Abstract
Purpose:
We investigated the effects of exposure to electronic cigarettes (E-cig) vapor on the sizes of the no-reflow and myocardial infarction regions, and cardiovascular function compared to exposure to purified air and standard cigarette smoke.
Methods and Results:
Sprague Dawley rats (both male and female, 6 weeks old) were successfully exposed to filtered air (n = 32), E-cig with nicotine (E-cig Nic+, n = 26), E-cig without nicotine (E-cig Nic−, n = 26), or standard cigarette smoke (1R6F reference, n = 31). All rats were exposed to inhalation exposure for 8 weeks, prior to being subjected to 30 minutes of left coronary artery occlusion followed by 3 hours of reperfusion. Exposure to E-cig vapor with or without nicotine or exposure to standard cigarettes did not increase myocardial infarct size or worsen the no-reflow phenomenon. Exposure to E-cig Nic+ reduced the body weight gain, and increased the LV weight normalized to body weight and LV wall thickness and enhanced the collagen deposition within the LV wall. E-cig exposure led to cardiovascular dysfunction, such as reductions in cardiac output, LV positive and negative dp/dt, suggesting a reduction in contractility and relaxation, and increased systemic arterial resistance after coronary artery occlusion and reperfusion in rats compared to air or cigarette exposure.
Conclusions:
E-cig exposure did not increase myocardial infarct size or worsen the no-reflow phenomenon, but induced deleterious changes in LV structure leading to cardiovascular dysfunction and increased systemic arterial resistance after coronary artery occlusion followed by reperfusion.
Introduction
Electronic cigarettes (E-cig) have been proposed as a potentially less harmful alternative to traditional combustion tobacco products; their use has increased rapidly worldwide during the past 10 years. 1 E-cigs are electronic devices that can vaporize a liquid containing solvents, flavorants, and nicotine for user inhalation. They use an electrically powered heating element instead of burning tobacco leaves. E-cig vapor, even without nicotine, can contain chemical products including acrolein, propylene glycol, glycerol, aldehydes, and flavoring which can stimulate reactive oxygen species formation, inflammation, edema, and can damage respiratory tract tissues. The inflammatory agents released into the systemic circulation have the potential to contribute to microvascular damage. Such damage could lead to worsened outcomes following coronary artery occlusion (ie, a heart attack) and reperfusion. Smoking either traditional cigarettes or E-cig has been shown to increase markers of oxidative stress and worsen flow-mediated vasodilation after just a single use. 2 Additionally, in vitro and in vivo animal studies have shown negative effects of E-cig exposure including loss of endothelial barrier function associated with oxidative stress, inflammation, and death. 3,4
The effect of standard cigarette products on myocardial infarct (MI) size is controversial. A limited number of controlled studies have been performed and some results have been contradictory. 5 -7 Difficulties in determining appropriate baseline variables for different MI patients have hindered the power of many clinical studies to establish the effect of cigarette smoke on MI size. 8,9 Key indicators of the seriousness of an MI are the size of the area of the heart with impaired recovery of perfusion (ie, the no-reflow zone) and the area of dead or necrotic tissue. 10 There is a lack of data surrounding the effect of inhaled E-cig vapor on the sizes of the MI and no-reflow zones and on the overall impact on cardiac function. This study aimed to determine the effects of exposure to E-cig vapor (with and without nicotine), compared to the effects of combustion cigarette smoke on the size of the no-reflow zone, myocardial infarct size, cardiovascular hemodynamics, and cardiac function following reestablishment of patency of the large epicardial coronary artery occlusion in the setting of acute myocardial infarction. The rat model utilized in this study mimics a smoker or E-cig user who then develops an acute myocardial infarction.
Methods
All experimental protocols were approved by the Institutional Animal Care and Use Committees at the University of California, Irvine, and the Huntington Medical Research Institutes. Both institutes are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The studies were performed in accordance with the “Guide for the Care and Use of Laboratory Animals” (NIH publication No. 85-23, National Academy Press, Washington DC, revised 2011).
Sprague Dawley rats (both sexes, 6 weeks old) were used in this study. The exposures took place for 5 hours/day, 4 days/week for a total of 8 weeks (to mimic a patient who is a smoker prior to an acute heart attack) at the Air Pollution Health Effects Laboratory at the University of California, Irvine. Animals were exposed using a nose-only inhalation exposure system (In-Tox Products, Clinton MS) to accurately simulate firsthand smoke/vapor exposure (Figure 1). During exposures, animals were held in individual exposure tubes that connected to an exposure manifold. Rats were positioned with just the snout exposed to the exposure atmosphere to minimize dermal exposure. E-cig and combustion cigarette aerosols were produced using a custom-made smoking system which connected directly to the nose-only exposure manifolds. The aerosol entered at the exposure manifold and was delivered to the nose of each animal. A series of exhaust ports in the front of each animal nose cone promoted the flow of new aerosol in the animal’s breathing zone while eliminating exhaled air and minimized the potential for re-breathing with CO2 buildup. Between exposures, the rats were housed 2-4 per cage in an atmosphere-controlled room on a 12-hour light/dark cycle in an Association for the Assessment and Accreditation of Laboratory Animal Care (AAALAC) accredited animal housing facility at the University of California, Irvine vivarium.

Schematic of the E-cig exposure system. E-cig and combustion cigarette aerosols were produced using a custom-made smoking system (Mod-Type E-cig Unit) and pumped to the mixing chamber, in which mixed with purified dilution air and entered directly to the nose-only exposure manifolds. Temperature and relative humidity (% RH) were measured inside the exposure manifold. During exposures, animals were held in individual exposure tubes that connected to an exposure manifold. Rats were positioned with just the snout exposed to the exposure atmosphere to minimize dermal exposure. A series of exhaust ports in the front of each animal nose cone promoted the flow of new aerosol in the animal’s breathing zone while eliminating exhaled air and minimized the potential for re-breathing CO2 buildup.
The rats were randomized to the following exposure groups: 1) Purified air (negative control; n = 16 male/16 female), 2) E-cig vapor with 15 mg/mL nicotine generated by delivering roughly the same amount of nicotine as the standard cigarette (n = 13 male/15 female), 3) E-cig vapor without nicotine (n = 16 male/12 female), and 4) Standard nicotine-containing combustion cigarette smoke (positive control; n = 16 male/16 female) generated from 1R6F Standard Research Cigarettes (Center for Tobacco Reference Products, University of Kentucky). E-cig vapor in groups 2 and 3 was generated from commercially available mod-style vape units with stainless steel atomizers. The E-cig liquid procured for this study had a 50/50 (vol/vol) propylene glycol/vegetable glycerin (PG/VG) matrix to which were added tobacco flavor but did not contain nicotine (www.VaporFi.Com). For group 2, pure nicotine (L-Nicotine, Acros Organics, Lot: A0382410) was added to the PG/VG mixture in the laboratory to ensure consistent control of nicotine concentrations over the exposure period. The nicotine concentration, 15 mg/mL was chosen to match that used in the original NIDA Standard Research E-cigarette (SREC) produced at the time of the experiment. For all studies, the puff frequency was 2 seconds per 30 seconds (2 puffs/minute) at a flow rate of 1.67 liters per minute (equating to a 35 mL puff volume as described in the International Organization for Standardization (ISO) smoking conditions).
Following the 8-week exposure, the rats were transferred to Huntington Medical Research Institutes (Pasadena, CA) and were allowed to acclimate for 2 to 4 days before starting the myocardial ischemia/reperfusion phase of the study. The rats were anesthetized with an intraperitoneal injection of ketamine and xylazine, and mechanically ventilated. The chest was shaved and a baseline echocardiogram (15-MHz transducer and Sonos 5500 ultrasound system, Philips Medical System, Andover, MA) was obtained for measurement of LV end-diastolic and end-systolic internal diameters at the mid-papillary muscle level; LV fractional shortening measurements were also obtained. Arterial blood pressure (BP) and heart rate (HR) were obtained via the ascending aorta using a 2F high-fidelity catheter-tipped micromanometer (model SPR-869, Millar, Inc.). Following BP and heart rate measurements, the catheter-tipped micromanometer was advanced into the left ventricular (LV) cavity to record LV end-systolic pressure (LVESP), LV end-diastolic pressure (LVEDP), the maximal slope of LV systolic pressure increment (dP/dtmax), diastolic pressure decrement (dP/dtmin), and Tau (a measure of diastolic function).
Cardiac output was measured using a thermodilution technique previously described. 11 Briefly, a thermocouple catheter was introduced into the aortic arch via the right carotid artery. The tip of the temperature probe remained positioned in the aortic arch immediately distal to the aortic valve. A catheter (PE-50) was inserted through the right jugular vein and its tip placed into the right atrium for future bolus saline injection. For measurement of cardiac output, 0.2 mL cold saline solution (0°C) was injected into the right atrium through the catheter within the jugular vein and the thermodilution curve was recorded. Cardiac Output was measured using the PowerLab system (LabChart 8.0, PowerLab, ADInstruments). The results of 3 measurements were averaged in each animal. Systemic arterial resistance was measured by dividing mean systemic arterial pressure by cardiac output.
A catheter was placed into the jugular vein allowing delivery of fluids and dyes. A left thoracotomy was performed to expose the heart. The pericardium was opened, and the proximal left coronary artery was encircled with a 4-0 silk suture that was threaded through a flanged plastic tube. The coronary artery was occluded by pulling the ends of the suture through the tube and clamping. Successful occlusion was confirmed by cyanosis and akinesis of the LV anterior wall. Coronary artery occlusion occurred for 30 mins followed by clamp release and reperfusion for 3 hours.
Echocardiogram measurements were taken prior to coronary artery occlusion and again at 2.5 hours into the reperfusion period; hemodynamic measures (Millar catheter pressure readings in the aorta and LV) and cardiac output were also obtained at this time point. During the final minute of reperfusion, a fluorescent dye (Thioflavin S) was injected into the jugular vein. Thioflavin S is a dye that appears fluorescent in areas of tissue receiving reperfusion, when viewed under UV light. When heart slices are viewed under UV light, dark, non-fluorescent perfusion zones represent the anatomic zones of no-reflow (NR). At the end of reperfusion, the proximal coronary artery was briefly re-occluded, and a solution of blue pigment (Super Imperse Blue) was injected into the jugular vein to delineate the ischemic risk zone. The blue pigment circulates only to perfused areas and does not reach the ischemic zone (which appears pink when viewing the heart slices under white light). Finally, intravenous potassium chloride (KCl) was injected to stop the hearts in a relatively diastolic or relaxed state, while the rats were under deep anesthesia.
The rats were weighed. The hearts were excised, washed in saline, and weighed. The left and right ventricles were weighed separately. The LV was then transected into 4 transverse slices from apex to base for histology. The LV slices were imaged using white light to delineate the ischemic risk zone (pink) in contrast to the non-ischemic regions (blue) and subsequently by UV light to delineate the areas of perfusion by thioflavin S (fluorescent areas) and the no-reflow zones (non-fluorescent perfusion defects). The LV slices were then incubated in 1% of triphenyltetrazolium chloride (TTC), a chemical that stains viable myocardial cells brick red, while dead or necrotic cells appear white or pale yellow. The LV slices were re-photographed. The photographs were used for planimetry to measure the percentage of each LV that was at risk, infarcted, and/or contained no-reflow. Planimetered areas were normalized to the weight of each slice. The percentage of each LV mass determined to be at risk (ischemic); that demonstrated no-reflow phenomenon, and that demonstrated necrosis was calculated. MI size was expressed as the percentage of the ischemic risk zone mass that developed necrosis. The NR zone was expressed as the percentage of the LV ischemic risk zone mass.
Heart weight and LV weight were recorded and normalized to body weight. LV wall thickness was measured from histologic sections, to determine if left ventricular hypertrophy had developed. For the determination of the extent of collagen deposition in the LV, the formalin-fixed hearts were embedded in paraffin and 5 µm thickness slides were stained with picrosirius red to estimate the extent of interstitial collagen using Image J. The data were collected from each heart and then averaged by group. The collagen volume fraction was determined as the percentage of picrosirius red positive-stained area relative to total LV area. Using the Hematoxylin and Eosin stained LV wall slides, the LV Wall thickness was measured at 6 points (3 from the nonischemic zone and 3 form the infarcted zone) around the circumference of the LV. Measures of ischemic risk zone, no-reflow, infarct size, hemodynamics, echo, and pathologic/histologic measurements were performed in a manner blinded to the group.
Statistical Analysis
All data are reported as mean ± SEM. Outcome measures were compared among the 4 groups using one-way analysis of variance (ANOVA). The significance threshold was set at P < .05. If the one-way ANOVA yielded a significant group difference, pair-wise comparisons between exposure types were performed using the Holm-Sidak method to control the error rate for multiple comparisons. We observed sex differences in outcome measures empirically, which we confirmed using ANOVAs (see Supplementary Figure); we subsequently conducted exploratory analyses stratified by sex for selected outcome measures. Stratified analyses allow us to evaluate the association between exposure and outcome measures within males and females separately, with the goal of planning future studies designed and powered to investigate sex differences. While the sample sizes in the stratified analyses were small, we verified that the data were normally distributed using Shapiro-Wilk tests and then used ANOVAs to determine exposure group effects within males and females.
Results
A total of 120 adult Sprague Dawley rats were used in this study. Five rats (2 male E-cig Nic+; 2 female E-cig Nic−; 1 female combustion) were excluded from analyses due to the rats dying during the coronary artery occlusion/reperfusion surgical procedure, or due to missed occlusion of the coronary artery.
No significant differences were seen in ischemic risk area (AR/LV, expressed as % of left ventricular mass), infarct size (AN/AR, expressed as % of left ventricular ischemic risk zone), or no-reflow size (A-NR/AR, expressed as % of left ventricular ischemic risk zone) among the 4 exposure groups. The data are demonstrated in detail in Table 1 and Figure 2. Figure 3 shows the representative left ventricular slices to demonstrate the ischemic risk zone, infarct zone, and zone of no-reflow of the hearts.
Ischemia Risk Area, Infarct Size, and No-Reflow Zone by Exposure Group.

Abbreviations: LV, left ventricle; AR, ischemia risk area; AN, area of necrosis; A-NR, area of no-reflow.

The size of ischemic risk area (expressed as % of LV mass, panel A), infarct size (expressed as % of LV ischemic risk area mass, panel B) and no-reflow size (expressed as % of LV ischemic risk area mass, panel C) were comparable among the Air, E-cig Nic+, E-cig Nic−, and Cigarette groups.
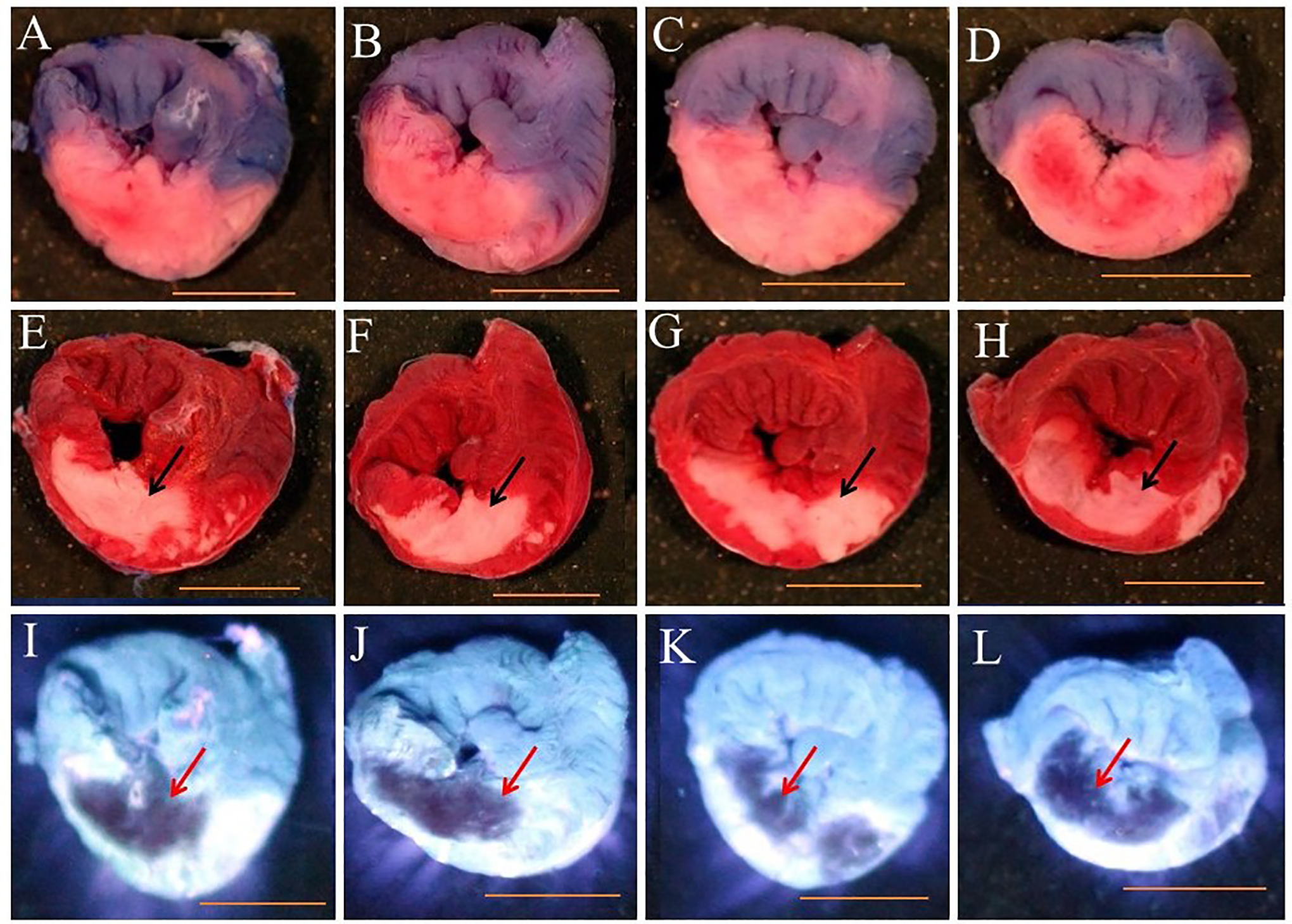
Representative left ventricular slices showing the ischemic risk zone (panels A, B, C, and D), infarct zone (panels E, F, G, and H), and zone of no-reflow (panels I, J, K, and L) of the heart. Heart slices in panels A, B, C, and D show the risk area. Blue represents the region that received blue dye during occlusion and is therefore the nonischemic area. The pink area did not receive blue dye and represents the ischemic risk area. Panels E, F, G, and H show infarct area. Heart slices are shown after incubation in TTC to visualize viable tissue (brick red) versus necrosis or infarct tissue (white area). The infarct areas appear as a homogeneous white area (black arrows). Panels I, J, K, and L show the no-reflow area (non-fluorescent). The heart slices are shown under UV light to the distinguish areas of no-reflow area (non-fluorescent) from the areas receiving flow during reperfusion (fluorescent). Non fluorescent perfusion defect that did not receive thioflavin S is visualized as dark zones (red arrows). Panels A, E, and I are an air exposed heart; panels B, F, and J are an E-cig Nic+ exposed heart; panels C, G, and K are an E-cig Nic− exposed heart; and panels D, H, and L are a cigarette exposed heart (Scale bar = 5 mm).
Body weights were lower in the E-cig Nic+ group (Table 2). Normalized LV weights to body weights were significantly higher in the E-cig Nic+ group compared to those seen in the other 3 groups (Table 2). The LV wall thickness was significantly thicker with more collagen deposition within the LV wall in the E-cig Nic+ group compared to the other 3 groups (Table 2; Figure 4).
Parameters of Left Ventricular Weight, Wall Thickness, and Collagen Deposition.
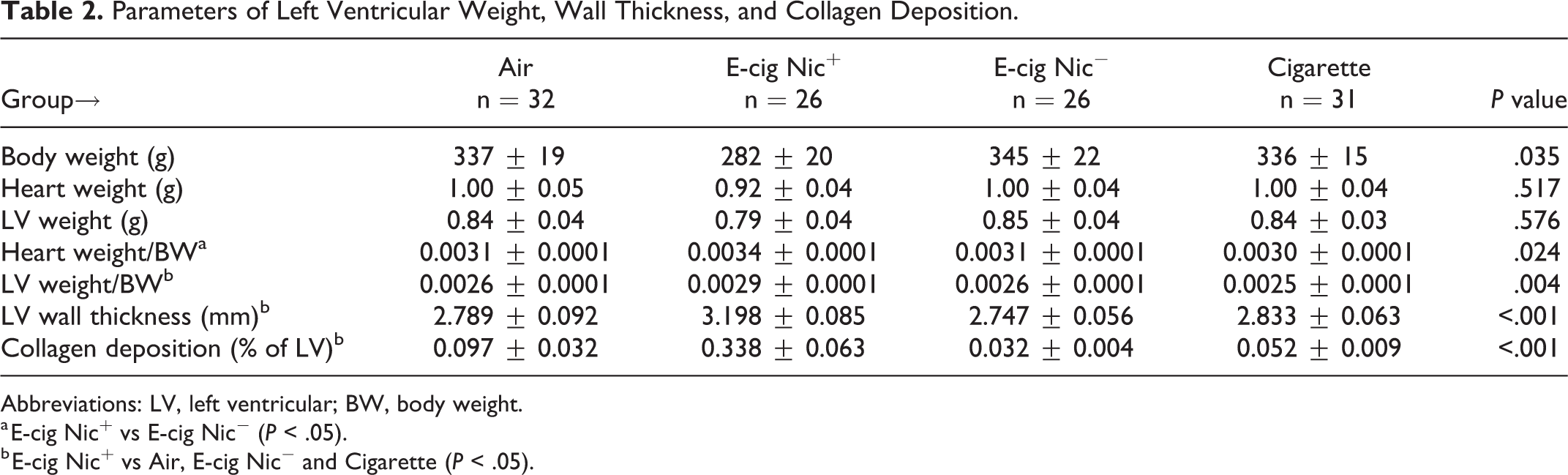
Abbreviations: LV, left ventricular; BW, body weight.
a E-cig Nic+ vs E-cig Nic− (P < .05).
b E-cig Nic+ vs Air, E-cig Nic− and Cigarette (P < .05).
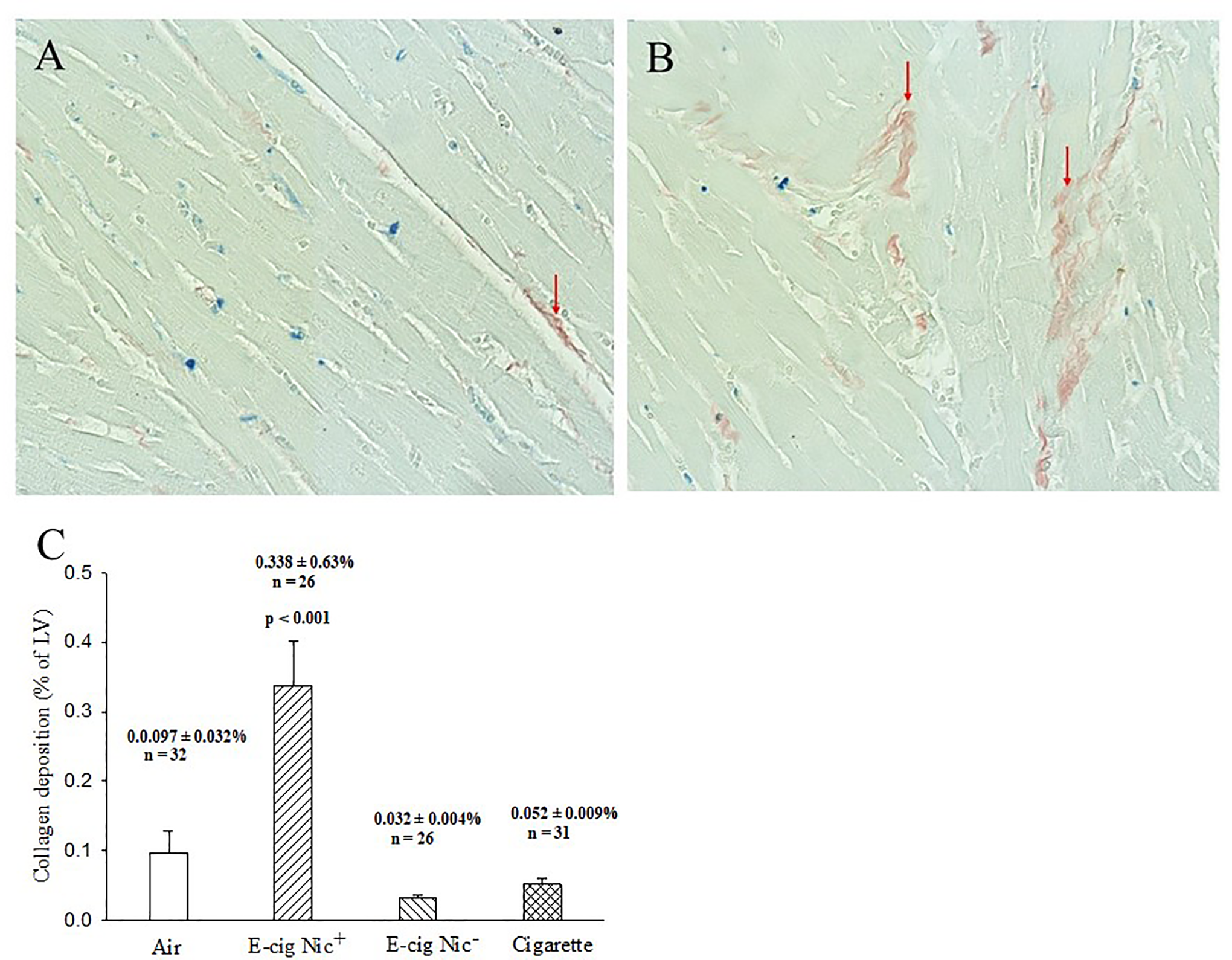
Panels A and B, Representative heart sections stained with picrosirius red stain, which stains collagen in red to demonstrate the collagen deposition in left ventricular wall (red arrows). (A) Exposure to air group (20X); (B) Exposure to E-cig with nicotine group (20X). Panel C, Significantly increased collagen deposition within the LV in the E-cig Nic+ group compared to the other groups (P < .001).
Significantly lower LV end diastolic pressure and negative LV dp/dt were recorded in the E-cig Nic+ group compared to the other 3 groups when measured prior to coronary artery occlusion (Table 3) suggesting that exposure to E-cig Nic+ affected the LV diastolic function. Heart rate, cardiac output, and positive/negative LV dp/dt were found to be lower in the E-cig Nic+ group following the occlusion/reperfusion cycle while systemic arterial resistance was significantly increased in the E-cig+ group (Table 4).
Hemodynamics at Baseline Prior to Coronary Artery Occlusion.

Abbreviations: BPM, beats per minute; LV Pes, left ventricular end-systolic pressure; LV Ped, left ventricular end-diastolic pressure.
a E-cig Nic+ vs E-cig Nic− and Cigarette (P < .05).
b E-cig Nic+ vs Air and Cigarette (P < .05).
Hemodynamics at 2.5 Hours After Coronary Artery Reperfusion.

Abbreviations: BPM, beats per minute; LV Pes, left ventricular end-systolic pressure; LV Ped, left ventricular end-diastolic pressure.
a E-cig Nic+ vs Air and E-cig Nic− (P < .05).
b E-cig Nic+ vs Cigarette (P < .05).
c E-cig Nic+ vs Cigarette (P < .05).
LV diastolic and systolic dimensions along with left ventricular fractional shortening (LVFS) were comparable among the 4 groups based on echocardiogram assessments prior to coronary artery occlusion (Table 5). However, smaller LV diastolic dimensions and higher LVFS were found in the cigarette group compared to E-cig Nic+ following the occlusion/reperfusion cycle (Table 5).
Cardiac Function Prior to Coronary Artery Occlusion and 2.5 Hours After Coronary Reperfusion.
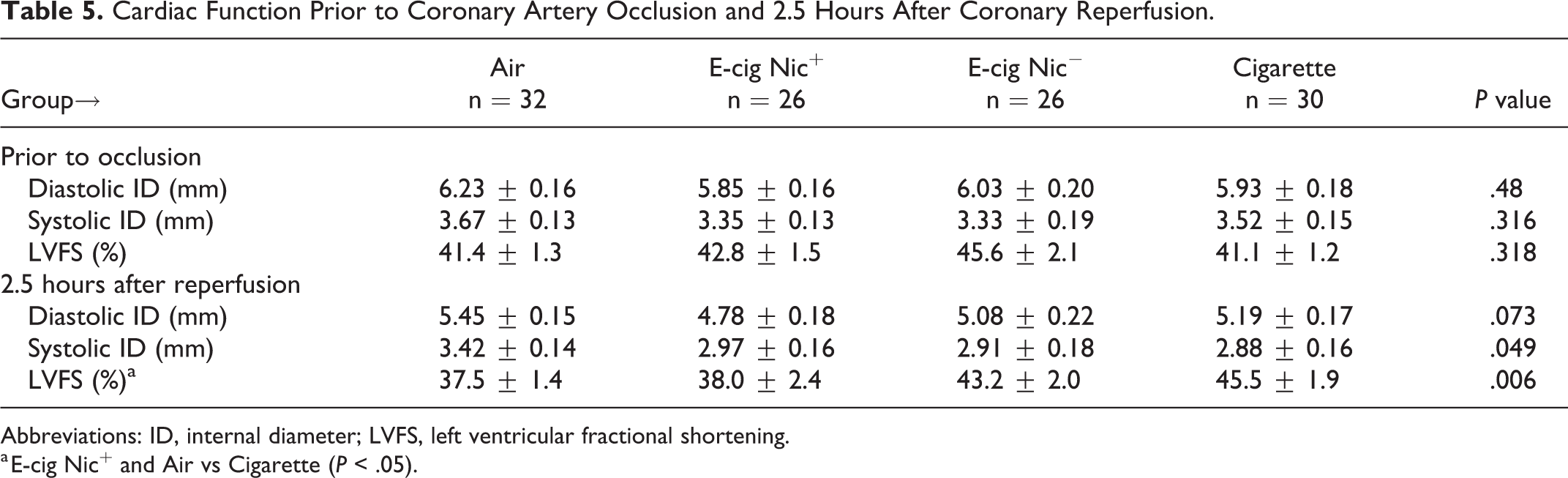
Abbreviations: ID, internal diameter; LVFS, left ventricular fractional shortening.
a E-cig Nic+ and Air vs Cigarette (P < .05).
Exploratory analyses examining selected outcome measures in males and females separately, yielded the following preliminary findings: myocardial infarct size and no-reflow area were smaller in female rats than male rats (Supplementary Figure); female rats exhibited significantly lower body weight, higher LV weight normalized to body weight, thicker LV wall and more collagen deposition within the LV wall in the E-cig Nic+ group compared to the other 3 groups (Supplementary Table); in male rats, there were no significant differences in the above parameters between groups, except for collagen deposition (Supplementary Table).
Discussion
Our study showed that exposure to E-cig, with or without nicotine, and exposure to standard tobacco cigarettes did not increase myocardial infarct size or worsen the no-reflow phenomenon compared to exposure to purified air. Exposure to E-cig with nicotine reduced the body weight gain, and increased the LV weight normalized to body weight. Exposure to E-cig with nicotine also increased LV wall thickness and enhanced the collagen deposition within the LV wall. These heart structure changes were not observed in the other 3 groups. E-cig exposure was associated with other cardiovascular dysfunction, such as reductions in cardiac output, LV positive and negative dp/dt, suggesting a reduction in contractility and relaxation; and an increase in systemic arterial resistance after coronary artery occlusion and reperfusion in the setting of experimental acute myocardial infarction.
Our exploratory analyses examining sex differences indicated that infarct size and no-reflow areas were smaller in female than male hearts. These studies agree with data from other investigators 12 -14 ; although an earlier study from our group examining older rats did not observe smaller infarct sizes in females than males. 15 The difference between our current findings and our previous study may be related to the younger animals and possibly influences of hormones in younger rats. Heart structure changes due to E-cigs were more prominent in females than males. We emphasize that these findings are preliminary and need to be validated in a future study designed to examine sex differences.
Exposure to E-cig may impact the LV mass, LV wall thickness, and fibrosis. Our present study demonstrated that exposure to E-cig with nicotine reduced body weight gain, increased LV mass normalized to body weight and increased LV wall thickness in female rats. Similarly, whole heart mass was found to be increased in mice exposed to nicotine-laced E-cig vapor compared to combustion cigarette-exposed mice following an 8-month exposure 16 which may be the result of the different particle size/mass distributions found in E-cig versus combustion cigarette aerosol. E-cigs have been found to produce elevated levels of ultrafine (<1.0 µm) particles 17 -21 which can easily penetrate into the deep lung and traverse the alveolar-capillary interface and gain direct access to vascular endothelial cells and the bloodstream. Combustion cigarettes generally produce larger particles than E-cigs (∼2.5 µm or larger) and are composed of incomplete combustion products. 2,22 -24 LV wall thickness has also been impacted by E-cig exposure. El-Mahdy et al 25 found no effect on LV wall thickness during both systole and diastole in mice exposed to E-cig (with or without nicotine) or to combustion cigarettes compared to air controls following 16 weeks of exposure; LV mass was also not affected by exposure. However, significantly increased LV wall thickness and LV mass were measured in mice exposed to E-cig+ and combustion cigarette compared to air controls following 32 weeks of exposure. 25 These LV thickness and mass increases seen in the E-cig Nic+ and combustion cigarette group progressively worsened until the study concluded at 60 weeks of exposure. 25 Moreover, 60-week exposure to E-cig (with or without nicotine) or combustion cigarettes significantly increased the heart weight and HW/BW (heart weight to body weight ratios) compared to air controls. 25 Taken together, El-Mahdy et al 25 demonstrated that E-cig and cigarette-induced cardiac hypertrophy with increased LV wall thickness and mass which progressively increased in an exposure time-dependent manner.
Studies investigated the potential effects of E-cigs on cardiac function are still in early stages and have yielded mixed results. Some studies reported that exposure to E-cig aerosol did not affect cardiovascular function, while others suggested negative cardiovascular consequences. For detailed information on the impact of E-cig vaping on the cardiovascular system, please read the excellent reviews covering these subjects. 26 -29 Our results suggest that E-cig exposure leads to cardiovascular dysfunction, such as reductions of cardiac output, and LV positive and negative dp/dt, which may indicate a reduction in contractility and relaxation, and increase in systemic arterial resistance after coronary artery occlusion and reperfusion. In the literature, the effects of E-cig exposure on cardiovascular function remain controversial. 27 Potential cardiovascular and pulmonary benefits are seen when smokers switch to using e-cigarette products from combustion cigarettes 30 which may be an argument for potential use of E-cig as a safer alternative to conventional tobacco products and use as a cessation tool. However, in a mouse model, 16 weeks of exposure to either E-cig or standard cigarette impaired endothelium-dependent and endothelium-independent vasodilation observed in isolated constricted aortic segments. 25 Additionally, systemic hypertension with elevations in systemic vascular resistance in rats exposed to either E-cig or standard cigarette have also been reported and increased exposure time worsened adverse health outcomes. 25 Thus, long-term vaping of E-cig can impair cardiovascular function.
A limitation of the present study is that the cotinine was not measured. Our current e-cigarette nose-only exposure protocol were based on our previously established parameters. 31 On the other hand, the nicotine vapor concentrations in our current study of 15 mg/mL, has previously been shown by Flores et al 32 to induce physiologic changes in rats and increase cotinine levels. In Flores et al's study, rats exposed to 12 and 24 mg/mL nicotine vapor displayed serum cotinine levels of 100 ± 20 and 130 ± 20 ng/mL, respectively, which were comparable to those observed in human daily smokers. 33
Conclusions
Our present study provides evidence that long-term E-cig exposure did not alter the substrate of the heart to increase myocardial infarct size or worsen the no-reflow phenomenon compared to exposure to purified air; but induced deleterious changes to LV structure and cardiovascular function. While E-cig effects are less harmful than those of smoking, they should not be considered a no-risk, harm-reduction alternative to traditional tobacco cigarettes. More investigations are needed to determine the potential time-course effects of varying exposure/usage levels in both males and females. The contribution of specific components of the E-cig, such as carbonyl compounds, nicotine, and flavorings, for mediating these cardiovascular effects remains unknown and needs to be addressed in the future. Studies are also needed to determine the molecular mechanisms and the principal pathways of E-cig exposure-induced cardiovascular impairment.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484231155992 - Effects of Electronic Cigarette Exposure on Myocardial Infarction and No-Reflow, and Cardiac Function in a Rat Model
Supplemental Material, sj-docx-1-cpt-10.1177_10742484231155992 for Effects of Electronic Cigarette Exposure on Myocardial Infarction and No-Reflow, and Cardiac Function in a Rat Model by Wangde Dai, Jianru Shi, Prabha Siddarth, Lifu Zhao, Juan Carreno, Michael T. Kleinman, David A. Herman, Rebecca J. Arechavala, Samantha Renusch, Irene Hasen, Amanda Ting and Robert A. Kloner in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
RAK and MTK made substantial contributions to the conception or design of the work. WD, PS, JS, LZ, JC, MTK, DAH, RJA, SR, IH, AT, and RAK contributed to the acquisition, analysis, or interpretation of data for the work; participated in critically revising the manuscript; approved the final version to be published; and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by University of California office of the president special research programs: Tobacco-Related Disease Research Program (grant number: 28IR-0057).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
