Abstract
Background:
The oral vasopressin-2 receptor antagonist, tolvaptan, causes aquaresis and improves symptoms in patients with congestive heart failure. However, few studies have explored the effect of tolvaptan in acute heart failure (AHF) patients ≥90 years old.
Methods:
This study enrolled 106 AHF patients treated with tolvaptan added to standard therapy. The subjects were divided into 2 groups: ≥90-year-old patients (over-90 group, mean 92 ± 2 years, n = 45) and <90-year-old patients (under-90 group, mean 76 ± 11 years, n = 61). Patients’ characteristics were assessed, and efficacy and safety were compared between the 2 groups.
Results:
The over-90 group had a lower proportion of male patients, lower body weight, and higher ejection fraction. The under-90 group had significantly higher prevalence of ischemic heart disease and diabetes mellitus. There were no significant differences between the 2 groups in total urine volume at 24 and 48 hours (1934 ± 983 mL vs 1816 ± 1028 mL, P = 0.58 and 3806 ± 1444 mL vs 4078 ± 1851 mL, P=0.47, respectively), the mean change in body weight (−3.0 ± 2.7 kg vs −2.6 ± 2.6 kg, P = 0.50), improvement of congestive symptoms, changes in serum sodium and creatinine levels, the incidences of hypernatremia (n = 0, 0% vs n = 1, 1.6%, P = 0.63), and worsening renal function (n = 9, 20% vs n = 17, 28%, P = 0.48).
Conclusion:
The efficacy and safety of tolvaptan in AHF patients aged more than 90 years were comparable to those of <90 years old. Tolvaptan provides a complementary therapeutic option for AHF patients aged more than 90 years.
Introduction
Heart failure (HF) is a major public health problem in developed countries. 1,2 As life expectancy grows, the proportion of elderly patients with HF has been increasing. Previous studies have shown that the number of Japanese patients with HF will reach 1.3 million by 2030. Furthermore, the number of such patients aged more than 85 years is estimated to reach 300 000 by 2035, and a large number of HF patients aged more than 90 years were included in the previous study from a nationwide administrative database. 3,4 Age is a strong and independent predictor of in-hospital and long-term mortality in HF patients. 1 Moreover, the rates of repeat hospitalization in HF patients are increased by aging of the patient. Elderly patients fare worse following HF events and are also considered to have more complications or comorbidities, such as chronic kidney failure, infection, anemia, hyponatremia, and dementia. 5 –7 Therefore, we need to provide more appropriate treatment for elderly patients with acute heart failure (AHF).
The oral vasopressin-2 receptor antagonist, tolvaptan (TLV), has an inhibitory effect on vasopressin-mediated water reabsorption in the distal nephrons and causes aquaresis. 8 This is a novel mechanism of action for producing free water diuresis. In the EVEREST trial, TLV improved dyspnea and reduced body weight in patients with AHF. 9 TLV has been used for volume overload HF patients along with loop diuretics in Japan. In addition, the Japanese post-marketing surveillance study confirmed the effectiveness of TLV in HF patients. 10
In elderly patients with HF, more than 80 years old as well, the efficacy and safety of TLV have been reported in the Surveillance In Heart faiLurE (SMILE) study. 11 However, the efficacy and safety for the higher age threshold of 90 years have not been examined. In this study, these points were investigated in AHF patients using an age threshold of 90 years in 2 hospitals.
Methods
Study Subjects and Design
This was a retrospective observational study based on our registry database, and patients were recruited from the Department of Cardiovascular Medicine of the University of Fukui Hospital and Hikone Municipal Hospital. This study included 137 patients admitted with AHF between June 2014 and June 2016. Symptoms and signs of HF were defined according to the Framingham criteria. 12 All patients were treated with TLV added to standard HF therapy, including diuretics, β-blockers, aldosterone antagonists, and angiotensin-converting enzyme inhibitors (ACE-Is) or angiotensin II receptor blockers (ARBs). The attending physicians decided on the initial therapy when the patients were admitted. Tolvaptan was administered when possible after hospitalization according to the attending physician’s judgment. Tolvaptan was discontinued if decongestion was achieved or hypernatremia (>150 mEq/L) occurred during hospitalization. A total of 31 patients were excluded due to the following exclusion criteria: discontinued TLV within 1 day due to the improvement of congestive symptoms; AHF after admission; serious general condition; malignancy; tracheal intubation; or hemodialysis. Thus, 106 patients were enrolled and evaluated (Figure 1). The investigation conformed to the principles outlined in the 1975 Declaration of Helsinki and later amendments. Ethics committee approval and informed consent from all patients were obtained. To obtain follow-up information, all patients were reevaluated after discharge from the hospital by reviewing their medical records, including clinical visits, conducting telephone interviews, and obtaining clinical information from the attending cardiologists. The patients who were included in this study were divided into 2 groups: patients ≥ 90 years old (over-90 group, mean 92 ± 2 years, n = 45) and patients under 90 years old (under-90 group, mean 76 ± 11 years, n = 61). Patients’ characteristics were assessed, and the efficacy and safety of TLV were compared between the 2 groups. The dose of oral furosemide was equivalent to half the dose of intravenous furosemide. 10 mg torasemide and 60 mg azosemide were equivalent to 40 mg intravenous furosemide. 13 The follow-up results were registered in the Universal Hospital Medical Information Network Clinical Trials Registry (UMIN 000023840).

Flowchart of the study population. A total of 137 hospitalized patients with AHF who were treated with TLV between June 2014 and June 2016 in our hospitals were included; 31 patients were excluded due to exclusion criteria, so 106 patients were enrolled. The population was divided into 2 groups: patients ≥ 90 years old (over-90 group, mean 92 ± 2 years, n = 45) and patients under 90 years old (under-90 group, mean 76 ± 11 years, n = 61). AHF indicates acute heart failure; TLV, tolvaptan.
Efficacy and Safety Assessments
Primary efficacy outcomes were assessed by total urine volume, the mean change in body weight, the percentage change in body weight, and improvement of congestive symptoms, such as dyspnea, lower limb edema, and pulmonary congestion. Secondary efficacy outcomes were the length of hospital stay, all-cause mortality within 30 days, and all-cause mortality and readmission for HF within 90 days. 14 Total urine volume was assessed at 24 and 48 hours after starting TLV. Dyspnea was assessed on the day starting TLV, 7 days later and at 48 hours after starting TLV. Body weight and lower limb edema were evaluated on the day of starting TLV and 7 days later. Pulmonary congestion was evaluated on the day of starting TLV and 7 and 14 days later. In addition, these congestive symptoms were assessed at discharge. The improvement of dyspnea was defined as a moderate or marked improvement from baseline according to patient-reported 7-point Likert scale. 15 Lower limb edema was evaluated by physical examination. Pulmonary congestion was evaluated with a chest X-ray. The improvement of lower limb edema and pulmonary congestion were judged by 2 different physicians.
Safety was evaluated by the incidences of hypernatremia and worsening renal function (WRF). Hypernatremia was defined as Na ≥ 150 mEq/L. The definition of WRF was an absolute increase in serum creatinine ≥ 0.3 mg/dL within 7 days after starting TLV. This period was based on evidence that almost all cases of WRF occur within 7 days. 16 The WRF prediction score was used to evaluate the risks of WRF and was evaluated as the sum of the following items: history of HF, history of diabetes, and systolic blood pressure (BP) > 160 mmHg at baseline were each assigned 1 point; plasma creatinine 1.5 to 2.4 mg/dL was assigned 2 points; and plasma creatinine >2.5 mg/dL was assigned 3 points. 17 The patients with a WRF prediction score ≥2 points were defined as high-risk patients for WRF. 18 Changes in serum sodium (ΔNa) and creatinine (ΔCre) levels from baseline to day 7 were also measured.
Statistical Analysis
Data are presented as frequencies and percentages for categorical variables, medians for non-normally distributed parameters, and mean ± standard deviation for continuous variables. Comparisons of continuous measurements between 2 or more groups were performed by the unpaired Student t test or the Mann–Whitney U test. Categorical variables were compared by the χ2 test or Fisher exact test. A value of P < 0.05 was considered significant. All statistical analyses were conducted with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). More precisely, it is a modified version of R commander programmed to add statistical functions used frequently in biostatistics. 19
Results
Patient Characteristics
Table 1 presents the characteristics of the subjects enrolled in this study. Compared with the over-90 group, the under-90 group included a higher proportion of male patients, a higher body weight, and higher prevalences of ischemic heart disease and diabetes mellitus. On the other hand, the over-90 patients had a higher ejection fraction (EF) and higher systolic BP. In addition, there were no significant differences in changes in systolic BP, diastolic BP, and pulse rate at 24 hours after starting TLV between the 2 groups. Likewise, no significant differences were found between the over-90 group and the under-90 group in the severity of HF, the severity of chronic kidney failure, history of HF, WRF prediction score, and the prevalence of WRF high-risk patients. The hemoglobin level was lower in the over-90 group than in the under-90 group. Medications on admission were also compared between the 2 groups, and treatment with ACE-Is or ARBs was more common in the under-90 group, while treatment with calcium channel blockers was more common in the over-90 group. There were no significant differences in the use of loop diuretics and aldosterone blockers and the dosage of furosemide between the groups. The prevalence of congestive symptoms and atrial fibrillation were similar in the 2 groups.
Patients Characteristics.a

Abbreviations: ACE-I, angiotensin-converting enzyme inhibitors; AHF, acute heart failure; ALT, alanine aminotransferase; ARB, angiotensin II receptor blockers; AST, aspartate aminotransferase; BMI, body mass index; BNP, brain natriuretic peptide; BP, blood pressure; BUN, blood urea nitrogen; eGFR, estimate glomerular filtration rate; IV, intravenous injection; K, potassium; LVDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; Na, sodium; NYHA, New York Heart Association; po, per os; TLV, tolvaptan; WRF, worsening renal function.
aValues are given as means (standard deviation) or percentage and frequency.
Table 1 also shows the mean TLV treatment period, the initial day of starting TLV after admission, the mean initial dose of TLV, and the total dose of TLV during hospitalization. No significant difference was observed in the administration of TLV between patients in the over-90 group and the under-90 group. In addition, the dose of furosemide equivalents at starting TLV and the use of carperitide were similar in the 2 groups.
Efficacy Assessments
Figure 2 shows the early effects of TLV on total urine volume and dyspnea. No significant differences were observed in the total urine volume at 24 and 48 hours between patients in the over-90 group and the under-90 group (1934 ± 983 mL vs 1816 ± 1028 mL, P = 0.58 and 3806 ± 1444 mL vs 4078 ± 1851 mL, P = 0.47, respectively, Figure 2A and B). There were no significant differences in the improvement of dyspnea at 48 hours after starting TLV between the over-90 group and the under-90 group (P = 0.51, Figure 2C). Figure 3 shows the mean change in body weight (A) and the percentage change in body weight (B). There were no significant differences in the mean change in body weight between the over-90 group and the under-90 group (−3.0 ± 2.7 kg vs −2.6 ± 2.6 kg, P = 0.50). The percentage change in body weight was also similar in the 2 groups (−6.2% ± 5.0% vs −4.4% ± 4.3%, P = 0.08). No significant differences were observed in the improvement of congestive symptoms between patients in the over-90 group and the under-90 group (P = 0.70, P = 0.37, and P = 1.0, respectively, Figure 4). Moreover, there were no significant differences in residual congestion at discharge (P = 1.0, P = 0.70, and P = 0.13, respectively, Table 1). The results of secondary efficacy outcomes are listed in Table 2. There were no significant differences between the 2 groups in the length of hospital stay, all-cause mortality within 30 days, and all-cause mortality and readmission for HF within 90 days.
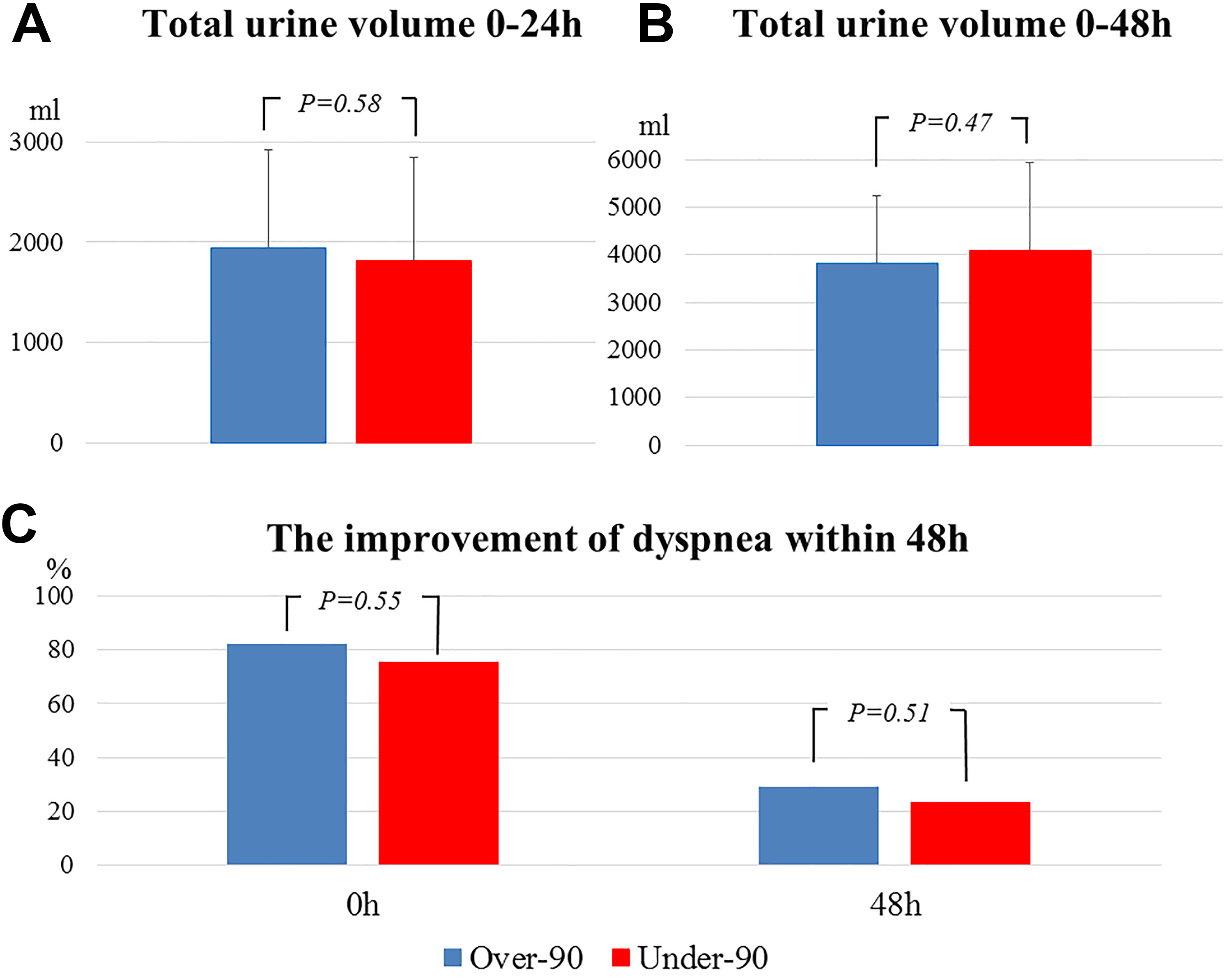
The early effects of TLV on total urine volume and dyspnea. No significant differences were observed in the total urine volume at 24 and 48 hours between patients in over-90 group and under-90 group (P = 0.58, P = 0.47, respectively, A and B). There are no significant differences in the improvement of dyspnea at 48 hours after starting TLV between the 2 groups (P = 0.51, C). TLV indicates tolvaptan.

Body weight changes. There are no significant differences in the mean change in body weight (A) between the over-90 group and the under-90 group (−3.0 ± 2.7 kg vs −2.6 ± 2.6 kg, P = 0.50). The percentage change (B) in body weight is also similar in the 2 groups (−6.2% ± 5.0% vs −4.4% ± 4.3%, P = 0.08).
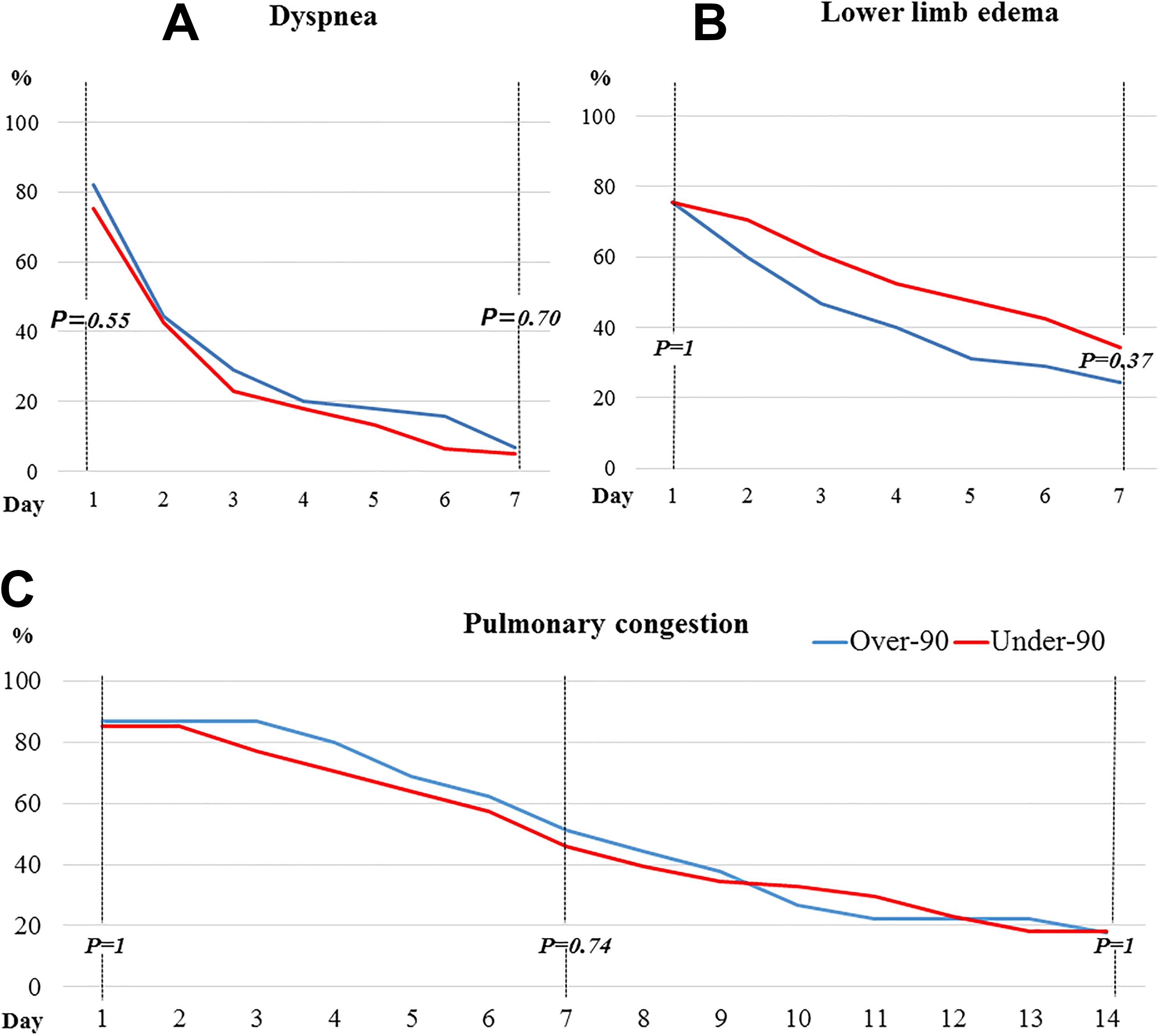
Changes in congestive symptoms and pulmonary congestion. There are no significant differences in the improvement of dyspnea (A), lower limb edema (B), and pulmonary congestion (C) between the over-90 group and the under-90 group (P = 0.70, P = 0.37, and P = 1.0, respectively).
Secondary Efficacy Outcomes.a
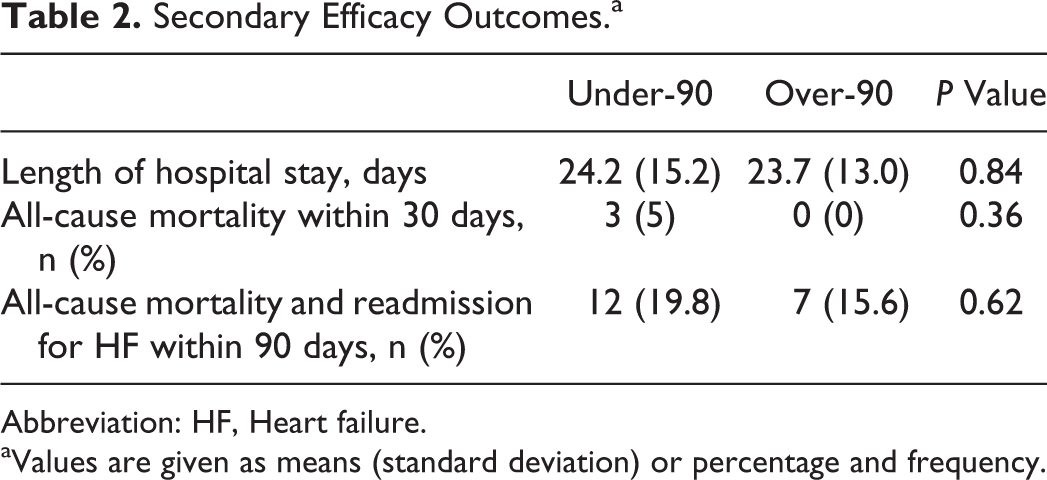
Abbreviation: HF, Heart failure.
aValues are given as means (standard deviation) or percentage and frequency.
Safety Assessments
As shown in Figure 5A, the incidence of WRF did not differ between patients in the over-90 group and those in the under-90 group (n = 9, 20% vs n = 17, 28%; P = 0.48). Furthermore, no significant difference was observed in the incidence of hypernatremia between the 2 groups (n = 0, 0% vs n = 1, 1.6%, P = 0.63). ΔNa and ΔCre values in the over-90 group were similar to those in the under-90 group (ΔNa 1.2 ± 4.8 mEq/L vs 2.4 ± 5.7 mEq/L, P = 0.34 and ΔCre 0.11 ± 0.32 mg/dL vs 0.04 ± 0.37 mg/dL, P = 0.40, respectively; Figure 5B).

Worsening renal function and hypernatremia after TLV administration. There are no significant differences in the incidences of WRF and hypernatremia in the over-90 group and the under-90 group (n = 9, 20% vs n = 17, 28%, P = 0.48; n = 0, 0% vs n = 1, 1.6%, P = 0.63, respectively; A). ΔNa and ΔCre values in the over-90 group are similar to those in the under-90 group (ΔNa 1.2 ± 4.8 mEq/L vs 2.4 ± 5.7 mEq/L, P = 0.34 and ΔCre 0.11 ± 0.32 mg/dL vs 0.04 ± 0.37 mg/dL, P = 0.40, respectively; B).TLV indicates tolvaptan; WRF, worsening renal function; ΔCre, the mean change of serum creatinine from baseline; ΔNa, the mean change of serum sodium from baseline.
Discussion
In the present study, the safety and efficacy of TLV were investigated in very elderly patients (age ≥90 years) with AHF, and the main findings were as follows: first, with respect to the efficacy of TLV, total urine volume, the mean change in body weight, and improvement of congestive symptoms in over-90 patients with AHF were comparable to those of under-90 patients; second, TLV was used without serious adverse events compared to the under-90 patients; and third, the use of a low starting dose of TLV contributed to the efficacy without serious adverse events in both groups.
Efficacy of TLV
A previous study showed that insufficient decongestion at discharge was significantly associated with poor mortality and readmission in AHF patients. 20 Therefore, it is necessary to reduce volume overload that results in congestion. The present results show that TLV improved congestive symptoms and reduced body weight in a similar manner in both over-90 and under-90 patients. Clinical differences between over-90 patients and younger patients were a higher prevalence of women, higher EF, lower BMI, and lower prevalences of ischemic heart disease and diabetes. Previous studies had shown similar clinical differences between octogenarians and younger patients. 1,11 This may be related to the high mortality rate in HF patients with diabetes and low EF, which consequently decreases the survival rate to the age of 90 years. 21,22 Additionally, the administration of TLV contributed to decongestion in elderly HF patients. 11,23,24 In this study, residual congestive symptoms in over-90 patients were almost not recognized at discharge, as in under-90 patients. Moreover, most of the over-90 patients in our study had a preserved EF. A previous study reported HF patients with preserved EF had poor short-term mortality and high readmission rate as those with reduced EF. 14 Our study demonstrated that short-term mortality and readmission rate in over-90 patients were comparable to those in under-90 patients. These findings collectively suggest that the use of TLV for over-90 patients may result in decreasing short-term mortality and the readmission rate in the same as under-90 patients.
The Incidence of WRF
Previous studies reported that the early administration of TLV within 24 hours of admission to elderly patients with AHF resulted in immediate decongestion and a lower incidence of WRF. 23,24 In the present study, AHF patients received TLV within a couple of days after admission (2.04 ± 2.30 days vs 2.18 ± 1.79 days, P = 0.09). The use of TLV was effective for urine output and improving dyspnea within 48 hours in a similar manner in both over-90 and under-90 patients. The present data support the idea that early use of TLV in over-90 patients with AHF may lead to immediate decongestion.
Several studies reported that between 27% and 40% of AHF patients experience WRF during hospitalization, and moreover, WRF is an independent predictor of poor outcomes in patients with AHF, with an increased length of hospitalization. 25,26 A history of HF or diabetes mellitus, admission creatinine > 1.5 mg/dL, and systolic BP > 160 mm Hg were reported to be independent factors associated with a higher risk of WRF. 17 Early use of TLV reduces the incidence rate of WRF in elderly patients and the high-risk population. 23,27 The majority of the present patients who were more than 90 years old were included in a high-risk population for WRF. In the present study, the incidence rate of WRF in over-90 patients was comparable to that in previous studies. The present data showed that administration of TLV to over-90 patients with AHF resulted in a lower incidence of WRF compared to previous studies without TLV usage. Thus, administration of TLV to over-90 patients with AHF in the early phase may become useful for achieving good outcomes in the same as under-90 patients with AHF.
Administration of Low-Dose TLV
In large postmarketing surveillance studies in Japan, the SMILE Study indicated that TLV needs to be started at a low dose of no more than 7.5 mg in elderly HF patients to prevent hypernatremia. 11 Several previous studies in elderly AHF patients reported that low-dose TLV contributed to immediate decongestion and a lower incidence of WRF. 23,27 The present data showed that starting TLV at a low dose for over-90 patients with AHF is as effective and safe as that for younger patients (<90 years old). Takasu et al reported that low-dose TLV therapy for elderly AHF patients with severe aortic stenosis resulted in decongestion without causing hemodynamic instability. 28 Their patients were given a lower dose of TLV (4.8 ± 1.8 mg daily) than in the present study (7.2 ± 1.8 mg daily).
In the present study, the changes in BP and pulse rate after the use of TLV in the over-90 group were as low as in the under-90 group. The data demonstrated that low-dose TLV therapy for very elderly AHF patients contributed to decongestion without causing remarkable changes in BP and pulse rate in the same as younger AHF patients.
Previous studies indicated that continuous administration of TLV after discharge reduced the risk of WRF and had a potential to improve mortality. 29 Moreover, Matsumoto et al reported that continuous TLV at a dose of 3.75 mg daily was an independent predictor for a good prognosis in HF patients after discharge. 30 Therefore, a lower daily dose of TLV should be considered for cases of continuous administration and patients with hemodynamic instability.
Clinical Implications
The present study demonstrated that administration of TLV within a couple of days after admission for over-90 patients contributed to immediate decongestion and a lower incidence of WRF, as in under-90 patients. Additionally, TLV therapy that was started at below 7.5 mg in over-90 AHF patients resulted in decongestion without adverse effects or causing remarkable changes in BP and pulse rate in the same as under-90 AHF patients. Moreover, TLV therapy may reduce short-term mortality and rehospitalization rate in over-90 AHF patients, as in under-90 patients. Therefore, our results might lead that TLV becomes one of the appropriate treatments for AHF patients aged more than 90 years.
Study Limitations
The present study has several potential limitations. First, it included a small number of patients, which may have led to selection bias. Second, this was a retrospective, observational study. The initial therapy and the administration of TLV were decided according to the attending physician’s judgment. Third, only patients receiving TLV therapy were evaluated, and not those who are not receiving. Therefore, the direct effects of TLV therapy for AHF patients could not be evaluated. Finally, the short-term effect of TLV was assessed, and TLV was not continued after discharge. The long-term prognosis in very elderly patients could not be evaluated. Hence, a future randomized, multicenter, large-scale study will be required to confirm the present results.
Conclusions
The early use of low-dose TLV in over 90 years old AHF patients contributed to immediate decongestion without adverse effects in the same as under-90 AHF patients. Additionally, there were no significant differences in short-term outcomes between AHF patients aged more than 90 years old and under-90 patients. We conclude that TLV provides one of therapeutic options for AHF patients aged more than 90 years old.
Footnotes
Authors’ Note
This author takes responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Acknowledgments
The authors would like to thank Hiromi Nishimura, Motoko Oku, Mari Kurata, and Yoshiko Kurose for providing excellent technical assistance. The first author would also like to express his gratitude to his wife for her moral support and constant encouragement.
Author Contributions
Yusuke Sato, Hiroyasu Uzui, Akira Nakano, Hiroshi Tada, and Tetsuji Morishita contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; and gave final approval. Moe Mukai, Yuichiro Shiomi, Kanae Hasegawa, Hiroyuki Ikeda, Naoto Tama, Yoshitomo Fukuoka, Kentaro Ishida, and Kenichi Kaseno contributed to conception; acquisition; drafted manuscript; critically revised manuscript; and gave final approval. Shinsuke Miyazaki contributed to conception and design; acquisition; drafted manuscript; critically revised manuscript; and gave final approval. All authors agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
