Abstract
Objective:
Abdominal aortic aneurysm (AAA) is a common condition that may be life-threatening when it is unrecognized. The aim of this study is to evaluate and compare the efficacy of ramipril and carvedilol on limiting AAA expansion in mouse model.
Methods and Results:
A total of 36 experimental AAA mouse model was induced with the continuous infusion of angiotensin II (Ang II) in 20-week-old male apolipoprotein E-deficient mice. They were randomly divided into 3 treatment groups and fed orally for 8 weeks; saline alone, ramipril (2.5 mg/30g/d), or carvedilol (3.125 mg/30g/d), respectively. Aortic diameter (AD) was measured by micro-computed tomography, and the level of biomarkers of aortic tissue such as monocyte chemoattractant protein-1 (MCP-1) and tissue inhibitor matrix metalloproteinase-1 (TIMP-1) was evaluated. After treatment, AD of both ramipril and carvedilol group was smaller than in the saline group. The percentage change of AD in both ramipril and carvedilol groups was significantly smaller than that of the saline group. Pathologic examination revealed relatively well-preserved aortic walls in the ramipril group compared to the carvedilol and saline groups. The level of MCP-1 was markedly decreased in both the ramipril and carvedilol groups compared to the saline group. The level of TIMP-1 was higher in the carvedilol group when compared to either the saline or ramipril groups.
Conclusions:
Ramipril and carvedilol treatment shows similar efficacy in limiting AAA expansion in mouse model. Future clinical research would be warranted to validate these results.
Introduction
Abdominal aortic aneurysms (AAAs) are defined as permanent and localized aortic dilations having diameters 1.5-times greater than normal (ie, more than 3 cm diameter in humans). 1 When an AAA ruptures, regardless of its size, the overall mortality remains at approximately 80% to 90% despite recent medical and technical advancements. 2,3 Surgical or endovascular repair has been well-established as a treatment option when an AAA is enlarged more than 5.5 cm in diameter. However, these invasive strategies do not provide enough therapeutic advantage over aggressive medical management in the case of smaller AAAs. 4,5 Moreover, there is little data for effective medical treatment in limiting the growth of AAA in patients with smaller AAAs.
Several approaches including hemodynamic management, inhibition of inflammation in atherosclerosis, and protease inhibition 1,6 -8 have been proposed in preventing AAA progression and rupture in addition to addressing various pathogenetic factors of aneurysm development. 9 Some studies have demonstrated that antihypertensive agents such as angiotensin converting enzyme (ACE) inhibitors and β-blockers have some efficacy on suppressing induced aneurysms in animal models and human studies. 10 -15
Angiotensin converting enzyme inhibitors have been shown to demonstrate pleiotropic and anti-inflammatory effects to reduce vascular inflammation on the atherosclerotic process independent of their antihypertensive effects. 13,16,17 However, it still remains to be determined how ACE inhibitors suppress AAA expansion and whether these observations are clinically applicable. 18,19
β-Blockers are thought to have some favorable hemodynamic effects on limiting aortic root dilatation 20,21 and suppressing AAA expansion in animal and/or human models. 22,23 However, a randomized controlled trial with the nonselective β-blocker, propranolol, has failed to show any positive efficacy because of several side effects and a high mortality rate. 24 More recently, third generation β-blockers such as carvedilol and nebivolol with higher cardiac selective, vasodilatory, and anti-inflammatory properties have shown more favorable outcomes on aortic elastic properties. 25
Despite some controversies regarding the pharmacological benefits of ACE inhibitors and newer β-blockers, there is little data demonstrating efficacy in patients with AAA. 16
In this study, we intended to investigate and compare the therapeutic effects of ramipril and carvedilol on the process of AAA expansion in a mouse model using staged micro-computed tomography (CT), histopathological examination, and biochemical analysis.
Materials and Methods
Animals
Male apolipoprotein E-deficient (apoE−/−) mice with a C57BL/6 background were obtained from the Central Animal Laboratory from Japan SLC, Inc (Hamamatsu, Japan). All mice were bred in a pathogen-free environment and also maintained on normal mouse chow diets under barrier conditions. They also had arbitrary access to water in controlled temperature facilities. 18,26 The animal care and all experimental procedures were approved by the Guidelines on Animal Experiments of Institutional Animal Care and Use Committee of Yonsei University Severance Hospital (Approval No. 2012-0158). All animal experiments were also performed in accordance with the Korean Government Animal Protection and Management Law.
Induction of AAA by Infusion of Angiotensin II
Alzet miniosmotic pumps (Model 2004; DURECT Corporation, California) were implanted into the 20-week-old C57BL/6 apoE−/− male mice. The minipumps were inserted into a subcutaneous space in the back of the neck utilizing microsurgery under general anesthesia with ketamine using a previously established protocol. 26,27
The implanted minipumps were filled with a solution of Angiotensin II (Ang II, CalBiochem, La Jolla, California) and were designed to deliver it subcutaneously 1000 ng/min/kg for 4 weeks to promote the development of an atherosclerotic aortic aneurysm (Figure 1 A). 26,28 -30

A, Overall experimental design for treatment and analysis of mouse AAA model: angiotensin II (Ang II)-induced AAA mice are randomly assigned to 3 treatment groups including saline, ramipril, and carvedilol. Vital sign measurement, micro-CT, histopathologic examinations, and biomarker evaluation are performed at each stage. B, The induced mouse AAA by Ang II infusion for 4 weeks by gross autopsy (left panel) and serial micro-CT for size measurement (right panel): Yellow circle indicates aorta at baseline (apoE−/−) and red circle indicates Ang II-induced AAA. AAA indicates abdominal aortic aneurysm; CT, computed tomography.
Treatment Drugs
After successful formation of the suprarenal AAA by Ang II infusion for 4 weeks, 36 mice with AAA were divided into 3 experimental groups, each group consisting of 12 AAA mice. The control group was treated with normal saline of 0.2 mL/30g/d. The ACE inhibitor group was treated with ramipril 4.2 mg/30g/d. The β-blocker group was treated with carvedilol 3.125 mg/30g/d. Each drug was suspended in 0.5% methylcellulose and administered by gavage for 8 weeks. 16 Each dose of test drug was consistent with other experimental studies in mice in which these agents had exerted significant pharmacologic effects. 18
Experimental Parameters
During the overall experimental period, various examinations including vital sign measurement, micro-CT for aortic size, histologic staining, and biochemical tests were performed and analyzed at each stage (Figure 1 A).
Vital sign measurement
Systolic blood pressure (SBP), diastolic blood pressure (DBP) and heart rate were measured on the conscious mice by a tail cuff photoplethysmogrpahy using the BP-2000 Blood Pressure Analysis System (Visitech Systems, Apex, North Carolina). Systolic blood pressure, DBP, and heart rates were measured repeatedly prior to Ang II administration as well as before and after the medical intervention period. Pulse pressures were also calculated of each stage.
Measurement abdominal aortic diameter by micro-CT
Micro-CT scans were performed to get aortic images using the NFR Polaris G90 (Nano Focus Ray, Seoul, Korea) on the mice under anesthesia. To acquire the tomographic datasets and to optimize micro-CT scan for AAA lesions in the mice, different scanning parameters (exposure time, number of projections, and beam conditions) were evaluated; the optimized scans were performed with constant source conditions of 65 kV and 115 μA. To assess the postscanned specimen, the radiographic data sets were imported into the hardware-based back projection reconstruction software supplied by NFR 4D-CT Control, producing an image series of 16-bit axial slices with slice-spacing equivalent to the pixel size for each sample. The pre/postenhanced micro-CT images were reviewed and measured by an independent radiologist (Figure 1 B).
Histopathological evaluation and immunohistochemical staining
Microsection of aortic tissue in a formalin-fixed paraffin-embedded block was performed for histopathological examination. They were stained with the hematoxylin–eosin (H-E), elastic-Verhoeff’s Van Gieson (EVG) for elastin fibers, and trichrome (TRC) staining for collagen at each stage of the baseline (apoE−/−), after 4 weeks of Ang II infusion, and after 8 weeks of treatment with either vehicle (saline) or test drugs (ramipril and carvedilol). Additionally, immunohistochemical staining for α-smooth muscle actin (α-SMA) using Actin antibody (1:200; cat ab5694 Abcam, Cambridge, Massachusetts) was performed with the Ventana BenchMark XT autostainer (Ventana Medical System, Inc, Tucson, Arizona). In addition, CD68 immunohistochemical staining for macrophages was performed, and the findings were checked each on an average field of ×200 and ×400 to count the number of macrophages. Histopathological image analysis was performed to confirm AAA development and to evaluate the therapeutic effect on AAA expansion by estimating the distribution of inflammatory cells and elastic fiber grade, collagen infiltration, smooth muscle proliferation, dissected aneurysm, and intimal proliferation. 31 All the histopathological stained slides were referred for semiquantitative analysis under light microscopy by an independent pathologist without any experimental knowledge (Kim S. H.).
Enzyme-linked immunosorbent assay and cytokine assay for associated biomarkers
Frozen aortic tissue in liquid nitrogen was homogenized with lysis buffer and incubated on ice for 30 minutes, after which it was centrifuged at 12 000 rpm for 15 minutes. The supernatant was used for an enzyme-linked immunosorbent assay (ELISA) test of MCP-1 (a pro-inflammatory marker) and tissue inhibitors of metalloproteinases-1 (TIMP-1, an endogenous inhibitor of the matrix metalloproteinase [MMP]). 32 -34 The concentration of MCP-1 and TIMP-1 in aortic tissue was measured using the mouse MCP-1 ELISA kit (Cat No. EMMCP1; Thermo scientific, Waltham, Massachusetts) and the TIMP-1 ELISA kit (Cat No. MTM100; R&D systems, Minneapolis, Minnesota), respectively.
Statistical Analysis
All values are expressed as mean (standard deviation [SD]). The null hypothesis for the variables measured in each treatment group and other statistical differences among the groups are determined by the analysis of variance test for normally distributed data. A comparison among different treatment groups is performed by the Kruskal-Wallis test in case of non-normally distributed continuous variables. Statistical significance is defined at P value < .05. If the difference among treatment groups is not significant and the data do not fit to constraints of the analysis form, post hoc testing was performed using the parametric testing for multiple groups. All statistical analyses were performed with SPSS 21 (SPSS Inc, Chicago, Illinois).
Results
Changes in vital sign: blood pressures, heart rates, and pulse pressure
Vital signs including both blood pressures (SBP/DBP) and heart rates were measured using tail-cuff plethysmography before and after 8 weeks of drug treatment to evaluate any correlation between the aneurysm-suppressing effect and the hemodynamic changes induced by the study drugs. The SBP in both ramipril and carvedilol groups was significantly lower while it did not change in the saline-treated mice. There was no significant difference in the DBP among treatment groups. Heart rate in the carvedilol group was markedly decreased compared to the saline and ramipril groups. In addition, the pulse pressure in the ramipril and carvedilol groups was also significantly lower than in the saline group. The lowest pulse pressure was observed in the carvedilol group (Figure 2).

Changes of vital signs including blood pressure, heart rate, and pulse pressure. A, Systolic blood pressure after 8-week treatments with ramipril and carvedilol decreases compared to the treatment with saline. B, Diastolic blood pressure after treatments are not different between groups. C, Heart rate markedly decreases in carvedilol treatment group. D, Pulse pressure significantly decreases in ramipril and carvedilol treatment group than saline group. *P < .01, **P < .05.
Changes in abdominal aortic diameter: micro-CT angiography findings
The mean aortic diameter (AD) consistently increased by more than 50% over baseline as seen on micro-CT performed at the end of the continuous infusion of Ang II for 4 weeks (baseline: 0.93 ± 0.07; Ang II: 1.46 ± 0.35 mm, P < .001; Figure 3 A). After 8 weeks of treatment, mean AD on the micro-CT was significantly increased in saline group (Ang II: 1.46 ± 0.35; saline: 3.60 ± 6.25, P = .011). The mean AD of both the ramipril and carvedilol groups was significantly smaller than that of the saline group (saline: 3.60 ± 6.25; ramipril: 1.51 ± 0.31; carvedilol: 1.53 ± 0.53 mm, P = .034). There was no statistical difference in the average AD between mice treated with ramipril and carvedilol. The percentage change in AD in both the ramipril and carvedilol groups was significantly smaller than that of the saline group (saline: 28.92% ± 23.47%; ramipril: 2.01% ± 9.56%; carvedilol: 8.50% ± 12.73%, P = .002, Figure 3 B).
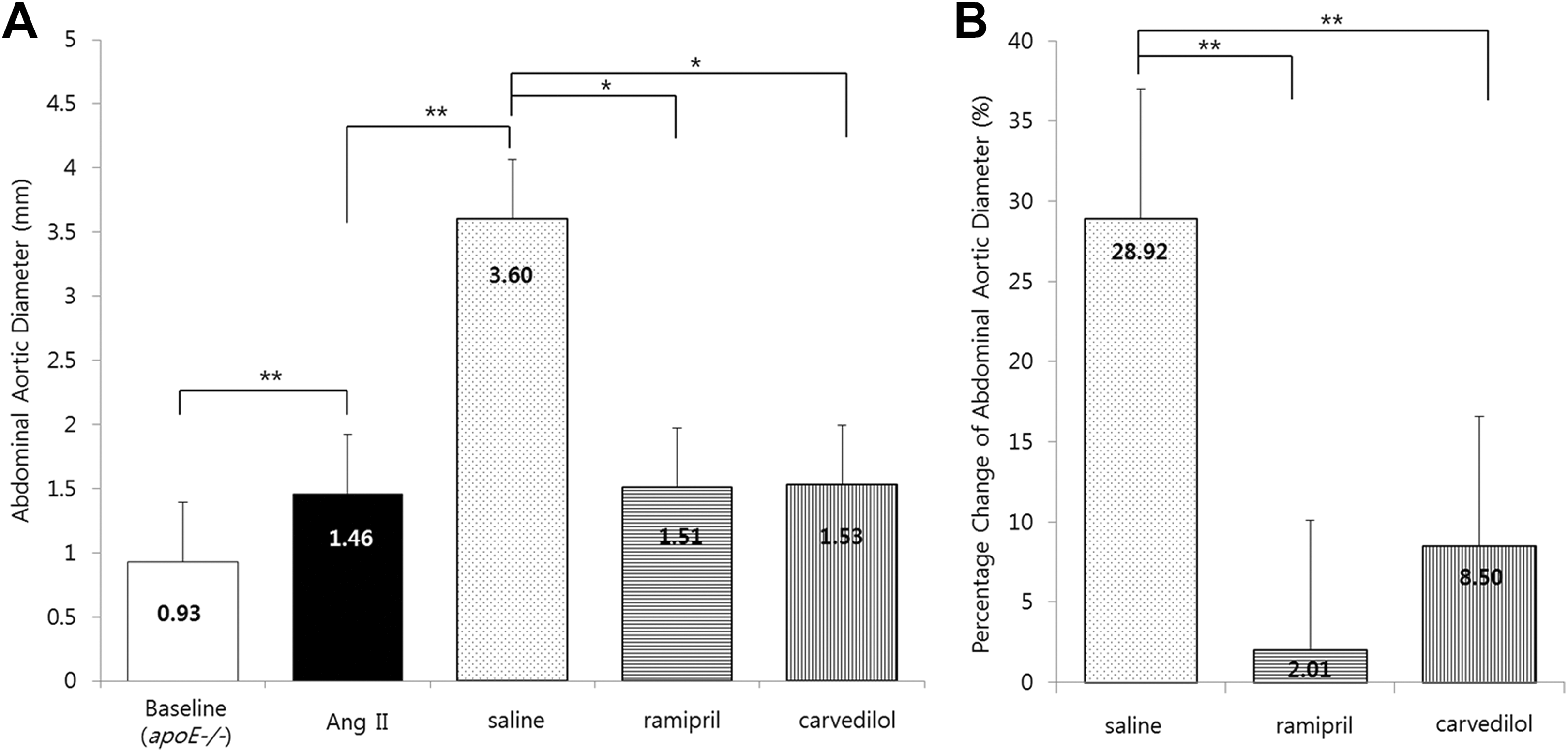
Changes of aortic diameter (AD) at each stage using micro-computed tomography(CT) measurement. A, Mean AD is markedly increased at angiotensin II (Ang II) state compared to baseline. After 8-weeks treatment, mean AD is smaller in ramipril and carvedilol than saline group. B, The percentage changes of aortic diameter (%ΔAD) are also smaller in ramipril and carvedilol than saline group.
Histopathological examination
To find any changes in the histopathological characteristics of the aortic tissues by the Ang II-induced AAA development or by pharmacological treatment, histopathological examinations with various staining were analyzed. At the end of Ang II infusion, the histolopathological examination by H-E, EVG, TRC, and α-SMA demonstrated aneurysmal dilatation which was associated with a chronic transmural inflammatory response and extensive destruction of the medial elastic lamellae when compared to the pathology of the initial baseline aorta (apoE−/−). After the 8-week treatment period, the pathological characteristics among treatment groups were comparable. On examining the transverse sections of aortic tissue, both drug and saline treatment groups showed medial and adventitial thickening with fibrosis due to wall injury when compared to the baseline (Figure 4). From the high-power field of EVG staining, relatively well-preserved aortic wall elastin was observed only in the ramipril group. In addition, a slight regression of thickened media with unstable plaques was noted in the carvedilol group (Figure 5). From CD68 immunohistochemial staining, the number of macrophages which are regulated by MCP-1 in ramipril and carvedilol group is significantly lower than in saline group (saline: 292.00 ± 73.02, ramipril: 100.00 ± 21.63, carvedilol: 74.67 ± 29.27, P = .003).

Histopathology of transverse sections of formalin-fixed aortic tissue with low power field (H-E, EVG, TRC, and α-SMA, 12 times LPF). Angiotensin II (Ang II) infusion state and all treatments group with saline, ramipril, and carvedilol show medial and adventitial thickening with fibrosis due to wall injury compared to b(apoE−/−) stage. H-E indicates hematoxylin-eosin; EVG, elastic-Verhoeff’s Van Gieson; TRC, trichrome; α-SMA, α-smooth muscle actin.
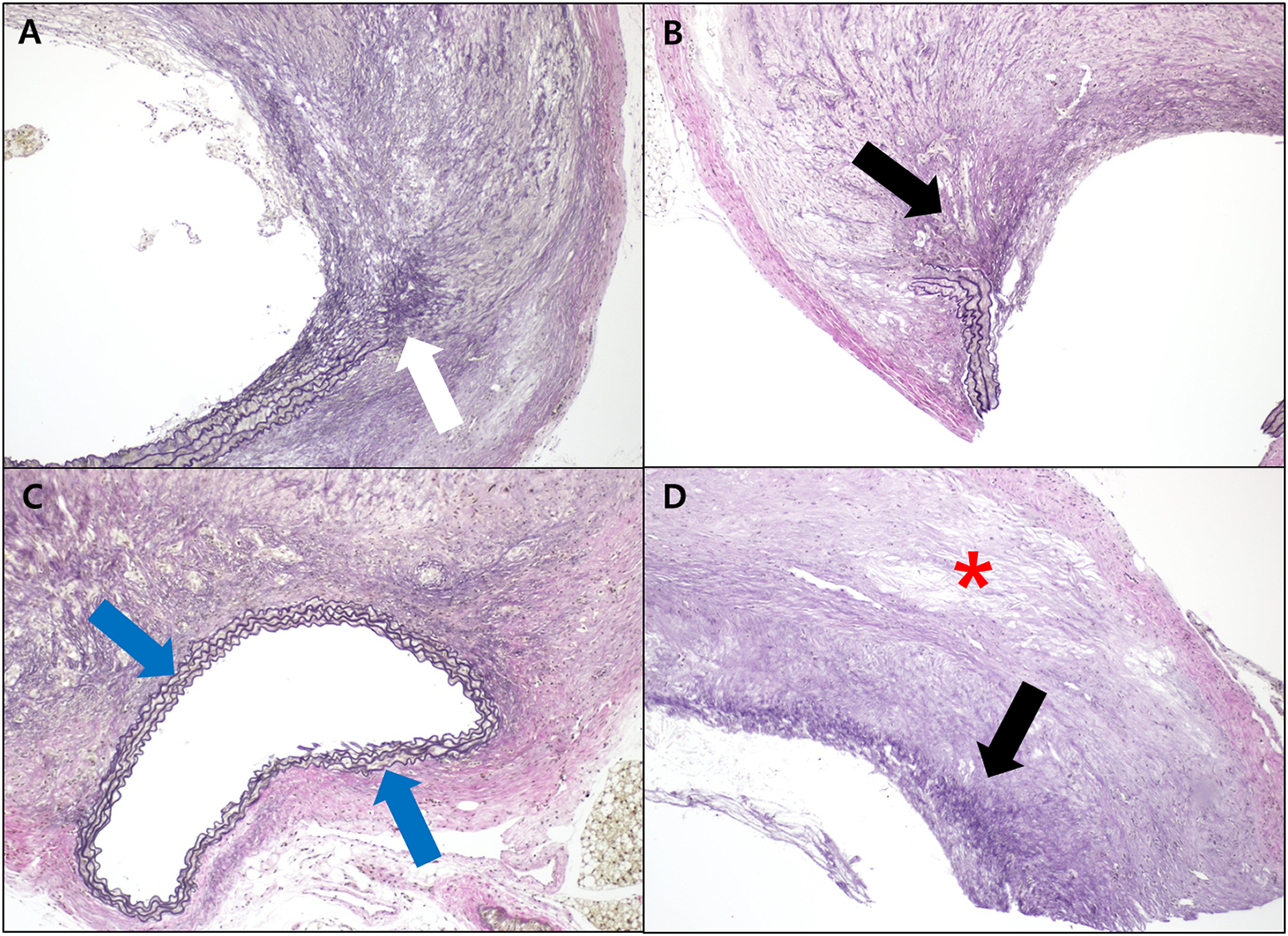
A-D, Pathology of transverse sections of formalin-fixed aortic tissue with high power field (elastic-Verhoeff’s Van Gieson [EVG], 100 times HPF); (A) Angiotensin II (Ang II) state, (B) saline group, (C) ramipril group, (D) carvedilol group: EVG stain of Ang II state (A) shows obvious destruction of internal elastic lamina (white arrow). After for 8-week treatment, preserved or reversed elastic layer is observed in ramipril group (blue arrows), not in carvedilol and saline group (black arrows). A slight regression of thickened media with unstable plaques was noted in the carvedilol treated group (red asterisk).
Changes in biomarker: MCP-1 and TIMP-1
The expression of MCP-1 and TIMP-1 was quantitated by ELISA testing. Analysis shows that expression and protein release of MCP-1 were significantly decreased in all treatment groups compared to the Ang II infusion state. After the treatment period, the level of MCP-1 of the ramipril and carvedilol groups was markedly decreased compared to the saline group (Ang II infusion: 192.72 ± 138.40, saline: 135.34 ± 90.00, ramipril: 64.59 ± 9.81, carvedilol: 64.98 ± 18.08 pg/mL, P = .006). In addition, the expression of TIMP-1 in the carvedilol group was significantly higher than in the ramipril and saline groups (Ang II infusion: 1890.82 ± 1018.08, saline: 1254.44 ± 426.78, ramipril: 1168.80 ± 219.95, carvedilol: 2080.49 ± 685.92 pg/mL, P = .002). Carvedilol treatment stimulated TIMP-1 secretion by 1.8-fold over ramipril treatment (Figure 6).

Changes of biomarkers (MCP-1, TIMP-1): (A) The level of MCP-1 expression of aorta markedly decreases in the ramipril and carvedilol groups compared to the saline group. (B) The level of TIMP-1 expression of aorta is significantly increased in the carvedilol group compared to saline and ramipril group. MCP-1 indicates monocyte chemoattractant protein-1, TIMP-1, tissue inhibitor of matrix metalloproteinase-1.
Discussion
AAAs are known to be one part of the spectrum of atherosclerotic diseases because it frequently coexists with generalized atherosclerosis. 2 Although hypertension seems to be weakly associated with AAA development unlike other forms of the atherosclerotic occlusive disease, it is known to be a major risk factor for the progression and rupture of AAA. 17 Based on the hypothesis of proteolytic degradation of connective tissue in the aortic wall, biomechanical wall stress may contribute to AAA expansion over time. 10,35 Immune-mediated vascular inflammatory response is also considered a contributor for the progression and complications of AAA. 16 Therefore, diverse regimens with antihypertensive properties including ACE inhibitors, β-blockers, and medications with anti-inflammatory properties such as doxycycline, statins, or antiplatelet agents have been previously examined as pharmacological agents that may limit the expansion of AAAs. 36
Because Ang II and its type 1 (AT1) receptor have a key role in the development of AAAs, ACE inhibitors are considered to reduce vascular inflammation independent of their effects on lowering blood pressure by blocking renin and angiotensin pathway. 31 Attenuation of AAA progression by ACE inhibitors was demonstrated by decreasing the MMP-9 in AT1 receptor-knockout mice. 37 The pleiotropic and anti-inflammatory effects of ACE inhibitors may contribute to the antihypertensive effect inversely based on in vitro studies. 16,38,39 Ramipril as a representative ACE inhibitor exerts comprehensive anti-inflammatory effects on vessel wall inflammation in AAA disease by reducing mRNA expression of multiple proinflammatory cytokines and aortic specific wall proteins. 16 In addition, angiotensin receptor blocking agents may reduce free radicals which increase MMP and accelerate atherosclerotic plaque formation. 37
β-Blockers decrease the blood pressure by lowering heart rates and provide benefits in preventing cardiovascular events. 25,40,41 β-Blockers may play a protective role against the development of atherosclerosis which results from both hemodynamic modification and antioxidative effects in animal and human studies. 18,42,43 Treatment with propranolol in the hypertensive rat AAA model shows favorable effect in limiting aneurysms. 22 Unfortunately, propranolol treatment in patients with small AAAs has not demonstrated clinical effect and mortality benefits in clinical trials. 24,44 Carvedilol is a newer generation β-blocker which is differentiated from traditional β-blockers by its vasodilatory and atheroprotective properties through simultaneous blocking of β1 and α1 adrenergic receptors. 18 The newer β-blockers have been shown to inhibit aortic lipid deposition in the hypercholesterolemic rat. 45 They have also been shown to provide superb cardioprotection due to multiple-functional neurohormonal antagonistic actions 46 and immune-modulating effects in the aortic walls of apoE−/− mice model. 47
Our study demonstrates that the inhibitory effect of ramipril and carvedilol treatment on mouse AAA expansion was similar to each other but superior to saline treatment. As far as we are aware, this study is the first to utilize micro-CT angiography to evaluate aorta size in the experimental animal AAA model.
Although pathologic evaluation demonstrating the therapeutic effect on AAA expansion is important, there has been little histological data showing the clinical impact of ramipril and carvedilol on aortic elastic properties and aneurysmal wall stability in the AAA model. 25,41 In our study, pathological evaluation including various staining of aortic wall tissue demonstrates relatively well-preserved elastic laminae in the ramipril group and slight regression of medial thickening with unstable plaques in the carvedilol group. These findings seem to be consistent with previous studies presuming the anti-inflammatory and pleiotrophic effects of ramipril secondary to the modulation of macrophages and T-cell expression. 48
The atherosclerotic degeneration of aortic aneurysms is known to be associated with the inflammatory biomarkers. In progression of AAA, macrophages are detected in the proximity of aortic smooth muscle cells. Apoptotic smooth muscle cells attract monocytes and other leukocytes by producing MCP-1 which seems to be a modulator of macrophage cytotoxicity. 33,35 Oxidized low-density lipoprotein (oxLDL) stimulates MMP-1 expression in vascular endothelial cells. The lack of coordination in the secretion of MMP-1 and TIMP-1 (an endogenous inhibitor of MMP) leads to an increased collagen-degrading activity that may contribute to destabilization of atherosclerotic plaques. 34 Both ramipril and carvedilol may inhibit the progression of atherosclerosis by reducing oxLDL uptake by macrophage within the atheromatous plaque. 46,48 In our study, the level of MCP-1 in the ELISA test is markedly decreased in the ramipril and carvedilol groups when compared to the saline group. Furthermore, the level of TIMP-1 in the ELISA test is significantly elevated in the carvedilol group compared to the saline and ramipril groups. This would seem to indicate that ramipril and carvedilol are effective in limiting AAA expansion by inhibiting the inflammatory process. 33,35,38 TIMP-1 as an endogenous inhibitor of MMP is closely related to vascular smooth muscle cells (VSMCs) in medial plaque layer and its downregulation due to overexpression of micro-RNA in VSMCs at AAA formation is introduced. 49 From our pathologic findings, we carefully suggest that carvedilol is related to regression of medial plaque while ramipril is related to preserving the elastic laminae regarding possible mechanism about limiting AAA.
The main limitations of our study include a relatively small number of animals and a short period for follow-up and treatment. Angiotensin II-induced AAA model is commonly used because it is similar to human AAA development, but there is somewhat challenging point to evaluate the various drug effect due to its mechanism. We could not be sure that the exact dosage of treatment drugs administrated to the mice was standardized because the drugs were administered with food.
In conclusion, ramipril and carvedilol have favorable effects in reducing the expansion of Ang II-induced AAA in the apoE−/− mice model using micro-CT angiography. The histological and biochemical evidence of ramipril and carvedilol in limiting AAA expansion via anti-inflammatory, antioxidant properties, as well as due to hemodynamic modulation was demonstrated. These findings could help elucidate the potential roles of ramipril and carvedilol in the medical treatment for patients with AAA.
Footnotes
Acknowledgments
The authors acknowledge the Korean Ministry of Science, ICT and Future Planning, Korean Ministry of Health and Welfare, National Research Foundation of Korea, Korean Ministry of Science, ICT and Future Planning, and Korea Health Technology R&D Project through the Korea Health Industry Development Institute, Korean Ministry of Health and Welfare.
Author Contributions
S. M. Park contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; M. Hong contributed to conception, contributed to interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; S. H. Kim contributed to design, contributed to interpretation, drafted manuscript, and critically revised manuscript; S Jung contributed to conception, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; B. K. Kim contributed to conception, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; D. Choi contributed to conception and design, contributed to interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research were supported by the research grants from Korean Ministry of Science, ICT and Future Planning (NRF-2015M3A9C6031514 and NRF-2015R1A2A2A01002731) and research grants from Korean Ministry of Health and Welfare (HI15C2782 and HI17C0882).
