Abstract
Background:
Patients suffering from heart failure (HF) and iron deficiency (ID) have worse outcomes. Treatment with intra-venous (IV) ferric carboxymaltose has been shown to reduce HF rehospitalizations and to improve functional capacity and symptoms in patients with HF and reduced ejection fraction (HFrEF). However, IV ferric carboxymaltose is significantly more expensive than IV sodium ferric gluconate complex (SFGC) limiting its availability to most HF patients around the globe.
Methods:
A retrospective analysis comparing patients admitted to internal medicine or cardiology departments between January 2013 to December 2018 due to acute decompensated HF (ADHF) and treated with or without IV SFGC on top of standard medical therapy.
Results:
During the study period, a total of 1863 patients were hospitalized due to ADHF with either HFrEF or HF with preserved ejection fraction (HFpEF). Among them, 840 patients had laboratory evidence of iron deficiency (absolute or functional) and met the inclusion criteria. One hundred twenty-two of them (14.5%) were treated with IV SFGC during the index hospitalization. Patients treated with IV iron were more likely to have history of ischemic heart disease, atrial fibrillation, and chronic kidney disease. The rate of readmissions due to ADHF was similar between the groups at 30 days, 3 months, and 1 year.
Conclusion:
High risk patient hospitalized to ADHF and treated with IV SFGC showed comparable ADHF readmission rates, compared to those who did not receive iron supplementation.
Keywords
Introduction
Heart Failure (HF) constitutes one of the biggest burdens on the public health system, with incidence of 20 per 1000 persons above the age of 65 and up to 80 per 1000 persons above 85 years. 1 Acute decompensated heart failure (ADHF) is the most common cause for hospitalizations among patients above the age of 65. 2 Even with the advances in the treatment and management of HF, the prognosis of these patients remains poor and results in impaired quality of life (QOL), repeated hospitalizations, and poor life expectancy. 3,4
Previous studies have shown how iron deficiency (ID) among HF patients is associated with worse New York Heart Association (NYHA) class, and higher risk for recurrent hospitalizations. 5 -7 The pathophysiology of ID in HF is presumed to be a combination of renal dysfunction, drugs for HF, impaired absorption, and hemodilution. 5,6,8
Unlike intra-venous (IV) sodium ferric gluconate complex (SFGC), IV ferric carboxymaltose has been broadly studied in HF patients. Mostly in patients with reduced ejection fraction (HFrEF) and stable chronic heart failure. It has been shown to reduce the risk of hospitalizations up to 60%, improve exercise ability, and QOL. 9 -11 Subsequently, the latest 2021 ESC guidelines recommended treatment with IV ferric carboxymaltose in patients with HFrEF and ID (class IIa recommendation, level of evidence A). 12
The recommendation of treatment with intra-venous iron in patients with stable HFrEF and evidence of ID rely on 2 randomized placebo-controlled studies (CONFIRM-HF and FAIR-HF) that investigated the treatment with IV ferric carboxymaltose. 9,10 Recently, IV ferric carboxymaltose was investigated in patients hospitalized for ADHF with an evidence of ID at discharge. 13 All these trials excluded patients with preserved ejection fraction HF (HFpEF) and some of them excluded patients unable to perform a 6-minute walk (being their primary outcome). 9,10,13 A recent small retrospective analysis found that IV SFGC is safe and cheaper option. However, it did not affect the 30-days or 6 months readmission rates, in comparison to patients who did not receive iron supplementation. 14
Among iron deficient patients with chronic kidney disease (CKD) and inflammatory bowel disease, treatment with IV SFGC was associated with improved QOL and functional ability. 15 -17
In this study we examined the effect of a more affordable IV SFGC on ADHF readmissions in patients admitted due to ADHF and evidence of ID.
Methods
Study Design and Data Sources
This retrospective study was conducted in the Rambam Medical Health Care Campus (RHCC) in Haifa, Israel. RHCC is an academic hospital, in northern Israel, serving around 2 million people. RHCC has developed, and using, its own electronic medical records system, “Prometheus.” Data were retrieved from Prometheus using MDClone software (Beer-Sheba, Israel). MDClone extracts data from the electronic medical records, including hospital admissions, coded diagnoses, medications, laboratory tests, and demographics. The system allows the retrieval of a wide range of variables, in a defined time frame, around an index event. 18
Study Population
The study population included all adult (>18 years) patients admitted due to primary diagnosis of ADHF between January 2013 to December 2018. The other inclusion criteria were: N-terminal pro b-type natriuretic peptide (NTproBNP) level >300 pg/ml or >800 pg/ml in the presence of atrial fibrillation. Hemoglobin level of 8-13 mg/dL on admission and ferritin level <100 mcg/L or Ferritin 100-400 mcg/L and transferrin saturation <20% on index admission or in the previous 3 months. Similarly, to previous studies protocols.
9,10,11
Treatment with IV loop diuretics.
Patients were excluded from the analysis if they had a cardiogenic shock or required IV vasopressors; had active bleeding, active gastrointestinal or genitourinary malignancy; loss of blood due to major surgery requiring blood transfusion up to 3 months prior to the index hospitalization; treatment with blood transfusion in the past 30 days from any reason; hemochromatosis; chronic liver disease; hemolytic anemia; myelodysplastic disorder; chronic lung disease; and HIV/AIDS disease. We also excluded patients receiving immunosuppressive therapy, erythropoietin, renal dialysis, or intravenous iron in the previous 30 days.
Outcomes
The primary outcome of our study was 30-day, 3-month, and 1-year readmission due to ADHF. Secondary outcomes were 30-day, 3-month, and 1-year all-cause mortality. Readmission due to ADHF was defined as admission with a primary diagnosis of ADHF, as recorded in the hospital’s electronic medical records. Only the first readmission was counted.
Statistical Analysis
Qualitative variables were expressed as numbers and percentage. Quantitative variables were descripted by median and interquartile range (IQR). Mann-Whitney U test were used to compare continuous variables. Chi-squared test was used to analyze differences between dichotomous variables. The cumulative survival curves for ADHF readmission were constructed using the Kaplan-Meier method, and curves were compared by the log-rank test. In this study we used all the available data from our databases within the study time frame. Missing data were handled using list-wise deletion. P-value <0.05 was considered statistically significant.
Data analysis was conducted with Statistical Package for the Social Sciences, version 23.0 (IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp). Graphs were generated with MedCalc Statistical Software version 14.8 (MedCalc Software, Ostend, Belgium).
Ethical Aspects
The Institutional Review Board approved this study (approval number 0348-19). The need for written informed consent was waived due to the specific study design.
Results
During the study period a total of 1863 patients were admitted due to primary diagnosis of ADHF and met the inclusion criteria. Out of them, 573 met one or more of the exclusion criteria, and 450 patients had insufficient data. Eight hundred and forty patients were included in the final analysis. One hundred twenty-two (14.5%) were treated with IV SFGC on top of the standard medical therapy during their admission and 718 were given only the standard treatment for ADHF.
Among patients treated with IV SFGC, the median dosage was 125 mg (IQR 125-250 mg) given over 2 days (IQR 2-4 days). No adverse effects were recorded among patients treated with IV SFGC. Serum phosphorus levels at admission and before discharge were available in 675 patients (80.4%). Serum phosphorus decreased by 0.26 mg/dL in 109 patients treated with IV SFGC and by 0.28 mg/dL in 566 who did not receive this treatment (P = 0.48).
Patients treated with IV iron were slightly younger (71 vs. 74 years, P = 0.06), they were more likely to be male (59.8% vs. 49.9%, P = 0.05), and had similar median left ventricle ejection fraction (LVEF) (55% in both groups). More of the patient treated with IV iron had history of ischemic heart disease (IHD) (45.1% vs. 33.7%, P = 0.02), atrial fibrillation (AF) (58.2% vs. 44.4%, P = 0.007), and CKD (52.5% vs. 39.8%, P = 0.01). Further comorbidities and laboratory results are outlined in Table 1.
Clinical and Laboratory Characteristics of the Entire Study Cohort.

Abbreviations: IQR, interquartile range; HF, heart failure.
a Statistically significant values (P < 0.05) are given in bold.
Regarding clinical endpoints, there was no difference between the 2 groups after a follow-up of 30 days, 3-months, and 1-year, not in admissions due to ADHF and nor in all-cause mortality rates (Table 1 and Figure 1). A subgroup analysis of patients with LVEF over or under 40% did not show significant difference between the groups (Figures 2 and 3).
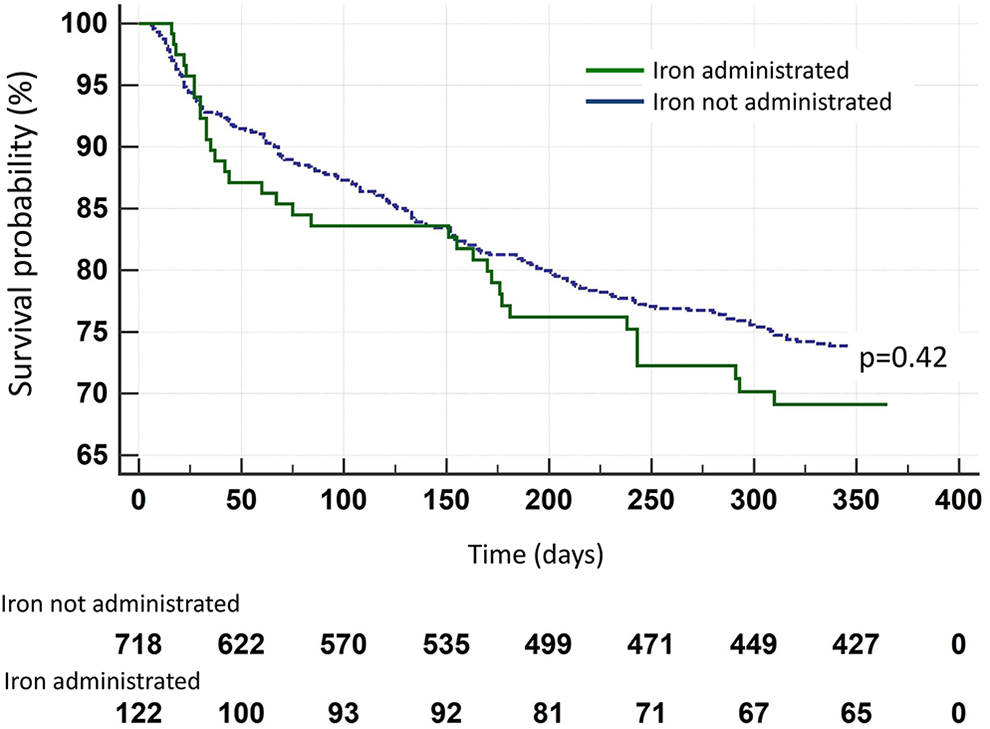
Readmission due to acute heart failure.

Readmission due to acute heart failure in patients with LVEF under 40%. LVEF indicates left ventricular ejection fraction.

Readmission due to heart failure in patients with LVEF of 40% or above. LVEF indicates left ventricular ejection fraction.
Discussion
ID among patients with HF is a major concern and treatment with IV iron was demonstrated in placebo-controlled studies to reduce the risk of rehospitalizations, improve exercise capacity and symptoms, upgrade functional capabilities, and QoL. 9 -11,13 In those trials only IV ferric carboxymaltose was investigated and only patients with HFrEF were included. The replenishment in the IV route has been shown to be safe and more effective than oral administration. 19,20 However, IV ferric carboxymaltose is more expensive than IV SFGC limiting its availability to most HF patients around the globe.
Socioeconomic level and low or high income has an important factor for patient’s life expectancy and longevity, especially in HF patients in which state-of-the-art medications have raised the monthly medical expense dramatically. 21 SFGC’s price in Israel is substantially low (about 2 US dollars for 62.5 mg and 12 US dollars for the entire iron replenishment treatment plan during admission) compared to ferric carboxymaltose with the lowest price listed of 18.4 US dollars per 200 mg dose, reaching over 110 US dollars for comparable dose, not including maintenance dosing. That alone is, in fact, an underestimation, as it does not include additional costs beside the drug itself. A recent study estimates a full treatment cost of up-to 2,112 US dollars (in the USA) and up to 242 British pounds (in the United Kingdom). 22 In this study, ferric carboxymaltose treatment was considered to be at least cost-effective and even cost-saving in some countries. However, not all healthcare systems offer a full payment on the iron treatment, and as stated above, heavy costs reduce compliance. IV SFGC is a well-established drug. It has been found to be well tolerated and effectively improve hemoglobin level, response to erythropoietin and prevents overall ID complications in patients with CKD and inflammatory bowel disease. 16,23
Another type of IV iron that is widely available is iron sucrose. It is less expensive then ferric carboxymaltose, however may have slightly higher adverse reactions rate compared to SFGC as demonstrated by some studies and reports. 24,25 Its effectiveness in HF patients is limited showing increased maximal oxygen consumption (VO2) in a small randomized trial (18 patients). 26
The data regarding treatment with SFGC in HF patients is scarce mainly showing its safety during HF admissions. Furthermore, previous study examined, as a secondary outcome, the rate of readmissions after 30 days and 6 months compared to standard of care, and in concordance to our findings, found no differences between the groups. 14 However, the study was relatively small (123 patients). To the best of our knowledge, this is the first large scale study to examine IV SFGC during index admission in both HFpEF and HFrEF patients.
In our study, patients admitted with ADHF with ID, and treated with IV SFGC were more likely to have AF, CKD, and IHD. All these comorbidities are known risk factors for HF re-admissions. 12 AF increases the risk for recurrent hospitalizations and mortality in HF patients, as shown by previous meta-analysis and a recent review by the European Society of Cardiology. 27,28 Similar effect was seen in patients with CKD and IHD. 29 -31 Therefore, higher rate of readmissions was expected in the IV SFGC group. However, there was no significant difference in recurrent HF hospitalizations between the 2 groups suggesting a possible positive effect of this treatment.
The role of ID replenishment in HFpEF is less established and underrepresented in previous IV Iron randomized studies. Therefore it is yet to be recommended by the ESC guidelines. 9,10,12,13 In a subgroup analysis of HFpEF and HFrEF patients, our results did not show different results. Studies aiming to study this distinct population may show other results. Regarding mortality, our study showed up to 25% all-cause mortality at 1 year following hospitalization in both group in line with previous studies. 3-4
The lack of advantage in outcome for the IV SFGC group might be related to the low median dose administered being 125 mg per day for a median of 2 days. This dose is lower than previous studies in which the total amount per protocol was from 300 to 1000 mg. 23,25
Limitations
This is a retrospective study with unmatched population. However, this also may be its strength, as it provides an analysis of “real world” data. Patients were treated for different duration during hospitalization and unlike the FAIR HF protocol, where iron stores were replenished during hospitalization and after discharge, data on further IV iron treatment after discharge was not available. IV ferric carboxymaltose was not funded by health care authorities during the study period and therefore it is unlikely that a significant number of patients were administered with such treatment after discharge. It is reasonable to assume that patients that were treated during hospitalization were more likely to be treated after discharge which also supports our conclusion. We included only readmissions to our medical center, it is possible that some of the patients were referred to other hospitals across the country.
Conclusion
High risk patient hospitalized to ADHF and treated with IV SFGC showed comparable ADHF readmission rates, compared to those who did not receive iron supplementation. A randomized control trial is underway to further examine SFGC possible benefit (NCT04063033).
Footnotes
Author Contributions
Itay Borreda and Robert Zukermann contributed equally. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
