Abstract
Although decreased protein kinase G (PKG) activity was proposed as potential therapeutic target in heart failure with preserved ejection fraction (HFpEF), randomized clinical trials (RCTs) with type-5 phosphodiesterase inhibitors (PDE5i) showed neutral results. Whether specific subgroups of HFpEF patients may benefit from PDE5i remains to be defined. Our aim was to test chronic sildenafil therapy in the young male ZSF1 obese rat model of HFpEF with severe hypertension and metabolic syndrome. Sixteen-week-old ZSF1 obese rats were randomly assigned to receive sildenafil 100 mg·Kg−1·d−1 dissolved in drinking water (ZSF1 Ob SIL, n = 8), or placebo (ZSF1 Ob PL, n = 8). A group of Wistar-Kyoto rats served as control (WKY, n = 8). Four weeks later animals underwent effort tests, glucose metabolism studies, hemodynamic evaluation, and samples were collected for aortic ring preparation, left ventricular (LV) myocardial adenosine triphosphate (ATP) quantification, immunoblotting and histology. ZSF1 Ob PL rats showed systemic hypertension, aortic stiffening, impaired LV relaxation and increased LV stiffness, with preserved ejection fraction and cardiac index. Their endurance capacity was decreased as assessed by maximum workload and peak oxygen consumption (V˙O2) and respiratory quotient were increased, denoting more reliance on anaerobic metabolism. Additionally, ATP levels were decreased. Chronic sildenafil treatment attenuated hypertension and decreased LV stiffness, modestly enhancing effort tolerance with a concomitant increase in peak, ATP levels and VASP phosphorylation. Chronic sildenafil therapy in this model of HFpEF of the young male with extensive and poorly controlled comorbidities has beneficial cardiovascular effects which support RCTs in HFpEF patient subgroups with similar features.
Keywords
Introduction
Population ageing, increased toll of comorbidities such as the metabolic syndrome, diabetes mellitus and hypertension and improved care and outcomes of ischemic heart disease have jointly contributed to a change in heart failure epidemiology, most of heart failure patients now present with preserved ejection fraction (HFpEF). 1 HFpEF is a syndrome of complex pathophysiology and considerable heterogeneity in which the unifying feature seems to be impaired cardiovascular reserve to any form of hemodynamic stress including exercise which heralds effort intolerance and acute lung edema. 2,3 Some authors have proposed chronic pro-inflammatory state, endothelial dysfunction and decreased protein kinase G (PKG) activity as unifying pathways that lead to increased cardiovascular stiffness, the hallmark of HFpEF. 4 Type 5 phosphodiesterase inhibitors (PDE5i) that potentiate cyclic guanosine monophosphate (cGMP) effects and PKG activity were consequently investigated as therapeutic targets. Off-label administration of PDE5i attenuated pulmonary hypertension secondary to HFpEF, 5 chronic and acute experimental therapy in animal models also reduced cardiovascular remodelling 6 and improved diastolic function, 7 respectively. However, the results obtained in a larger heterogeneous population of HFpEF patients were discouraging in the RELAX randomized clinical trial (RCT). 8 From many potential explanations for neutral outcomes, the fact that HFpEF remains a syndrome that encompasses a largely heterogeneous set of patient phenotypes that warrant clustering and tailored therapeutic strategies remains the most consensual hypothesis. Still, steps to patient cluster tailored therapy are yet to be taken. 9,10
Recently, we characterized an experimental model of HFpEF featuring left ventricular (LV) and aortic stiffening with effort intolerance associated with severe metabolic syndrome and systemic arterial hypertension in young ZSF1 obese male rats. 11 -14 This model mimics a subset of younger male HFpEF patients in whom the toll of poorly controlled or previously untreated comorbidities is high. This seems to be a growing group among HFpEF patients. 15 The aim of the present work is to evaluate in this experimental model the effect of chronic PDE5i therapy on exercise capacity, vascular function, LV hemodynamics and myocardial histology, bioenergetics and PKG activity as assessed by phosphorylation of Vasodilator-stimulated phosphoprotein (VASP).
Methods
Animal Model
Nine-week-old male Wistar Kyoto (WKY, n = 8) and ZSF1 obese rats (ZSF1 Ob, n = 16; Charles River, Barcelona, Spain) were kept in individually ventilated chambers and fed ad libitum with recommended diet (LabDiet® 5008, International Product Supplies Ltd.), in a controlled environment with a 12:12 h light-dark cycle at 22°C room temperature. At their 16th week of life, ZSF1 Ob were randomized to receive either sildenafil 100 mg·Kg−1·d−1 (Sildenafil citrate, PF-01000000-10, Pfizer, Inc.) dissolved in the drinking water (ZSF1 Ob SIL, n = 8) or placebo (ZSF1 Ob PL, n = 8). On the 20th week, all animals underwent oral glucose tolerance and insulin resistance testing, metabolic cage studies and peak effort and endurance testing. After 48 h, hemodynamic evaluation was performed and samples were collected for aortic ring preparation, LV myocardial adenosine triphosphate (ATP) level quantification, immunoblotting and histological analysis. Sildenafil administration was interrupted 24 h prior to hemodynamic evaluation and sample collection. All animals received humane care. Experimental procedures complied with the Faculty of Medicine of Porto guidelines, Portuguese law on animal welfare, EU Directive 2010/63/EU for animal experiments and the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH publication no. 85-23, revised 2011).
Peak Effort Test and Endurance Capacity
Effort testing with maximum O2 consumption (V˙O2max) determination was performed on a closed-chamber treadmill with a stimulation grid coupled to a gas analyser (LE8700C and LE405, Panlab®, Harvard Apparatus). The fresh air flow was 700 mL·min−1 and treadmill inclination were 10°. Animals were weighted at the beginning and end of experiments while O2 and CO2 concentrations, velocity, distance and number of shocks were continuously recorded. Tests were interrupted whenever animals became incapable to maintain the rhythm and stayed on the stimulation grid longer than 3 s. Each animal underwent a peak effort test and an endurance test separated by 24 h. After an initial adaption period at 15 cm/s, peak effort testing to assess consisted of changing velocity to 30 cm/s and then stepping up 5 cm/s every 60 s, whereas endurance testing consisted of stepping up velocity by 5 cm/s every 15 min to assess fatigue. Data analysis was performed with Metabolism software (version 2.2.01, Panlab®, Harvard Apparatus).
Hemodynamic Evaluation
Animals were sedated and received analgesia by intraperitoneal injection of midazolam 5 mg·kg−1 and fentanyl 100 µg·kg−1, respectively, and were then anesthetized with sevoflurane (8% for induction and 2.5%-3% for maintenance; Penlon Sigma Delta), orotracheally intubated and mechanically ventilated to achieve normocapnia (TOPO, Kent scientific). Fluid replacement with warm Ringer’s lactate 32 mL·kg− 1·h−1 (NE-1000, New Era Pump Systems) was instituted through a dorsal foot vein, rectal temperature was held at 38°C on a heating pad and electrocardiogram was continuously recorded. Surgical preparation consisted of a left thoracotomy, apical insertion of a pressure-volume catheter in the LV (SPR-847, Millar Instruments) and placement of an ascending aorta flow probe (2.5PS, Transonic) for continuous real-time cardiac output (CO) measurement. Data was continuously acquired (MPVS 300, Millar Instruments), recorded at 2000 Hz (ML880 PowerLab 16/30, AD instruments) and analyzed offline (PVAN 3.5, Millar Instruments). Hemodynamic recordings were made after a stabilization period of 15 min, with ventilation held at end-expiration. Transient inferior vena cava occlusions were obtained to derive end-systolic and end-diastolic pressure-volume relationships. Upon completion of LV function assessment, the catheter was advanced into the ascending aorta as recognized by the pressure tracings and simultaneous acquisitions of aortic pressure and flow were recorded for 6 mins. Volume signal was calibrated for parallel conductance by 40-60 µL 10% saline injection and for slope factor α by simultaneous measurement of CO by the flowmeter. Upon experiment completion, animals were euthanized by exsanguination under anesthesia. Blood samples were collected, organs were excised and weighed, and tibia length was measured. Tissue samples were snap frozen in liquid nitrogen and stored at −80°C. Organ weights were normalized to tibial length, considering the differences observed in body weight between groups. Time constant of isovolumic relaxation was computed by the monoexponential method. Systemic vascular resistance was computed neglecting central venous pressure. Characteristic aortic impedance (Zc) was determined in the frequency domain by means of Fourier analysis of the measured pressure and flow waves (MATLAB R2014b, The MathWorks, Inc.). The average of harmonics 4-10 was employed. Backward and forward pressure and flow waves were determined in the time-domain according to the corresponding Zc. The augmentation index was calculated as the proportion of pulse pressure due to reflected waves after determining the shoulder point. Compliance was determined by monoexponential fitting of diastolic pressure decay from the dicrotic notch onward for known systemic vascular resistances. To account for large differences in body weight between groups, volumes were indexed for body surface area (BSA) as estimated by 9.1*body weight in grams2/3.
Aortic Ring Preparation
Two 1.5 mm rings were isolated from ascending aorta samples (n = 7 per group) and mounted between metal pins in an organ bath system (770MO, Danish Myo Technology). After stabilization, a passive-length tension relationship was obtained by progressive monoaxial stretching of the vascular rings, at intervals of 20%, from slack length (L0) up to 200% of L0. Strain (ε) was defined as percent increase in length from L0. Exponential fitting of the curve enabled derivation of vascular stiffness constant. By dose-response curves we assessed vascular reactivity to phenylephrine (10−9 to 10−5, at 0.5 log unit intervals) and then endothelial function by responsiveness to acetylcholine and direct vasodilator response to nitric oxide (NO) donor sodium nitroprusside (both, 10−9 to 10−4, at 0.5 log unit intervals) with relaxation assessed as percentual decrease of tension after pre-contraction with phenylephrine.
Glucose Metabolism and Metabolic Cage Studies
In successive days, all rats underwent oral glucose and insulin resistance testing upon a 12 h fast. Glycemia was recorded at baseline and 15, 30, 60, 90 and 120 min (Freestyle-Mini) after a 1 g·Kg−1 glucose gavage or a 0.5 U·Kg−1 intraperitoneal insulin injection, respectively. In the following day animals underwent a 24 h acclimatization period in metabolic cages. Water and energy intake and urine output were then recorded for 24 h (Techniplast, Buguggiate).
Myocardial Histology
Sirius red- and hematoxylin-and-eosin-staining was performed in 3 μm-thick-sections of left ventricular myocardium fixated in 4% formaldehyde (n = 7 per group). Images were acquired with a microscope (Olympus XC30 Digital color Camera) and analyzed with the aid of software (Cell^B, Olympus and ImageJ) to determine cardiomyocyte cross-sectional area and myocardial fibrosis as previously described. 11
PhosphoVASP/VASP Immunoblotting
Myocardial phosphorylation of VASP on Ser219 was evaluated as a surrogate of protein kinase G (PKG) activity, as previously described. 16 Briefly, 60 µg of left ventricular myocardium protein (n = 6 per group) were separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) with a 10% polyacrylamide gel and then electroblotted onto nitrocellulose membranes (Bio-Rad). Blots were blocked for 1 h with 5% bovine serum albumin in Tris-buffered saline (TBS; 50 mM Tris-HCl, 150 mM NaCl, pH 7.4) and then transferred to 1% bovine serum albumin in TBS-T (0.1% Tween 20) to which primary antibodies to VASP (Cell Signaling, #3132), phospho-VASP (Ser239, Cell Signaling, #3114) and GAPDH (Abcam, ab8245) were added and incubated overnight at 4°C. Immunoblots were subsequently washed and incubated in 0.5% non-fat dry milk in TBS-Tween 20 with 700 nm and 800 nm infra-red dye-conjugated antibodies (IRDye® 680LT and 800 W, LI-COR) for 60 min at room temperature and protected from light. Membranes were washed and imaged by scanning at 800 and 700 nm with Odyssey Infrared Imaging System (LICOR Biosciences).
Myocardial ATP Levels
LV myocardium (25-60 mg) was homogenized, and proteins precipitated in 10% methanol. An additional volume of ultrapure water with 5% 2-chloro-adenosine (2-CA) was added. The sample was then left on ice for 10 min and vortexed every 2 min to solubilize adenine nucleotides after which it was centrifuged at 16000 g (−5°C) for 20 min (Mikro 200 R, Zentrifugen). The precipitate was stored at −80°C for total protein quantification by Bradford’s method while the supernatant was dehydrated in a liquid nitrogen concentrator for 2 to 4 hours and then rehydrated on ice with 700 µL of mobile phase (100 mM KH2PO4, 1.5 mM TBA, 40% methanol, pH = 6.5 adjusted with KOH). Samples (20 µL) then underwent ultra-high-performance liquid chromatography at 4°C (uHPLC, Flexar FX-10 Ultra High Performance LC 10,000 PSI, PerkinElmer) with a 3 µm particle specially treated octadecyl-coupled silica surface column (SupelcosilTM LC18-T column, 15 cm × 4.6 mm, Sigma-Aldrich). Separation was carried out at 1 mL/min and 2800-4200 psi by reversed-phase ion pairing. Optical density of ATP was recorded at 256 nm. Quantification was performed based on standard curves for ATP and 2-CA as internal control for extraction efficiency (Chromera® software, version 3.2.0, Perkin Elmer). All chemical products were obtained of Sigma-Aldrich (Sintra, Portugal).
Statistical Analysis
Assumption of normality was checked by Shapiro-Wilk’s test and of homogeneity of variances by Levene’s test. When assumptions were met data were analyzed by parametric one-way ANOVA. Otherwise, they were analyzed by non-parametric Kruskal-Wallis test. Post hoc comparisons were performed with Student-Newman-Keuls method. Coefficients derived by regression in pressure-volume hemodynamic analysis and passive length-tension curves of aortic rings were compared between groups with analysis of covariance. Differences were considered significant at two-tailed P < 0.05. Data are mean ± SEM or median (interquartile range) according to distribution.
Results
Hemodynamics and Morphometrics
ZSF1 Ob rats showed systemic arterial hypertension, elevated systemic vascular resistance and elevated afterload which were significantly attenuated by sildenafil as assessed by mean arterial pressure, systemic vascular resistance index and effective arterial elastance, respectively. No differences were observed between groups regarding heart rate, cardiac index, end-diastolic volume or ejection fraction. Systolic function indexes such as maximum rate of pressure rise and the slope of the end-systolic pressure-volume relationship (end-systolic elastance) were increased in both ZSF1 Ob groups but preload recruitable stroke work, which is a geometry and chamber size-independent index of contractility was unchanged as well as ventricular-vascular coupling (Table 1). Untreated ZSF1 Ob showed delayed relaxation, elevated end-diastolic pressure and upward-shifted end-diastolic pressure volume relationship which were attenuated by sildenafil (Representative loops in Figure 1, panel A). Regarding central arterial hemodynamics, ZSF1 Ob PL showed increased aortic impedance and augmentation index along with decreased compliance compared with WKY, which was attenuated by chronic therapy with sildenafil (Figure 1, panels B-D). Representative aortic pressure and flow along with corresponding forward and backward (reflected) wave tracings are presented in panel E of Figure 1.
Hemodynamics.a

Abbreviations: BSA, body surface area; HR, heart rate; MAP, mean arterial pressure; SVRI, systemic vascular resistance for indexed volumes; EDP, end-diastolic pressure; dP/dtmax, maximum rate of pressure rise; τ, time-constant of isovolumic relaxation by the monoexponential method; CI, cardiac index; EF, ejection fraction; EDVi, end-diastolic volume for indexed volumes; Eai, effective arterial elastance for indexed volumes; Eesi, end-systolic elastance for indexed volumes or end-systolic pressure-volume relationship slope; VVC, ventricular-vascular coupling by Ea/Ees; PRSW, preload recruitable stroke work; β i, left ventricular chamber stiffness constant derived from end-diastolic pressure-volume relationships. BSA was estimated as 9.1 (BW in g)2/3.
a Values are mean ± SEM or median (interquartile range), n = 8 per group.
b P < 0.05 vs WKY.
c P < 0.05 vs ZSF1 Ob PL, by one-way ANOVA or Kruskal-Wallis with Student-Newman-Keuls as post hoc comparison method.

In vivo hemodynamic evaluation. Representative pressure-volume loops (panel A) of Wistar-Kyoto control animals (WKY, light gray lines), placebo-treated ZSF1 obese (ZSF1 Ob PL, darker gray lines) and sildenafil-treated ZSF1 obese rats (ZSF1 Ob SIL, black lines); aortic input impedance for indexed flow (Zci, panel B), aortic compliance for indexed volumes (Ci, panel C), and augmentation index (panel D) in WKY (white circles and bars), ZSF1 Ob PL (dark gray squares and bars) and ZSF1 Ob SIL (black triangles and bars); and, finally, representative joint (solid black lines) and decomposed forward (dashed light gray lines) and backward (dashed gray lines) pressure and flow tracings are depicted in panel E. In panel A, transient inferior vena cava occlusions, linear end-systolic pressure-volume relationship and exponential end-diastolic pressure-volume relationship are represented. In the basal loop, effective arterial elastance (Ea) is plotted (dashed lines). In the insert on the lower right, the end-diastolic pressure-volume relationship is shown in detail. In panel E pressure and flow tracings are shown in the upper and lower subpanels, respectively, WKY, ZSF1 Ob PL and ZSF1 Ob SIL tracings are presented from left to right. Values are mean ± SEM or represented as point-plot/box-plot, according to distribution, n = 8 per group. *P < 0.05 vs WKY; † P < 0.05 vs ZSF1 Ob PL by one-way ANOVA or Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls.
ZSF1 Ob rats showed larger body weights, LV weights and lung weights which were unchanged upon chronic sildenafil therapy (Table 2). Although no differences were observed on gross morphometry between sildenafil-treated and placebo-treated ZSF1 Ob rats, on histological analysis cardiomyocyte hypertrophy was significantly reduced by sildenafil (Figure 2, panel A) and only placebo-treated ZSF1 Ob rats showed significantly increased fibrosis area, as assessed by Sirius red staining (Figure 2, panel B).
Morphometry.a

Abbreviations: TL, tibial length; LV, left ventricle.
a Values are mean ± SEM, n = 8 per group.
b P < 0.05 vs WKY by one-way ANOVA.

Hematoxylin-eosin stained (panel A, ×250 magnification) and Sirius-red stained histological sections (panel B, ×100 magnification) of Wistar-Kyoto (WKY), placebo-treated ZSF1 obese (ZSF1 Ob PL), sildenafil-treated ZSF1 obese rat myocardium (ZSF1 Ob SIL) and corresponding statistical analysis, from left to right, respectively. Notice increased cardiomyocyte area (panel A) and Sirius red staining denoting collagen volume fraction (panel B) in ZSF1 Ob PL compared with WKY and the attenuation in ZSF1 Ob SIL. Values are mean ± SEM or represented as point-plot/box-plot, according to distribution, n = 7 per group. *P < 0.05 vs WKY; † P < 0.05 vs ZSF1 Ob PL by one-way ANOVA or Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls.
Vascular Function
ZSF1 Ob presented an upward shifted passive length-tension relationship compared with WKY denoting higher aortic stiffness (Figure 3, panel A), which was attenuated by sildenafil. Developed tension and receptor sensitivity to phenylephrine was increased while endothelium-dependent vasodilation induced by acetylcholine and response to direct vasodilation by NO donor sodium nitroprusside was impaired in both ZSF1 Ob groups (Table 3 and Figure 3, panels B-D).

Passive-length tension relationship (panel A), contraction with phenylephrine (panel B) and subsequent relaxation with acetylcholine (panel C) and sodium nitroprusside (panel D) in Wistar-Kyoto (WKY, white circles, light gray dashed lines), placebo-treated ZSF1 obese (ZSF1 Ob PL, dark gray squares, dark gray dash-dotted lines) and sildenafil-treated ZSF1 obese rats (ZSF1 Ob SIL, black triangles, solid black line). In panel A, vascular stiffness constant derived from exponential fitting is shown as insert in point-plot/ box-plot format. Half-maximum effective dose (ED50) is presented as dotted vertical lines on panels B-D. In panels C and D vasodilatory response is reported as percent relaxation from maximum developed force upon phenylephrine. For detailed results please refer to Table 3. ε, strain; L, length; L0, slack length. Values are mean ± SEM, n = 7 per group. *P < 0.05 vs WKY; † P < 0.05 vs ZSF1 Ob PL by Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls.
Aortic Ring Preparation.a
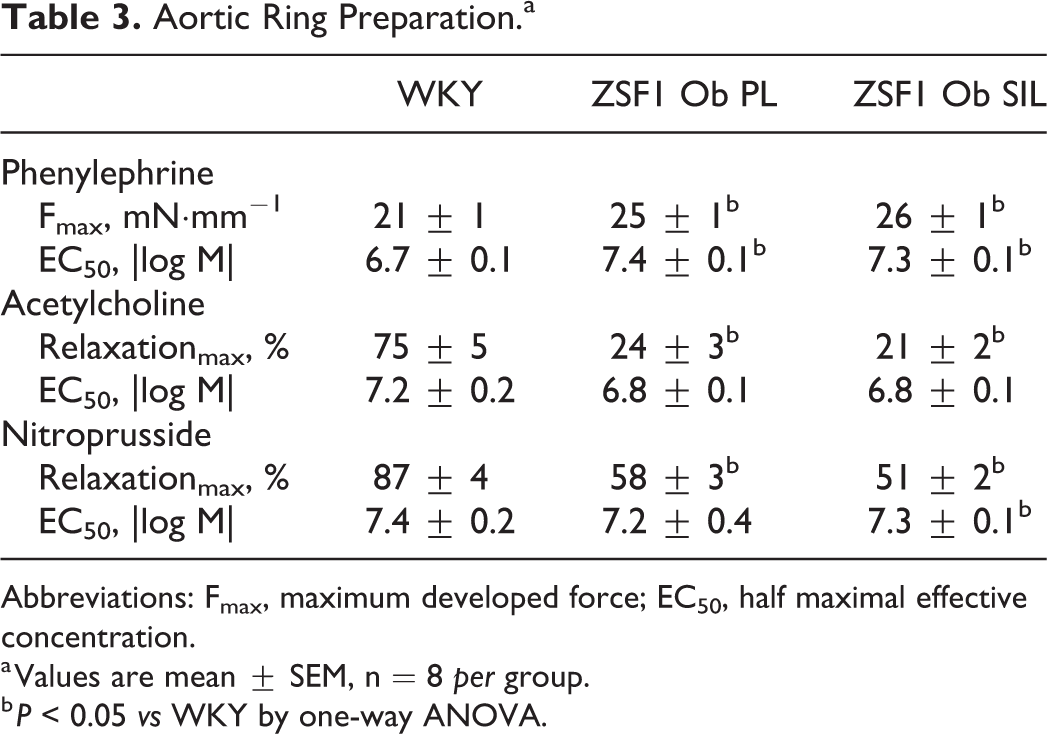
Abbreviations: Fmax, maximum developed force; EC50, half maximal effective concentration.
a Values are mean ± SEM, n = 8 per group.
b P < 0.05 vs WKY by one-way ANOVA.
Effort Tolerance and Myocardial ATP Levels
ZSF1 Ob showed lower V˙O2max and maximum workload compared with WKY without a significant effect of sildenafil in peak effort testing (28 ± 1, 14 ± 1 and 15 ± 1 mL·min−1·Kg−1 for V˙O2max, and 87 ± 7, 46 ± 3 and 48 ± 4 m·Kg−1 for maximum workload, in WKY, ZSF1 Ob PL and ZSF1 Ob SIL, respectively). Contrastingly, in endurance effort testing, sildenafil improved maximum workload and the achieved peak V˙O2 while attenuating the rise in respiratory quotient (RQ) (Figure 4, panels A-C). Myocardial ATP levels were decreased in ZSF1 Ob PL compared with WKY and restored by sildenafil in ZSF1 Ob SIL (Figure 4, panel D). No differences were found in 2-CA concentration and protein quantities between groups (33 ± 5, 45 ± 5 and 36 ± 4 µg·mL−1, for 2-CA, and 1.6 ± 0.3, 2.2 ± 0.3 and 1.9 ± 0.2 mg, for proteins, in WKY, ZSF1 Ob PL and ZSF1 Ob SIL, respectively).

Endurance effort test (panels A-C) and myocardial adenosine triphosphate (ATP) levels (paned D) in Wistar-Kyoto (WKY, white circles and bars), placebo-treated ZSF1 obese (ZSF1 Ob PL, gray squares and bars) and sildenafil-treated ZSF1 obese rats (ZSF1 Ob SIL, black triangles and bars). Peak V˙O2 (panel A), maximum workload (panel B), and maximum respiratory quotient achieved during endurance effort testing before fatigue (panel C) are represented alongside myocardial ATP levels (panel D). *P < 0.05 vs WKY; † P < 0.05 vs ZSF1 Ob PL by one-way ANOVA or Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls, n = 8 per group.
Metabolism
ZSF1 rats showed markedly impaired glucose tolerance and pronounced insulin resistance which were marginally improved by sildenafil therapy, nevertheless insulin resistance was significantly reduced (Figure 5). No significant differences were observed in calorie intake (193 ± 7, 160 ± 8, 180 ± 8 kcal·Kg−1·day−1), water intake (90 ± 5, 85 ± 12 and 102 ± 14 mL·Kg−1·day−1) or diuresis (57 ± 3, 73 ± 11 and 76 ± 11 mL·Kg−1·day−1) among WKY, ZSF1 Ob PL and ZSF1 Ob SIL, respectively.

Oral glucose tolerance (panel A) and insulin resistance tests (panel B) in Wistar-Kyoto (WKY, white circles and bars), placebo-treated ZSF1 obese (ZSF1 Ob PL, gray squares and bars) and sildenafil-treated ZSF1 obese rats (ZSF1 Ob SIL, black triangles and bars). Glycemia time-response and analysis of area under curve (AUC) are presented in the left and right subpanels, respectively. Values are mean ± SEM or represented as point-plot/box-plot, according to distribution, n = 8 per group. *P < 0.05 vs WKY; † P < 0.05 vs ZSF1 Ob PL by one-way ANOVA or Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls.
VASP Phosphorylation
VASP phosphorylation on Ser219 was significantly attenuated only in placebo-treated ZSF1 Ob rats compared with WKY, while sildenafil-treated ZSF1 Ob rats showed intermediate levels of phosphorylation (Figure 6). No inter-group difference in total VASP signal was observed.

Left ventricular myocardial phosphorylation of Vasodilator-stimulated phosphoprotein (VASP) on Ser219 in Wistar-Kyoto (WKY, white circles), placebo-treated ZSF1 obese (ZSF1 Ob PL, gray squares) and sildenafil-treated ZSF1 obese rats (ZSF1 Ob SIL, black triangles). Representative gel scans of phospho-VASP (pVASP), VASP and the loading control glyceraldehyde-3-phosphate dehydrogenase (GAPDH) in 3 samples per group along with pVASP/VASP point-plot/box-plot in relative units (RU) with WKY as reference. Logarithmic scale was employed to enhance visualization of differences given the low levels found in ZSF1 Ob PL. *P < 0.05 vs WKY by Kruskal-Wallis with post hoc comparisons by Student-Newman-Keuls, n = 6 per group.
Discussion
With this work, we describe the beneficial effects of sildenafil on vascular function, hemodynamic status, myocardial histology, effort tolerance, myocardial bioenergetics and PKG activity in the ZSF1 Obese rat model of HFpEF, which mimics a subset of younger male HFpEF patients with poorly controlled comorbidities.
RCTs of sildenafil in HFpEF are contradictory. While Guazzi et al showed positive effects in HFpEF patients with pulmonary hypertension, 5 the RELAX trial enrolling 216 patients with HFpEF diagnosis had disappointing results on exercise capacity, clinical status, quality of life, LV remodeling or diastolic function. 8 Author’s possible explanations are the higher levels of hypertension and LV mass as well as the concomitant presence of pulmonary hypertension in Guazzi’s study. 8 Indeed, recently Guazzi’s positive results were corroborated in an independent sample of HFpEF patients with pulmonary hypertension. 17 Furthermore, in the RELAX trial the effects on exercise capacity were hampered by an abnormally high proportion of patients with chronotropic incompetence and an inferior dose and duration of therapy. More recently, the potential role of mitochondrial dysfunction and endoplasmic reticulum stress caused by sildenafil 18 and pharmacogenomics of liver metabolizing enzymes 19 were pointed out as other possible explanations. Moreover, a preclinical work helps explain the potential causes of failure of PDE5i in estrogen-deficient postmenopausal females. 20 Nevertheless, the lack of therapeutic success in HFpEF has been largely attributed to the fact that this syndrome groups a vast heterogeneity of patient phenotypes that is still far from resolved and therapeutic strategies should be personalized according to an eagerly awaited effective clustering of these patients. 9 Indeed, in more homogeneous patient groups such as severe symptomatic aortic stenosis with preserved ejection fraction and resistant hypertension sildenafil showed favorable hemodynamic effects, such as decreased pulmonary capillary wedge pressure and improved LV stroke volume 21 and reduced blood pressure and peripheral vascular resistance with improved diastolic function, 22 respectively, suggesting that in selected HFpEF cases there may be some clinical benefit. Animal models offer a unique possibility to investigate response to therapy by partly mimicking homogeneous subgroups of HFpEF patients. In Zucker diabetic fatty rats long course preventive therapy with vardenafil, a more potent and selective PDE5i, resulted in decreased cardiac remodeling, increased plasma cGMP levels, enhanced relaxation and decreased LV cardiomyocyte stiffness. 23 Although this model does not involve systemic arterial hypertension and might be questioned as valid HFpEF model since there is yet no documentation of effort intolerance, 3 it constitutes a further preclinical argument to support beneficial effects of PDE5i. 24 Contrarily, the ZSF1 obese rat model has well-documented effort intolerance. 12 It is a young adult genetic male model of severe uncontrolled and untreated systemic arterial hypertension, obesity, metabolic syndrome and diabetes mellitus so it closely mimics a subgroup of younger, middle-aged, HFpEF patients that is increasing in prevalence. 15 Of note, ZSF1 obese rats are in sinus rhythm and lean ZSF1 littermates are also hypertensive, so, we used healthy WKY rats as control group. In the current work a shorter course of sildenafil, only 4 weeks compared with 25 weeks in Mátyás’s work, 23 started after HFpEF is established 12 and not as preventive therapy was able to reduce systemic arterial hypertension, to increase aortic compliance and decrease the impact of reflected waves on LV afterload as assessed by the augmentation index, to ameliorate diastolic LV function, reduce myocardial hypertrophy and interstitial fibrosis, and, finally, to modestly improve peak V˙O2 and maximum workload during endurance effort. Along with functional and structural improvement, corroborating Mátyás’s findings with vardenafil 23 we also found partly restored PKG activity, as appraised by VASP phosphorylation. Regarding dosage and duration of sildenafil therapy in the current study, we must highlight that 4 weeks of therapy in the rat equate to years of therapy in humans and that 100 mg·Kg−1·day−1 is a high dose compared with previous studies carried out in hypertensive rat models. 25 Nevertheless, sildenafil undergoes extensive first-pass hepatic metabolism, 26 the selected dose range is safe and may have incremental effects in endothelial function 27 and natriuresis. 28
Attenuation of systemic arterial hypertension, systemic vascular resistance, aortic stiffness and wave reflection by sildenafil were expected 29 and may partly explain improved effort tolerance 30 while the absence of effect in endothelial dysfunction and response to NO as assessed by vascular response to acetylcholine and nitroprusside, respectively, may be explained by drug withholding 24 h before evaluation and are in accordance with the lack of improvement in endothelial function, assessed with the non-invasive flow-mediated dilation method in brachial artery in hypertensive patients. 22 Both chronically decreased afterload and direct myocardial effects of sildenafil, confirmed by enhanced VASP phosphorylation and thus partly restored PKG activity in the LV myocardium, may explain the overall cardiac effects. Sildenafil therapy modestly improved peak V˙O2 and effort capacity during endurance suggesting these may partly translate into improved functional performance. Nevertheless, the determinants of exercise capacity in HFpEF are complex and seem to be mainly determined by systemic inflammation and skeletal muscle mass 31 which may partly explain the discreet improvement of only around 10% in both peak V˙O2 and workload. Alternatively, a longer course of therapy might have achieved better results. Interestingly, chronic sildenafil therapy also prevented the rise in RQ suggesting enhanced oxidative metabolism of fatty acids and restored myocardial ATP levels. These metabolic and bioenergetic effects may be partly due to direct myocardial effects of sildenafil. Indeed, improved oxidative phosphorylation and mitochondrial function involving multiple subcellular pathways are known effects of sildenafil in ischemia and genetic cardiomyopathy models 32 -34 and preconditioning with sildenafil improved functional recovery of the rat heart after cardioplegic arrest which was abolished by ATP-sensitive mitochondrial K+ channel blockade. 35
Myocardial bioenergetics impairment in HFpEF has been reported with P 31 cardiac magnetic resonance spectroscopy showing decreased phosphor-creatine content with or without concomitant ATP reduction and proposed by Phan et al to be one of the determinants of effort intolerance and pulmonary congestion in HFpEF. 36 In heart failure progression, cardiomyocytes undergo a series of metabolic changes, which limit the ability to adapt to variations in substrate bioavailability and favor glucose usage by non-oxidative pathways instead of fatty acids. 37 In parallel, cardiomyocyte energy stores become depleted. 38 Although this is far from established, multiple mechanisms link bioenergetic depletion to cardiac dysfunction and it is appealing to assume that impaired bioenergetics partly underlie myocardial dysfunction. 39 In this work we demonstrate that myocardial ATP levels are indeed reduced in the ZSF1 obese rat model of HFpEF and improved by sildenafil therapy, through a precise analytical laboratory method. The levels of ATP measured by the uHPLC procedure in healthy control rats conform with previous reports. 40 All care was taken to snap freeze samples in liquid nitrogen as fast as possible upon collection and to carry out all experimental steps at negative temperatures to preserve the stability of adenosine nucleotides. We did not observe differences in the amounts of proteins or 2-CA between groups, which ensures the robustness of the experimental methodology.
As for systemic effects on glucose metabolism, sildenafil significantly decreased insulin resistance, as previously described in high fat-fed mice 41 and patients with prediabetes. 42 More recently this effect was linked to increased levels of cGMP. 43 It is also known the effect of insulin in the glucose uptake by skeletal muscle during exercise, which represents one of the abnormalities associated with exercise intolerance in HFpEF patients. 44,45 Therefore, this slightly augmentation in insulin sensitivity mediated by enhanced cGMP-PKG signaling pathway, assessed by VASP phosphorylation can partly explain the improved exercise capacity in ZSF1 Ob treated with sildenafil.
In conclusion, chronic treatment with sildenafil reduced blood pressure, systemic vascular resistance and aortic stiffness, it improved myocardial diastolic function and compliance, increased myocardial ATP levels and the activity of PKG, as assessed by VASP phosphorylation status, as well as modestly restored endurance effort capacity and peak V˙O2 in the ZSF1 obese rat model of HFpEF. A model which mimics a subset of younger male patients with HFpEF in whom the role of poorly controlled or untreated comorbidities seems to dominate pathophysiology. The clinical translation to younger patients with metabolic syndrome, refractory hypertension, or other similar subgroups of HFpEF patients remains to be established and should be investigated.
Footnotes
Author Contributions
Sara Leite and Liliana Moreira-Costa contributed equally to the work. All authors contributed to the study conception and design. S. Leite and L. Moreira-Costa contributed to acquisition, analysis, interpretation and drafted the manuscript. R. Cerqueira and C. Sousa-Mendes contributed to acquisition, analysis, and interpretation. A. Angélico-Gonçalves, D. Fontoura, and F. Vasques-Nóvoa contributed to acquisition and analysis. A. Leite-Moreira and A. Lourenco contributed to acquisition, analysis, interpretation, and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the European Union (FP7-HEALTH-2010-MEDIA; ERA-CVD/0001/2016-MINOTAUR), grants from Portuguese Foundation for Science and Technology (PTDC/DTP-PIC/4104/2014, NORTE-01-0145-FEDER-000003-DocNET, and SAICT-PAC/0047/2015; 01/12/2016-NETDIAMOND) and by Portuguese Foundation for Science and Technology Grant SFRH/BD/126088/2016 (to D. Fontoura), SFRH/BD/145216/2019 (to L. Moreia-Costa) and SFRH/BD/110404/2015 (to S. Leite).
