Abstract
Background:
Cilostazol is a drug of choice for the treatment of intermittent claudication that also affects innate and adaptive immune cells. The purpose of our study was the evaluation of cilostazol’s impact on the immune and angiogenic response in murine models of hind limb ischemia.
Methods:
We used 108 immunodeficient NOD.CB17-Prkdcscid/J mice and 108 wild-type CB17 mice. At day 0 (D0), all animals underwent hind limb ischemia. Half of them in both groups received daily cilostazol starting at D0 and for the next 7 postoperative days, while the rest of them served as controls, receiving vehicle. Interleukin (IL) 2, IL-4, IL-6, IL-10, IL-17A, tumor necrosis factor α (TNF-α), and interferon γ (IFN-γ) serum concentrations were measured by flow cytometry on postsurgery days D1, D3, D5, and D7. On D7, both groups underwent positron emission tomography scan with 68Ga-RGD. Mice were euthanatized and gastrocnemius muscles were obtained for histological evaluation.
Results:
There was a statistically significant augmentation (P < .05) in IL-4, IL-10, IL-6, and IFN-γ concentrations in treated CB17 animals, while IL-2 was significantly suppressed. Significant difference was detected between the CiBisch and Bisch groups on D1 and D7 (P < .05) in CD31 staining. In treated NOD.CB17 animals, TNF-α, IL-6, and IFN-γ presented significant augmentation, while 68Ga-NODAGA-RGDfK uptake and CD31 expression were found significantly lower for both legs in comparison to the control.
Conclusion:
Cilostazol seems to significantly increase angiogenesis in wild-type animals during the first postoperational week. It also influences immune cells, altering the type of immune response by promoting anti-inflammatory cytokine production in wild-type animals, while it helps toward inflammation regression in immunodeficient animals.
Introduction
Angiogenesis is a physiological process involving the growth of new blood vessels from preexisting vessels, 1 with macrophages 2 and T cells 3 playing an important role in peripheral arterial ischemia and revascularization. Cilostazol is a selective inhibitor of phosphodiesterase 3A (PDE-3α), with proven effectiveness in intermittent claudication (IC) treatment, since it increases the walking distance in IC patients. 4 Cilostazol acts on platelets, vascular smooth muscle cells, and endothelial cells by increasing the levels of cyclic adenosine monophosphate (cAMP), 5 which plays a key role in angiogenesis stimulation and regulates multiple inflammatory mechanisms. 6 In particular, high cAMP levels inhibit leukocyte and platelet adhesion in vitro 7,8 and suppress interleukin (IL) 23 production, 9 while they increase the production of IL-6 and IL-10. 10
Cellular sources of these cytokines are T lymphocytes, mainly CD4+ helper T (Th) cells; this subset can be further subdivided into Th1 and Th2. The produced cytokines are known as Th1-type cytokines (that tend to support pro-inflammatory responses) and Th2-type cytokines that counteract Th1-mediated immune action. However, the precise assessment of immune activation is quite complicated, as it includes additional cytokines such as tumor necrosis factor α (TNF-α), interferon γ (IFN-γ), IL-1, IL-2, IL-6, and IL-10. 11
Molecular imaging of angiogenic biomarkers may offer means for detecting angiogenesis both in native revascularization and in response to proangiogenic therapies. In particular, cell adhesion integrins have been utilized as biomarkers of neovascular proliferation since they control vascular endothelial cell migration and invasion. The αvβ3 integrin is ideally suited for the detection of angiogenesis because expression of integrin αvβ3 receptor quickly increases upon ischemia by activated endothelial cells. In contrast, the receptor is absent on quiescent endothelial cells, and only very low levels thereof are found in normal vessels. 12
Multiple tracers of αvβ3 integrin at different imaging modalities (such as positron emission tomography scan with Gallium) have been used to provide visual information on vascularization, in other clinical entities where angiogenesis may significantly affect their progress, like in cerebral ischemic stroke 13 and peripheral arterial disease (PAD). 14
The purpose of our study was to investigate the impact of cilostazol on revascularization and the functional capacity of ischemic muscles after induction of surgical hind limb ischemia in vivo. We hypothesized the counterregulatory effect of cilostazol on inflammation, and for this purpose, we tested its interaction with innate immunity cells and their derivatives (cytokines) applying a comparative evaluation of immunodeficient and wild-type animal models with hind limb ischemia.
Materials and Methods
Study Design
All experiments were performed in the Biomedical Research Foundation of the Academy of Athens. The facility is registered as “breeding” and “experimental” facility (Reg. Numbers: EL BIO 01 and EL 25 BIO 03, respectively) according to the Greek Presidential Decree 56/2013, which harmonizes National Legislation with the European Directive 2010/63 on the protection of animals used for scientific purposes. All applicable national guidelines for the care and use of animals were followed. The study protocol was approved by the Department of Agriculture and Veterinary Service of the Prefecture of Athens (Permit Number: 6653/17-12-2015). Cages were kept in the same animal room with high efficiency particulate air (HEPA) filtered air supply, 15 air changes per hour (ACH), light intensity of 300 lux measured 1 m above the floor in the middle of the room, and color temperature of 4100 K as well as positive air pressure of 0.6 Pa within the room. Room conditions were continuously monitored through the central building management system of the animal facility.
Animals were bred and maintained in a specific pathogen-free, temperature-, and humidity-regulated unit (21°C ± 2°C; 55% ± 10%), and a 12–12 hour light–dark cycle with lights off at 19:00 hours and no twilight period. Mice were housed in individually ventilated cages (Seal Safe 1284 L, H-Temp; Techniplast, Varese, Italy) receiving 70 air changes per hour, at a stocking density of 4 to 5 mice per cage unless otherwise stated: caging dimensions (L × W × H): 365 mm × 207 mm × 140 mm, floor area = 530 cm2.
All mice had ad libitum access to filtered tap water in drinking bottles and a vacuum-packed pelleted rodent chow that contained 18.5% protein, 5.5% fat, 4.5% fiber, and 6% ash (4RF22; Mucedola, Milan, Italy). The bedding in each cage comprised of ∼250 g of autoclaved corncob bedding (Rehofix MK 2000; J. Rettenmaier & So, Rosenberg, Germany). The cages were cleaned and autoclaved once a week.
A total of 108 immunodeficient NOD.CB17-Prkdcscid/J and 108 wild-type CB17 male mice, 10 to 12 weeks old, were used. We used only male animals to reduce the variability of the outcome caused by sex differences and to thereby decrease group sizes. After surgery, each mouse strain cohort was randomly divided into 2 groups (receiving or not cilostazol; 54 mice/group) and further each group to 4 subgroups (day 1, day 3, day 5, and day 7; 12 mice/time point), and all data collected were analyzed in a blinded manner. NOD.CB17-Prkdcscid/J mice:
Nisch group (n = 54), immunodeficient animals with ischemia and
CiNisch group (n = 54), immunodeficient animals with ischemia administered cilostazol. CB17 mice:
Bisch group (n = 54), wild-type animals with ischemia and
CiBisch group (n = 54), wild-type animals with ischemia administered cilostazol.
Surgical Procedure
All animals underwent hind limb ischemia under general anesthesia with 2% isoflurane (IsoFlo; Abbott, Abbott Park, Illinois) by an experienced veterinary surgeon. Since the model can be affected by bias, every surgical procedure was recorded and evaluated right after the end of the procedure.
Briefly, a small skin incision was made in the right inguinal region (Supplementary Figure 1); the femoral artery was dissected from the nerve and vein; electrocoagulation resulted in permanent occlusion and complete transaction of the right femoral artery. Further, the proximal end of the artery shifted proximally into the surrounding tissue and the distal end shifted distally, creating a distance of 3.1 ± 0.8 mm between both ends of the vessel. At the day of surgery (day 0), 60 mg/kg of cilostazol (Cilostazol, Code: 19340; Urquima S.A., Barcelona, Spain) was administered to all animals of the CiNisch and CiBisch groups via esophageal gavage. These animals continued receiving a daily dosage of 30 mg/kg of cilostazol via esophageal gavage every day postoperatively (D1 to D7; Supplementary Figure 2). The selection of dose was based on previous animal studies. 1,15 Our aim was to obtain a discernible effect with the lowest possible dose, thereby avoiding drug toxicity.
Mice from the specific postoperative day subgroup (D1, D3, D5, and D7) were macroscopically evaluated for limb functionality and ischemic severity every other day postoperatively, that is, on D 1, D3, D5, and D7. In terms of functionality, a pole test was performed to examine the animals’ ability for vertical movement.
At each postoperative day, all mice of the relative group were euthanized by exsanguinations via cardiac puncture after induction of anesthesia with isoflurane 2%. The gastrocnemius muscle (Supplementary Figure 1) was excised and fixed in 10% buffered formalin overnight and then embedded in paraffin blocks. Blood was centrifuged at 8000 rpm for 15 minutes at 6°C, serum was isolated, aliquoted, and stored at −80°C until analysis. On D7, 6 mice were used for the biodistribution study.
Macroscopic Evaluation of Ischemic Severity
During D1, D3, D5, and D7 postoperatively, the ischemic limb was macroscopically evaluated and scored using graded morphological scales for the necrotic area and induced ischemia as follows:
Grading for necrosis: 0, absence of necrosis; I, necrosis limiting to toes (toes loss); II, necrosis extending to a dorsum pedis (foot loss); III, necrosis extending to a crus (knee loss); and IV, necrosis extending to a thigh (total hind limb loss).
Grading for ischemia: 0, no necrosis; 1, one nail discoloration; 2, two or more nail discolorations; 3, one toe discoloration; 4, two or more toe discolorations; 5, foot necrosis; 6, leg necrosis; and 7, autoamputation of leg.
To detect motor deficits due to possible dysfunction caused by ischemia, the pole test was conducted during (D1, D3, D5, and D7) post-surgery days. 16 Mice were placed head upward on the top of a vertical rough-surfaced pole (diameter 8 mm; height 55 cm), and the latency to descend (locomotor activity time) was scored for 3 trials performed for each animal at 15-minute intervals. 17
Vertical movement on poles is not typical, and therefore, we initiated a 3-day training period prior to surgery to accustom mice to the task. Our goal was to ensure that response time was due to limb function rather than unfamiliarity with the test.
Limb functionality was evaluated using the Tarlov scoring system, 18 and results were expressed according to the following grading system: 0, no movement; 1, barely perceptible movement, no weight bearing; 2, frequent and vigorous movement, no weight bearing; 3, weight bearing, 1 or 2 steps; 4, walking with only mild deficit; 5, normal but slow walking; and 6, full and fast walking.
Cytokine Analysis Using the Cytometric Bead Array
Cytokine analysis was performed using the Mouse Th1/Th2/Th17 Cytometric Bead Array kit (BD Biosciences, San Jose, California) which allowed for the simultaneous detection of IL-2, IL-4, IL-6, IL-10, TNF-α, IFN-γ, and IL-17A. Analysis was performed per the manufacturer’s instructions. Briefly, serum samples were thawed and diluted (1:2 vol/vol) with assay diluent, and 50 μL of each diluted sample was added to equal volume of the bead capture mix. Next, 50 μL of phycoerythrin-conjugated detection antibody was added to the sample-capture bead mixture, and samples were incubated for 3 hours in the dark, with occasional shaking. After 2 washing steps, the samples were analyzed on a BD FACS Canto II. In parallel, cytokine standards of known concentrations were run alongside the samples, thus allowing for a standard curve for quantitative analysis. Three separate experiments were performed for each sample. Results were analyzed with FCAP Array Software (BD Biosciences).
Radiolabeling of NODAGA-c(RGDfK) With Gallium-68 and Biodistribution Studies
All commercially obtained reagents and solvents were of analytical grade and were used without further purification. NODAGA-c(RGDfK) was purchased from piCHEM (Raaba-Grambach, Austria). A lower activity commercial 68Ge/68Ga generator (250 MBq) was acquired from Eckert & Ziegler (Berlin, Germany). For the quality testing, high-performance liquid chromatography (HPLC) was performed using a Waters 600 Controller pump, a Waters 996 Photodiode Array detector, and a γ-RAM radioactivity detector to measure radioactive flow on a Waters μ-Bondapack C18 RP (3.9 mm i.d. × 300 mm) cartridge column (Waters, Eschborn, Germany). The HPLC solvents of analytical grade were filtered through 0.22-µm membrane filters (Millipore, Milford, Massachusetts) and degassed before use. The ultraviolet detection wavelength was 295 nm for all experiments, and radioactivity measurements were conducted in an automated well-typed γ-counter NaI(Tl) crystal (Packard). Tissue and blood samples were measured on a Packard COBRA II Auto-Gamma counter (Canberra, Germany).
Gallium-68 was eluted from the 68Ge/68Ga generator with 7 mL 0.1 N HCl and trapped onto an acidic cation exchange resin. Metal impurities (such as Zn, Fe, Ti, and Ge) were removed with washing steps of appropriate buffer solution containing acetone (80% vol/vol) and 0.15 M HCl (20% vol/vol). 18 Subsequent desorption of purified gallium-68 from the cation exchanger was achieved with 400 μL of acetone solution (97.6% vol/vol) containing 0.15 M HCl (2.4% vol/vol). For a typical preparation of gallium-68 labeled NODAGA-RGDfK, 50 μL of NODAGA-RGDfK was mixed with 350 μL of sodium acetate buffer (0.2 mol/L, pH 4) and 100 μL of gallium-68 eluate (∼50 MBq). The mixture was then incubated for 15 minutes at 90°C. For the quality control of radiolabeling, 10 μL aliquots of the reaction solution were analyzed by reverse-phase HPLC (RP-HPLC), applying a linear gradient system at a 1 mL/min flow rate from 0% B to 100% B in 20 minutes, where solvent A = 0.1% trifluoroacetic acid (TFA) in H2O and solvent B = 0.1% TFA in acetonitrile (ACN). The radiolabeled complex was used without further purification. The radiotracer solution for the biodistribution studies was prepared by dilution with water for injection to a final volume of 1.5 mL.
The radiochemical purity of 68Ga-NODAGA-RGDfK, as determined by RP-HPLC, was over 98%. The specific activity of the 68Ga-labeled peptide was approximately 1 GBq/μmol and was intravenously administered via the tail vein. Each mouse received 100 μL/2.0 ± 0.5 MBq/3.47 nmol of the gallium-68-labeled imaging reporter.
The biodistribution study was performed at 30 minutes (n = 6 mice/group). At the end of the protocol, all mice were euthanized by cardiac puncture, and the heart, lungs, liver, kidneys, stomach, intestine, spleen, tail, and left (control) and right (operated) hind limbs were removed, weighed, and counted in an NaI well counter (Packard COBRA II auto gamma counter Canberra, Germany). All measurements were corrected for background and radioactive decay. Tissue distribution data were calculated as the percentage of injected dose per gram (% ID/g), 19 using an appropriate standard. Stomach and intestine were not emptied before measurements. The % ID in whole blood was estimated assuming a whole blood volume of 6.5% of the total body weight of the animals.
Tissue Collection and Histological Analysis
On D1, D3, D5, and D7 postoperatively, the gastrocnemius muscle was excised and fixed in 10% buffered formalin overnight. Tissue samples were then embedded into paraffin blocks. Serial sectioning (5-µm thickness) was performed with a microtome (Leica RM2255—Fully Automated Rotary Microtome, Leica Biosystems GmbH, Wetzlar, Germany). For each staining, 3 nonconsecutive slices at equal intervals of 50 µm were used per mouse, and mounting was applied on poly-D-lysine-coated slides. After rehydration with ethanol solutions of decreasing concentrations, slides were used for conventional hematoxylin and eosin staining.
All slides were observed under ×4, ×10, ×20, and ×40 magnification in a bright-field microscope (Leica DM LS2, Wetzlar, Germany), and digital pictures were acquired and stored in a lossless format using a Leica DC 500 microscope camera (Heerbrugg, Switzerland) and “Altras Soft Imaging System” software (Munster, Germany). Observations and grading of the operated limb on D1, D3, D5, and D7 were performed in all 3 slides per mouse by 1 investigator blinded to the research protocol. Five areas, with cell inflammatory infiltration in each slide, were photographed with a ×20 objective (magnification ×200). Histopathologic assessment was based on the research work of Akdemir et al, 20 and leukocyte infiltration was graded as follows: 0, normal; 1, light infiltration; 2, pronounced infiltration; and 3, massive infiltration. Data were subjected to statistical analysis.
Sections were also used for immunohistochemical staining with antibodies directed against Mac-3 (BD Pharmingen, Franklin Lakes, New Jersey) and CD31 (BD Biosciences). One slide from each animal was analyzed, and a minimum of 10 images were captured using a light microscope with a ×40 objective (magnification ×400). The number of Mac-3+ cells on each slide was manually counted and summed. Data were subjected to statistical analysis, and morphological aspects were qualitatively evaluated as described by Souza et al. 21 Results for CD31 are given as percentage of the positively stained tissue area.
Statistical Analysis
All data were analyzed by GraphPad Prism 6 (GraphPad Software, La Jolla, California) and expressed as mean ± standard deviation. For each experimental day, data were analyzed using the Shapiro-Wilk test to confirm values’ normal distribution. Depending on the results, we used Student t test for 2 samples with 1 variable in case of normal distributions or the nonparametrical Mann-Whitney U test in order to compare the differences in each postoperative day (D1, D3, D5, and D7) between the 2 immunodeficient groups (CiNisch and Nisch). The same analysis was applied in the 2 wild-type groups’ comparison (CiBisch and Bisch). A P value <.05 was considered statistically significant.
Results
Animal Health Status After Surgery
On days D1, D3, D5, and D7 after surgery, all mice were macroscopically evaluated. No signs of infection were observed in any group, a fact that demonstrates no major systemic effect of surgery on the health of these mice. We graded score zero (0) for necrosis, depicting the absence of necrosis for all experimental groups and score zero for ischemia (0), since the animals presented no foot necrosis or toe discoloration (data not shown).
Functional Scoring
In all experimental groups, the active use of the right foot of each mouse improved from D1 to D7, as evidenced by the gradually decreased time required to descend from the top of the vertical pole to the floor. In both immunodeficient and wild-type groups, comparison between cilostazol-treated and untreated animals along time revealed no statistical significance for any of the postoperative days from D1 to D7 (P > .05; Figure 1A).

A, Results of latency in vertical movement for all experimental groups on postoperative days 1, 3, 5 and 7. No statistically significant differences were found between groups (P > .05). B, Limb functionality evaluated by Tarlov scoring system for all experimental groups on postoperative days 1, 3, 5, and 7. No statistically significant differences were found between the groups (P > .05).
Similarly, mice showed improved functional outcomes with accelerated improvement in the Tarlov score during the post-surgery period; however, the addition of cilostazol did not add any benefit (Figure 1B).
Cytokine Profile
We next searched for possible effects of cilostazol in serum levels of TNF-α, IFN-γ, IL-2, IL-4, IL-6, and IL-10 in both NOD-CB17 and CB17 mice. Levels of IL-17A remained near zero value in all experimental groups throughout the study and they were excluded from the analysis.
First, in immunodeficient animals, cytokine analysis revealed statistically significant differences in TNF-α, IL-6, and IFN-γ levels between the CiNisch and Nisch groups following a similar pattern. In particular, the concentrations of the aforementioned cytokines were abruptly increased on D1 in the CiNisch group compared to Nisch group (P < .001) and were gradually decreased along time till D7, when low levels of these 3 cytokines were detected in both groups. Concentrations of IL-4, IL-2 and IL-10 did not differ between groups during all days of experimentation.
Regarding wild-type CB17 animals, IL-6 and IFN-γ were significantly elevated in CiBisch rather than Bisch mice during the experimentation period (P < .001). Tumor necrosis factor α showed a delayed increment on D5 and D7 in the CiBisch group (P < .001). Interleukin 4 was found significantly higher in the cilostazol-treated group of wild-type mice but only on D1 (P < .001) along with IL-10 levels from D1 to D5 (P < .001). Contrarily, the Bisch group showed higher IL-2 levels than the CiBisch group on D1, D3, and D5. All data are presented in Figure 2.
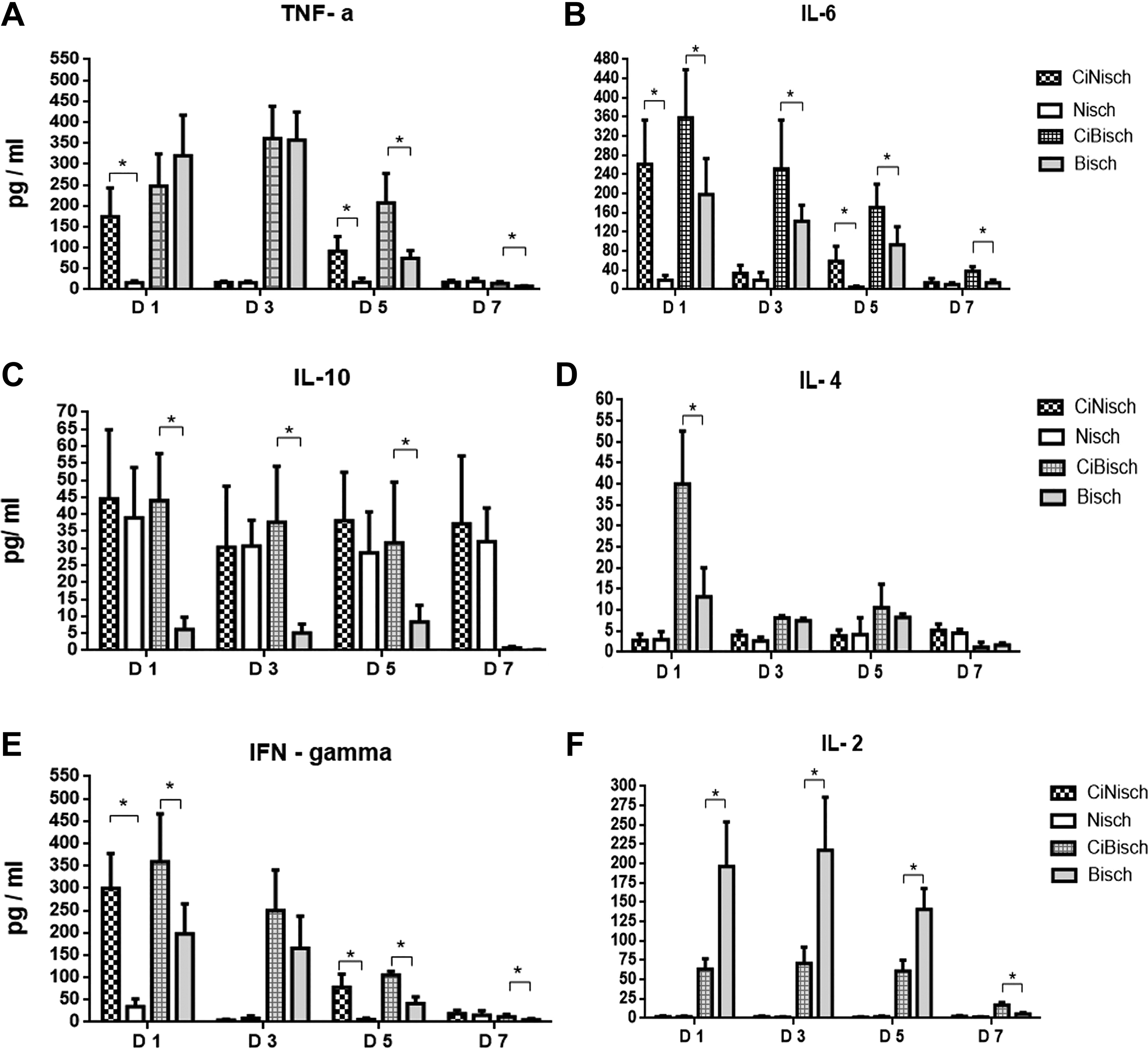
Cytokines plasma levels measured in all experimental groups on postoperative days 1, 3, 5 and 7. A, For tumor necrosis factor α (TNF-α) concentrations, statistically significant differences were observed between CiNisch and Nisch groups on D1 and D5 (P < .001) as well as between CiBisch and Bisch groups on D5 and D7 (P < .001). B, For interleukin (IL) 6 concentrations, statistically significant differences were observed between CiNisch and Nisch groups on D1 and D5 (P < .001) as well as between CiBisch and Bisch groups on all postoperative days (P < .001). C, For IL-10 concentrations, no statistically significant differences were observed between CiNisch and Nisch groups on each postoperative day (P > .05), while between CiBisch and Bisch groups, there were statistically significant differences on D1, D3, and D5 (P < .001). D, For IL-4 concentrations, no statistically significant differences were observed between CiNisch and Nisch groups on each postoperative day (P > .05), while between CiBisch and Bisch groups, statistically significant differences were observed on D1 (P < .001). E, For IFN-γ concentrations, statistically significant differences were observed between CiNisch and Nisch groups on D1 and D5 (P < .001) as well as between CiBisch and Bisch groups on D5 and D7 (P < .001). F, For IL-2 concentrations, statistically significant differences were observed between CiNisch and Nisch groups on D1 and D5 (P < .001) as well as between CiBisch and Bisch groups on all postoperative days (P < .05).
Biodistribution of 68Ga-NODAGA-RGDfK
The results for the ex vivo biodistribution of 68Ga-NODAGA-RGDfK are presented in Table 1. Nisch group showed a significantly higher uptake of 68Ga-NODAGA-RGDfK for both legs than their cilostazol-treated counterparts (left hind limb P < .001 and right hind limb P < .001). The uptake values for all other tissues (heart, liver, spleen, lung, stomach, and intestine) were slightly higher in CiNisch mice, with the exception of kidneys, where the differences in mean uptake values achieved statistical significance (P < .05).
Ex Vivo Biodistribution of 68Ga-NODAGA-RGDfK in all Experimental Groups at the End of the Study.a

a Data are presented as the percentage injected dose per gram (% ID/g ± standard deviation, SD) for each tissue evaluated.
Contrary to immunodeficient mice, the uptake of 68Ga-NODAGA-RGDfK in wild-type mice was higher in cilostazol-treated animals than controls in both legs (left hind limb, P = .048 and right hind limb P > .05). Besides, spleen uptake of the peptide was significantly increased in the CiBisch rather than the Bisch group (P < .001). No statistical difference was found for the remaining tissues evaluated between the CiBisch and Bisch groups (P > .05).
Histological Evaluation
Hematoxylin and eosin staining of gastrocnemius muscles in immunodeficient animals revealed a marked increase in leukocyte infiltration in the Nisch group on D3 and D7 compared to CiNisch (Figures 3A and 4).

A, Leukocyte infiltration measured in all experimental groups on postoperative days 1, 3, 5, and 7. A statistically significant difference was observed between CiNisch and Nisch groups on D3 and D7 (P < .05), while there was no statistically significant difference between CiBisch and Bisch groups on each postoperative day (P > .05). B, Immunohistochemical staining with antibodies against CD-31 in all experimental groups on postoperative days D1, D3, D5, and D7. A statistically significant difference was observed between CiNisch and Nisch groups on D1, D5, and D7 (P < .05), while between CiBisch and Bisch groups, there was a statistically significant difference on D1 and D7 (P < .05). C, Macrophage numbers measured in all experimental groups on postoperative days 1, 3, 5, and 7. A statistically significant difference was observed between CiNisch and Nisch groups on D1 and D7 (P < .05), while between CiBisch and Bisch, there was a statistically significant difference on D7 (P < .05).

Representative pictures of the histological analysis of the right gastrocnemius muscle for CiNisch (panels A-D) and Nisch groups (panels E-H) hematoxylin and eosin (H&E) staining (×20). Leukocyte infiltration was evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.
Statistically significant difference was observed between the CiNisch and Nisch groups during D1, D5, and D7 (P < .05) in immunohistochemical staining with an antibody against CD31 (Figures 3B and 5). Accordingly, macrophages stained with Mac-3 were found more abundant in untreated animals on D1 (P < .05) and D7 (P < .01; Figures 3C and 6).

Immunohistochemical staining with antibody against CD31 for CiNisch (panels A-D) and Nisch groups (panels E-H). Sections were counterstained with hematoxylin and eosin (H&E) staining (×20). Results are given as percentage of the positively stained tissue area and were evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.

Immunohistochemical staining with antibody against macrophages for CiNisch (panels A-D) and Nisch groups (panels E-H). Sections were counterstained with hematoxylin and eosin (H&E) staining (×20). Macrophage numbers were evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.
Moreover, in wild-type animals, leukocyte infiltration was low with less extensive inflammatory areas, and no differences were detected between the Bisch and CiBisch groups (Figures 3A and 7). There was also a statistically significant difference between the CiBisch and Bisch groups on D1 and D7 (P < .05) in immunohistochemical staining with an antibody against CD31 (Figure 3B and 8). On the other hand, macrophage (Mac-3+) infiltration was considerably increased in untreated animals only on D7 (Figure 3C and 9).

Representative pictures of the histological analysis of the right gastrocnemius muscle for CiBisch (panels A-D) and Bisch groups (panels E-H) hematoxylin and eosin (H&E) staining (×20). Leukocyte infiltration was evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.
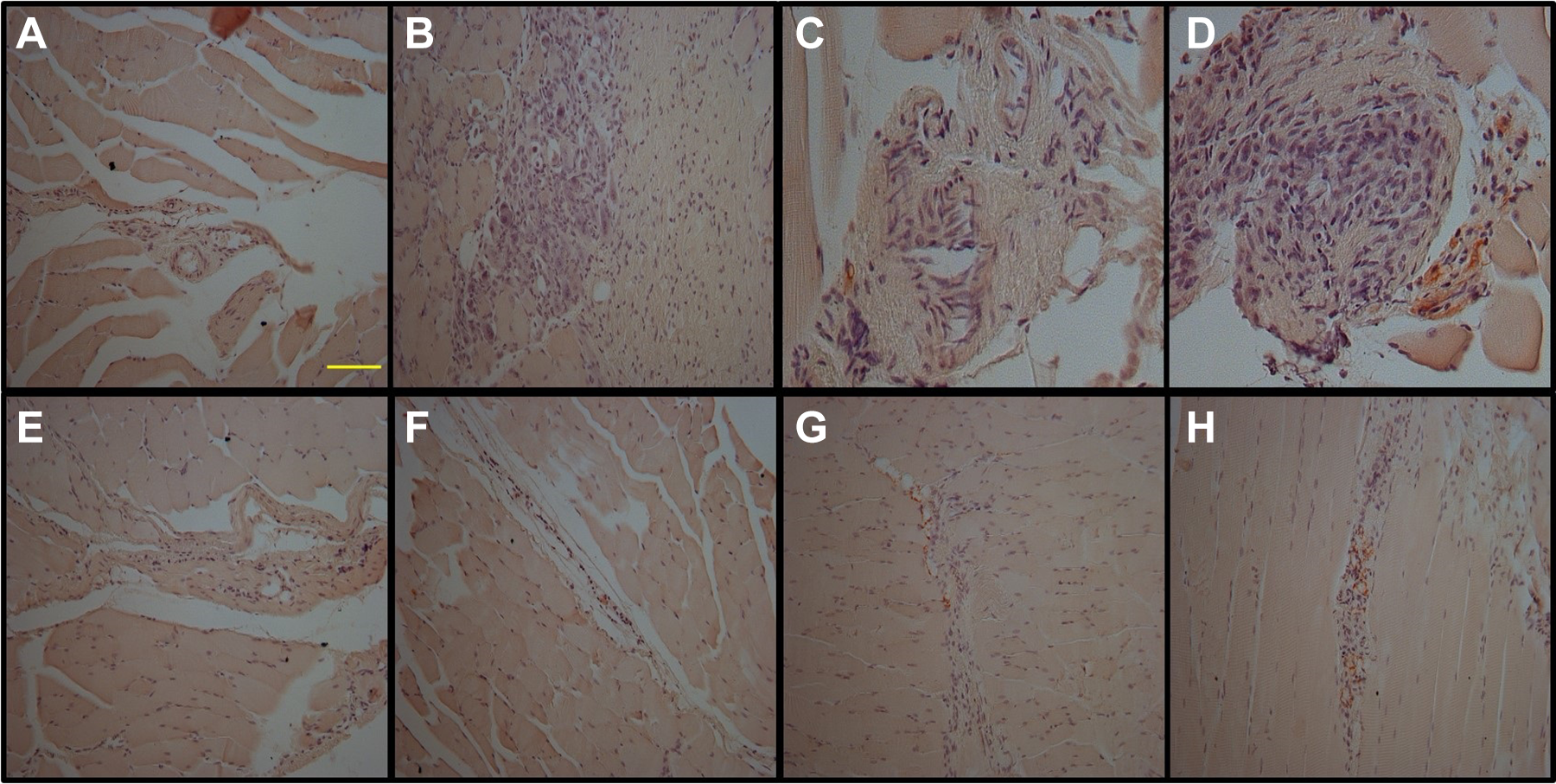
Immunohistochemical staining with antibody against CD31 for CiBisch (panels A-D) and Bisch groups (panels E-H). Sections were counterstained with hematoxylin and eosin (H&E) staining (×20). Results are given as percentage of the positively stained tissue area and were evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.

Immunohistochemical staining with antibody against macrophages for CiBisch (panels A-D) and Bisch groups (panels E-H). Sections were counterstained with hematoxylin and eosin (H&E) staining (×20). Macrophage numbers were evaluated on each postoperative day (D1: A and E; D3: B and F; D5: C and G; and D7: D and H), scale bar = 0.1 mm.
Discussion
To our knowledge, this is the first study to mechanistically investigate the impact of cilostazol on inflammation and angiogenesis during the acute phase of experimental peripheral arterial ischemia. In immunodeficient mice (lacking functional B and T cells) with hind limb ischemia, cilostazol therapy was associated with suppressed inflammation and angiogenesis, while in wild-type mice, cilostazol therapy induced angiogenesis and blunted the inflammatory response compared to untreated counterparts.
We conducted a variation in the experimental protocol of Goto et al, 22 producing the least possible ischemic damage in order to avoid mortality of immunodeficient animals induced by extensive ischemic damage. 3 The severity of ischemia was confirmed with the clinical and functional evaluation of the operated leg. Over the 7 postoperative days, the gradually decreased time needed by the animals to descend the pole validates the functional deficit of the limb.
This work used 68Ga-NODAGA-RGDfK binding in endothelial cell receptors as a proxy for early proangiogenic events. The potential of angiogenesis requires monitoring of later stage events such as the generation of perfusable vessels. In this context, we examined the gastrocnemius muscle for in vivo angiogenesis at early stages by detecting αvβ3 integrin receptor with 68Ga-NODAGA-RGDfΚ. This is a very sensitive and accurate method for the quantitative evaluation of angiogenesis development, along with the semiquantitative method of immunohistological detection of CD31 expression, a molecule signifying the formation of new vessels. We assumed that distal ischemic tissue provides more accurate manifestations of angiogenesis in PAD.
Regarding the angiogenic effects exerted by cilostazol via immune system components, we focused on changes in cytokine secretion and inflammatory cell infiltration, both of which regulate angiogenesis at a very early stage.
Previous evidence has connected the anti-inflammatory action of cilostazol with inhibition of plasmacytoid dendritic cell activation, antigen presentation by peripheral blood mononuclear cells, 6 T-cell proliferation, and IFN-γ, IL-17, and TNF-α production. 23 In agreement to that, our cilostazol-treated immunodeficient animals showed remarkably reduced inflammatory cell infiltration.
The connection between the anti-inflammatory effects of cilostazol and the lack of functional improvement can be explained by the relatively small degree of injury caused by the surgery. The limb was functional (at least partially), and this is because it is nourished by collateral circulation via the deep femoral artery and other branches; therefore, as expected, occlusion of the femoral artery did not cause severe ischemia.
One important finding of our study was the abrupt increase in TNF-α, IL-6, and IFN-γ blood levels shortly after cilostazol administration, which was gradually alleviated (Figure 2). This may implicate an acute pro-inflammatory cytokine release by cilostazol, which does not persist but requires further investigation.
Concerning the absence of anti-inflammatory response in immunodeficient animals, this is obviously attributed to the lack of their ability to regulate the production of anti-inflammatory (IL-4 and IL-10) cytokines.
In cilostazol-treated wild-type mice, we found a completely different response compared to their immunodeficient counterparts. With the exception of IL-2, all other cytokines (IL-4, IL-10, IL-6, and IFN-γ) showed significant augmentation.
By design, we assessed the contribution of the immune system to the possible angiogenic effects of cilostazol. For this purpose, we used an immunodeficient along with a wild-type murine model. According to literature, angiogenesis was retarded in athymic C57BL/6J-nu mice when femoral arterial resection led to extensive limb necrosis in 10 days. Recovery of blood flow remained severely impaired, resulting in toe necrosis later on. 3 This study underscored the contributory role of T-cell infiltration to angiogenesis in a severe limb ischemic environment leading to necrosis. Similarly, in other immunodeficient models of arteriogenesis, C57BL/6 CD4−/− 24 and C57BL/6 CD8−/− 25 mice appeared with reduced collateral flow in comparison to wild-type groups, during D7 postoperatively of a relevant ischemic muscle induction. Our results are complementary to the abovementioned reports, since this is the first study of hind limb ischemia without any leg foot or toe necrosis in an immunodeficient animal model. Our effort was targeted in the study of the pro- and inflammatory environment that leads to the regulation of angiogenesis in its very early stage. Indeed, mice lacking functional immune cells showed increased angiogenetic response to mild hind limb ischemia. Surprisingly, this response was suppressed by cilostazol administration.
Cilostazol is well known for its anti-inflammatory actions and that was confirmed in our study with the downregulation of inflammatory cell infiltration in all treated groups. It seems that, besides its anti-inflammatory effects, cilostazol may trigger Th2 response over Th1, as depicted by the persistently high concentrations of IFN-γ and other pro-inflammatory cytokines (eg, IL-6, TNF-α), which mediate the promotion of angiogenesis in wild-type animals suffering from peripheral ischemia. Additionally, we observed increased expression of CD31 in wild-type animals (Figure 8). These findings are in accordance with previous study, 26 where Th2 and Th17 conditioned media enhanced angiogenesis and blood perfusion in ischemic injuries, in part due to their ability to directly stimulate endothelial sprouting. 26 In immunodeficient mice, where Th1/Th2 response is absent, cilostazol may even induce vascular regression. In the absence of Th2-biased response, endothelial sprouting is not properly regulated, possibly leading to nonfunctional angiogenesis and lower blood flow as is mentioned in various reports. 24,25 One other possible explanation could be the reduced sensitivity of vascular endothelial cells to PDE inhibitors. 7,27 Therefore, the reduced production of vascular endothelial growth factor from endothelial and mesothelial cells may explain the low αvβ3 and CD31 expression (Figure 5). Another plausible explanation for the discrepancy of findings between immunodeficient and wild-type animals in the present study may also be related to the ambiguous role of IFN-γ. In some studies, IFN-γ induced vascular regression only in activated endothelial cells in tumor vasculature and not in preexisting normal blood vessels, 28 while it enhanced angiogenesis in the presence of proangiogenic M1 macrophages. 29 In other studies, M1/Th1 responses have been considered to be “antiangiogenic,” 30 while further reports have shown that M1 macrophages are proangiogenic. 31 It is unclear whether the suppressed macrophages in our cilostazol-treated groups were pro- or antiangiogenic.
Histological analysis revealed a common pattern of gradual increase in macrophage infiltration which peaked at D7, a phenomenon that was more pronounced in immunodeficient than in wild-type mice. Particularly, in immunodeficient mice, cilostazol had a greater effect on the reduction in the observed inflammation by reducing significantly the extent of macrophage infiltration.
Although we tried to design an optimal model for testing the effect of cilostazol in peripheral ischemia, our study has 2 major limitations. First, with relevance to the age and health status of the animals, our study was performed on healthy young mice, while in most cases of severe PAD, patients are aged and PAD is accompanied with underlying diseases such as diabetes, hypercholesterolemia, and hypertension. The extrapolation of our findings to humans should be performed with caution since there is no variation in collateral circulation in mice, while it greatly varies between humans. 32 Second, we did not evaluate the functional capacity of the infiltrating inflammatory cells which could better highlight the impact of cilostazol on Th1/Th2 response along with the use of the semiquantitative scoring systems for the histological evaluation.
Conclusion
In conclusion, cilostazol seems to significantly affect immune cells and promote the production of anti-inflammatory cytokines and the suppression of IL-2 in wild-type animals, altering the type of the immune response, while our cilostazol-treated immunodeficient animals showed an abrupt increase in TNF-α, IL-6, and IFN-γ blood levels shortly after cilostazol initiation, which gradually alleviated. The cilostazol-induced reduction in reduced inflammatory cell infiltration requires further investigation.
Regarding the angiogenic effects of cilostazol, the immune system seems to play a mediating but still not well-defined role. Although there is a significant association between anti-inflammatory and angiogenic effects of cilostazol, the presence of an “inflammatory environment” is a prerequisite background for cilostazol to exert its action. Unambiguously, our preliminary data require further investigation.
Supplemental Material
supplementary_file - Cilostazol Mediates Immune Responses and Affects Angiogenesis During the Acute Phase of Hind Limb Ischemia in a Mouse Model
supplementary_file for Cilostazol Mediates Immune Responses and Affects Angiogenesis During the Acute Phase of Hind Limb Ischemia in a Mouse Model by Efthymios Paronis, Michalis Katsimpoulas, Nikolaos P. E. Kadoglou, Claire Provost, Marianna Stasinopoulou, Christos Spyropoulos, Elpida Poulaki, Aurelie Prignon, Ioannis Kakisis, Nikolaos G. Kostomitsopoulos, Penelope Bouziotis, Ioannis V. Kostopoulos, Ourania Tsitsilonis and Andreas Lazaris in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
The authors would like to thank Libytec Pharmaceuticals Company for providing the pure cilostazol substance. The authors would like to acknowledge Mr Stavros Xanthopoulos (Institute of Nuclear & Radiological Sciences & Technology, Energy & Safety, NCSR Demokritos) for excellent technical assistance.
Author Contributions
Katsimpoulas and Kadoglou contributed equally to study conduction. Paronis and Katsimpoulas contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Kadoglou, Provost, Spyropoulos, Poulaki, Prignon, Bouziotis, and Kostopoulos contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Stasinopoulou contributed to acquisition, analysis, or interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Kakisis, Kostomitsopoulos, Tsitsilonis, and Lazaris contributed to conception or design; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
