inflammatory response and infarct size, leading to a decreased risk of adverse cardiovascular events.11,12
Based on the findings of these studies, we hypothesized that SGLT2-I may have a beneficial effect on LVR following AMI in T2DM patients, potentially leading to a favorable prognosis. Hence, we investigated the impact of SGLT2-I on LVR and the long-term cardiovascular outcomes in T2DM patients with AMI who have undergone successful PCI.
Patients and Methods
Ethics Approval
Approval for the research protocol in this study was granted by the Ethics Review Committee at the Second Affiliated Hospital of Anhui Medical University (YX2021-001). The research methods in this study followed the ethical principles set forth in The Code of Ethics of the World Medical Association (Declaration of Helsinki), and all subjects gave their consent after being informed.
Participants
This retrospective active-comparator observational study conducted at a single center, included a cohort of 505 consecutive T2DM AMI patients within the age range of 18 to 90, encompassing both ST-segment elevation myocardial infarction (STEMI) and non-ST-segment elevation myocardial infarction (NSTEMI), and undergoing successful PCI between January 2021 and January 2023. The diagnostic criteria and treatment protocols for STEMI and NSTEMI adhere to current guidelines.13,14 Patients were divided into two groups depending on whether they were discharged with SGLT2-I treatment or not: SGLT2-I users versus non-SGLT2-I users. SGLT2-I in this study refers only to dagliflozin and engegliflozin. The exclusion criteria include: (1) patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m2, (2) individuals who have not undergone PCI or have experienced unsuccessful PCI during the acute phase, (3) patients who have received coronary artery bypass grafting (CABG), (4) a history of malignant tumors, (5) pregnancy, and (6) discontinuation of SGLT2-I during the follow-up period. The recommended optimal drug therapy, as outlined in the guidelines, was administered to each patient (Figure 1).

Study flow.
Echocardiography Procedure
All participants in the study received baseline echocardiography during their hospitalization and had a follow-up echocardiographic assessment at six months using the same standardized protocol. Cardiac indices, such as dimensions and volumes, were measured following the guidelines established by the American Society of Echocardiography. 15 Echocardiography was performed using the GE ultrasound system (Vivid E9, GE Healthcare, Inc.) with a 2.5 to 3.6 MHz four-dimensional volume transducer. A biplane Simpson technique was used to evaluate the left ventricular ejection fraction (LVEF), left ventricular end-diastolic volume (LVEDV), and left ventricular end-systolic volume (LVESV) in the apical four-chamber view.
Definition of Endpoints
The primary endpoint was the LVR index (LVRI), which was determined by comparing the percentage change in LVEDV after six months to the initial measurement. A positive LVR (LVRI > 0%) signified an increase in LVEDV from baseline to the six-month follow-up assessment. 16 The presence of pathological LVR (LVRI > 20%) was defined as a 20% or greater increase in LVEDV between baseline and six-month follow-up. 17 The secondary endpoint in our study was defined as the composite of major adverse cardiovascular events (MACE) according to the current standards, 18 which included all-cause mortality, hospitalization for HF, nonfatal MI, and nonfatal stroke throughout the duration of the study. Data regarding MACE was acquired through telephonic inquiries or retrieval from the medical records database.
Statistical Analyses
Continuous variables were presented as the mean and standard deviation (SD), and data analysis was performed using Student's t-tests. Categorical variables were displayed as case number (percentage) and evaluated using either the Pearson chi-square test or the Fisher exact test. Univariate and multivariate logistic regression analyses were conducted to determine the protective effect of SGLT2-I treatment on positive LVR. Kaplan-Meier graphs were used to examine the occurrence of MACE in different groups, with significance evaluated through the log-rank test. Subgroup analyses were conducted based on variables including age, sex, type of myocardial infarction (STEMI/NSTEMI), presence of multivessel coronary artery disease, hypertension, and LVEF. Statistical tests were conducted with a two-tailed approach, where significance was determined at a level of P < .05. The analyses were performed using SPSS software version 26.0 (SPSS Inc., Chicago, IL, USA).
Results
Baseline Characteristics
Out of the initial 505 T2DM patients with AMI screened, 16 were excluded due to lack of undergoing successful PCI, 5 were excluded because they died during hospitalization, 21 were excluded for discontinuation of SGLT2-I during follow-up, 22 were excluded for failure to comply with the follow-up protocol, and 18 were excluded by other exclusion criteria, which included 6 who had undergone CABG, 7 with an eGFR below 30 mL/min/1.73 m2, and 5 with a history of malignant tumors. The final study cohort consisted of 423 AMI patients, with 239 SGLT2-I users and 184 non-SGLT2-I users. Their mean age was 61.90 ± 13.32 years. Table 1 provides a summary of the basic characteristics of the individuals involved. In comparison to the non-SGLT2-I users, SGLT2-I users had higher synergy between percutaneous coronary intervention with taxus and cardiac surgery (SYNTAX) scores and LVESV but lower LVEF. Regarding admission medical therapy, there were no significant disparities observed except for a marginally elevated rate of statin usage and a reduced rate of sulfonylurea usage among SGLT2-I users. With the exception of statin, β-blocker and sulfonylurea, the remaining medications prescribed upon discharge were comparable between the two groups.
Baseline Characteristics.

Values Are Shown as the Means ± SD, Number (Percentage)
STEMI, ST-segment elevation myocardial infarction; SBP, systolic blood pressure; DBP, diastolic blood pressure; SYNTAX score, synergy between percutaneous coronary intervention with taxus and cardiac surgery score; NSTEMI, non-ST-segment elevation myocardial infarction; LDL-C, low-density lipoprotein cholesterol; Cr, creatinine; cTnI, cardiac troponin I; NT-proBNP, N terminal pro B type natriuretic peptide; LVEF, left ventricular ejection fraction; LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume; ACEI, angiotensin-converting enzyme-inhibitors; ARB, angiotensin II receptor blockers; ARNI, angiotensin receptor blockade neprilysin inhibitor; GLP-1RA, glucagon-like peptide-1 receptor agonists; DPP4 inhibitor, dipeptidyl peptidase-4 inhibitor;
Changes of Echocardiography Parameters and Prevalence of LVR
After a six-month follow-up period, there was an improvement in left ventricular systolic function in both groups under observation. SGLT2-I users exhibited a greater increase in LVEF compared to non-SGLT2-I users (ΔLVEF 2.17 ± 6.66 vs 0.55 ± 9.51, P = .040). Additionally, echocardiographic parameters indicating structural cardiac changes showed significant improvements in SGLT2-I users compared to non-SGLT2-I users, with smaller dilation in LVEDV (ΔLVEDV 1.42 ± 18.10 vs 6.36 ± 14.75, P = .003) and LVESV (ΔLVESV −2.16 ± 14.89 vs 2.87 ± 16.02, P < .001) (Table 2).
Comparison of Changes of Echocardiography Parameters and Prevalence of LVR in the Two Groups.

Values are shown as the means ± SD, number (percentage).
LVEF, left ventricular ejection fraction; LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume; ΔLVEF = LVEF6 months - LVEFat baseline; ΔLVEDV = LVEDV6 months-LVEDVat baseline; ΔLVESV = LVESV6 months-LVESVat baseline; LVRI, left ventricular remodeling index.
The LVRI, which quantifies the percentage change in LVEDV after six months from baseline, was found to be significantly lower in SGLT2-I users compared to non-SGLT2-I users (3.49 ± 19.71% vs 7.06 ± 15.15%, P = .042). Additionally, a higher proportion of non-SGLT2-I users experienced positive LVR (64.67% vs 50.63%, P = .004) and a greater incidence of pathological LVR (19.57% vs 12.13%, P = .036) compared to SGLT2-I users (Table 2). Furthermore, the treatment of SGLT2-I was significantly linked to a reduced risk of positive LVR in both univariate and multivariate logistic regression analyses (P < .05 for all models) (Table 3).
Univariate and Multivariate Logistic Analyses for Positive LVR Risk According to the SGLT2-I Administration.

Model 1: Unadjusted
Model 2: Adjusted for age, sex
Model 3: Model 2 + smoking, hypertension, dyslipidemia
Model 4: Model 3+ STEMI/NSTEMI + syntex score + multivessel disease or not
Model 5: Model 4+ LDL + Cr + cTnI + NT-proBNP + HbA1c
Model 6: Model 5+ β-block at discharge, ACEI/ARB at discharge, ARNI at discharge
The subgroup analyses presented in Figure 2 demonstrate that treatment with SGLT2-I results in a significant reduction in the risk of LVR in specific patient cohorts. Specifically, patients aged over 60 years experienced a 46% decrease in risk (OR 0.54; 95% CI: 0.32-0.92, P = .023), male patients saw a 42% reduction in risk (OR 0.58; 95% CI: 0.37-0.91, P = .019), and STEMI patients had a 42% lower risk of LVR with SGLT2-I treatment (OR 0.58; 95% CI: 0.36-0.94, P = .028). Additionally, patients with multivessel disease experienced a 55% decrease in the risk of LVR with SGLT2-I therapy (OR 0.45; 95% CI: 0.24-0.84, P = .012). It is noteworthy that the use of SGLT2-I led to a substantial 85% decrease in the risk of LVR in individuals with an LVEF below 40% (OR 0.15; 95% CI: 0.04-0.66, P = .012).

Subgroup analyses of SGLT2-I for positive LVR.
The Occurrence of MACE During Follow-up in the Two Groups
During the follow-up, non-SGLT2-I users had a higher incidence of total MACE, hospitalization for HF and nonfatal MI compared to SGLT2-I users. But no discernible distinction was observed in all-cause mortality and nonfatal stroke (Table 4 and Figure 3). The Kaplan-Meier analysis revealed that the group not taking SGLT2-I had a significantly lower rate of MACE-free survival compared to SGLT2-I users (log-rank P = .005, hazard ratio 2.760, 95%CI: 1.556-4.896, depicted in Figure 4).
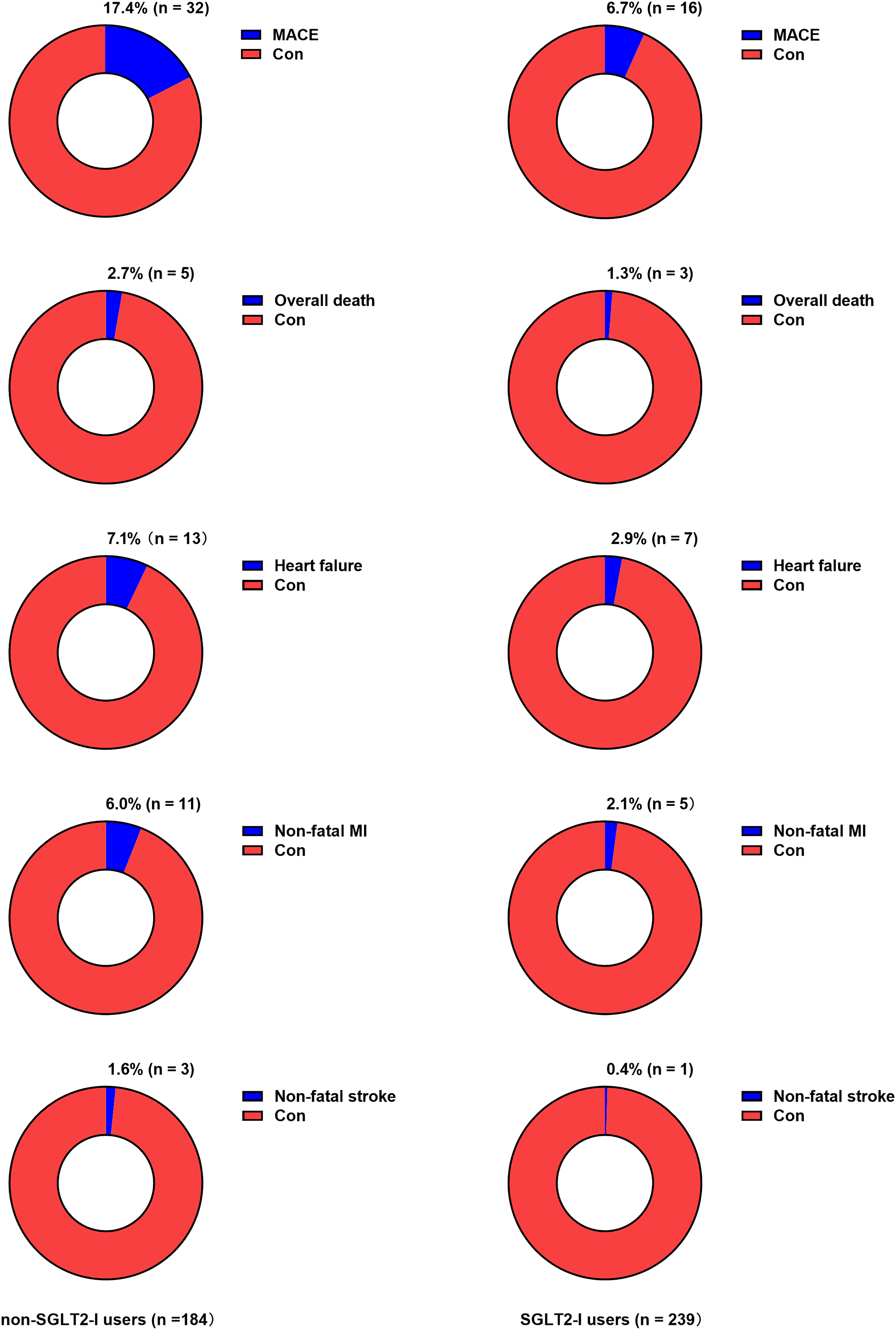
Presentation of adverse cardiovascular events during follow-up.

Kaplan-Meier curves of MACE.
The Occurrence of MACE According to the SGLT2-I Treatment During Follow-up.

Values are shown as the number (percentage).
MACE was described as the composite of all-cause mortality, hospitalization for HF, nonfatal MI, and nonfatal stroke; MACE, major adverse cardiovascular events; MI, myocardial infarction.
Discussion
This study represents the first evaluation of the efficacy of SGLT2-I treatment in preventing and reversing LVR in T2DM AMI patients undergoing PCI. The findings indicated that SGLT2-I users demonstrated a significantly lower LVRI compared to non-SGLT2-I users, which means a smaller dilation in LVEDV after a 6-month follow-up period. Additionally, a higher proportion of non-SGLT2-I users developed both positive and pathologic LVR compared to SGLT2-I users. Our research also suggests that certain individuals may experience a notable reversal of LVR following the administration of SGLT2-I. Furthermore, along with improvement in cardiac structure, a more pronounced enhancement in LVEF was also found in these patients. The subgroup analysis demonstrated that SGLT2-I was correlated with a decreased risk of LVR in T2DM AMI patients, especially in individuals of older age and male gender, those presenting with STEMI, as well as those with multivessel disease and heart failure characterized by reduced ejection fraction (HFrEF).
In recent studies, researchers have demonstrated an increasing interest in the cardioprotective properties of SGLT2-I. Clinical trials have indicated that treatment with SGLT2-I significantly enhances clinical outcomes and reduces cardiovascular mortality in patients with both chronic and acute HF.19–21 The potential mechanism of SGLT2-I in the management of HF partly lies in its ability to ameliorate adverse cardiac remodeling, which is a crucial pathophysiological process in HF and an independent predictor of mortality and morbidity in patients with cardiovascular disease. 22 The SUGAR-DM-HF trial revealed a decrease in LVEDV index in patients with HFrEF and type 2 diabetes who were treated with SGLT2-I. 5 Furthermore, a randomized controlled trial conducted by Santos-Gallego observed that giving SGLT2-I led to notable improvements in LV volumes, LV mass, LVEF, and quality of life in nondiabetic patients with HFrEF. 6 A meta-analysis conducted in prior studies has also demonstrated that empagliflozin enhances indicators of ventricular remodeling and cardiac function while exerting positive effects on atrial remodeling. 23
In the context of AMI, despite the success of primary PCI, it does not entirely eliminate the occurrence of LVR, which continues to be the primary contributor to adverse clinical outcomes such as HF and mortality. After AMI, the presence of ischemic apoptosis and necrosis in cardiomyocytes triggers immune and inflammatory reactions within the infarcted area, ultimately activating sympathetic nerve pathways and causing degradation of the extracellular matrix. Simultaneously, myocardial fibroblasts proliferate and produce collagen, leading to an accumulation of collagen in the myocardium that perpetuates cardiac remodeling.24–26 There is a scarcity of clinical research examining the impact of SGLT2-I on LVR in AMI patients. The EMMY trial represents the first investigation into the efficacy and safety of SGLT2-I treatment in this patient population. The results indicate that SGLT2-I is associated with a significant reduction in LVEDV and a corresponding improvement in LVEF following a 26-week treatment regimen. 27
The potential mechanisms by which SGLT2-I mitigates LVR include the suppression of inflammation, attenuation of sympathetic activation, and antifibrotic effects. Inflammation is an essential driver of LVR after AMI. 28 SGLT2-I reduces the secretion of interleukin-1β (IL-1β), a key inflammatory mediator, by inhibiting the NLRP3 inflammasome in macrophages.29,30 Furthermore, SGLT2-I therapy has been associated with lower levels of inflammatory markers in T2DM patients with AMI compared to those not treated with SGLT2-I. 12 Sympathetic nerve activity also significantly contributes to LVR post-AMI. 31 The EMBODY trial demonstrated that SGLT2-I could potentially enhance cardiac nerve function in patients with AMI and T2DM. 32 Additionally, SGLT2-I has been shown to decrease levels of tyrosine hydroxylase and noradrenaline in both the heart and kidneys. 33 Cardiac fibrosis serves as the primary pathological mechanism underlying LVR. 34 Empagliflozin has been observed to reduce fibroblast activation and extracellular matrix remodeling, thereby exerting antifibrotic effects. 35 Furthermore, SGLT2-I has also demonstrated efficacy in mitigating cardiomyocyte apoptosis 36 and ischemia/reperfusion injury, 37 as well as enhancing energy metabolism in cardiomyocytes, 38 contributing further to the amelioration of LVR.
However, there is ongoing debate surrounding the effectiveness of SGLT2-I in enhancing the long-term outcomes of patients with AMI. This uncertainty stems from findings of the recent EMPACT-MI trial, which indicated that SGLT2-I did not significantly decrease the occurrence of adverse clinical events in AMI patients, except for reducing hospital admissions for HF to some extent. 39 However, a more favorable outcome was observed in patients with AMI combined with T2DM. Previous clinical trials have demonstrated the effectiveness of SGLT2-I in reducing infarct size, 12 in-hospital arrhythmia burden, 40 mortality, and enhancing long-term outcomes in T2DM patients with AMI. 41 Furthermore, SGLT2-I was identified as an independent protective element against MACE in T2DM AMI patients who underwent PCI. 42 Our present results align with those findings, as we observed an increased incidence of total MACE, hospitalization for HF, and nonfatal MI in the cohort that did not receive SGLT2-I compared to those who did. However, no discernible distinction was observed in other clinical event risks. Nevertheless, this observation may also be attributed to constraints in case numbers and follow-up duration, as there were relatively few overall deaths and instances of nonfatal stroke reported during a mean follow-up period of 25 ± 8 months (ongoing).
Strengths and Study Limitations
This study represents a novel exploration of the effectiveness of SGLT2-I in the management of LVR in T2DM AMI patients who have undergone PCI. However, the single-center observational design of the study, limited sample size, and potential impact of regional healthcare variations may restrict the generalizability of these findings. Patients treated with SGLT2-I showed a reduction in LVRI and decreased LVEDV dilation six months post-treatment compared to those not receiving SGLT2-I therapy, providing valuable insights. Yet, like most clinical studies, the LVR evaluation endpoint was set at six months, and the long-term efficacy requires further validation through clinical follow-ups. Moreover, the inherent challenges of retrospective studies, such as potential selection bias and inevitable exposure factor influences, warrant careful consideration. Additionally, the short follow-up duration limits the assessment of long-term adverse clinical events post-AMI with SGLT2-I treatment. Nevertheless, the study results offer valuable insights into the relationship between early intervention with SGLT2-I and LVR as well as prognosis in T2DM AMI patients.
Conclusion
Given the cardioprotective effects of SGLT2-I in HF, T2DM, and AMI, prompt initiation of SGLT2-I therapy in T2DM patients post-AMI may confer substantial benefits due to their elevated risk profile. Although current evidence on the cardioprotective effects of SGLT2-I in T2DM patients with AMI is limited, the available data suggest a promising role for these agents in mitigating adverse LVR and improving outcomes in this patient population. Future studies are warranted to further explore the mechanisms and clinical benefits of SGLT2-I in this setting.
Footnotes
Author Contributions
JW was a major contributor in writing the manuscript. FX, HPZ, and YLW contributed to the case collection. YJ and CC analyzed and interpreted the patient data. XJ and CLY performed the statistical analysis of the data. JLC and HL contributed to the design of this study. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The research protocol for this study received approval from the Ethics Review Committee of the Second Affiliated Hospital of Anhui Medical University (YX2021-001). The methodologies utilized in this study adhered to the ethical guidelines outlined in The Code of Ethics of the World Medical Association (Declaration of Helsinki), and all participants provided informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
All participants signed an informed consent form for the publication of the study results.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
