Abstract
Low-density lipoprotein cholesterol (LDL-C) is a major cardiovascular risk factor, but other lipid variables such as triglycerides (TRGs), high-density lipoprotein cholesterol (HDL-C) and lipoprotein a [Lp(a)] also affect cardiovascular risk. Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors significantly lower LDL-C concentration but also modestly improve the concentrations of TRGs and HDL-C and more robustly decrease Lp(a) levels. The review presents the associated mechanisms of the beneficial effects of PCSK9 inhibitors on the other than LDL-C lipid variables, including the effects on lipid/apolipoprotein secretion and clearance and the heteroexchange between lipoproteins, as well as the possible effects on other variables involved in lipid metabolism such as sortilin. Proprotein convertase subtilisin/kexin type 9 inhibitors improve the overall lipid profile, and these beneficial effects may play a role in the reduction of cardiovascular risk.
Keywords
Introduction
Proprotein convertase subtilisin/kexin type 9 (PCSK9) is a key protein in lipid and lipoprotein metabolism, since it has a paramount role in the degradation of low-density lipoprotein (LDL) receptors on the hepatic cells surface leading to enhanced LDL particles catabolism and reduction of serum LDL cholesterol (LDL-C) levels. 1 The anti-PCSK9 monoclonal antibodies bind circulating PCSK9, thus preventing PCSK9-induced degradation of LDL receptors. The administration of these drugs on the top of conventional lipid lowering treatment substantially decreases LDL-C concentration by approximately 50% in various groups of high-risk patients. 2 Recently, the Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk (FOURIER) trial showed a significant risk reduction of the composite cardiovascular end point with the PSCK9 inhibitor evolocumab. 3 This significant risk reduction is mainly attributed to the substantial decrease in LDL-C concentration, which is a major cardiovascular disease risk factor. The FOURIER trial is the only completed outcome trial with PCSK9 inhibitors and we have to wait for the results of the Evaluation of Cardiovascular Outcomes After an Acute Coronary Syndrome During Treatment With Alirocumab to confirm a class effect. However, PCSK9 inhibitors also affect other lipid and lipoprotein variables, effects that may also play some role in the reduction of cardiovascular risk.
Thus, the aim of the review is to present the effects of PCSK9 inhibitors on other than LDL-C lipid variables, specifically on serum triglycerides (TRGs), high-density lipoprotein cholesterol (HDL-C), and lipoprotein a [Lp(a)].
Effects of PCSK9 Inhibitors on Serum TRG Concentration
Hypertriglyceridemia increases cardiovascular risk especially when it is combined with decreased levels of HDL-C. 4 Thus, the reduction of TRG concentration could reduce cardiovascular events, possibly beyond the reduction achieved by LDL-C lowering. Various therapeutic regimens, with fibrates and omega-3 fatty acids as the main components, are available for the treatment of hypertriglyceridemia and associated cardiovascular risk markers. 5 –16
Serum PCSK9 concentration affects the metabolism of TRG-rich lipoproteins both in intestine and liver origin (Figure 1). 17,18 In fact, in most studies, PCSK9 levels are modestly correlated with TRG levels, 19 –21 while patients with PCSK9 gain-of-function mutations exhibit increased very-low-density lipoproteins (VLDLs) and lipoprotein remnants. 22 Furthermore, serum PCSK9 levels are predominantly related to intermediate density lipoproteins (IDLs) that are VLDL remnants, a finding which also imply the relation of PCSK9 with the metabolism of TRG-rich lipoproteins. 23 Additionally, serum PCSK9 concentrations are also correlated with TRG levels in proteinuric patients as well as in patients with chronic kidney disease who commonly exhibit abnormalities of TRG-rich lipoproteins. 24,25

Potential mechanisms of PCSK9 inhibitors-associated reduction of serum triglycerides (TRGs). Apo indicates apolipoprotein; LDL, low-density lipoprotein; LRP, LDL-related protein; PCSK9, proprotein convertase subtilisin/kexin type 9; VLDL, very low-density lipoprotein.
The reduction of PCSK9 with the PCSK9 inhibitors is associated with mild/moderate reductions of TRG levels (Table 1). 26 –31 For example, in the recently published FOURIER trial, evolocumab significantly reduced serum TRG levels compared with placebo (−16.7% vs −0.7%, P < .001). 3 Several mechanisms affecting both secretion and catabolism of TRG-rich lipoproteins can explain the PCSK9 inhibitors-mediated reduction of TRGs. Thus, the drugs’-associated profound increase of LDL receptors activity results in an increased catabolism not only of LDL but also VLDL particles. 32 It has been also suggested that PCSK9 can also affect other lipoprotein receptors activity, 33 which may play a role in the clearance of TRG-rich lipoproteins, such as the VLDL receptors and apolipoprotein (apo) E2 receptors (though their role in TRG metabolism in humans is unclear), 34,35 the LDL-related protein, which contributes to the catabolism of both chylomicrons and VLDL remnants, 36 and the cluster of differentiation 36 (CD36, also known as platelet glycoprotein 4), which is a scavenger receptor that may increase the catabolism of chylomicron remnants. 37,38
Effects of Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitors on Serum Triglyceride Levels (vs Placebo).

Additionally, a decreased secretion of TRG-rich lipoproteins has also been suggested to participate in the PCSK9 inhibitors-associated decrease of TRG levels (Figure 2). 39 In fact, LDL receptors directly affect apoB stability through enhanced autophagic degradation in hepatocytes. 40 Thus, PCSK9 inhibitors-associated increased LDL receptors expression leads to an increased degradation of apoB resulting in a reduced synthesis of apoB-containing lipoproteins. 39 Furthermore, the drugs’-associated increased cholesterol pool in hepatocytes (through the profound increase of LDL receptors) results in a reduction of the sterol regulatory element–binding protein-1C pathway, which leads to decreased synthesis of cholesterol and decreased fatty acid synthase and acetyl-CoA carboxylate activity, as well as of the non-sterol receptor element pathways leading to decreased apoB, apoE, diglyceride acyltranferase (DGAT1), and microsomal transfer protein (MTP) synthesis, which affect the synthesis of TRG-rich lipoproteins. 41 A reduction of de novo lipogenesis may also play a role.
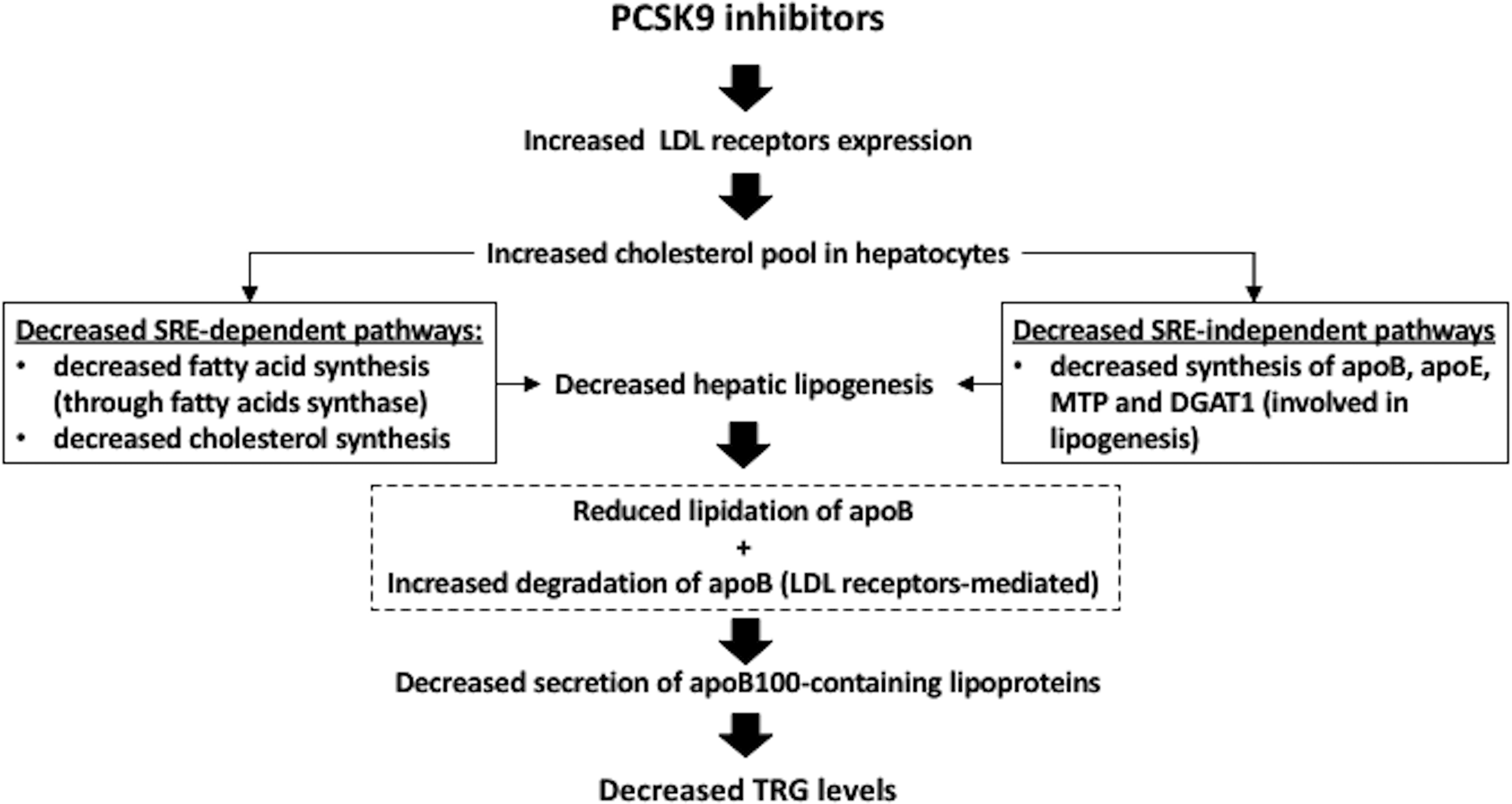
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors and hepatic secretion of triglycerides (TRGs)-rich lipoproteins. Apo indicates apolipoprotein; DGAT, diglyceride acyltransferase; LDL, low-density lipoprotein; MTP, microsomal transfer protein; PCSK9, proprotein convertase subtilisin/kexin type 9; SRE, sterol regulatory element.
There is also evidence that these drugs can also decrease the secretion of apoB48-containing lipoproteins in the intestine. In fact, PCSK9 is expressed in the intestine and experimental studies have suggested that PCSK9-null mice are protected from postprandial lipemia. 42,43 Experimental studies have also suggested that PCSK9 can increase the secretion of intestinal apoB48-containing lipoproteins with similar mechanisms with those previously reported in hepatocytes, including the decreased degradation of apoB and the increased activity of enzymes implicated in lipogenesis, such as fatty acid synthase, MTP, DGAT2, and stearoyl-CoA desaturase. 43
Kinetic studies have also revealed interesting findings. In a recently published study in healthy normolipidemic individuals, the administration of alirocumab (150 mg/2 weeks) was associated with a significant decrease in the mean fractional catabolic rate (FCR) of both LDL-C and LDL-apoB and IDL-C and IDL-apoB (associated with a shift toward smaller LDL particles). 44 Additionally, a reduction in the production rate of LDL-apoB was found possibly due to increased FCR of IDL-apoB. However, alirocumab administration was not associated with any significant change in VLDL-apoB and VLDL-triglycerides metabolism. This absence of LDL receptor-mediated increased clearance of VLDL particles after drug administration could be ascribed to the normal TRG concentrations of the study population leading to a rapid lipolytic conversion of VLDL to IDL and LDL particles. However, no change in lipolytic activity and apoCIII levels was found after alirocumab administration. It has been also speculated that the observed decrease in apoE concentrations (due to increased fractional removal of apoE-containing IDL and LDL particles) can contribute to the absence of increased LDL receptors-mediated clearance of VLDL particles. Additionally, no change in VLDL production or postprandial lipemia was found possible due to the normal triglyceride concentrations of the study population. 44 On the other hand, a recently published 2 × 2 factorial trial of the effects of atorvastatin (80 mg daily) and subcutaneous evolocumab (420 mg every 4 weeks) for 8 weeks in 81 healthy normolipidemic nonobese men showed that evolocumab administration was followed by an increased fractional catabolism of VLDL-apoB, IDL-apoB, and LDL-apoB, while a decreased production rate of both IDL-apoB and LDL-apoB was also observed. 45 Furthermore, drug administration was associated with a decrease of free PCSK9 concentrations and apoB levels, while no significant change of lipoprotein lipase and cholesterol ester transfer protein (CETP) mass or measures of insulin resistance was found. 45 Thus, the latter study confirmed the PCSK9 inhibitors-associated increased catabolism of TRG-rich lipoproteins, even in normolipidemic individuals, which can explain the observed reduction of TRG levels. However, relative studies in patients with hypertriglyceridemia or mixed dyslipidemia can better delineate the underlying mechanisms of the TRG-lowering effect of PCSK9 inhibitors.
Another study evaluated the effect of alirocumab (150 mg/2 weeks) on lipoprotein particle concentration and size in hypercholesterolemic individuals, using nuclear magnetic resonance spectroscopy. The study showed that alirocumab was followed by a reduction in total VLDL and chylomicron particles by 36.4% compared with an increase by 33.4% in the placebo group (P < .001), mainly due to a decrease in medium and small VLDL particles leading to an increase in VLDL particles size (+10.8% vs + 0.8% P < .01). 46 Another recently published study (using the vertical auto profile method) showed that alirocumab administration (150 mg/2 weeks, n = 74) was associated with reductions of VLDL-cholesterol by 26.1% to 32.4% and of triglyceride-rich remnant particles-cholesterol by 42.1% to 52.5%. 47 Additionally, the cholesterol content of VLDL subfractions VLDL1+2 and VLDL3 was similarly reduced, and a reduction in both apoCII and apocIII (apolipoproteins that play a significant role in TRG metabolism 48 –50 ) concentrations was noticed. 47
Effects of PCSK9 Inhibitors on Serum HDL-C Levels
The HDL has multiple pleiotropic effects against atherosclerosis, and decreased serum HDL-C concentration is conventionally considered a cardiovascular risk factor. 51 It should be mentioned that HDL particles are heterogeneous in lipid and protein composition, as well as in size and function, and multiple factors through oxidation and inflammation might render HDL particles malfunctional or proatherogenic. 52 –58 Many drugs affect HDL concentration and composition and through this mechanism (among others) may alter cardiovascular risk. 9,11,15,59 –66 However, the apparent failure of specific HDL-C-raising therapies [niacin and cholesteryl ester transfer protein (CETP)] raises questions about the HDL-C hypothesis. 67,68
PCSK9 gain-of-function mutations are associated with increased HDL-C and apoA1 levels. 69 Furthermore, it should be mentioned that PCSK9 levels are positively related to HDL-C levels. 70,71 These observations can be explained by the fact that in normal situations, a decreased LDL receptors’ activity (but also of VLDL receptors and apoE2 receptors) is observed in the presence of PCSK9 leading to a decreased clearance of LDL particles from the circulation. 72,73 This mechanism can also decrease the catabolism of apoE-rich HDL particles leading to increased HDL-C levels. Thus, PCSK9 inhibition through this mechanism is expected to decrease HDL-C levels (Figure 3).

Potential mechanisms of PCSK9 inhibitors-mediated increase in high-density lipoprotein (HDL) cholesterol levels. Apo indicates apolipoprotein; CETP, cholesterol ester transfer protein; LDL, low-density lipoprotein; PCSK9, proprotein convertase subtilisin/kexin type 9; TRG, triglycerides.
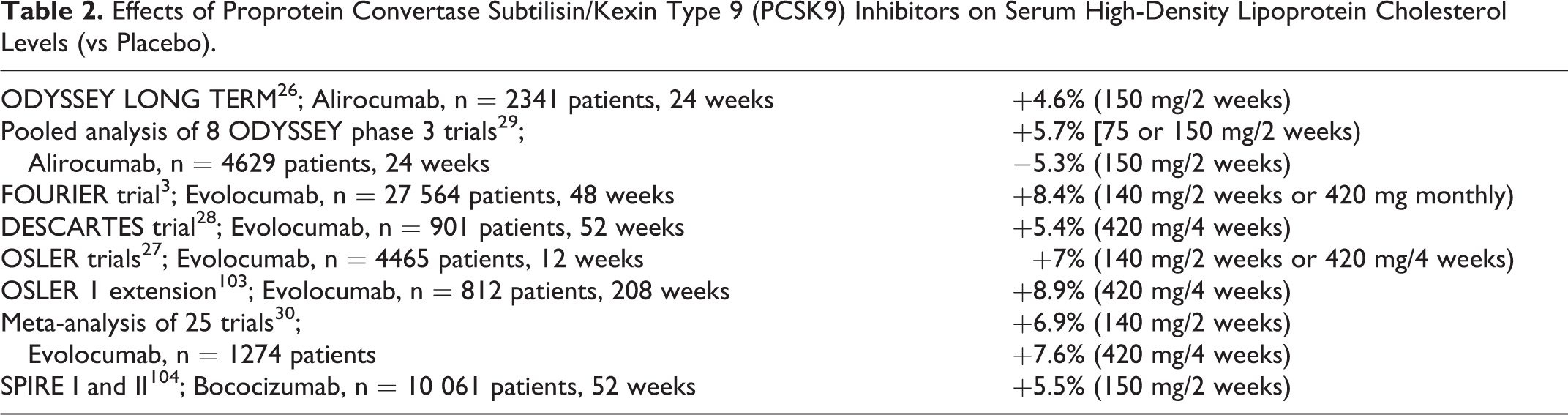
However, most clinical trials have shown that PCSK9 inhibitors can modestly increase HDL-C and apoA1 levels (usually <10%; Table 2). For example, in the Long-term Safety and Tolerability of Alirocumab Versus Placebo on Top of Lipid-Modifying Therapy in High Cardiovascular Risk Patients With Hypercholesterolemia (ODYSSEY Long Term) study, alirocumab administration (150 mg/2 weeks) was associated with an increase in HDL-C and apoA1 concentration by 4.6% and 2.9% 26 , respectively, while in the Open Label Study of Long Term Evaluation Against LDL-C Trial (OSLER), increases in HDL-C and apoA1 levels by 7% and 4.2% were noticed with evolocumab administration (140 mg/2 weeks or 420 mg/4 weeks). 27 In the recently published FOURIER trial, evolocumab significantly increased HDL-C (+8.4%) and apoA1 (+6.5%) compared with placebo (+0.3 and +2.1%, respectively, both P < .001). 3
Effects of Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitors on Serum High-Density Lipoprotein Cholesterol Levels (vs Placebo).
These increases in HDL-C can be attributed to the marked decrease of LDL particles leading to a decreased transfer of cholesterol from HDL to LDL particles (a mechanism similar to that observed in statin-treated patients; Figure 3). 72,73 Additionally, the decrease in the concentrations of TRG-rich lipoproteins observed with PCSK9 inhibitors administration results in a decrease in CETP activity leading to a decreased heteroexchange of lipids between HDL particles and TRG-rich lipoproteins (that is a decreased transfer of cholesterol from HDL to VLDL particles). 72 –74 Interestingly, PCSK9 levels are positively correlated with CETP activity independent of lipid lowering treatment and this mechanism can also explain the increase of HDL-C noticed with PCSK9 inhibitors administration (Figure 3). 75 The final result of the above mechanisms is a modest increase of HDL-C levels observed during the treatment with PCSK9 inhibitors (Figure 3).
It is worth mentioning that PCSK9 levels are associated with small HDL in males. 76 The increase in HDL-C with PCSK9 inhibitors administration is followed by an increase in total HDL particles (by 11.2% vs 1.4% in placebo-treated patients, P < .01) due mainly to an increase in the large HDL particles (by 44.6%), while small increases are observed in median (by 17.7%) and small HDL particles (by 2.8%), 46 a finding consistent with the previously reported drugs-associated reduced CETP activity.
It should be mentioned that HDL-C levels are not always indicative of HDL functionality. Indeed, HDL functionality, as measured by reverse cholesterol transport or efflux capacity, is considered more clinically important for the protective role of HDL compared with HDL-C concentration. 77 –79 Currently, we have limited data of the effects of PCSK9 inhibitors on HDL functionality. An experimental study showed that THP-1 macrophage cholesterol efflux capacity of serum was reduced in PCSK9-knockout mice, but the fatty streak lesion volume was similar to wild-type controls. 80
Effects of PCSK9 Inhibitors on Lp(a) Concentrations
Elevated Lp(a) levels have seen associated with an increased risk of coronary heart disease, stroke, and aortic stenosis. 81 –83 Commonly used hypolipidemic drugs, including statins, do not affect Lp(a) concentration, 84,85 whereas the cardiovascular benefit of the newer CETP inhibitors that significantly lower Lp(a) concentration has not been established. 68
Interestingly, increased Lp(a) levels are observed in patients with familial hypercholesterolemia due to PCSK9 gain-of-function mutations, suggesting a role of LDL receptors in Lp(a) catabolism. 86 However, conflicting data concerning the correlation of Lp(a) concentrations with PCSK9 levels have been reported. 87,88 In patients with coronary artery disease serum levels of PCSK9 levels were positively correlated with serum levels of Lp(a) (r = .195, P = .05). Indeed, multivariate regression analyses showed that serum PCSK9 was a significant predictor of serum levels of Lp(a) (β = .235, P = .01). 89
As shown in Table 3, PCSK9 inhibitors can decrease Lp(a) levels by approximately 20% to 30% (a decrease which was little affected by the dose of the drugs), though the underlying mechanisms are not well delineated (Figure 4). 3,90,91 Some studies have shown that these reductions of Lp(a) levels were well correlated with LDL-C reductions suggesting a prominent role of LDL receptors in Lp(a) clearance. 90,91 However, statins can also increase LDL receptors activity but they do not affect Lp(a) levels. 85 It has been speculated that in cases of a marked supraphysiological increase in LDL receptors activity observed with PCSK9 inhibitors administration and in the face of drugs’-mediated low circulating LDL particles, the LDL receptors-mediated clearance of Lp(a) is markedly increased. In fact, it has been demonstrated that the overexpression of LDL receptors in HepG2 cells increases Lp(a) binding via the LDL portion of Lp(a) and not the apo(a) portion. 90,92 This LDL receptors-mediated increased catabolism of Lp(a) was also confirmed by a recently published study, which showed that alirocumab was followed by a decreased Lp(a) concentration due to increased apo(a) clearance. 44 On the other hand, evolocumab administration was followed by a reduction in Lp(a) levels even in patients with homozygous familial hypercholesterolemia due to LDL receptors null mutations, in whom no change in LDL-C was observed. 93 Thus, other mechanisms may play a role. As described above in the triglycerides section, PCSK9 inhibitors can significantly decrease the concentrations of apoB-containing particles, resulting in a decreased availability of these particles to couple covalently to apo(a) leading to decreased synthesis of Lp(a). 17 Finally, recent findings have implicated sortilin (SORT1), which is a type I membrane glycoprotein ubiquitously expressed in many tissues that participate in protein transport between the Golgi apparatus, endosome, lysosome, and plasma membrane and is involved in multiple biological processes such as lipid metabolism. 94 It has been shown that sortilin is a high-affinity sorting receptor for PCSK9, which is colocalizes with PCSK9 in the trans-Golgi network and enables its secretion from hepatocytes. 95 Sortilin-deficient mice display decreased levels of circulating PCSK9, whereas circulating sortilin and PCSK9 levels are elevated and correlated in healthy patients 95 and in patients with coronary artery disease. 96 Statin treatment increases plasma PCSK9 and decreases sortilin levels. 97 It has recently been described that the overexpression of sortilin, which (similar to PCSK9) is also capable to bind LDL and to regulate LDL receptors activity, leads to increased Lp(a) internalization even in fibroblasts with defective LDL receptors, while PCSK9 was not able to affect this sortilin effect. Furthermore, the overexpression of sortilin may also increase the secretion of apo(a) by HepG2 cells. Interestingly, PCSK9 affects the internalization of apo(a) to a similar extent. 98,99 Thus, a sortilin-mediated effect of PCSK9-inhibitors on Lp(a) clearance is a possible mechanism of serum Lp(a) lowering with these drugs.
Effects of Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitors on Serum Lipoprotein a [Lp(a)] Levels (vs Placebo).


Potential mechanisms of PCSK9 inhibitors-associated decrease in lipoprotein a [Lp(a)] concentration. Apo indicates apolipoprotein; LDL, low-density lipoprotein; PCSK9, proprotein convertase subtilisin/kexin type 9.
Effects of PCSK9 Inhibitors on Inflammatory Markers
Patients with subclinical inflammation have increased CVD risk. 100 The beneficial effect of statins, apart from their mechanism of action that leads to LDL-C reduction, has been also attributed to the reduction of subclinical inflammation. 12,101 PCSK9 inhibitors have not shown in clinical trials to significantly decrease inflammatory markers. A meta-analysis of 16 treatment arms (2546 participants) did not show any significant effect of PCSK9 inhibitors on high-sensitivity C-reactive protein (hsCRP) levels (weighted mean difference: 0.002 mg/L, 95% confidence interval: −0.017 to 0.021; P = .807). 102 This effect was not affected by the type or dosing frequency of the PCSK9 inhibitor. Additionally, no association of changes in hsCRP levels with the observed alterations in plasma LDL-C concentrations or cumulative dosage of the drug was found. 102 This nonsignificant effect of PCSK9 inhibitors on hsCRP can be attributed to the fact that the populations studied would be expected to have normal levels of hsCRP since in most studies they have been treated with statins, which diminish the vascular inflammatory response. Additionally, studies with statin-intolerant patients included a run-in phase with statin therapy.
Conclusions
The LDL-C is a major cardiovascular risk factor, but other lipid variables such as TRG, HDL-C, and Lp(a) also affect cardiovascular risk. PCSK9 inhibitors significantly lower LDL-C concentration. Additionally, these drugs improve modestly the serum concentrations of TRG and HDL-C and more robustly decrease serum Lp(a) levels. Thus, PCSK9 inhibitors improve the overall lipid profile, and these beneficial effects may have played a role on the reductions of cardiovascular outcomes shown in the recently published FOURIER trial.
Footnotes
Authors’ Contribution
Filippatos T. contributed to conception and design, acquisition, analysis, or interpretation, drafted the manuscript, gave the final approval, and agrees to be accountable for all aspects of work ensuring integrity and accurarcy; Kei A. contributed to acquisition, analysis, and interpretation, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Rizos C. contributed to acquisition, analysis, and interpretation, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Ellisaf M. S. contributed to conception and design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This review was written independently. Professor MS Elisaf reports honoraria from MSD, Novartis, Chiesi, Bayer, Astra Zeneca, Pfizer, Abbott, Mylan, Sanofi, Amgen, Boehringer Ingelheim, Eli Lilly, GSK, Angelini, Winmedica and grants and personal fees from MSD and Astra Zeneca. The authors have given talks and attended conferences sponsored by various pharmaceutical companies, including Bristol-Myers Squibb, Novartis, Chiesi, Bayer, Astra Zeneca, Pfizer, Abbott, Mylan, Sanofi, Amgen, Boehringer Ingelheim, Eli Lilly, GSK, Angelini, Winmedica and MSD.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
