Abstract
Inotropic support in ischemic acute heart failure (AHF) is controversial. We tested a therapeutic principle for AHF by combining a low dose of omecamtiv mecarbil (OM; 0.25 mg/kg bolus plus 0.25 mg/kg/h) with a low dose of dobutamine (Dobut; 1.25 µg/kg/min). In 10 pigs subjected to myocardial ischemia by left coronary microembolization, this cotreatment increased cardiac power (CP) from 0.48 ± 0.14 to 0.81 ± 0.22 W (P < .05). When the drugs were given as a monotherapy, CP increased from 0.57 ± 0.11 to 0.65 ± 0.15 W (OM; n = 5; not significant) and from 0.40 ± 0.07 to 0.70 ± 0.10 W (Dobut; n = 5; P < .05). Dobut counteracted OM-mediated impairments in early relaxation and diastolic shortening. In a second protocol using the same doses, we assessed cardiac efficiency in 5 healthy pigs by relating myocardial oxygen consumption (MVO2) to the pressure–volume area. Here, the increases in cardiac work and MVO2 were matched, leaving cardiac efficiency unaltered by this drug combination. Low-dose cotreatment with OM + Dobut produces an appropriate hemodynamic effect with improved CP at doses that do not affect cardiac efficiency. This outcome is mainly attributed to the inotropic effect of Dobut.
Introduction
The use of inotropic support in ischemic acute heart failure (AHF) is controversial. The European Society of Cardiology guidelines give the weakest recommendation (class IIb) at the lowest level of evidence (C) for such treatment. 1 The reluctance stems from the well-known arrhythmogenic effects, 2 increased myocardial energy demands, 3 and elevated mortality seen in clinical trials following inotropetherapy. 4 The adverse events are particularly prominent at a high dosage, which is often necessary to reach the desired treatment goals. Patients with AHF typically have a previous history of cardiovascular disease (CVD), with impaired sensitivity in the adrenergic pathway 5 and/or are on oral β-blockers at hospital admission. 1 These challenges have led to R&D for new inotropes that do not act on the adrenergic cAMP-mediated pathway. A leading drug in this pipeline is the myosin activator omecamtiv mecarbil (OM), which is currently under investigation in a phase III trial, GALACTIC-HF. 6 Omecamtiv mecarbil prolongs the systolic ejection time (SET), 7 which shortens diastole. 8 This finding has raised concerns related to ventricular filling 8 and myocardial blood flow 9 that are supported by the elevated troponins in clinical trials. 10,11 In addition, continuous activation of myosin ATPase by OM 7 causes substantial myocardial oxygen wastage when OM is given as a monotherapy for experimental AHF. 12
We aimed to assess the therapeutic efficacy of a low-dose cotreatment with omecamtiv mecarbil and dobutamine (OM + Dobut) in a pig model of ischemic AHF. We hypothesize that the drugs potentiate systolic unloading and limit the adverse events observed with both drugs at high dosages. The outcome was assessed by surrogate end points such as systolic unloading, diastolic relaxation, pressure–volume relations, and myocardial oxygen consumption (MVO2).
Methods
Experimental Animals
All experiments were conducted in accordance with the Consensus Author Guidelines for Animal Use developed by The International Association of Veterinary Editors. A total of 15 castrated male domestic pigs (Sus scrofa domesticus) weighing 25.7 ± 2.0 kg (mean ± standard deviation [SD]) were used. The animals were held in an approved animal facility as previously described. 8
General Instrumentation
Induction of anesthesia, intubation, and general instrumentation for all animals is described elsewhere. 8 Schematic overview of the experimental groups are given in Figure 1.
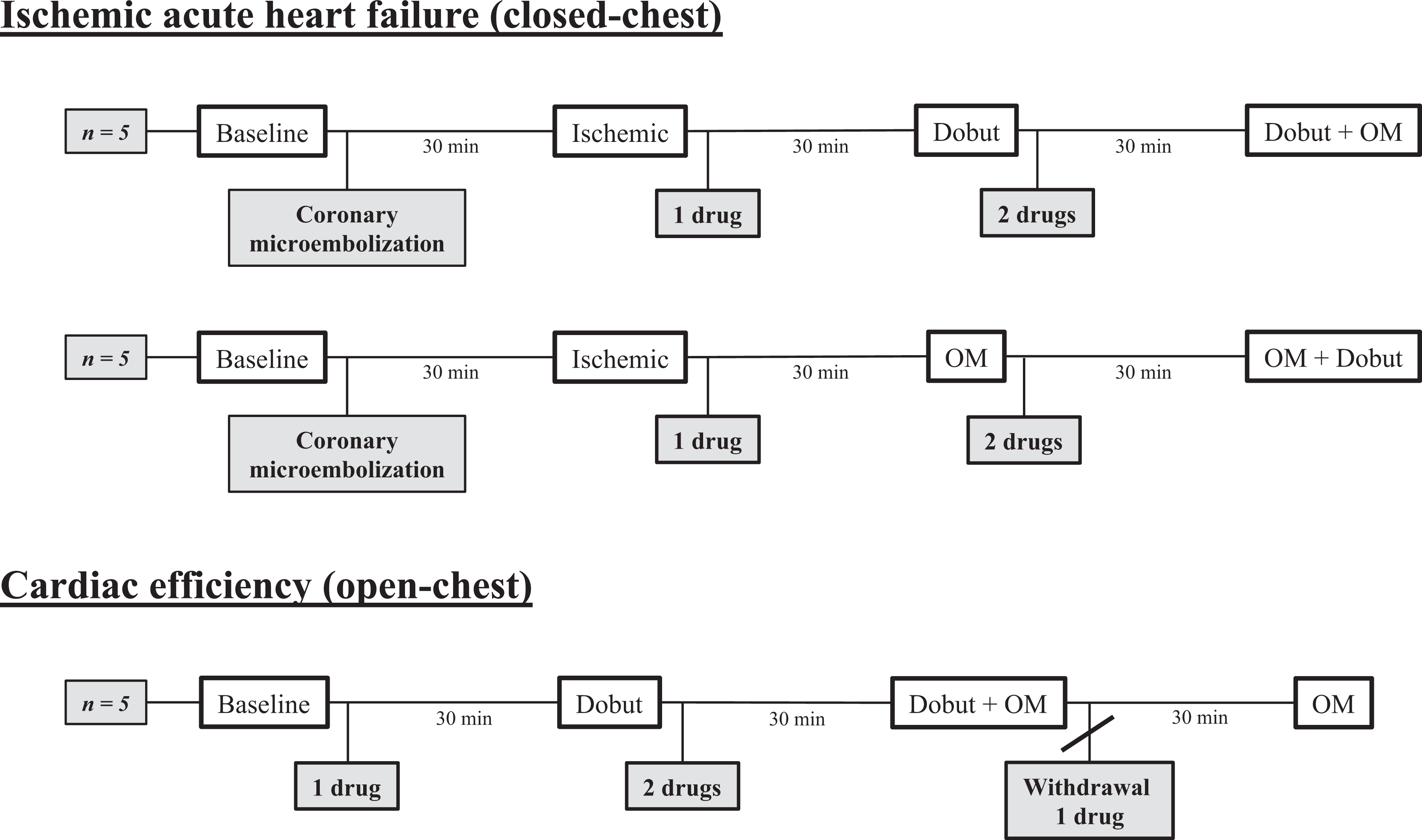
Schematic diagram summarizing the 2 protocols used. OM, omecamtiv mecarbil (0.25 mg/kg bolus plus 0.25 mg/kg/h); Dobut, dobutamine (1.25 µg/kg/min).
Closed-Chest Model of Ischemic AHF
Coronary microembolization is a reliable and clinically relevant method to induce ischemic ventricular dysfunction. 13 In the present study, we used a protocol as previously described. 14 Throughout the experiments, continuous infusion of 0.9% NaCl (10 mL/kg/h) was administered to maintain the circulating volume. Glucose (1.25 g/L) was added to the infusion to maintain blood glucose levels. After general instrumentation, a Swan-Ganz catheter was placed in the pulmonary trunk for assessment of central venous and pulmonary arterial pressure, as well as cardiac output (CO) by thermodilution. The right femoral artery was cannulated to enable catheterization of left coronary artery main trunk and the following coronary microembolization protocol. Transthoracic short-axis echocardiography (Vivid I; GE, Chicago, Illinois) was used for calculations of left ventricular (LV) volumes.
Open-Chest Model for the Assessment of Cardiac Energetics
The open-chest model is previously described in our group. 15 Healthy pigs were used to assess cardiac energetics. Due to increased fluid loss in this open-chest model, a higher NaCl 0.9% volume was infused (20 mL/kg/h). Glucose (1.25 g/L) was added to the infusion to maintain blood glucose levels. After general surgical preparation, we advanced with (1) median sternotomy, (2) pericardial removal, (3) hemiazygos vein ligation, (4) dissection to free the pulmonary trunk, (5) dissection to free the coronary arteries, (6) suture of 3 sonomicrometric crystals (Sonometrics Corporation, London, Ontario, Canada) into the myocardium, and (7) great cardiac vein catheterization via the superior vena cava using a pediatric central venous catheter (Arrow 24G; eSutures). Thereafter, flow probes (Medistim, Horten, Norway) were placed around the main pulmonary and coronary arteries (right branch, circumflex, and left anterior descending) for measurements of CO and coronary blood flow, respectively. The sonomicrometric crystal dimensions (apex to basoseptal and basolateral to basoseptal) were calibrated to endocardial LV dimensions from epicardial echocardiography (Vivid I; GE).
Experimental Protocol
After surgical preparation and stabilization in the closed-chest protocol (n = 10), baseline recordings were performed before LV ischemia by coronary microembolization was induced as described previously. 8 Level of ischemic AHF was aimed at reduction in the stroke volume by approximately 30% and the pulmonary capillary wedge pressure rise to 15 to 20 mmHg. An average of 16.1 ± 6.3 mL of microspheres was injected to reach this level of heart failure. Second recordings were performed approximately 30 minutes after the last injection under stable hemodynamics. The animals were then randomly divided into 2 groups to receive either Dobut (1.25 µg/kg/min; n = 5) or OM (0.25 mg/kg bolus plus 0.25 mg/kg/h; n = 5) as the first treatment. Monotherapy recordings were performed 30 minutes after the start of drug infusion. The second drug was added for combination therapy, and final recordings were carried out after 30 minutes of infusion.
A group of healthy animals (n = 5) was used for the assessment of cardiac energetics. We performed an open-chest surgical preparation as described above before baseline recordings. Dobut (1.25 µg/kg/min) was infused for 30 minutes before new recordings, and OM (0.25 mg/kg bolus plus 0.25 mg/kg/h) in combination with Dobut was infused before the next recordings. Dobut was then withdrawn before final measurements after 30 minutes of OM infusion alone. Finally, the left ventricle was weighed after euthanasia by intravenous pentobarbital sodium injection. Euthanasia was performed according to the regulations on the use of animals in experiments (Norwegian legislations).
Left Ventricular Energetics
Cardiac efficiency was assessed by relating LV work (pressure–volume area [PVA]) to MVO2 at multiple workloads. Multiple workloads were achieved by a stepwise reduction in preload by inflating a balloon catheter situated in the vena cava as previously described. 12 Calculation of PVA and MVO2 is described in detail elsewhere. 3,12
Hemodynamics
Methods for pressure, flow, and CO measurements are described earlier by our research group. 8 All LV volumes were calculated using the bullet formula, 16 where Volume = 5/6 × Area × Length. End-diastolic and end-systolic areas were measured with short-axis transthoracic echocardiography, and the long-axis diameter (length) was calculated as 1.37 times the short-axis diameter obtained by echocardiography. 17
For the closed-chest model, values from transthoracic echocardiography were used to calculate the volumes. For the open-chest model, endocardial end-diastolic diameters were obtained at steady-state hemodynamics before preload reductions using epicardial echocardiography for the calibration of the sonomicrometric crystal-derived short-axis dimension. Dimensions from crystal signals were used for volume estimations with the same formula (bullet) at each preload. Hemodynamic data were recorded and analyzed using ADI LabChart software (ADI, Dunedin, New Zealand).
Statistical Analysis
Power analysis (G*Power) was carried out, and results from previous studies were considered to estimate the number of animals needed. Calculations and statistical analyses after the experimental protocols were performed using a spreadsheet (Microsoft Excel; Microsoft, Redmond, Washington) and a statistical package (GraphPad Prism 7; GraphPad, La Jolla, California). Values are presented as the mean ± SD (Figures 2 –4). Repeated measure one-way analysis of variance followed by Tukey test for multiple comparisons was used on bar graphs in Figures 3 and 4. Analyses of covariance were used on cardiac energetics data (linear regression Figure 4). P values < .05were regarded as statistically significant.
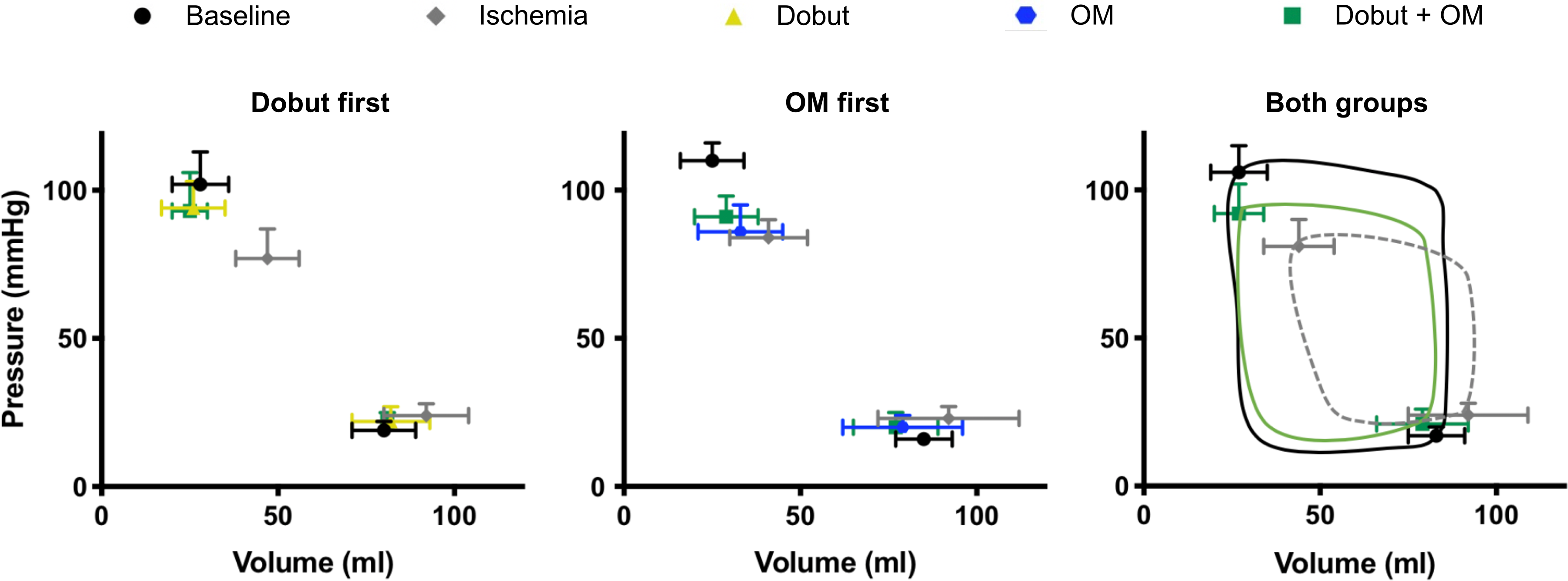
Pressure–volume relations from ischemic acute heart failure (AHF) pigs. Left ventricular (LV) end-systolic and end-diastolic pressure–volume relationships (ESPVR and EDPVR) in healthy pigs (black) subjected to ischemic AHF by left coronary microembolization (gray). The left panel shows data from 5 pigs in which dobutamine (Dobut; yellow) were given as the first treatment. The middle panel shows 5 pigs in which omecamtiv mecarbil (OM; blue) were given first. In both groups, the second inotrope was subsequently added for the assessment of OM + Dobut cotreatment (green). The right panel shows data for both groups together with an illustration of LV pressure–volume loops. All data are presented as the mean ± standard deviation. OM indicates omecamtiv mecarbil.
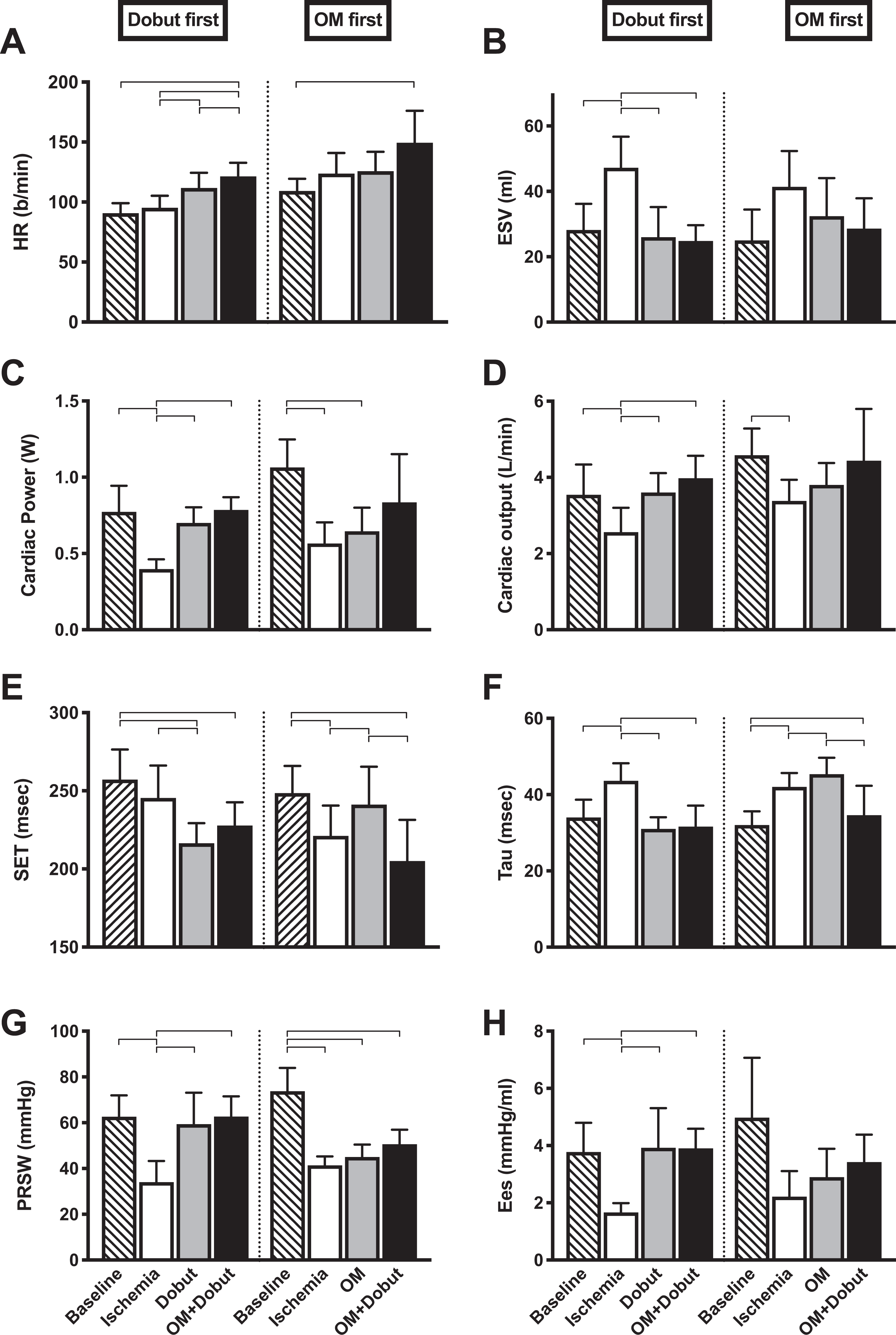
Hemodynamic indices from ischemic acute heart failure pigs. Following baseline recordings (stripe), the animals were subjected to left ventricular (LV) ischemia (blank) by coronary microembolization. The pigs received either dobutamine (Dobut; n = 5, gray, left stack) or omecamtiv mecarbil (OM; n = 5, gray, right stack) as the first drug. The second drug was subsequently added for final recordings of the cotreatment (OM + Dobut; black). HR, heart rate; ESV, end-systolic volume obtained by transthoracic echocardiography of the LV short axis; CP, cardiac power is cardiac output multiplied by LV-developed pressure; CO, cardiac output measured by thermodilution; SET, systolic ejection time is the time between peak positive and peak negative derivatives of LV pressure (dP/dtmax and dP/dtmin, respectively); Tau, the time constant of LV isovolumetric relaxation calculated by Weiss method; PRSW, preload recruitable stroke work is the slope of the relation between end-diastolic volume and stroke work during rapid preload reductions; Ees, end-systolic elastance is the slope of the end-systolic pressure–volume relation. Bars indicate mean values with standard deviations. Brackets indicate statistical significance. P values < .05 were considered statistically significant.
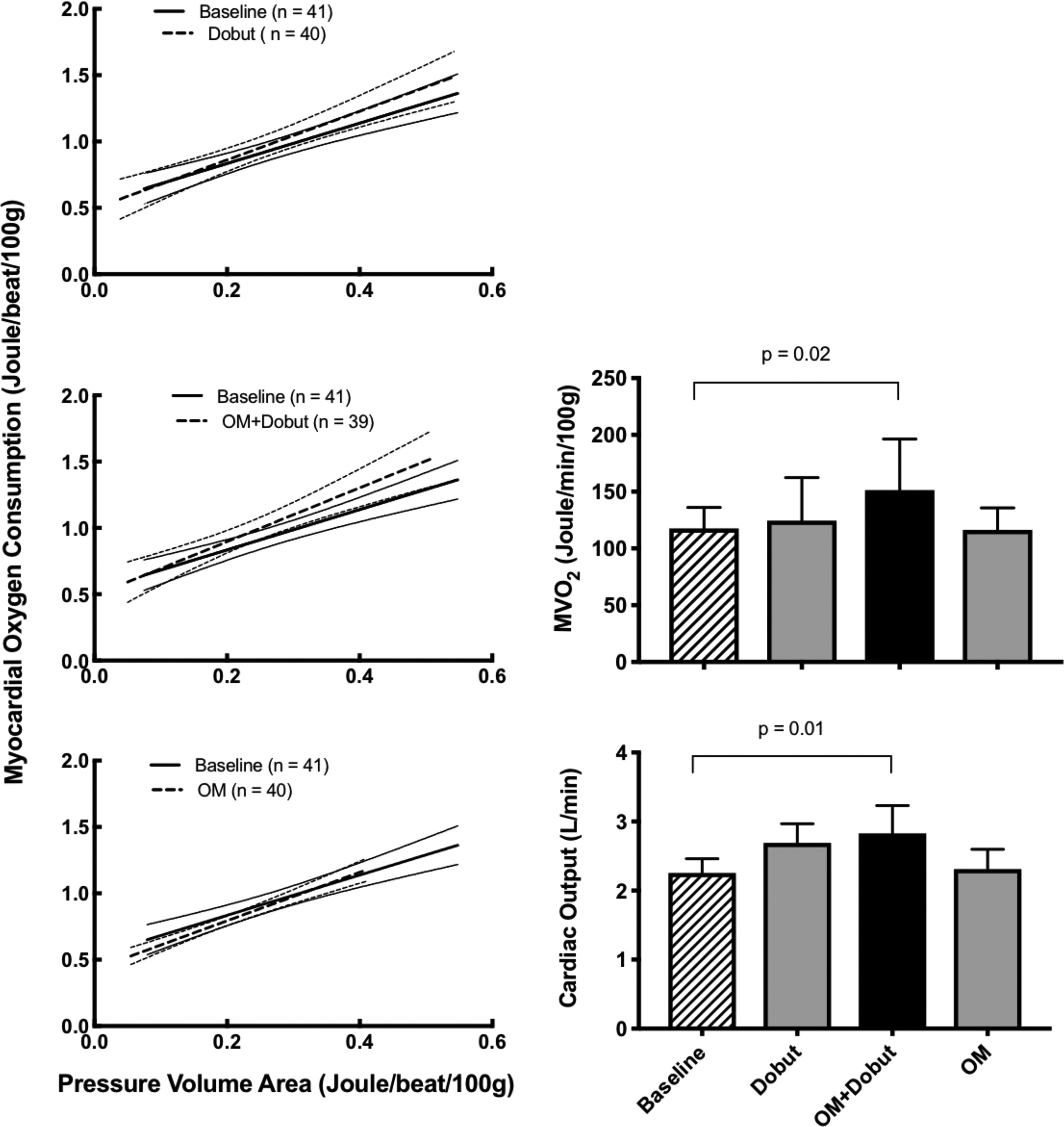
Cardiac energetics in healthy pigs. Cardiac efficiency data from 5 pigs at baseline (striped bar, solid line), after receiving dobutamine (Dobut; gray bar, dotted line), after adding omecamtiv mecarbil (OM + Dobut); black bar, dotted line), and after withdrawal of Dobut (OM; gray bar, dotted line). Left panels show regression lines including 95% confidence intervals of the relationship between left ventricular oxygen consumption (MVO2) and total mechanical work (pressure–volume area [PVA]) at multiple workloads. At each timepoint, 7 to 9 recordings of the PVA–MVO2 relationship were carried out by a stepwise reduction in preload. None of the regressions were significantly different. The right panels show steady-state MVO2 and cardiac output measurements from the same pigs at each intervention. Values are presented as the mean ± standard deviation.
Results
Inducing myocardial ischemia by coronary microembolization substantially impaired systolic function as seen by a halving of end-systolic elastance and preload recruitable stroke work (Figure 3G, H). This led to ventricular dilatation (Figure 3B), the rightward shift of the pressure–volume relationship (Figure 2) and reduced CO (Figure 3D). Monotherapy with OM did not restore systolic function in the ischemic hearts (Figures 2 and 3B-H). However, its impact on the heart was recognized by characteristic prolongations of SET and impaired early relaxation (SET and Tau; Figure 3E, F). Low-dose Dobut as monotherapy did restore systolic function as seen by a normalization of the pressure–volume relationship (Figures 2 and 3B) and that CO increased to preischemic levels (Figure 3D). Also, Dobut restored early relaxation as seen by a normalization of Tau (Figure 3F).
The relationship between total LV work and MVO2 was measured in 5 nonischemic pigs. As seen from the overlapping confidence interval, none of the drug protocols had an impact on this relationship over a broad range of cardiac workload (Figure 4, left panels). This suggests that cardiac efficiency was maintained during all treatments and not depending on workload. At steady-state workload (Figure 4, right panels), both MVO2 and CO increased when OM and Dobut were combined.
Discussion
Effect of the Drugs on the Ischemic Heart
The OM dose selected in the present study is comparable to that used in the ATOMIC-HF trial 11 for the treatment of AHF. Although this phase II trial did not reach its primary end points, that is, dyspnea relief, the treatment did improve cardiac function by moderately decreasing LV systolic dimensions. Such systolic unloading was not seen in the present study using pigs subjected to ischemic AHF. This discrepancy could not be explained by different sensitivity to OM between humans and pigs since the functional significance of the drug (prolongation of SET) was comparable. More likely, the further impairment of diastolic function in the ischemic ventricle by OM counteracted any systolic improvement in the present study. In contrast, low-dose Dobut monotherapy did, to a large extent, restore cardiac function back to preischemic levels. This is in line with others giving Dobut to dogs also subjected to coronary microembolization. 13 When the drugs were combined, minimal additive effects were observed compared to administration of Dobut alone. However, the combination was well tolerated, and this dual treatment restored cardiac power (CP) to preischemic levels (Figure 3C). This is clinically important because CP is the superior early survival predictor in patients hospitalized with cardiogenic shock, 18 and in vivo animal experiments can help guide further clinical and preclinical studies. Of interest was that Dobut counteracted the unwanted effects of OM on diastole. This is evident by a normalization of relaxation rate and a relatively prolonged filling time by shortening the SET. However, a chronotropic effect was seen by this combination treatment that may be a limitation when treating patients with tachycardia. To our knowledge, this is the first study to assess this dual inotropic drug target approach for improving systolic function in the ischemic heart.
Impact of the Drugs on Cardiac Efficiency
An attempt to use OM as a sole drug in experimental AHF revealed that the drug caused substantial myocardial oxygen wastage that was suggested to be mediated by hyperactivity in myosin ATPase. 12 Such trends were also observed in a study that gave OM to conscious dogs. 19 Here, a mismatch of 33% increase in MVO2 versus only a 22% increase in CO was observed following 24 hours of drug infusion. 19
However, catecholamines also cause myocardial oxygen wastage, particularly prominent in high doses. 20 This oxygen wastage is likely mediated by a metabolic switch toward myocardial fatty acid oxidation 21 as well as altered intracellular calcium handling. 3 However, during low-dose Dobut infusion, such oxygen waste is not clear. 20
In the present study, we did not observe any significant alteration in cardiac efficiency by the selected low dose of Dobut, by the low dose of OM, or when the drugs were combined. When OM and Dobut were combined, the heart responded with a matched increase in MVO2 and CO. In addition, when using gold standard methodology (PVA–MVO2 relationship), there was no indication of surplus MVO2 for any of the interventions. This was seen by that data obtained during treatment substantially overlapped with baseline recordings over a broad range of cardiac workloads.
Effect of the Drugs on Myocardial Perfusion
Concerns have been raised regarding the safety of OM in relation to myocardial perfusion. 9 The drug prolongs systole, increases MVO2, 12 and reduces relaxation speed, 8 which may underlie the cardiac troponin elevation observed in 2 clinical trials. 10,11 Our study did not show any indication of myocardial malperfusion when OM was combined with Dobut. Blood gas analysis showed that the oxygen saturation in blood drained from the great cardiac vein was never below 21%, and myocardial lactate uptake was present at all measurement points (range 0.95-1.74 g/min). However, a net global lactate uptake in the myocardium does not exclude regional lactate release from ischemic regions. 22
Limitations
Our study was carried out in healthy juvenile pigs, which are different from typical AHF patients with old age and a previous history of CVD. An animal model in which coronary perfusion is truly challenged, such as coronary stenosis and tachycardia, is warranted to gain the necessary knowledge on the safety of this cotreatment scheme in ischemic heart disease. In addition, the time frame of this study is shorter than the typical clinical time course of AHF. This precludes the use of troponin as a quantitative measure of myocardial damage in this study. This is unfortunate since elevated troponin is observed in clinical trials using omecamtiv. Also, activation of lipoprotein lipase and the subsequent initiation of fatty acid metabolism by adrenergic stimulation occur over time. Thus, a longer study period using this cotreatment protocol for assessing cardiac efficiency is warranted.
Cardiac energetics was not assessed in a heart failure model like the coronary microembolization cohort. The rationale to use a separate nonischemic protocol is, by our experience, the most sensitive setup to detect any surplus MVO2.
The closed-chest AHF cohort aims to reassemble the clinical setting of patients admitted to the intensive care unit with ischemic AHF. Sternotomy and the following cardiac instrumentation as required for assessing energetics is a considerable surgical trauma. This impacts general hemodynamics, thus an induction of severe myocardial ischemia in addition to this often leads to hemodynamic collapse in the need for inotropes. This would preclude the predrug measurements.
Furthermore, ischemia is often complicated with episodes of arrhythmia. This limits the accuracy of the PVA–MVO2 recordings substantially. Also, the accuracy of the work-independent assessment of energetics by regression analysis is dependent on the range of workloads. This is carried out in an unloading protocol by restricting venous return. A compromised circulation (ie, AHF) does not allow much unloading before organ malperfusion sets in.
Previously, our group have documented that therapeutic levels of omecamtiv impacts cardiac efficiency quantitatively similar in both preischemic and ischemia-reperfusion induced AHF. 12 Thus, the fact that no impact on cardiac efficiency was seen in an optimized preischemic model, we see it as highly unlikely that this conclusion would differ in an AHF model. Finally, this study assessed the therapeutic effects using only single doses. This may hamper a general qualitative statement on the potential of this therapy in the clinical setting.
Conclusions
Combination treatment with low-dose OM and Dobut is well tolerated in the ischemic heart. This drug combination does not aggravate cardiac efficiency, as it does not alter the MVO2-work relation. However, the data does not support our hypothesis that this cotreatment potentiates systolic function, as the restoration of cardiac function is almost exclusively ascribed to the inotropic effect of Dobut.
Footnotes
Acknowledgments
The authors thank the technical staff at the surgical research laboratory at The Faculty of Health Sciences, UiT–The Arctic University of Norway, for assisting the experiments.
Author Contributions
Rødland: Planned protocol, executed experiments, analyzed data and drafted the manuscript. Rønning: Planned protocol and executed experiments. Kildal and Myrmel: evaluated data. How: Produced initial idea, planned protocol and analyzed data. All authors: revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: UiT–The Arctic University of Norway and The Regional Health Authorities of Northern Norway (Helse Nord) provided financial support for the project.
