Abstract
Introduction:
Cholesterol efflux capacity (CEC) is impaired following acute myocardial infarction (AMI). CSL112 is an intravenous preparation of human plasma-derived apoA-I formulated with phosphatidylcholine (PC). CSL112 is intended to improve CEC and thereby prevent early recurrent cardiovascular events following AMI. AEGIS-I (ApoA-I Event Reducing in Ischemic Syndromes I) was a multicenter, randomized, double-blind, placebo-controlled, dose-ranging phase 2b study, designed to evaluate the hepatic and renal safety of CSL112. Here, we report an analysis of a pharmacokinetic (PK) and pharmacodynamic (PD) substudy of AEGIS-I.
Methods:
AMI patients were stratified by renal function and randomized 3:3:2 to 4, weekly, 2-hour infusions of low- and high-dose (2 g and 6 g) CSL112, or placebo. PK/PD assessments included plasma concentrations of apoA-I and PC, and measures of total and ABCA1-dependent CEC, as well as lipids/lipoproteins including high density lipoprotein cholesterol (HDL-C), non-HDL-C, low density lipoprotein cholesterol (LDL-C), ApoB, and triglycerides. Inflammatory and cardio-metabolic biomarkers were also evaluated.
Results:
The substudy included 63 subjects from AEGIS-I. CSL112 infusions resulted in rapid, dose-dependent increases in baseline corrected apoA-I and PC, which peaked at the end of the infusion (Tmax ≈ 2 hours). Similarly, there was a dose-dependent elevation in both total CEC and ABCA1-mediated CEC. Mild renal impairment did not affect the PK or PD of CSL112. CSL112 administration was also associated with an increase in plasma levels of HDL-C but not non-HDL-C, LDL-C, apoB, or triglycerides. No dose-effects on inflammatory or cardio-metabolic biomarkers were observed.
Conclusion:
Among patients with AMI, impaired CEC was rapidly elevated by CSL112 infusions in a dose-dependent fashion, along with an increase in apoA-I plasma concentrations. Findings from the current sub-study of the AEGIS-I support a potential atheroprotective benefit of CSL112 for AMI patients.
Introduction
There is a robust inverse association between serum high-density lipoprotein cholesterol (HDL-C) concentration and the incidence of coronary heart disease (CHD). 1 -5 Following several observational studies that established this inverse association, researchers have sought interventions that increase the level of endogenous HDL-C in order to improve cardiovascular outcomes. Medications that increase serum HDL-C concentration, such as cholesteryl ester transfer protein inhibitors (CETPi) and niacin, have failed to demonstrate an improvement in cardiovascular outcomes. 6 -8 Findings such as these have challenged the hypothesis that raising the levels of endogenous HDL-C would lead to clinical benefit. 9 Dyslipidemia denotes an imbalance between atheroprotective lipoproteins and pro-atherogenic triglyceride-rich lipoproteins such as very low density lipoproteins (VLDL), lipoprotein a Lp(a), and their daughter species as well as small dense low density lipoproteins (LDL). It has been reported that low HDL-C concentrations coexist with inflammation and other proatherogenic conditions to increase predisposition to major adverse cardiovascular events (MACE). Interestingly, a recent observational study in which more than 1 million US male veterans were followed longitudinally, found a U-shaped relationship between serum HDL-C concentration and total mortality with an HDL concentration of 50 mg/dL shown to be associated with the lowest mortality. 10 This relationship however was quantitative and failed to account for qualitative alterations in HDL particles occurring in the population under study. Indeed, the vascular effects of HDL can be highly variable in patients with cardiovascular disease, owing to HDL dysfunction. 11 -13 In patients with diabetes mellitus, coronary disease, chronic kidney disease, metabolic syndrome and other cardiovascular risk factors, the function of HDL is impaired; therefore, static measurements of serum HDL-C fail to account for the disruption in its functional properties. 14 -16 Improved understanding of HDL function has allowed a more nuanced approach to targeting its pathways for cardiovascular risk reduction.
HDL is critical in mediating reverse cholesterol transport. 17,18 Efflux of unesterified cholesterol from an atherosclerotic lesion is the initial critical step of reverse cholesterol transport, and since plaque cholesterol is associated with instability and appears to drive processes associated with plaque instability, this efflux may act as a biological counter-mechanism to plaque instability. 19 -23 Serum cholesterol efflux capacity (CEC) has emerged as a tool for assessment of HDL functionality. Observational data from the Dallas-Heart Study, MESA study, and EPIC-Norfolk study have shown that CEC has an inverse relationship with the incidence of CHD in both healthy individuals and those with atherosclerosis. 24 -27 Retrospective analysis of the EPIC Norfolk and IDEAL studies reveals that concentrations of HDL-C above 70 mg/dl may be associated with an increased risk of MACE; however, a chief protein component of HDL, Apolipoprotein A-I (apoA-I), remained negatively correlated to the occurrence of MACE. 28 Rather than raising the quantity of HDL-C, several studies have highlighted the importance of improving HDL quality, or function, assessed by CEC. 29,30 ApoA-I is critical in facilitating CEC and has thus been investigated for its therapeutic potential in CHD. Further, it has been shown that HDL particles in inflammatory states do not function as anti-oxidative agents. Rather, they support oxidation of cholesterol, augmenting the inflammatory process. Deranged HDL particles carry increased concentrations of serum amyloid A, ceruloplasmin, secretory phospholipase A-2, and a lower concentration of apoA-I. 31
CSL112 is a novel infusible human plasma-derived apoA-I, which has been associated with improved CEC with up to 2.5-fold and 4.3-fold elevations in total and ATP-binding cassette transporter 1 (ABCA1)-mediated CEC observed upon CSL112 administration in the phase 2b AEGIS-I (ApoA-I Event Reducing in Ischemic Syndromes I) trial. 32 -34 The AEGIS-I trial demonstrated that 4 weekly infusions of CSL112 are feasible, well tolerated, and not associated with any significant alterations in hepatic or renal function in 1,258 patients with acute myocardial infarction (AMI). 34 The present study is a detailed pharmacokinetic (PK) and pharmacodynamic (PD) substudy of the AEGIS-I trial, which further investigated the effect of CSL112 on markers of HDL functionality in patients with AMI.
Methods
Study Design
The AEGIS-I study was a phase 2b multicenter, randomized, dose-ranging, placebo-controlled trial of CSL112 (apolipoprotein A-I [human]) in patients with acute myocardial infarction (AMI). 34 The trial assessed safety, PK and PD of placebo, 2 or 6 g CSL112 administered at weekly intervals for 4 weeks in patients with normal renal function [estimated glomerular filtration rate (eGFR) ≥ 90 mL/minute/1.73 m2] or mild renal impairment (RI) [eGFR ≥ 60 and < 90 mL/minute/1.73 m2]. The design of the AEGIS-I study is detailed in the study of Gibson et al. 34
AEGIS-I utilized sparse sampling, which enabled a PK/PD analysis by calculation of summary statistics of PK and PD for a limited number of time points. A PK/PD substudy was undertaken in a subset of subjects participating in AEGIS-I in which frequent sampling was utilized for detailed characterization of the PK/PD profile of CSL112. Subjects participating in the PK/PD substudy gave separate consent to provide additional blood samples. The PK/PD substudy formed part of the overall AEGIS-I trial, which was conducted in accordance with the International Conference on Harmonization Good Clinical Practice guidelines. The study protocol was approved by the Independent Ethics Committees or Institutional Review Boards of the participating centers, as previously reported. 34
Subjects who had experienced an AMI in the previous 7 days were stratified by renal function and assigned randomly in a ratio of 3:3:2 to 2 g CSL112, 6 g CSL112 or placebo, respectively, with equal allocation of patients with normal renal function and mild RI to each treatment group. Frequent sampling was conducted following the first and last (fourth) doses: blood samples were taken before the start of the infusion, at the end of the infusion (2 hours), and 4, 6, 12, 24, 48, 96 hours post-infusion. A further sample at 168 hours was also obtained: for the first infusion this collection time point was the predose sample prior to the second infusion; for the last infusion, an additional sample was taken 168 hours after the final infusion.
Study Drug and Administration
CSL112 is an investigational drug containing human purified apoA-I, formulated with phosphatidylcholine (PC) and stabilized with sucrose. 35 CSL112 is supplied sterile and lyophilized in an evacuated vial, and was reconstituted with 50 mL of water for injection and administered intravenously at doses of 2 g (1 vial) or 6 g (3 vials) over 2 hours. The placebo comparator was 0.9% sterile saline, and was administered intravenously at the same infusion rate as CSL112.
Pharmacokinetic Endpoints
The PK endpoints investigated in this study related to the plasma concentration-time profiles of the main constituents of CSL112: apoA-I and PC. ApoA-I and PC were measured by immunoturbidimetric and enzymatic assays, respectively, as previously described. 36 Validation of these assays for CSL112 has been described previously. 37 Means ± standard deviations (SD) of unadjusted and baseline-corrected data by treatment received (2 g or 6 g CSL112, or placebo) and renal function group were calculated. Baseline correction was performed using a predose sample obtained prior to the first infusion.
To calculate PK parameters for apoA-I and PC, a noncompartmental analysis (NCA) using a combined linear and logarithmic trapezoidal rule (linear up-log down) was performed on unadjusted and baseline-corrected plasma concentration-time data using Model 202 (constant infusion) in WinNonLin® Version 6.3 (Phoenix Build 6.3.0.395). The following plasma PK parameters were calculated for unadjusted and baseline-corrected plasma apoA-I and PC: maximum concentration in plasma (Cmax); time to reach maximum concentration in plasma (Tmax); area under the plasma concentration-time curve from zero to a predefined time point, t (AUC0-t); area under the plasma concentration-time curve from baseline to the last quantifiable point before the analyte first returns to baseline (AUC0-last); terminal half-life (t1/2). PK parameters were calculated as follows: any predose concentration below the limit of quantification (BLQ) and/or BLQ occurring post-dose but before the first quantifiable value were set to zero; any BLQ occurring in between the profile or at the end of the profile was set to missing; if ≥2 BLQs occurred in succession, the profile was deemed to have terminated at the final quantifiable concentration and subsequent values were treated as missing; any not available, no result or no sample was set to missing.
Pharmacodynamic Endpoints
The principal PD endpoints of interest were ex-vivo measures of CEC, including total, ABCA1-dependent and ABCA1-independent CEC. Total and ABCA1-independent CEC were measured after incubation of apoB-depleted serum in vitro with J774 macrophages preloaded with radiolabeled cholesterol with and without cyclic AMP (cAMP) induction, respectively, as previously described. 38 Under standard culture conditions, J774 macrophages do not express ABCA1; ABCA1-independent CEC was assessed under these conditions. Addition of cAMP causes expression of ABCA1, thereby enabling assessment of total CEC. ABCA1-dependent CEC was calculated by subtracting ABCA1-independent CEC from total CEC. CEC is typically expressed as %efflux/4 h reflecting the percentage of cholesterol effluxed from J774 macrophages over the assay incubation period of 4 hours.
Further lipid biomarkers assessed included total cholesterol, HDL cholesterol (HDL-C), non-HDL cholesterol (non-HDL-C; calculated by subtraction of HDL-C from total cholesterol), low-density lipoprotein cholesterol (LDL-C), apolipoprotein B (apoB), and triglycerides. HDL-C was measured in plasma samples depleted in apoB by precipitation with polyethylene glycol. ApoB was measured by an immunoturbidimetric method on a Roche Modular P analyzer (Roche Diagnostics, Indianapolis, IN) using a polyclonal anti-apoB antibody. Triglycerides were measured via the colorimetric quantification of glycerol following hydrolysis of triglycerides by lipoprotein lipase; glycerol blanking was employed to avoid overestimation of triglycerides by endogenous glycerol sources. The other lipid biomarkers were assessed using standard enzymatic methods.
Means ± SD of unadjusted and baseline-corrected, in addition to predose-corrected (last infusion only), lipid biomarker data by treatment received and renal function group were calculated, and time courses of these biomarkers were presented on linear scales. Non-compartmental PD analysis of biomarkers was performed using Model 220 (baseline effect) in WinNonlin™ Enterprise Version 5.2. PD parameters calculated included: time to reach maximum response (TmaxR), maximum response (Rmax) at TmaxR, area under the effect curve from zero to a predefined time point, t (AUEC0-t), whereby t can be 24, 48, 96 and 168 hours post infusion. PD parameters were determined for first and last infusions, respectively.
Inflammatory, Metabolic and Cardiac Biomarkers
Also reported are inflammatory (high sensitivity C-reactive protein [hsCRP], interleukin-6 [IL-6] and fibrinogen) metabolic (hemoglobin-A1c [HbA1c]) and cardiac (troponin I, N-terminal pro-B-type natriuretic peptide [NT-proBNP]) biomarkers from the PK/PD study population. hsCRP was measured using the Roche Tina-quant® hsCRP immunoturbidimetric assay. IL-6 was assessed using the Quantikine® HS IL-6 solid-phase ELISA (R&D systems Minneapolis, MN) and Roche Diagnostics Modular P analyzer. Fibrinogen was measured using the Kamiya Biomedical K-Assay immunoturbidimetric assay (Kamiya Biomedical, Tukwila, WA) and the Roche Diagnostics Modular P analyzer. HbA1c was assessed using the Roche Diagnostics HbA1c immunoturbidimetric inhibition immunoassay and the Roche Modular P analyzer. Troponin I was measured by 2 methods: a conventional high sensitivity assay with a lower limit of quantification (LLOQ) of 6 pg/mL (Siemens TnI-Ultra™ Sandwich ELISA and the ADVIA Centaur® XP instrument), and an ultra-high sensitivity assay with a LLOQ of 0.4 pg/mL (Singulex TnI Sandwich Immunoassay and Erenna® instrument, Singulex Inc, Alameda, CA). NT-proBNP was measured by an electrochemiluminescence (ECL) sandwich immunoassay (Elecsys proBNP II assay; Roche Diagnostics). hsCRP, IL-6, fibrinogen, and Troponin I (Siemens assay) were measured at baseline, at 24 to 48 h after the first infusion and on study days 29 and 112 during the AEGIS-I trial. HbA1c was measured at baseline and at day 112 only; Troponin I (Singulex assay) and NT-proBNP were assessed on days 29 and 112 only.
Statistical Analyses
Due to the number of blood samples required for frequent sampling, the PK/PD substudy was designed to include only a subset of the 1,258 patients randomized in the AEGIS-I study. A minimum number of 48 subjects (including patients who received CSL112 2 g, CSL112 6 g and placebo in a 3:3:2 ratio) was deemed sufficient to assess and characterize the PK and PD of apoA-I and of PC.
Differences in baseline characteristics between patients receiving CSL112 2 g, CSL112 6 g and placebo were assessed using chi-square, ANOVA, and Kruskal-Wallis tests for categorical, parametric continuous, and nonparametric continuous variables, respectively. In the PK and PD analyses, ANOVA and a paired t test were used to assess fold differences in Cmax and AUC between the first and last infusions, calculated using arithmetic mean or median of the within-patient ratios. In the PD analysis, associations between CEC and CSL112 dose/apoA-I exposure were assessed by linear regressions.
Results
Baseline Characteristics
The PK/PD substudy included 63 subjects enrolled in the AEGIS-I trial: 36 patients with normal renal function, 26 with mild (RI) and 1 with moderate RI (eGFR ≥ 30 and < 60 mL/min/1.73 m2). Baseline patient characteristics and lipid, metabolic, and cardiac biomarkers for all patients included in the PK/PD substudy are shown in Table 1 and Table 2, respectively. Statistical comparisons showed no differences in subject characteristics between CSL112 dose and placebo groups; characteristics were also similar to the overall AEGIS-I study population. 34
Baseline Patient Characteristics in the AEGIS-I PK/PD Substudy.a

Abbreviations: BMI, body mass index; RI, renal impairment.
a Data presented as mean (standard deviation) unless otherwise stated.
b P value displayed is an F test of an overall difference between the 3 treatment groups.
Baseline Biomarker Levels in the AEGIS-I PK/PD Substudy.a

Abbreviations: ApoA-I, apolipoprotein A-I; CEC, cholesterol efflux capacity; HDL, high-density lipoprotein cholesterol; hsCRP, high-sensitivity C-reactive protein; IL-6, interleukin-6; LDL-C, low density lipoprotein cholesterol; non-HDL-C, non-high-density lipoprotein cholesterol.
a Data presented as mean (standard deviation) unless otherwise stated.
b P value displayed is an F test of an overall difference between the 3 treatment groups.
c Assessed by Siemens assay.
Pharmacokinetic Endpoints
Following administration of CSL112, rapid, dose-dependent increases in plasma apoA-I were observed that peaked at the end of the infusion (2 hours) followed by a biphasic decline to baseline at approximately 48 hours for the 2 g dose and approximately 168 hours for the 6 g dose. Similar exposures were observed in patients with normal renal function and those with mild RI, and between the first and last infusions (Supplementary Table S1), confirming that no dose-adjustment is required in the case of mild RI. Following the first infusion, baseline corrected AUC0-24 and Cmax were approximately 3- to 4-fold higher with the 6 g dose versus the 2 g dose (Table 3). These parameters were approximately 3-fold higher with the 6 g dose versus the 2 g dose following the last infusion. The median fold ratios presented in Table 3 indicate slightly greater apoA-I exposure (AUC and Cmax) after the last infusion than the first, which suggests a minimal cumulative effect on apoA-I exposure following repeated dosing. Following the first and last infusions of 6 g CSL112, the half-life of apoA-I was 53.9 and 104 hours, respectively.
Baseline-Corrected PK Parameters for Apoa-I and PC Following the First and Last Infusions.a

Abbreviations: ApoA-I, apolipoprotein A-I; AUC, area under the curve; AUC0-last, area under the plasma concentration time curve from time point zero to the last quantifiable timepoint before the analyte first returns to baseline or zero; Cmax, maximum concentration; NA, not applicable (The study was not designed to specifically evaluate the PK of an endogenous analyte); ND, not determined; IQR, interquartile range; PC, phosphatidylcholine; t1/2, half-life; Tmax, time of maximum concentration.
a Values presented as arithmetic mean (SD) unless otherwise stated. IQR values shown in parentheses are 25% and 75% quartiles.
The PK profile of PC following CSL112 infusion was similar to that of apoA-I, but with a shorter half-life (Figure 1B). Rapid, dose-dependent, increases in plasma PC were observed, which peaked at 2 hours, followed by a biphasic decline to baseline at approximately 24 hours. Similar to apoA-I, AUC0-24 and Cmax were approximately 3-fold higher with the 6 g dose versus the 2 g dose following both the first and last infusions. There was no apparent PC accumulation following repeated dosing, although there was greater variation in PC measurements (Table 3). Further, the half-life of PC was noticeably shorter than that of apoA-I: 9.73 ± 7.20 versus 53.9 ± 38.7 hours, respectively (following the first infusion of CSL112 6 g). Similar PK results were observed between normal renal function and mild RI patients, although half-life estimates increased with dose and were slightly higher in patients with mild RI (Supplementary Table S2). Unadjusted PK parameters for apoA-I and PC following the first and last infusions are reported in Supplementary Table S3.

Changes in apolipoprotein A-I and phosphatidylcholine following CSL112 infusion. Presented are mean ± standard deviation of baseline-corrected plasma concentration-time profiles for apolipoprotein A-I (apoA-I) (A) and phosphatidylcholine (PC) (B). Total study population: 63 (placebo [n = 18], 2 g CSL112 [n = 24], 6 g CSL112 [n = 21]). Normal RI: 36 patients; Mild RI: 26; Moderate RI: 1.
Pharmacodynamic Endpoints
Cholesterol efflux capacity is suppressed following an AMI
Prior work has shown that measures of cholesterol efflux are suppressed in the days immediately after MI and then rise in the subsequent month. 39,40 Measurements of placebo-treated patients in the current study have confirmed and extended the latter finding. Because of the baseline variability and the small number of subjects in that group (n = 17) CEC AUEC0-72 was compared rather than predose CEC AUEC0-72 for ABCA1-dependent CEC rose 1.6-fold (P = .009) between the first and last infusion times in the placebo arm (Table 4). ABCA1-independent CEC and total CEC also rose, but to a lesser extent. In contrast, apoA-I concentration was unchanged between the first and last infusion times, suggesting that the functionality rather than the amount of apoA-I was altered during the weeks after MI.
Unadjusted Apoa-I and CEC Exposures Following the First and Last Infusions in Placebo Patients Only.a

Abbreviations: ApoA-I, apolipoprotein A-I; AUC, area under the curve; AUEC, area under the effect curve; CEC, cholesterol efflux capacity; SD, standard deviation.
aBolded values are <.05 (statistically significant).
CSL112 rapidly and strongly increases cholesterol efflux capacity in AMI patients
Infusion of CSL112 resulted in immediate, strong, dose-dependent increases in total, ABCA1-dependent and ABCA1-independent CEC (Figure 2). CEC profiles were similar between the first and last infusions, although there were small increases (approximately 11% of baseline) in predose CEC over baseline levels at the last infusion with both doses of CSL112 (data not shown). Following administration of the 2 g and 6 g CSL112 doses at 2 hours, there were approximately 2-fold and 3-fold increases relative to baseline in total CEC and 3-fold and 6-fold increases in ABCA1-dependent CEC relative to baseline; increases in CEC were similar between patients with normal renal function and those with mild RI (Table 5). Unadjusted ApoA-I and CEC concentrations showed comparable elevations between the first and last infusions (Supplementary Table S4).

Elevations in cholesterol efflux capacity following CSL112 infusion. Presented are mean + standard deviation baseline- (first infusion) and predose-corrected (last infusion) total (A), ABCA1-dependent (B), and ABCA1-independent (C) cholesterol efflux capacity. Total study population: 63 (placebo [n = 18], 2 g CSL112 group [n = 24], 6 g CSL112 [n = 21]). Normal RI: 36 patients; Mild RI: 26; Moderate RI: 1.
Baseline-Corrected PD Parameters for CEC by CSL112 Dose and Renal Function After the First Infusion.a

Abbreviations: AUEC, area under the effect curve; CEC, cholesterol efflux capacity; Rmax, maximum response.
a Shown are means (standard deviation).
b Immediately after the end of infusion.
c Due to high variability in ABCA1-dependent cholesterol efflux (including zero at baseline), the summary statistic for this parameter is the median.
TmaxR for ABCA1-dependent and ABCA1-independent CEC was at approximately 2 hours (end of infusion) for both CSL112 doses. Both ABCA1-dependent and ABCA1-independent CEC returned to baseline after approximately 48 hours following the 6 g dose. However, following infusion of the 2 g dose, ABCA1-dependent CEC returned to baseline at approximately 12 hours, whereas ABCA1-independent CEC returned to baseline at approximately 24 hours, supporting the dose-response relationship associated with CSL112 infusion.
CSL112 dose strongly correlates with increase in total and ABCA1-independent cholesterol efflux capacity (Figure 3). Rmax for CEC was dose-proportional for ABCA1-independent efflux but saturated for total and ABCA1-dependent efflux. This saturation was due to assay saturation observed in some samples in the 2 g and 6 g CSL112 dose groups collected at 2 hours. The saturation was determined if the result exceeded 75% CEC of a saturating lipid-free apoA-I control. This indicates very high lipid-free/poor apoA-I concentrations in these samples resulting in saturation of the activity of the ABCA1 transporter in the assay. Furthermore, the effect of CSL112 dose on CEC appeared to be independent of renal function (Figure 3). There were positive linear relationships between apoA-I exposure following infusion of CSL112 and total, ABCA1-dependent, and ABCA1-independent CEC (Figure 4). These relationships were similar following the first and last infusions.

Dose-proportionate changes in total and ABCA1-independent cholesterol efflux capacity after CSL112 infusion. Presented are the relationships between CSL112 dose and baseline-corrected maximum response (Rmax) and baseline-corrected area under the effect curve in the first 24 hours (AUEC0-24) for cholesterol efflux capacity (CEC), total (A) and ABCA1-independent (B), following the first infusion. Data points are shown by renal function. Green circle represents mean. A non-linear profile may be a result of the dose reaching saturation level. Total study population: 63 (placebo [n = 18], 2 g CSL112 group [n = 24], 6 g CSL112 [n = 21]). Normal RI: 36 patients; Mild RI: 26; Moderate RI: 1.

Correlation between apolipoprotein A-I and CEC exposure following infusion of CSL112. Presented are the linear relationships between predose-corrected apolipoprotein A-I (apoA-I) exposure, area under the curve in the first 24 hours post infusion (AUC0-24), and predose-corrected cholesterol efflux capacity (CEC), area under the effect curve in the first 24 hours (AUEC0-24): total (A), ABCA1-dependent (B), and ABCA1-independent CEC (C), following the first and last infusions of CSL112.
CSL112 increases HDL-C levels
Following CSL112 administration, there were rapid, dose-dependent increases in plasma HDL-C levels, with a broad peak at 2 to 6 hours, followed by a gradual decline (Figure 5A). HDL-C levels returned to baseline after approximately 48 hours following 2 g CSL112 infusion; in comparison, levels remained elevated at 96 hours following 6 g CSL112 infusion. Following the first infusion, the maximum increase in HDL-C from baseline was observed to be 4.6 ± 3.7 mg/dL (at 6 hours) with the 2 g CSL112 dose, and 13.4 ± 5.1 mg/dL (at 4 hours) with the 6 g CSL112 dose; similar results were observed following the last infusion. Alongside this, there were small dose-dependent increases over placebo in total cholesterol following CSL112 infusion, but these declined to/past baseline levels within 24 hours (Figure 5B). The change in total cholesterol levels in relation to that of HDL-C was explained by the decrease in non-HDL-C following the first infusion (Supplementary Figure S1A).
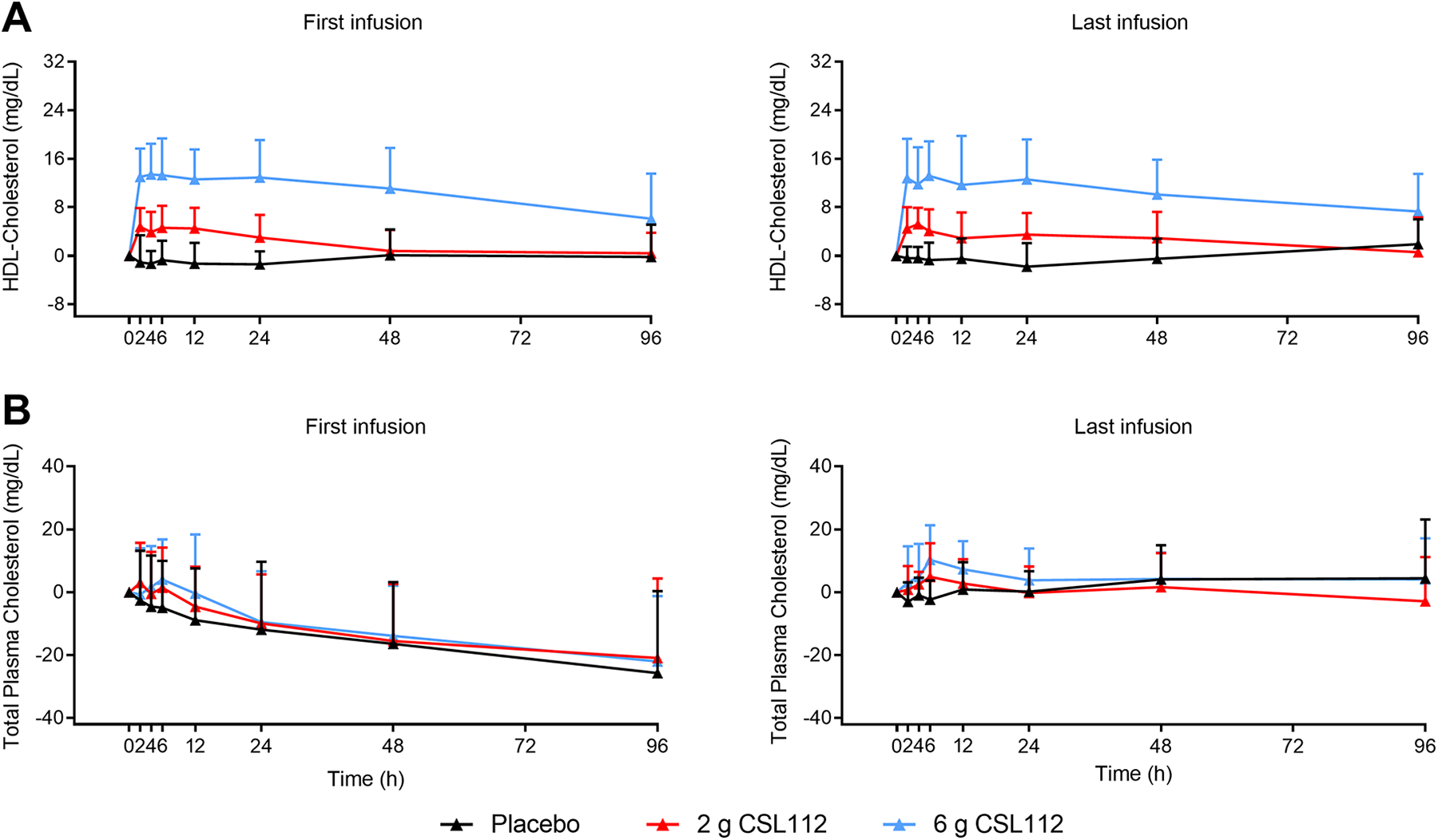
Changes in HDL and total cholesterol following infusion of CSL112. Presented are mean + standard deviation pre-dose-corrected plasma concentration-time profiles for HDL cholesterol (A) and total cholesterol (B) following the first and last infusions.
CSL112 does not increase atherogenic lipids/lipoproteins
There were no increases in the atherogenic lipids/lipoproteins non-HDL-C, LDL-C, and apoB following infusion of CSL112. Consistent with the main study results (data not shown), small dose-dependent decreases in non-HDL-C, LDL-C and apoB were observed following the first infusion of CSL112—this effect was less evident following the last infusion (Supplementary Figure S1). There were small transient increases in triglycerides peaking at 12 hours after both first and last infusions (Supplementary Figure S1).
Inflammatory, Cardiac and Metabolic Biomarkers
Levels of inflammatory biomarkers (hsCRP, IL-6, and fibrinogen) for all treatment groups declined from baseline through the course of the trial, suggesting that they were elevated post-AMI, which is to be expected (Supplementary Figure S2A-C). There was no discernable change in HbA1c between baseline and the end of the study (Supplementary Figure S2D). Troponin-I declined through the course of the study, which again is to be expected in post-AMI patients (Supplementary Figure S2E). There was no numerical post-infusion increase in NT-proBNP in any treatment group (Supplementary Figure S3). Overall, there was significant variability in the inflammatory, cardiovascular and metabolic biomarker data, preventing assessment of a dose-effect of CSL112 on these biomarkers.
Discussion
In this study we demonstrate the detailed PK and PD profile of CSL112 following 4 weekly intravenous infusions in the target population for CSL112, i.e., patients who have had an AMI. When administered over a period of 4 weeks, both the 2 and 6 g infusions of CSL112 were associated with a rapid increase in plasma apoA-I concentration, which peaked (Tmax) at 2 hours for both doses, followed by a biphasic decline. Increases in plasma apoA-I were dose-dependent, with achieved Cmax and AUC0-24 observed to be 3- to 4-fold higher following administration of the CSL112 6 g versus the 2 g dose. The increases achieved with the 6 g dose were sustained above baseline for approximately 7 days before returning to close to predose levels; there was evidence of little accumulation. The PK findings thus confirmed the rationale for the weekly dosing interval, which balances avoidance of accumulation with practical considerations in an AMI population. The peak in plasma apoA-I concentration was accompanied by a concomitant peak in plasma level of PC, the stabilizing phospholipid component of CSL112; however, plasma PC was cleared more rapidly and reached close to baseline levels within 24 hours.
In terms of the PD findings, patients in the treatment groups showed dose-dependent elevations in total- and ABCA1-dependent CEC, which correlated closely with elevations in apoA-I. Two- and 3-fold elevations in total CEC immediately (t = 2 hours) following infusion of CSL112 2 g and 6 g were observed, respectively, but CSL112 was shown to predominately promote CEC via the ABCA1 transporter, with 3- and 6-fold elevations observed in Rmax in relation to the 2 and 6 g doses, respectively (Table 5). Importantly, elevations in both apoA-I concentration and CEC were consistent following repeated administration. These increases were substantially higher than those observed in the placebo group, which showed a small gradual increase in CEC (Figure 2 and Table 4) through the study period, suggesting that CEC is suppressed post-AMI. Only a few studies have sequentially assessed CEC post-AMI. This prior work has suggested that MI causes an acute fall in CEC, a hallmark of HDL dysfunctionality, and that it is gradually restored in the following month. 27,39,41 Subjects in the placebo cohort (n = 110) in the dal-ACUTE trial showed a total CEC elevation of 7.2% four weeks following acute coronary syndrome. 40 CEC measurements of placebo-treated patients in the current study support the latter finding by a gradual restoration of CEC at 1 month. The biological basis of CEC depression post-MI is not presently clear, but recent work has suggested 2 mechanisms which may each contribute to loss of HDL function. Firstly, oxidative modification of HDL, particularly via leukocyte-derived myeloperoxidase (MPO) has been shown to reduce CEC. 42 -44 Other work has demonstrated that serum amyloid A (SAA), an acute phase reactant elevated after MI, may also impair HDL function. 45 Our study supports the concept of the pathological alteration of apoA-I, as the increase in CEC in placebo-treated patients between the first and last infusions was accompanied by minimal change in apoA-I concentration. This suggests that the functionality, rather than the amount of apoA-I, was altered during the weeks after MI. Although these findings warrant further study of the length and magnitude of CEC suppression following AMI, these observations also support a potential benefit of substantially elevating CEC via the ABCA1 transporter, which is preferentially expressed in cholesterol-laden macrophages such as those found in atherosclerotic plaque. 46
The observed PK and PD profile of CSL112 was in line with previous observations. 33,36,37,47 The biphasic kinetics observed upon CSL112 administration are in agreement with earlier studies using radiolabeled apoA-I, which explains the drug kinetics using a 2-compartment model consisting of central and peripheral (delayed distribution in peripheral tissues) compartments. 48 -50 In addition, our findings extend the previous knowledge concerning the effect of CSL112 on ABCA1-mediated CEC among healthy individuals, subjects with moderate RI, and patients with stable atherosclerotic disease. 33,47 It has previously been shown that CSL112 can be safely administered to individuals with mild to moderate RI, and is associated with favorable PK and PD in these individuals. 36,51,52 This observation is important because CSL112 contains a small amount of sucrose as a stabilizer, and high doses of sucrose have been associated with sucrose nephropathy. The present study extends the latter observations, supporting the potential of CSL112 for restoring the impaired cholesterol removal in AMI patients irrespective of mild RI.
Other objectives of the present study were to assess the effects of CSL112 on HDL-C, pro-atherogenic lipids/lipoproteins, and markers of inflammation and cardiac/metabolic biomarkers. Findings in relation to elevation of HDL-C are in-line with the expected pharmacology of CSL112, as HDL transports cholesterol mobilized from tissues. Increased transport of cholesterol to the liver (i.e., through reverse cholesterol transport) could theoretically result in increased generation of pro-atherogenic lipoproteins, particularly LDL C and VLDL-C in the liver, resulting in increased plasma levels of these lipoproteins. However, the present study provides no evidence that apoB-containing lipoproteins are elevated following CSL112 infusions, and in fact there was a progressive decline in apoB levels after the first infusion. There were small transient increases in triglycerides in the current study, the significance of which is unknown. Another investigational apoA-I-based therapy (MDCO-216) was associated with dose-dependent increases in triglycerides. 53 However, the presence of elevated triglycerides is observed in individuals with the apoA-I Milano variant (of which MDCO-216 is a recombinant form) 53 and was not demonstrated in a study with CER-001, another recombinant wild-type apoA-I, in which there were non-significant reductions in triglycerides. 54 Although highly variable, the non-lipid/lipoprotein biomarkers demonstrated that inflammatory and cardiac markers, e.g., hsCRP and Troponin, declined through the course of the study in all groups, which is to be expected following an AMI.
These data are important in the context of the continued development of CSL112, as they support the selection of 6 g dose for further development (in terms of the favorable PK and PD profile), and that 4 weekly infusions have a sustained impact on CEC. The data also provide additional support to the HDL function hypothesis. The CSL112 6 g dose and dosing regimen (once weekly for 4 weeks) are currently being investigated in the ongoing phase 3 AEGIS-II trial (NCT03473223), for the reduction of cardiovascular risk in the period shortly after AMI, i.e., up to 90 days. Despite current standard of care, this is a period of particularly high rates of major adverse cardiovascular events (MACE). 55,56 In order to address residual cardiovascular risk, research in recent years has focused on the atheroprotective role of HDL-C, with the traditional “HDL hypothesis” arguing that raising HDL-C may potentially reduce MACE. However, recent evidence has raised questions regarding the validity of HDL hypothesis, and cholesterol efflux, as assessed by CEC, has emerged as a key metric of serum HDL function. 57 -60 A wealth of data support a robust inverse association of CEC with cardiovascular disease risk in MI patients independent of HDL-C levels and other risk factors. 41,61 -63 This is also a parameter that current standard of care has limited impact on, with a 16-week course of pravastatin or atorvastatin showing no effect on CEC. 29 Consequently, CSL112 has been developed with the aim of reducing MACE risk by targeting CEC; the clinical efficacy of this approach will be tested in the AEGIS-II trial, which is currently in the enrollment phase.
Limitations
Our study has limitations. First, the study did not account for potential confounding by lipid-lowering medications on parameters such as apoA-I and CEC; however, concomitant medication use was similar between CSL112 and placebo groups in the AEGIS-I study. 34 Second, due to the need for repeated sampling, the sample size used was relatively small. Nevertheless, the sample size was deemed sufficient to assess PK and PD, and the findings are consistent with prior studies of the PK and PD of CSL112 in different population. 33,36,37,47,52 Third, our study did not take into account HDL particle size and composition based variation which may have occurred secondary to HDL remodeling following CSL112 infusion. 64 It has been hypothesized that the compositional heterogeneity of the HDL particle ascribes many subspecies of HDL with distinctive anti-thrombotic, anti-inflammatory, and anti-oxidant properties based on the associated proteins. The proteins linked to HDL play a significant role in downstream metabolism and function of HDL via interactions with receptors, enzymes and other proteins. 65 -69 The structural composition of HDL in regard to other proteins such as, Apo M, ApoC-II, ApoE, ApoJ, ApoD, and Paraoxonase 1 (PON 1) was not assessed. 70 -73 ApoM attaches to small lipophilic ligands and has been shown to be associated with small, dense pre-beta HDL particles involved in cellular cholesterol efflux. 74 PON1 has anti-inflammatory properties and associates with small dense lipid-poor HDL particles. 75 As CSL112-sourced apoA-I joins the endogenous pool post CSL112 administration, and since it goes through the same pathway of metabolism as endogenous apoA-I, it is difficult to monitor the PK of the specific components deriving from CSL112 versus that from the endogenous. This most likely resulted in a wide range of estimated CSL112 half-life in current and previous reported analyses given the high level of endogenous apoA-I. 32,33,76 Further studies are required to elucidate the interactions of HDL proteins in order to fully assess its functionality and the effect of these interactions on CEC. Lastly, it remains unclear whether improvements in CEC in AMI patients will lead to substantial clinical benefits.
Conclusion
Cardiovascular risk assessment may be more accurate if risk estimation relies on more markers of HDL metabolism than HDL-C. Given that high plasma apoA-I and CEC have been shown to lower cardiovascular risk, their measurement may be a valuable alternative marker. 30,77 -79 However, it might also be useful to develop novel biomarkers that provide more information about HDL functionality. The results of the present study may have important consequences for the development of pharmacological therapies that target the reverse cholesterol transport pathway to reduce risk of atherosclerotic disease, especially immediately after an acute MI. Therapies that predominantly raise plasma HDL-C but do not significantly affect CEC may fail to have significant beneficial effects on atherosclerosis. Notably, such interventions may even increase risk of atherosclerotic disease when achieving very high levels of HDL-C and hence HDL particle size. In contrast, given the present results, it can be hypothesized that interventions primarily raising plasma CEC through infusion of small lipid-poor apoA-I particles, could have a significant effect on atherogenesis. Among patients with AMI, CEC was rapidly elevated by CSL112 infusions in a dose-dependent fashion, which correlated with increase in apoA-I plasma concentrations. Findings from the current sub-study of AEGIS-I support a potential athero-protective benefit of CSL112 for AMI patients via substantial elevation in CEC, which is being evaluated in the ongoing AEGIS-II trial. The findings also support the dosing rationale being utilized in the AEGIS-II study.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221121507 - CSL112 (Apolipoprotein A-I [Human]) Strongly Enhances Plasma Apoa-I and Cholesterol Efflux Capacity in Post-Acute Myocardial Infarction Patients: A PK/PD Substudy of the AEGIS-I Trial
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221121507 for CSL112 (Apolipoprotein A-I [Human]) Strongly Enhances Plasma Apoa-I and Cholesterol Efflux Capacity in Post-Acute Myocardial Infarction Patients: A PK/PD Substudy of the AEGIS-I Trial by C. Michael Gibson, Syed Hassan A. Kazmi, Serge Korjian, Gerald Chi, Adam T. Phillips, Sahar Memar Montazerin, Danielle Duffy, Bo Zheng, Mark Heise, Charles Liss, Lawrence I. Deckelbaum, Samuel D. Wright and Andreas Gille in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
All authors contributed to the design of the study and/or assisted with the data analysis/interpretation of the data. All authors assisted in the preparation of the manuscript, reviewed the manuscript, and provided their approval for submission. All authors agree to be accountable for all aspects of the work presented.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Gibson has received research grant support and consulting support from the manufacturer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by CSL Behring.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
