Abstract
Background:
The effect of oral beta-blocker therapy on long-term mortality in patients with ST-segment elevation myocardial infarction (STEMI) who are treated with primary percutaneous coronary intervention (PCI) and who have preserved left ventricular ejection fraction (LVEF) remains unclear.
Methods:
We searched MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials for studies evaluating the effect of oral beta-blocker therapy in patients with STEMI who underwent primary PCI and who had preserved LVEF. The primary outcome was all-cause mortality. Randomized controlled trials and the observational studies that reported an adjusted hazard ratio (or hazard ratio in the propensity score-matched patients) with follow-up duration equal to or more than 6 months were included. Pooled hazard ratio with 95% confidence interval (CI) was calculated using a random effect model.
Results:
No randomized controlled trials met the inclusion criteria. Seven observational studies totaling 10 857 patients met the inclusion criteria. Follow-up duration ranged from 6 months to 5.2 years. Preserved LVEF was defined as 40% in 4 studies and 50% in 3 studies. Based on the pooled estimate, oral beta-blocker therapy was associated with a reduction in all-cause mortality (combined hazard ratio 0.79, 95% CI 0.65-0.97).
Conclusion:
This meta-analysis demonstrates that oral beta-blocker therapy is associated with decreased all-cause mortality in patients with STEMI who are treated with primary PCI and who have preserved LVEF. This supports the current American College of Cardiology Foundation/American Heart Association 2013 Guideline for the Management of STEMI.
Keywords
Introduction
A number of randomized trials predating the era of reperfusion have shown that beta-blocker therapy reduces long-term mortality in patients with acute myocardial infarction. 1 –4 Similarly, the benefit of beta-blocker therapy was demonstrated in the era of thrombolysis. 5 –7 Based on this evidence, the current guideline from the American College of Cardiology Foundation/American Heart Association recommends oral beta-blocker as a Class I recommendation in all patients with ST-segment elevation myocardial infarction (STEMI) who have no contraindication. 8 In the same line, the current guideline from the European Society of Cardiology recommends oral beta-blocker as a Class I recommendation in patients with STEMI having heart failure or left ventricular dysfunction and as a Class IIa recommendation in other patients. 9
The treatment of STEMI has markedly advanced since the role of oral beta-blocker therapy was first established. Primary percutaneous coronary intervention (PCI) has been shown to significantly decrease long-term mortality compared to thrombolysis. 10 Therefore, one may speculate that the magnitude of oral beta-blocker therapy may be reduced in the current era of primary PCI. Observational studies that assessed the effect of oral beta-blocker on long-term mortality in patients with STEMI undergoing primary PCI demonstrated mortality benefit, but these benefits were mainly observed in high-risk patients such as those with decreased left ventricular ejection fraction (LVEF) or those on diuretics. 11 –13 These findings were consistent with the established mortality benefit of beta-blocker in patients with low LVEF following acute myocardial infarction. 14
In contrast to the data in patients with reduced LVEF, the effect of beta-blocker therapy in patients with STEMI undergoing primary PCI who have preserved LVEF has not been clarified. Given this uncertainty, we conducted a meta-analysis to evaluate the effects of oral beta-blocker therapy on long-term mortality in patients with STEMI who underwent PCI and who had preserved LVEF.
Methods and Materials
We searched MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials from 1996 to January 2015 for studies evaluating the efficacy of oral beta-blocker therapy in patients with STEMI. Our search included both medical subject headings and key words including (1) “myocardial infarction” and (2) “β-blocker” or “beta-blocker” or “adrenergic beta-antagonists” or “beta adrenergic receptor blocking agent.” We had no language restrictions. Studies were also identified through a search of references cited in the screened original articles and relevant review articles.
Two investigators (NM and YK) independently screened all titles and abstracts of the identified articles. After initial screening, full-text articles of potentially relevant studies were reviewed by both investigators to determine their eligibility. Disagreements were resolved through discussion between the two reviewers. The following inclusion criteria were used for study selection: (1) randomized controlled trial or observational study that investigated the effect of oral beta-blocker therapy on long-term mortality in patients with STEMI undergoing primary PCI who were discharged alive; (2) hazard ratio of all-cause death with 95% confidence interval (CI) in patients with preserved LVEF; (3) an adjusted hazard ratio (or hazard ratio in the propensity score-matched patients) is available for an observational study; and (4) median follow-up duration equal to or more than 6 months.
Data were abstracted including the following information: first author’s name, year of publication, study period, country, study design, number of patients, clinical characteristics of the study population, type of beta-blocker and dose, definition of preserved LVEF, duration of follow-up, an adjusted hazard ratio of all-cause death with 95% CI (or equivalent) in patients with preserved ejection fraction, clinical variables used for multivariate adjustment, or propensity score matching. When the potentially eligible studies did not report an adjusted hazard ratio with 95% CI in patients with preserved LVEF, authors were contacted. Data abstraction was performed by one author (NM) and verified by another (YK).
Hazard ratios of all-cause death were used as the measure of efficacy of oral beta-blocker therapy. The natural log of the hazard ratios of each study were pooled across the studies using the inverse variance method. The standard errors were estimated from the CI provided in each original study. The I2 index was used to summarize the proportion of the total variability in the estimate. 15 We used random-effect model to calculate the pooled hazard ratio, which allows for heterogeneity between studies. We also performed subgroup analyses to evaluate associations between the effect size and study characteristics such as definitions of preserved LVEF and follow-up duration. Publication bias was estimated visually by funnel plot and with the Begg test. All analyses were performed using R software version 3.0.1 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
The results of our search strategy are shown in Figure 1. Of the 4951 articles, 124 potentially eligible articles were reviewed in full text. As a result, 7 observational studies totaling 10 857 patients met the inclusion criteria. 11,12,16 –20 No randomized controlled trials met the inclusion criteria. Additional data were obtained through contact with the authors in the 2 studies. 11,19 Four studies that reported only nonadjusted hazard ratio for all-cause mortality in patients with preserved LVEF were not included in the analysis. 13,21 –23

Flow diagram of the systematic overview process.
The characteristics of the included studies are shown in Table 1. Among the 7 studies, 1 was a retrospective analysis of patients included in randomized trials comparing PCI versus thrombolysis, 11 4 were prospective multicenter registries, 12,16 –18 and 2 were single-center studies. 19,20 Three studies specifically included patients with preserved LVEF, 18 –20 and the other 4 studies reported the results of subgroup analyses of the patients with preserved LVEF. Two studies that included patients with non-ST-segment elevation acute myocardial infarction or acute coronary syndrome reported the results of subgroup analyses of the population with STEMI. 18,19 All of the patients except for 4 patients in Raposeiras-Roubín et al study 19 underwent primary PCI. Preserved LVEF was defined as >40% in 4 studies 12,16,17,20 and >50% in 3 studies. 11,18,19 Mean follow-up duration ranged from 6 months to 5.2 years. Of the 7 studies, 3 studies reported the type of beta-blocker, and carvedilol was most commonly used in all of the 3 studies.12,16,18
Characteristics of the Studies Included in the Meta-Analysis.
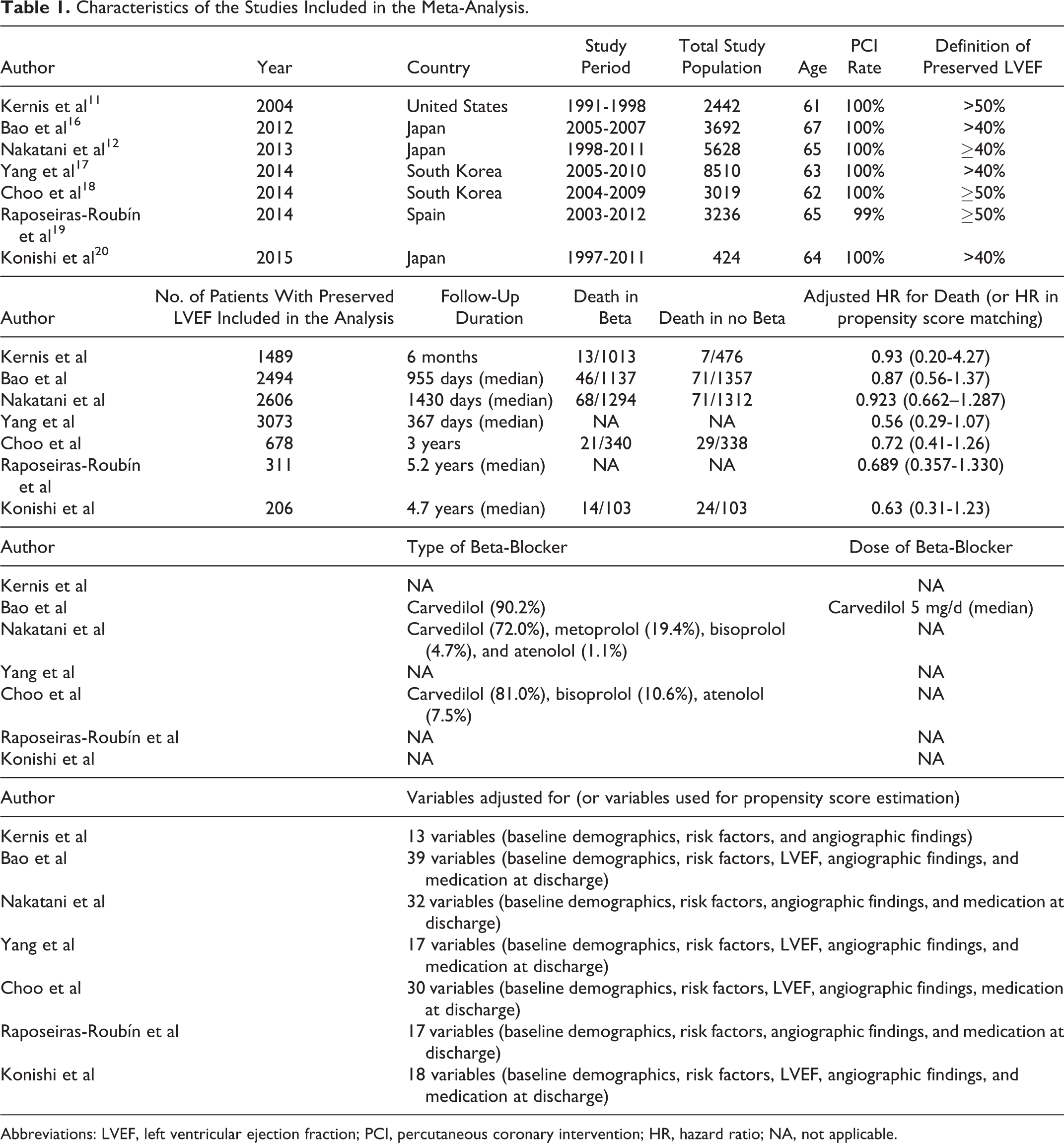
Abbreviations: LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; HR, hazard ratio; NA, not applicable.
Figure 2 shows the Forest plot reporting the hazard ratio estimate with 95% CI for all-cause mortality in patients with preserved LVEF. Pooled analysis of the 7 studies revealed a significant reduction in all-cause mortality in patients on oral beta-blocker therapy (combined hazard ratio 0.79, 95% CI: 0.65-0.97). There was no heterogeneity among the studies (I2 = 0%). Subgroup analysis showed that the pooled estimate of the hazard ratio did not vary substantially with the exclusion of the study with follow-up duration less than 1 year (combined hazard ratio 0.79, 95% CI 0.64-0.97). 11 Another subgroup analysis stratified according to the definition of preserved LVEF showed similar pooled estimates of hazard ratio (LVEF >40%; combined hazard ratio 0.82, 95% CI 0.65-1.03, LVEF >50%: combined hazard ratio 0.72, 95% CI 0.48-1.09).
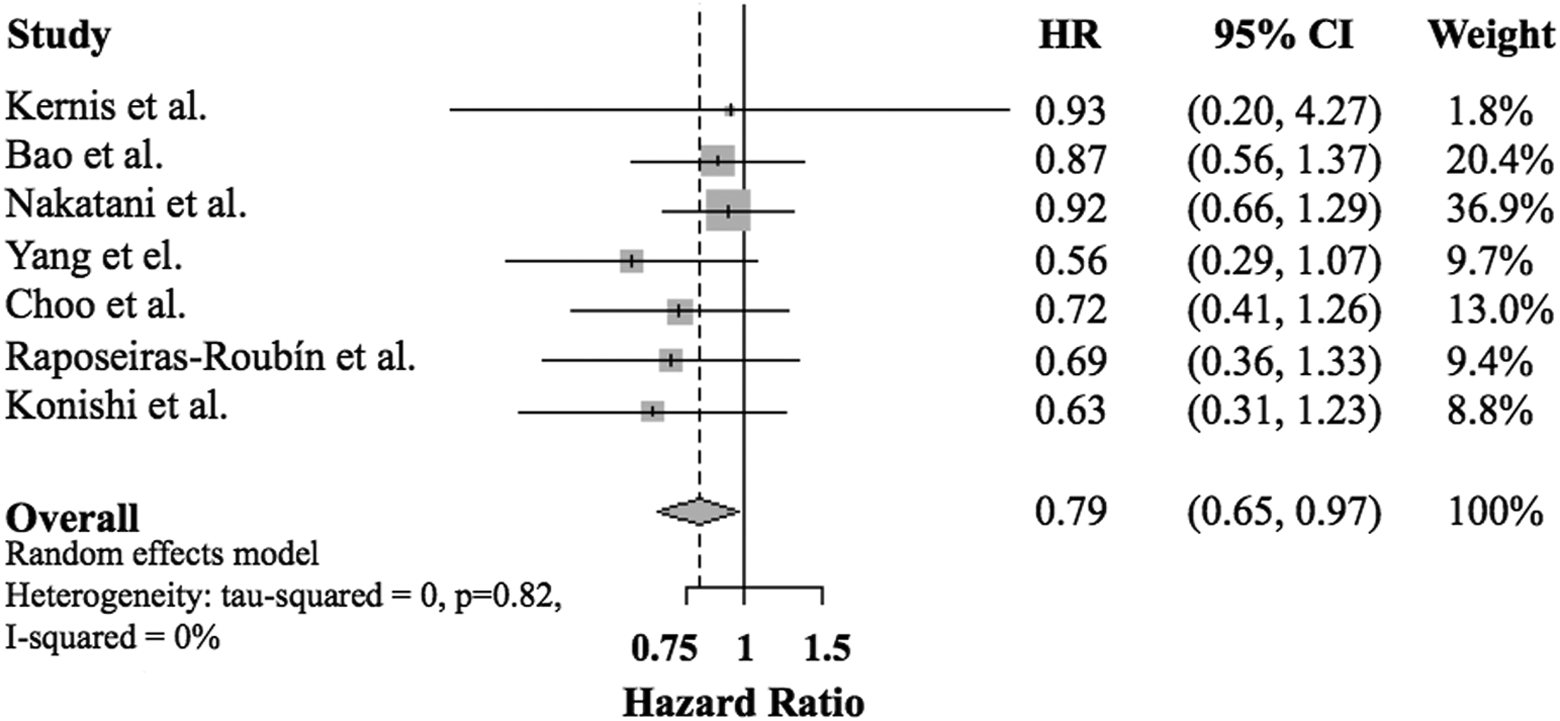
Forest plot reporting the hazard ratio estimate with 95% confidence interval for all-cause mortality in patients with ST-segment elevation myocardial infarction (STEMI) having preserved left ventricular ejection fraction.
Funnel plot is shown in Figure 3. Visual assessment of Funnel plot suggested a possible publication bias. The Begg test showed that P value was .18.

Funnel plot of all studies included in the meta-analysis.
Discussion
In the present meta-analysis of 7 observational studies consisting of 10 857 patients with STEMI undergoing primary PCI who have preserved LVEF, we found that oral beta-blocker was associated with decreased all-cause mortality. To our knowledge, this is the first meta-analysis that has evaluated the association between oral beta-blocker therapy and mortality in this specific population.
Based on the results of randomized trials predating the era of reperfusion, oral beta-blocker therapy is indicated for patients with STEMI according to the current guidelines. 8,9 Observational studies in the era of PCI that assessed the effect of oral beta-blocker on long-term mortality in patients with STEMI demonstrated mortality benefit, but these benefits were mainly observed in high-risk patients such as those with decreased LVEF or those on diuretics. 11 –13 In fact, not a single study included in this meta-analysis showed statistically significant reduction in an adjusted all-cause mortality in patients on oral beta-blocker therapy who have preserved LVEF. However, all of the studies showed a similar trend for decreased mortality in patients on oral beta-blocker therapy. This similar trend for decreased mortality observed in all studies and the lack of heterogeneity among studies suggest that the statistically significant reduction in all-cause mortality observed in this meta-analysis was led by increased number of study population. Although the definition of preserved LVEF ranged from 40% to 50% among studies, subgroup analyses showed similar trends toward reduction in all-cause death in either subgroup. The result of our meta-analysis supports the use of oral beta-blocker therapy in patients with STEMI who are treated with primary PCI and have preserved LVEF, as recommended by the current guidelines.
To date, there has been no randomized controlled trial of oral beta-blocker therapy in patients with STEMI undergoing PCI, likely reflecting the clear recommendation for beta-blocker in patients with STEMI on the current guidelines. The first randomized controlled trial assessing the efficacy of oral beta-blocker therapy (carvedilol) in patients with STEMI who have preserved LVEF (≥40%) after primary PCI is now ongoing (NCT01155635). This study is planning to recruit 1300 patients and is hoping to be completed in 2018.
In our meta-analysis, we evaluated the effect of oral beta-blocker therapy in specific patients with STEMI who underwent primary PCI and who had preserved LVEF. This population partially overlaps with patients having stable coronary artery disease with prior myocardial infarction. Several studies have evaluated the effect of oral beta-blocker therapy in patients with stable coronary artery disease having prior myocardial infarction. Bangalore et al evaluated the effect of oral beta-blocker using propensity score matching in 6758 patients with stable coronary artery disease having prior myocardial infarction from the Reduction of Atherothrombosis for Continued Health (REACH) Registry. 24 They reported that all-cause mortality and cardiovascular mortality were not significantly different in patients with beta-blocker use compared to those without beta-blocker use at a median follow-up of 44 months (hazard ratio for all-cause mortality 0.93, 95% CI 0.80-1.08, P = .34; hazard ratio for cardiovascular mortality 0.91, 95% CI 0.76-1.09, P = .31). Bauters et al also evaluated the effect of oral beta-blocker using propensity score matching in 1678 patients with stable coronary artery disease, of which about a half of the patients had prior myocardial infarction. 25 Their study demonstrated that the use of beta-blocker was associated with a lower risk of cardiovascular mortality at 2-year follow-up (hazard ratio 0.43, 95% CI 0.22-0.82, P = .011). Bauters et al theorized that the disagreement regarding mortality benefit of beta-blocker between their study and REACH registry beta-blocker analysis might be related to the difference in the covariates used to estimate the propensity score, and LVEF was controlled in their study but not in the study by the REACH investigators. 25 Since patients with a low LVEF are also more likely to receive beta-blocker, the lack of adjustment for LVEF might have disadvantaged the beta-blocker group in the analysis by the REACH investigators. A post hoc analysis from the Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance (CHARISMA) trial showed that beta-blocker use in patients with prior myocardial infarction but no heart failure was associated with better clinical outcomes driven by lower risk of recurrent myocardial infarction without statistically significant difference in mortality (hazard ratio for all-cause mortality 0.77, 95% CI 0.52-1.14, P = .197). 26 These lines of evidence suggest a potential clinical benefit of oral beta-blocker therapy in patients with stable coronary artery disease having prior myocardial infarction; however, further studies are warranted to elucidate the role of beta-blocker in patients with stable coronary artery disease having prior myocardial infarction.
There are several limitations in this meta-analysis. First, we used an adjusted hazard ratio (or hazard ratio in the propensity score-matched patients) as a measure of effect, since only observational studies were available to address this clinical question. Multivariate analysis or propensity score matching can control measured confounding variables, but it cannot account for unmeasured confounders. Patients who did not receive beta-blockers were more likely to have had a contraindication to beta-blocker such as hypotension, bradycardia, or decompensated heart failure. Therefore, we cannot exclude the possibility that these potential biases might have inflated the mortality benefit associated with oral beta-blocker therapy observed in this meta-analysis. In addition, several studies used a subgroup analysis of the propensity-matched population, which is another potential cause of bias. Second, beta-blocker therapy was defined as a beta-blocker prescription at the time of discharge, which may not be representative of long-term beta-blocker therapy. The studies included in this meta-analysis only enrolled patients who were discharged alive. Finally, we could not exclude the possibility of existence of publication based on the visual assessment of Funnel plot, although Begg test did not show significant publication bias. Considering the lack of randomized control trial assessing the efficacy of oral beta-blocker therapy in this specific population, this meta-analysis would be the current most comprehensible evidence supporting oral beta-blocker therapy in patients with STEMI who are treated with primary PCI and have preserved LVEF, as recommended by the current guidelines. However, our results should be considered as hypothesis generating because of these study limitations.
Conclusion
This meta-analysis demonstrates that oral beta-blocker is associated with decreased all-cause mortality in patients with STEMI who are treated with primary PCI and who have preserved LVEF. We conclude that it would be prudent to prescribe oral beta-blocker in all patients with STEMI, unless contraindicated, as recommended by the current guidelines, while awaiting the results of a randomized controlled trial.
Footnotes
Acknowledgments
The authors would like to thank Kamyar Arasteh for statistical analysis support and Raposeiras-Roubín Sergio for providing data for this analysis.
Author Contribution
Misumida, N and Kanei, Y contributed to conception or design, acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Harjai, K and Kernis, S contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
