Abstract
Introduction
Heart failure (HF) is a complex clinical syndrome that negatively impacts the quality of life, and places a huge and costly burden on the global healthcare system probably caused by high readmission rates.1,2 As the first agent of angiotensin receptor-neprilysin inhibitor (ARNI), Sacubitril/Valsartan (S/V) has been proven to significantly reduce all-cause readmission and mortality of patients diagnosed with HF with reduced ejection fraction (HFrEF).3,4 In clinical practice, however, many patients cannot achieve the standard dose (200 mg, twice a day [b.i.d.]), or even the lowest approved dose (≤50 mg b.i.d.) due to several factors (ie, hypotension, hyperkalemia, renal dysfunction).5,6 Given novel-dose S/V (below the standard dose) would be used to treat HF patients in the real world, the clinical effects were of particular concern to clinicians. A clear and early benefit of S/V was to reduce hospital readmissions of HF patients due to any causes. 7 McMurray et al and Desai et al pointed out that S/V was superior to enalapril in improving HF patient outcomes.3,8 Lately, Carnicelli et al reported that patients with higher adherence to S/V showed a significant reduction in all-cause readmission at 3 months or 1 year. 9 Unfortunately, there remained poor understanding of novel-dose S/V. 10 It would be of importance and interest to estimate the effects of novel-dose S/V on patients’ readmissions.
Moreover, left ventricular reverse remodeling (LVRR) has the potential to play a role in the beneficial effects of S/V. 11 Especially, some previous studies have demonstrated a strongly favorable effect conferred by S/V on LVRR, meanwhile, some have reported a positive association between LVRR and clinical outcomes of HF patients.11–13 It was well known that LVRR was pivotal to the progression of HFrEF patients, while far fewer studies have examined its mediation effects on the relationship between S/V and patients’ outcomes. 14 Herein, this study would estimate the effects of novel-dose S/V on hospital readmissions, construct a risk prediction model, as well as investigate the possible role of LVRR. The findings of our study would hopefully provide crucial insights into the novel-dose S/V, and assist clinicians in selecting treatment options for patients to improve therapeutic outcomes.
Materials and Methods
Data Sources
The present study complied with the Declaration of Helsinki and was approved by the ethics committee of Xinqiao Hospital, Army Medical University (Third Military Medical University) in Chongqing, China. We retrieved the data of 2424 patients diagnosed with HF from Xinqiao Hospital from December 29th, 2017 to September 17th, 2021. Patients would be included in our study if the following criteria were met: (1) age ≥18 years, (2) left ventricular ejection fraction (LVEF) <50%, (3) echocardiography was performed as historical data, and another would be performed as a comparison, (4) receiving novel-dose S/V, and would not discontinue therapy during the study period. Exclusion criteria included: (1) underwent cardiac transplantation therapy before or during the follow-up and (2) loss of follow-up. The flowchart of patients’ selection is shown in Figure 1. The informed consent in the study was waived for its retrospective design by the ethical committee.
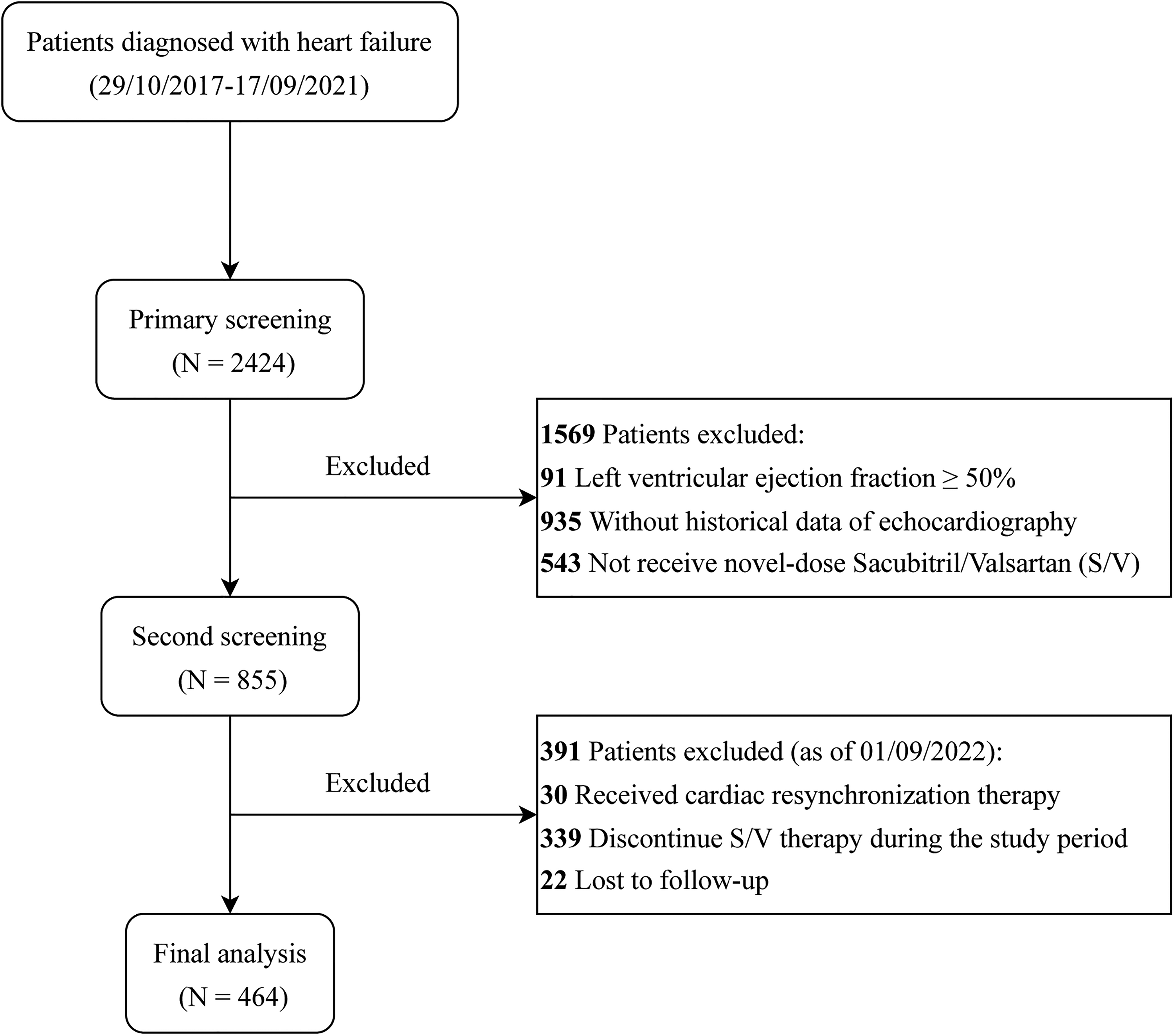
The flowchart of patients’ selection.
Data Collection
HF-associated raw data was to be mainly collected from the medical records as follows: (1) demographic characteristics, ie, identification number, sex, date of birth, height, weight, etc, (2) comorbidities, ie, dilated cardiomyopathy (DCM), myocardial infarction (MI), hypertension, coronary heart disease (CHD), valvular heart disease (VHD), chronic obstructive pulmonary disease (COPD), diabetes mellitus (DM), severe renal impairment (SRI), (3) electrocardiogram, ie, premature atrial contractions (PACs), premature ventricular contractions (PVCs), ventricular tachycardia (VT), (4) echocardiography, ie, LVEF, left ventricular end-diastolic diameter (LVEDD).
All included patients were followed up about 1 year (as of September 1st, 2022). The primary outcome was all-cause readmission, and other outcomes of interest was all-cause death.
Statistical Analysis
Given the study aimed to estimate the effects of different novel-dose levels, all patients were divided into 3 groups based on the time-weighted average dose, that was, lowest dose (≤50 mg b.i.d.), lower dose (50-<100 mg b.i.d.) and low dose (100 + mg b.i.d.), respectively. Then we split the dataset into a training dataset and a testing dataset with a ratio of 6:4 stratified by dose groups, which were used to develop a predictive model (nomogram) and assess the performance, respectively.
The competing risk analysis was performed as detailed in the supplementary files. Further, two variable selection methods were combined to determine the potential predictors for multivariable competing risk analysis, that was, least absolute shrinkage and selection operator (LASSO) approach with 5-fold cross-validation and multivariable regression using stepwise backward selection based on Akaike's Information Criterion.
Meanwhile, nomograms based on the multivariable regression models would be constructed to predict all-cause readmission of individuals with HR receiving novel-dose S/V by weighting prognostic factors. Accordingly, a web-based dynamic nomogram would be built as a decision-making tool. The performance of the nomogram was evaluated by exploiting discrimination (ie, concordance index [C-index] based on bootstrap, the time-dependent area under the curve (AUC)), calibration (ie, calibration curve), and net benefit (ie, decision curve analysis).
Moreover, nonlinear causal mediation analysis was used to further clarify the association between novel-dose S/V and all-cause readmission through LVRR as the potential mediation. LVRR was defined as an improvement ≥10% in LVEF accompanied by a reduction of LVEDD ≥ 10%, or a LVEF increase ≥10% with at least 6-month follow-up as reported.15–17 Mediation analysis commonly decomposed the total effect into direct effect and indirect effect through mediators.18,19 All statistical analyses were performed with R-4.2.1. Two tailed p-value <.05 was considered as significant.
Results
Study Population
A total of 464 patients with a median follow-up of 660 days (interquartile range [IQR]: 17-1494) were entered into our dataset, which comprised 122, 173, and 169 receiving lowest (≤50 mg b.i.d.)-, lower (50-<100 mg b.i.d.)-, and low-dose (100 + mg b.i.d.) S/V, respectively. 278 patients (median follow-up 646 days, IQR: 21-1487) and 186 patients (median follow-up 687 days, IQR: 17-1494) were divided into the training dataset (model development) and testing dataset (model performance), respectively. The baseline characteristics of these patients were summarized in the ESM_pdf: Appendix Table S1. In general, a greater proportion of these three groups of patients (lowest-, lower-, and low-dose S/V) were male, or those aged <55 years old, or diagnosed with DCM or VHD.
Novel-Dose Related Cumulative Incidence
Gray's Test showed that there was a statistically significant difference in all-cause readmission (p-value < .001) across the 3 novel-dose groups, whereas no significant difference in all-cause death (p-value = .307). As illustrated in Figure 2, the estimated cumulative incidences of all-cause readmission of patients receiving the lowest-dose S/V were 4%, 10%, 15%, 25%, 30%, 43% at 30 days, 3 months, 6 months, 1 year, 2 years, 3 years, respectively. The corresponding estimates for patients taking lower-dose S/V were 0%, 1%, 2%, 4%, 11%, 18%, and for those with low-dose S/V were 0%, 2%, 3%, 4%, 6%, 8%, respectively.

Cumulative incidence curves for all-cause readmission and death stratified by novel-dose Sacubitril/Valsartan on the basis of all dataset.
Risk Prediction Model
Univariate competing risk analysis (Table 1) revealed that age, BMI, VHD, PACs, PVCs, VT, COPD, DM, SRI, and dose were identified as the potential prognostic factors on all-cause readmission of HF patients. Then LASSO approach and stepwise backward elimination method suggested that age, VT, DM, and dose were significantly associated with patients’ readmission, while VHD and COPD were marginally associated variables. As shown in Table 1, our study constructed two models to predict primary outcome of HF patients with novel-dose S/V therapy, that was, Model 1 (age, VT, VHD, COPD, DM, and dose) and Model 2 (age, VT, DM, and dose).
Univariate and Multivariate Competing Risk Analysis of Patients’ Characteristics Influencing Primary Outcome (All-Cause Readmission) Based on Training Dataset.

Note: aVariables from least absolute shrinkage and selection operator approach and Akaike's Information Criterion, bModel 1, cModel 2.
Abbreviations: BMI, body mass index; DCM, dilated cardiomyopathy; CHD, coronary heart disease; MI, myocardial infarction; VHD, valvular heart disease; PACs, premature atrial contractions; PVCs, premature ventricular contractions; VT, ventricular tachycardia; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; SRI, severe renal impairment; HR, hazard ratio; CI, confidence interval.
C-index was 0.752 (95% confidence interval (CI): 0.724, 0.789) and 0.702 (95%CI: 0.663, 0.743), and time-dependent AUC was more than 0.677 and 0.556 over the follow-up period for Model 1 and 2, respectively. The calibration curves depicted that the observed and predicted values of the 2 models were almost consistent (as shown in the ESM_pdf: Appendix Figure S2). Decision curves were illustrated in the ESM_pdf: Appendix Figure S3, and exhibited that clinical net benefit gained from Model 1 was higher than two hypothetical scenarios (none readmission, all readmission) and Model 2 when the threshold probabilities were within the range of 0%-31% and 0%-7% for 30-day and 6-month readmission, respectively. Model 1 also had a wider range of threshold probabilities (0%-10%, 17%-20%, 23%-33%, 39%-50%) in predicting 3-month readmission. Therefore, Model 1 had considerable discriminative and calibrating abilities, and clinical utility, which was displayed as a static nomogram (Figure 3) and a web-based dynamic nomogram (https://haoxx.shinyapps.io/DynNom_HF/).

Nomogram for predicting 30-day, 3-month and 6-month cumulative incidence of patients with heart failure (in accordance with Model 1 developed by training dataset).
The nomogram implied that patients aged 75 years and above were at the highest risk of having novel-dose-related readmission, followed by those aged <55y, 65-<75y, 55-<65y. Then all-cause readmission mainly occurred in patients with the lowest-dose S/V, followed by the lower- and low-dose. Lastly, other factors associated with an increase in all-cause readmission included the history of VT, VHD, COPD, and DM.
Causal Mediation Effects
An omnibus mediation effect analysis (Figure 4) indicated that LVRR had a significant negative indirect effect on the difference of novel-dose S/V in all-cause readmission, accounting for 20.3% (−0.01735/−0.08557) of the total effect. Then the relative mediation effect analysis showed that the odds of having readmission for the lower- and low-dose were respectively on average 0.45 (95% CI: 0.37, 0.56) and 0.14 (95% CI: 0.10, 0.19) times that for the lowest-dose in total effect, while the corresponding odds of indirect effect of LVRR were 0.92 (95% CI: 0.91, 0.94) and 0.978 (95% CI: 0.971, 0.984) times.

Estimated mediation effects (with 95% confidence intervals) of left ventricular reverse remodeling on the association between novel-dose sacubitril/valsartan (S/V) and hospital readmission of patients with heart failure.
Discussion
Although S/V has been recommended in clinical practice guidelines for patients with HFrEF, some were only able to tolerate novel dosage (below the standard) dominantly due to hypotension. Our study was an attempt to assess the effects of novel-dose S/V on hospital readmissions in these patients, establish clinical prediction models, as well as explore the possible mediation effect of LVRR. Berg et al stated that the efficacy and safety of different S/V levels seemed to be consistent no matter whether patients obtained the maximum dose. 20 Consistently, our study showed significantly different cumulative incidences of all-cause readmission. The risk of all-cause readmission for patients with the lowest-dose S/V were much higher than those with the lower- and low-dose, and mostly occurred within 1 year. However, in our small-sized cohort, no difference in all-cause death among the 3 novel-dose S/V was observed. The clinical effects of novel-dose S/V remain to be further investigated.
Reducing hospital readmission was one of the primary targets of HF treatment. However, it remained unsolved how to predict which patients with HF would suffer hospital readmission. In this ARNI therapy cohort, we built a nomogram model, which revealed that age, VT, VHD, COPD, DM, and dose were associated with an increased risk of all-cause readmission. As expected, patients who had taken the lowest-dose S/V were at the highest risk of all-cause readmission, followed by those who had taken lower- and low-dose, which was in accordance with cumulative incidence curves. Previous studies have reported some factors associated with hospital readmission of HF patients, such as male gender, age, DM, VT, VHD, COPD, renal dysfunction, etc. Similarly, our study suggested that patients aged 75 + y were the high-risk population, followed by those aged <55y, 65-<75y, 55-<65y. The elderly patients were mostly observed to have low degrees of physical activity and depression and anxiety, as well as concomitant chronic diseases, which were likely to result in increasing disease risk and aggravating clinical outcomes.21,22 Whellan et al pointed out a dichotomous relationship between age and risk of death or readmission, namely, risk of readmission or death decreased as age increased up to 55 years of age, then increased in those older than 55 years. 23 Thus, there might be a tick-shaped relationship between age and all-cause readmission in patients taking novel-dose. Future studies might help more finely identify the high-risk population. In addition, patients with VT, or COPD, or DM were at significantly higher risk of all-cause readmission. This might indicate that targeted treatments of these comorbidities would provide a potential opportunity to improve outcomes in patients with HF.
Reverse cardiac remodeling refers to the recovery of ventricular function and reduction in cardiac volumes. Reverse remodeling has become a primary objective in HF treatment. Guideline-directed medical and device therapies have been proven to result in reverse cardiac remodeling. Numerous studies have highlighted the role of RAAS blockade in promoting reverse remodeling in patients with HFrEF. The SOLVD and Val-HeFT studies have demonstrated the beneficial effects of ACEI and ARB on LVRR, respectively.24–27 Since the landmark of the PARADIGM-HF study, the data supporting the link between ARNI and LVRR was growing. 3 A meta-analysis including more than 10,000 population showed that ARNI has significant improvement over ACEI/ARB on the left LV dimensions and EF. 28 The EVALUATE-HF and PROVE-HF trials both observed the reduction of multiple atrial and ventricular parameters of remodeling after initiation of ARNI.29,30 However, the relations between ARNI dosage and the LVRR are not addressed. In our study, we classified the patients into different dosage groups and revealed that the under-target novel dose remained effective on LVRR.
Mechanistically, LVRR might be a part of the explanation for reduced readmission. The mediation effects of a variety of covariates on all-cause readmission were explored. The omnibus mediation effect analysis revealed that novel-dose S/V was negatively related to all-cause readmission through its effect on LVRR, which has been identified as a critical mediator accounting for approximately 20.3% of the total effect. Meanwhile, our study demonstrated that the lower- and low-dose S/V decreased the risk of readmission by 55% (1-0.45) and 86% (1-0.14) as compared with the lowest-dose, respectively, whereas only on average 8% (1-0.92) and 2.2% (1-0.978) of the readmission risk were correspondingly reduced through the mediation of LVRR. Although LVRR had a significant negative indirect effect on the difference of novel-dose S/V in all-cause readmission, the relative effect caused by LVRR was similar between the lower- (or low-) and lowest-dose levels. It seemed that the novel-dose S/V might exert its effect through other undermined mechanisms.
Limitations
There were several limitations to our study. First, as a single-center retrospective study, our study performed internal validation for the prediction models, which still needed further external validation using an independent cohort. Second, this cohort recruited the patients taking S/V without tracing back the length of HF and the medication history. It remains unknown whether there are differences for the patients prescribed with S/V under circumstances between the recently hospitalized patients or those who were stable and transitioned to S/V. Moreover, the selection bias could not be ruled out because of the small sample size, especially for those aged over 75 years old, VHD and COPD. When an opportunity arises in the future, we hope, the reliability of the results will be further examined with a larger sample size.
Conclusions
Our study developed a clinically useful competing-risk nomogram for predicting all-cause readmission in HF patients receiving novel-dose S/V therapy. The findings revealed that the novel-dose S/V, especially the lowest, significantly affected hospital readmissions, in part mediated by LVRR. However, the clinical benefits of different novel-dose S/V and more potential novel mechanisms still require further in-depth examination.
Supplemental Material
sj-docx-1-cpt-10.1177_10742484231219603 - Supplemental material for Predicting Hospital Readmissions in Patients Receiving Novel-Dose Sacubitril/Valsartan Therapy: A Competing-Risk, Causal Mediation Analysis
Supplemental material, sj-docx-1-cpt-10.1177_10742484231219603 for Predicting Hospital Readmissions in Patients Receiving Novel-Dose Sacubitril/Valsartan Therapy: A Competing-Risk, Causal Mediation Analysis by Changchun Hou, MD, Xinxin Hao, PhD, Ning Sun, MD,Xiaolin Luo, MD, Zhichun Gao, MD, Ling Chen, MD, Xi Liu, MD, and Zhexue Qin, MD in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 82000295), the Joint Project of Chongqing Health Commission and Science and Technology Bureau (No. 2022MSXM115), and the Project for Young Talented Doctors of Xinqiao Hospital Affiliated to Army Medical University (No. 2022YQB093).
Authors’ Notes
Changchun Hou and Xinxin Hao contributed equally to this work.
Authors’ Contributions
ZX.Q., XL.L., and X.L. were involved in conception and design. CC.H. and L.C. carried out the acquisition of data. N.S. contributed to the follow-up of the patients. XX.H. and CC.H. performed the statistical analysis and drafted the manuscript. XL.L. and ZC.G. contributed to the interpretation of data. All authors contributed to the critical revision of the manuscript for important intellectual content. CC.H., XX.H., XL.L., and ZX.Q. were involved in the final revision of the manuscript. All authors read and approved the final manuscript.
Data availability
The data supporting this study's findings are available from the corresponding author upon reasonable request.
Conflict of interest
The authors declare that there is no conflict of interest.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
