Abstract
Background:
Direct-acting oral anticoagulants are indicated for the treatment of nonvalvular atrial fibrillation, but their use in patients after undergoing cardiac surgery is poorly defined despite a high prevalence of postoperative atrial fibrillation in this population.
Methods:
Patients diagnosed with postoperative atrial fibrillation were prospectively randomized to warfarin or apixaban. Safety, efficacy, and economic outcomes were evaluated until their 4- to 6-week postoperative appointment.
Results:
While this pilot study was not powered to determine a difference in safety or efficacy, adverse event rates were similar to the published literature. It was noted that a patient’s course of therapy when utilizing apixaban was significantly less costly than warfarin when including medication, bridging, and laboratory expenses.
Conclusion:
Apixaban and warfarin both appeared to be safe and effective for anticoagulation throughout the duration of this pilot study in treating postoperative atrial fibrillation after coronary artery bypass grafting. Apixaban was associated with significantly less expense when bridging and monitoring costs were included in addition to medication expense.
Keywords
Introduction
Atrial fibrillation (AF) after coronary artery bypass graft surgery (CABG) is common, occurring in approximately 20% of patients. 1 -5 Postoperative atrial fibrillation (POAF) adversely impacts patient outcomes, and patients who develop POAF are at increased risk of stroke, prolonged hospitalization, and other thromboembolic complications. Occurring in the early postoperative period, POAF is usually treated with pharmacologic rate control agents, potentially in combination with rhythm control agents or electrical cardioversion. When POAF is persistent or recurrent, anticoagulation is indicated. Postoperative AF after CABG is self-limiting and usually resolves within weeks of surgery at which time systemic anticoagulation may be discontinued if the patient remains in normal sinus rhythm. 6 Despite the normally limited duration of POAF, it carries similar thromboembolic risk as nonsurgical, nonvalvular AF. 7 -9
When anticoagulation is needed, warfarin therapy has been the mainstay of therapy for patients with POAF after cardiac surgery and is generally less expensive than newer direct-acting oral anticoagulants (DOAC). However, warfarin therapy involves international normalized ratio (INR) monitoring, dose adjustment to maintain therapeutic range, and substantial drug and food interactions. Because of the delayed onset of warfarin’s inhibition of clotting factors VII, IX, X, and II and the inhibition of protein C and S, a period of hypercoagulability exists after initiation of warfarin therapy. Patients with higher risk of thromboembolic events may require bridging with low-molecular-weight or unfractionated heparin after starting warfarin, which is associated with additional bleeding risk and increased cost. Apixaban is a factor Xa antagonist with a well-established safety and efficacy profile for patients with non-valvular AF. 10 While the use of apixaban for the treatment of AF after cardiac surgery has been described, little controlled randomized data exist. 11
The bridging agents and laboratory monitoring including travel expenses contribute to the overall expense of warfarin, but comparisons of the total costs of warfarin and apixaban have not been prospectively evaluated in this patient population. In this prospective, randomized controlled pilot study, we compared the early efficacy, safety, and cost of apixaban and warfarin for patients with AF after isolated CABG.
Methods
This study was conducted in accordance with the amended Declaration of Helsinki. The Sanford Health Institutional Review Board approved the protocol (STUDY00000511), and written informed consent was obtained from all patients. This study was registered with clinicaltrials.gov (registration number: NCT02889562) prior to patient enrollment.
Patients undergoing isolated CABG between 2016 and 2019 who developed new-onset persistent or recurrent POAF during the index hospitalization were eligible for inclusion. New-onset POAF was defined as AF identified by telemetry and confirmed by electrocardiogram within 7 days of surgery, with AF episodes lasting greater than 12 hours or multiple episodes of AF lasting longer than 30 minutes each and observed by telemetry. Patients younger than age 18, those with cognitive impairment, pregnant women, and prisoners were excluded. Also excluded were patients that had a stroke within 7 days, those with increased bleeding risk precluding anticoagulation at the physician’s discretion, patients with prosthetic valves or severe renal insufficiency (serum creatinine of >2.5 mg/dL or creatinine clearance less than 25 mL/min), and patients taking P2Y12 inhibitors during the study period were also excluded (Figure 1). This pilot study was not adequately powered to determine a statistically significant difference in safety or efficacy but was powered to determine a cost difference between study arms. Using a baseline cost estimate of US$378 per month for apixaban, 11 with an estimated current standard deviation of US$20 based on prescription cost variance, a sample size of 28 (56 total) patients per arm would detect a 4% difference between groups with 80% power and an α of .05. Patients meeting selection criteria were randomized to receive apixaban or warfarin in a 1:1 permuted block (block sizes: 6) randomization until the recruitment goal was met. Patients participated in the study until they returned for their second postoperative clinic appointment, scheduled between 4 and 6 weeks from CABG procedure. If the patient was in normal sinus rhythm at their postoperative clinic appointment, anticoagulation was discontinued. Baseline data, Society of Thoracic Surgery risk scores, and operative details were also collected and analyzed.
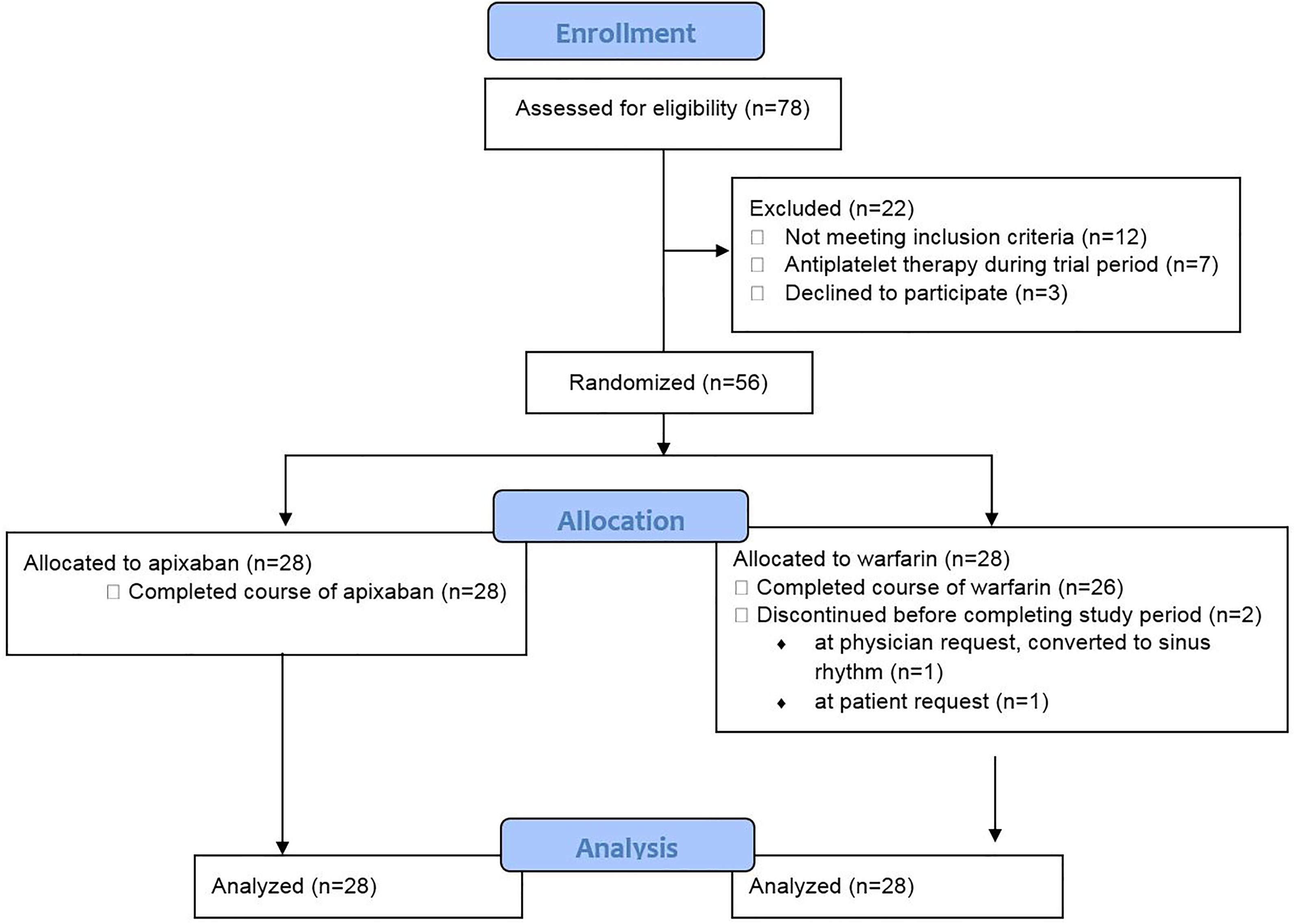
Consort diagram.
The following safety and efficacy outcomes were collected after the study period: clinically relevant bleeding as defined by the International Society for Thrombosis and Haemostasis, 12 thoracentesis and reoperation rates, the numbers of units of blood products given and the sources of any bleeding, stroke diagnosed by neurologist, and thromboembolic events. A pharmacoeconomic analysis was performed with a focus on cost of anticoagulant medications plus anticoagulation monitoring costs. Medication cost estimates were based on the institutional acquisition cost for hospital-supplied medication and wholesale acquisition cost (WAC) for patient therapy as provided by our medication wholesaler and confirmed by the manufacturer Bristol-Myers Squibb. 13,14 We measured cost this way because invoice cost varies widely between pharmacies based on contract agreements with wholesalers or manufacturers as well as other market forces. Direct patient costs would have been affected by their insurance plans, coverage tiers, copays, and deductibles. Other cost measurements such as average wholesale price may have been artificially inflated or associated with reimbursement rates than true cost to the health care system. 15 The number of inpatient and outpatient anticoagulant doses, INR laboratory tests, and outpatient laboratory locations were collected to estimate the direct and indirect costs of testing. Mileage associated with INR monitoring was calculated by determining the round-trip distance from the patient’s address in the electronic medical record to the laboratory utilized by the patient for INR testing. International normalized ratio-associated travel cost was then calculated using the Internal Revenue Service mileage reimbursement rate of US$0.58 USD per mile. 16 For patients bridged with unfractionated or low-molecular-weight heparin, medication cost and number of doses were included in the analysis in the same institutional acquisition cost and WAC pricing structure, similar to warfarin and apixaban. Total cost of medications, INR tests, and travel related to INR testing (when applicable) was calculated for each patient from the time of randomization to the postoperative clinic appointment.
Statistical Analysis
Categorical variables were summarized as frequencies (%) and analyzed with Pearson χ2 tests or Fisher exact tests where appropriate. Continuous data are presented as means ± standard deviation. The t tests and Wilcoxon analyses were used to analyze parametric and nonparametric continuous variables, respectively. Data were analyzed with SAS Enterprise Guide, version 7.1.
Results
Patient Presentation and Clinical Characteristics
Patient demographics and baseline clinical characteristics were similar between treatment groups (Table 1). More patients who were randomized to receive apixaban presented with an acute myocardial infarction (17 [60.7%] patients vs 9 [32.1%] warfarin patients; P = .032). Although not significant, more patients in the apixaban group had a prior stroke/transient ischemic attack (6 [21.4%] patients vs 2 [7.1%] patients; P = .252). There were no statistical differences in Society of Thoracic Surgery calculated mortality, morbidity or mortality, or stroke risk between the groups.
Characteristics of Patients and Past Medical History.a

Abbreviations: AF, atrial fibrillation; kg, kilogram; kg/m2, kilogram per meter squared; STS, Society of Thoracic Surgery.
a Data are mean ± standard deviation or n (%).
Operative Details and Hospital Course
Both on- and off-pump surgical techniques for isolated CABG were used at the discretion of the surgeon with similar rates of each technique between the groups (Table 2). There was no significant difference in the amount of time spent on cardiopulmonary bypass among surgeries performed on-pump, and the number of bypass grafts per patient was not significantly different between study groups (Table 2). Length of stay in the intensive care unit (ICU) was longer for the apixaban group (2.14 ± 1.41 vs 1.43 ±0.50; P = .016) with 3 patients experiencing reversible respiratory failure. There was no significant difference in total postoperative length of stay. The average onset of POAF was 2 days after surgery and not significantly different between the 2 groups (Table 2). Two patients in the warfarin arm were withdrawn from the study before discharge from the hospital—1 at patient request and 1 at the provider’s discretion due to resolution of AF (Figure 1). These patients were included in the analysis in an intent-to-treat manner, except for outpatient outcomes.
Operative Details and Hospital Course.a

Abbreviations: ASA, American Society of Anesthesiologists; CPB, cardiopulmonary bypass; ICU, intensive care unit.
a Data are mean ± standard deviation or n (%).
Clinical Outcomes
While not powered to determine a difference in efficacy and safety outcomes, complication rates between the study groups were similar (Table 3). No in-hospital or 30-day mortality occurred in either group, nor were strokes or thromboembolic events identified. Bleeding complications occurred in both the groups after CABG with similar frequency, and no patients enrolled in the study required reoperation for bleeding or experienced intracranial hemorrhage. The most common adverse events were pleural effusions requiring thoracentesis, which occurred with similar rates (8 [28.6%] in the apixaban arm and 6 [21.4%] in the warfarin arm, P = .645). Four patients received blood transfusions after initiating anticoagulation therapy (Table 3), 3 patients in the apixaban group and 1 patient who received warfarin (P = 1.000). Each of the 4 patients received a total of 2 units of packed red blood cells. No other blood products were administered to any patients during the study period.
Bleeding and Thromboembolic Complications After Randomization and Clinical Outcomes.a
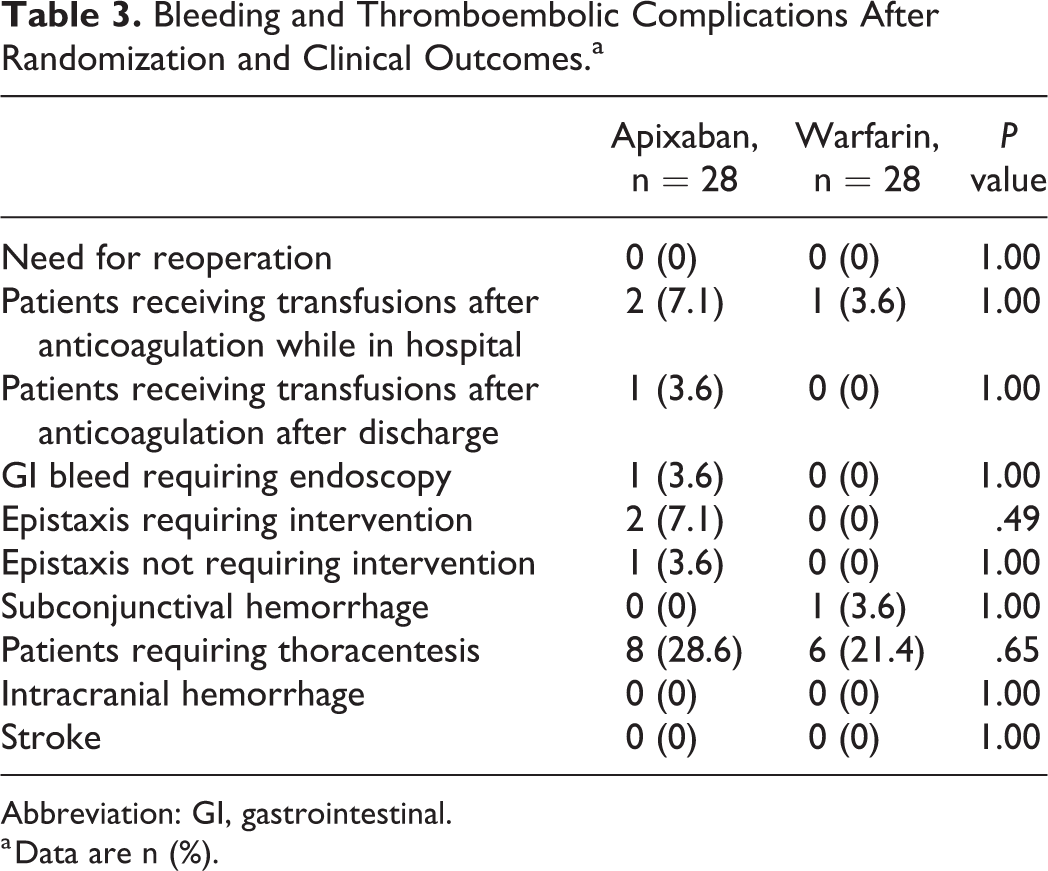
Abbreviation: GI, gastrointestinal.
a Data are n (%).
Three patients experienced epistaxis events in the apixaban group (compared to none in the warfarin group). Two required intervention, with 1 patient receiving expandable foam nasal packing and the other requiring the application of silver nitrate and topical cocaine. One patient developed a gastrointestinal bleed requiring endoscopy while on apixaban (compared to none in the warfarin group). There was one incidence of subconjunctival hemorrhage in the warfarin patient group (compared to none in the apixaban group). Patients in the warfarin group had a time in therapeutic INR range of 56.8% during the study period.
Cost Analysis
Total inpatient anticoagulant institutional acquisition cost was higher for the apixaban group (US$9.80 ± 8.27 vs US$0.75 ± 0.43, P <.001). However, this was outweighed when other costs associated solely with warfarin were included, such as daily INR monitoring (US$160.30 ± 92.82), any bridging that was done with low-molecular-weight heparin (US$25.14 ± 20.00, n=9) or unfractionated intravenous heparin, plus the associated monitoring of activated partial thromboplastin times (US$811.20 ± 258.10, n = 2). The total number of days of inpatient anticoagulant therapy were similar (3.4 ± 3.1 for apixaban vs 2.3 ± 1.8 for warfarin, P = 0.139).
Outpatient medication WAC was also higher for apixaban than warfarin (US$512.70 ± 92.49 vs US$5.62 ± 5.37, P = <.001). However, this too was offset by INR laboratory expense (US$398.20 ± 84.14), travel expenses associated with INR monitoring (US$130.20 ± 159.00), and the costs of bridging with enoxaparin (n = 3, US$148.40 ± 95.83) for patients taking warfarin. Outpatient days of therapy were similar (34.6 ± 6.2 for apixaban vs 36.0 ± 5.4 for warfarin, P = .387). Apixaban required no additional inpatient or outpatient monitoring and was not associated with these ancillary expenses.
To calculate the combined inpatient and outpatient costs associated with anticoagulation for POAF with apixaban or warfarin, the medication, INR laboratory monitoring, round-trip travel to the laboratory utilized by the patient, and any potential bridging were included. Apixaban was associated with a significantly lower total cost versus warfarin (US$522.50 ± 91.40 vs US$778.22 ± 348.80, P = .003).
Discussion
With a similar safety and efficacy profile, apixaban offers an alternative to vitamin K antagonists such as warfarin with less laboratory monitoring. However, studies are limited comparing these in patients that have recently undergone surgeries with a high risk of bleeding complications. In addition, AF is common after cardiac surgeries, such as CABG, occurring in approximately 20% of patients in the early postoperative period. 1 -5 While usually self-limiting, anticoagulation is recommended for patients with recurrent or persistent AF after CABG, as the thromboembolic risks of POAF are similar to that of non-surgical AF. 6 -9
The ICU length of stay was longer for patients randomized to receive apixaban (2.14 versus 1.43 days, P = .016; Table 2). This may have been due to significantly more patients having a history of myocardial infarction in the apixaban group (17 vs 9 patients, P = .032). Three patients on apixaban therapy had reversible respiratory complications prolonging their ICU course, and it is plausible that the postoperative ICU complications could be related to the numeric (but not statistically significant) difference in preoperative chronic lung disease rates (6 [21.4%] patients in the apixaban group vs 2 [7.1%] patients in the warfarin group, P = .252). These respiratory complications were reversible, and the overall hospital length of stay was not different for patients treated with apixaban or warfarin.
Bleeding events occurred in both groups, and while numerically more bleeding events were observed in the apixaban group, it did not reach statistical significance. Larger studies such as ARISTOTLE have shown apixaban to be the superior anticoagulant with regard to bleeding complications compared to warfarin. 10,17 This feasibility study was not powered to assess differences in bleeding events, and larger randomized data are needed in cardiac surgical patients.
Pleural effusions occur commonly after CABG even without anticoagulation, ranging from 42% to 89%, similar to this study. 18 -20 The rate of pleural effusions requiring thoracentesis after CABG is higher in patients who receive anticoagulation (warfarin: 13.2%, DOACs: 26.6%) than those patients who do not receive any anticoagulation postoperatively (1%-10%). 18,20,21 In our study, the incidence of thoracentesis was similar between the groups (apixaban 8 [28.6%] vs warfarin 6 [21.1%], P = .645). In a study in which isolated patients with CABG were given DOACS postoperatively, the prevalence of pleural effusions requiring thoracentesis was similar (26.6%) to that of the rates seen in this study population. 21
The issue of bridging patients until therapeutic oral anticoagulation is achieved is unique to warfarin. The decision to bridge is made by the multidisciplinary team caring for the patient, as bridging therapy carries additional bleeding risk. In this study, 11 (39.3%) patients received bridging therapy after the initiation of warfarin therapy compared to 62% of the warfarin patients in a 2015 retrospective analysis. 11 Three of the patients did not have therapeutic INRs when clinically ready for discharge and were bridged with enoxaparin as an outpatient. Apixaban reaches maximum concentrations 3 to 4 hours after administration; therapeutic effects are observed after initial dosing and are maintained when taken as prescribed. 22 Due to this, the use of apixaban obviates bridging as the time to therapeutic anticoagulation with apixaban is mere hours rather than the 3 to 5 days usually seen in clinical practice with warfarin.
Therapeutic effects of both warfarin and apixaban are dependent on patient adherence, but several additional factors may also affect the safety and efficacy profile of warfarin. Prescribed and over-the-counter medications may interfere with the therapeutic effects of warfarin. Consumption of alcoholic beverages or a diet with inconsistent intake of vitamin K content may also cause sub- or supra-therapeutic INR levels. Patients on warfarin also must have routine INR testing and adjust dosing accordingly to keep their INR within goal range. These factors may contribute to significant time outside the INR goal range of 2 to 3. Our site’s anticoagulation clinic manages approximately 4900 patients with an average time in therapeutic range (TTR) of 69.9% (calculated using the Rosendaal method). This is comparable to an analysis of the US Outcomes Registry for Better Informed Treatment of Atrial Fibrillation which evaluated 5210 patients over an average of 18 months and found an overall mean TTR of 69% among patients followed at anticoagulation clinics. 23 The first months of warfarin therapy will generally yield a TTR lower than this average as the dose/INR response patterns for a patient have not yet been established. As POAF is generally self-limiting, 25 of the 26 warfarin patients were in normal sinus rhythm at the postoperative appointment, an average of 36 ± 5.4 days after surgery, and warfarin was discontinued at that time. Due to this short duration of treatment, it was found that the TTR of the study patients was lower than average at 56.8%. Apixaban does not have dietary interactions and requires no laboratory monitoring, although it is dose adjusted based on renal function, body weight, and age. 22 Due to the rapid onset/offset of apixaban, it can be assumed the time a patient is therapeutic is equivalent to the patient’s compliance with the prescribed twice daily regimen.
Cost analysis of each therapy regimen included acquisition cost of medication as well as ancillary nonpharmaceutical costs such as laboratory cost. Travel expenses were also included in our analysis by measuring round-trip distance from the patient’s address of record to the laboratory. Expenses for INR monitoring contribute significant expense to anticoagulation therapy. Inpatient and outpatient phlebotomy and laboratory expenses over the duration of therapy averaged US$160.30 and US$398.20, respectively. There were also costs incurred to the patient associated with laboratory -associated travel, with patients traveling an average of 224.5 miles during that time traveling to and from the laboratory.
Bridging with low-molecular-weight or unfractionated heparin presents another cost unique to warfarin. Enoxaparin is typically less expensive compared to unfractionated heparin infusions, especially when accounting for additional laboratory monitoring required with heparin (Table 4). Policy at the facility dictates measuring the activated partial thromboplastin time every 6 hours which leads to considerable additional expense compared to apixaban. Unfractionated heparin is still a viable and common bridging agent, however, used at a rate of 36% in nonvalvular AF warfarin therapy. 24
Cost Analysis Per Patient (in US$).

Abbreviations: INR, international normalized ratio; n/a, not applicable to the medication.
a Inpatient medication cost based on institutional acquisition cost.
b Individual medication bridging costs shown are averages per patient bridged with that medication. Total costs include all costs averaged over all warfarin patients.
c Outpatient medication cost based on wholesale acquisition cost.
While medication acquisition cost is important, it does not always represent the total cost burden on a patient or the health care system as a whole. Laboratory monitoring presents a very significant expense in addition to the burden of travel cost traditionally incurred by the patient. When these factors are accounted for, DOACs are often a more cost-effective alternative.
In addition to lower cost, another important variable to consider is patient convenience. Because this is a difficult variable to quantify, it was left out of the analysis but should be acknowledged. Although this study did not collect patient satisfaction data, apixaban does not require INR monitoring, associated travel time, and potentially missed work. This may make apixaban more convenient for patients in an outpatient setting. Alternatively, apixaban is taken twice daily versus once daily for warfarin, which may be noted as a patient inconvenience.
Limitations
There are several limitations to this study. This was a pilot study, and clinical outcome analysis is limited by sample size. The assessment of these outcomes is included for transparency, but large prospective randomized trials comparing apixaban versus warfarin for assessment of must be relied upon for safety and efficacy. However, while nonsurgical data exists, there are no large, prospective, randomized trials of DOACs versus warfarin for patients with POAF to our knowledge.
Second, invoice costs are variable between institutions and pharmacies depending on contracts, which is why this study used the more standardized WAC; while this accurately depicts acquisition cost and cost to the health care system, it does not represent patient payment responsibility.
It should be noted that because the patient population studied is largely rural, data may be skewed toward longer travel distance, and this expense may be less significant for patients in an urban setting, although this may also be offset with public transportation expenses. Some patients chose to get INRs drawn at the facility where they were having follow-up office visits or cardiac rehabilitation therapy rather than a laboratory closer to their residence, which could overestimate travel costs. To account for this, and in an effort to keep the estimates conservative for analysis, if a patient used multiple laboratories, average mileage estimates were made using the laboratory closest to their address of record.
Finally, no data were collected for other non-vitamin K antagonists such as rivaroxaban or dabigatran; thus, no comparisons or inferences can be made for these medicines from this study.
Conclusion
This prospective, randomized pilot study of 56 patients suggests that apixaban use after CABG may be less costly when compared to warfarin when costs of inpatient and outpatient anticoagulation plus associated laboratory testing are considered. While this pilot study was not powered to evaluate the efficacy and safety profiles of these medications, bleeding complications were similar between groups. Postoperative atrial fibrillation in patients after cardiac surgery remains an important and common problem, and larger prospective studies that include all relevant expenses should be done to confirm the results of this study.
Footnotes
Acknowledgments
The authors wish to thank Danelle Staebler, MS and Carlina Grindeland, PharmD, BCPS, BCPPS for their assistance with statistical analysis. (This does not imply their endorsement of data or conclusions).
Author Contribution
Manuscript preparation/composition: T.W.C., A.B.B., A.M.P., M.G.S., Study design: T.W.C., D.D.L., M.L.L., C.M.D., Data collection: T.W.C., A.B.B., A.M.P., M.G.S., Analysis: T.W.C., D.D.L., M.L.L., C.M.D.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Bristol-Myers Squibb and Pfizer Alliance (no role in study design or the collection, analysis, or interpretation of data).
