Abstract
In patients with an acute coronary syndrome undergoing percutaneous coronary intervention, novel P2Y12 receptor inhibitors, prasugrel and ticagrelor, are proposed as “first-line” antiplatelet agents in the absence of contraindications and up to a year from the index event. However, de-escalation of treatment to clopidogrel occurs with a variable frequency in real-life practice, most commonly due to an increased bleeding potential, more frequent side effects, and a higher cost for the novel agents. Pharmacodynamic studies provide most of the data on guidance for de-escalation. Despite positive messages from recent trials and registries, lack of definitive efficacy or safety results of such a strategy remains an obstacle to suggest de-escalation in a routine basis. Carefully designed studies are likely to improve our understanding of the impact of de-escalation and help to better define its position in current pharmacotherapy.
The novel P2Y12 receptor inhibitors, prasugrel and ticagrelor, have been introduced in clinical practice for almost a decade as an alternative to clopidogrel in patients with acute coronary syndrome (ACS). Both agents exert a more potent and consistent antiplatelet action, which has contributed to an anti-ischemic benefit when tested clinically over clopidogrel, though at a cost of increased bleeding. 1,2 Prasugrel and ticagrelor are recommended by current practice guidelines as first choice agents over clopidogrel, in the absence of contraindications and for 1 year of treatment. 3,4 Despite the long-term benefits of strong platelet inhibition, including reduction in mortality by ticagrelor, it has been well appreciated that most of the stent thrombosis reduction using prasugrel or ticagrelor over clopidogrel is observed within the first month of the index event, which is recognized as the most vulnerable period. 1,2 This observation along with an increased bleeding potential, more common side effects, and a higher cost for the novel agents provide adequate reasoning for shortening the high-platelet inhibition period via switching treatment to clopidogrel in several cases in everyday clinical practice. 5,6 The modality of switching between oral P2Y12 inhibitors from a more intensive to a less intensive agent (ie, prasugrel or ticagrelor to clopidogrel) is defined as de-escalation. 5
The rationale, prevalence, pharmacodynamic and clinical data, and perspectives of de-escalation P2Y12 receptor inhibitor therapy represent the focus of this review.
Rationale
As the anti-ischemic benefit offered by the more potent platelet inhibition provided by novel agents over clopidogrel is apparent at least up to the analyzed time period in the respective clinical trials, the rationale for the 1-year recommendation of these agents has been supported. Shortening this period by de-escalating P2Y12 receptor inhibitor therapy may lead to an increased risk of stent thrombosis or other ischemic events. Following primary percutaneous coronary intervention (PCI), even in the contemporary era, reinfarction is not infrequent and it is strongly associated with cardiac and all-cause mortality. 7 On the other hand, increased bleeding potential and side effects represent the main limitation in the use of novel agents, which frequently urges practicing physician or the patient himself to switch to clopidogrel. The PREdicting bleeding Complications In patients undergoing Stent implantation and subsEquent Dual AntiPlatelet Therapy score is calculated according to baseline bleeding risk and may be used as a tool for the prediction of out-of-hospital bleeding and subsequent selection of a shorter than 12-month treatment duration in patients deemed at high bleeding risk. 8 However, no specific role for this score on de-escalation decision has been described. In case of a bleeding event while on prasugrel or ticagrelor, the severity of bleeding affects antiplatelet choice. If a severe bleeding occurs with no correctable cause, even discontinuation of the P2Y12 receptor inhibitor may be considered. 9 More often, if bleeding has resolved or is moderate or minor, de-escalation is a reasonable option permitting the completion of the required period for dual antiplatelet therapy (DAPT). Importantly, the 3 oral P2Y12 receptor inhibitors share—according to the respective summary of product characteristics—common warnings for precaution regarding conditions related to increased bleeding risk, lesions with a propensity to bleed, and comedication related to increased bleeding risk. 10 Nevertheless, diminishing the intensity of antiplatelet activity appears reasonable and commonly applied in clinical practice in case of a bleeding event. 11,12
Dyspnea is a common ticagrelor-associated side effect. Although in Platelet Inhibition and Patient Outcomes study ticagrelor was rarely (0.9%) discontinued because of dyspnea, de-escalation from ticagrelor to clopidogrel for this reason is more common in real-life practice. 13 Overall, in cases of even minor bleeding or side effects, a de-escalation strategy is expected to enhance patient’s adherence. 9 The need for concomitant anticoagulant treatment in clinical scenarios like atrial fibrillation or left ventricular thrombus formation represents common reason for prasugrel/ticagrelor de-escalation. 12,14,15 Physician’s and patient’s preferences emerge as frequent causes of de-escalation. Physicians also feel more comfortable with de-escalation given the better safety profile of current generation drug-eluting stents, which are indeed less thrombogenic and warrant shorter duration of DAPT and/or less potent agents. 16 Cost issues appear to play significant, though variable role in different registries involving de-escalation strategies. 12,14,17 In a recent systematic review of observational studies and registries in 19 132 ticagrelor-treated patients, the most common reasons for de-escalation among 586 patients involved the need for oral anticoagulant therapy (28%), bleeding (17%), intolerance (12%), patient unwillingness (10%), and dyspnea (10%). 18 Finally, a rather unexplored scenario of de-escalation involves post myocardial infarction (MI) patients with features of high ischemic and low bleeding risk in whom extending the 1-year mandatory DAPT is decided and the option of clopidogrel is selected. 19
Prevalence
The prevalence of de-escalation therapy varies widely (Table 1). The type of initial P2Y12 receptor inhibitor seems to impact on de-escalation prevalence, being more common among ticagrelor- than prasugrel-treated patients. 11,17 In case of prasugrel, prevalence ranges from 0.8% to 12.7% and 4.3% to 18%, while in case of ticagrelor, from 1% to 23% and 9.2% to 35.5%, for in-hospital and postdischarge de-escalation, respectively. Differences between the 2 agents in side effects (eg, dyspnea with ticagrelor) and dosing requirements (twice a day vs once daily for ticagrelor and prasugrel, respectively) affecting adherence and cost seem to be implicated. Timing of de-escalation may vary, for example, in hospital, at discharge, or early or late postdischarge, along with its prevalence. In a recent meta-analysis of observational studies, the prevalence of ticagrelor de-escalation reported was 22%, 20%, and 17% in hospital, at discharge, and postdischarge up to 1 year, respectively. 18
Prevalence of In-Hospital and Postdischarge De-Escalation of P2Y12 Inhibitors.

Abbreviations: ACS, acute coronary syndrome, C, clopidogrel, MI, myocardial infarction, NSTE, non ST elevation, P, prasugrel, PCI, percutaneous coronary intervention, STEMI, ST-segment elevation myocardial infarction, T, ticagrelor.
Several factors are associated with greater chances for de-escalation. In The TReatment with ADP receptor iNhibitorS: Longitudinal Assessment of Treatment Patterns and Events after Acute Coronary Syndrome registry, older age, prior stroke, low body weight, atrial fibrillation, higher risk for mortality and bleeding, and in-hospital bleeding predicted in-hospital de-escalation. 15,22 Gender (female), socioeconomic factors, increased bleeding risk, and in-hospital bleeding events were associated with postdischarge de-escalation. 17 Importantly, de-escalation was rarely preceded in the previous 7 days by a Global Utilization of Streptokinase and tPA for Occluded Arteries moderate/severe bleeding event.
Pharmacodynamic Studies
Pharmacodynamic studies involving de-escalation are depicted in Tables 2 and 3. When compared to values under prasugrel or ticagrelor, a significantly diminished platelet inhibition with de-escalation is apparent in all studies. Whether these changes are reflected into a different clinical outcome is not, however, clear as all of these studies were small in size, of short duration, and underpowered for clinical events. A signal of reduced bleeding potential was described in patients with low platelet reactivity while on prasugrel who switched to clopidogrel. 33 Importantly, pharmacodynamic studies have provided information on how to de-escalate for example, with or without a clopidogrel loading dose (LD). For de-escalation occurring within 30 days from the index event, an LD of clopidogrel is suggested. 5 In cases of a bleeding event or bleeding concerns, consideration of omitting an LD may be given. De-escalation beyond the 30 days from the index event is suggested without clopidogrel LD in case of prasugrel, but with an LD in case of ticagrelor initial treatment. Exception of the latter may be de-escalation reasoned by a bleeding event. The recent Switching Antiplatelet Therapy-4 study, which analyzed de-escalating from ticagrelor to clopidogrel with an LD, before starting maintenance dose (MD), revealed greater levels of platelet inhibition during the first 48 hours compared to switching directly to an MD regimen (without an LD). The latest results are suggestive of a drug–drug interaction between ticagrelor and clopidogrel. 37 Overall, the pharmacodynamic studies provide data for the timing of next clopidogrel dose, with most experts suggesting administration 24 hours after the last prasugrel or ticagrelor dose. 5
Pharmacodynamic Studies of De-Escalating From Prasugrel to Clopidogrel.
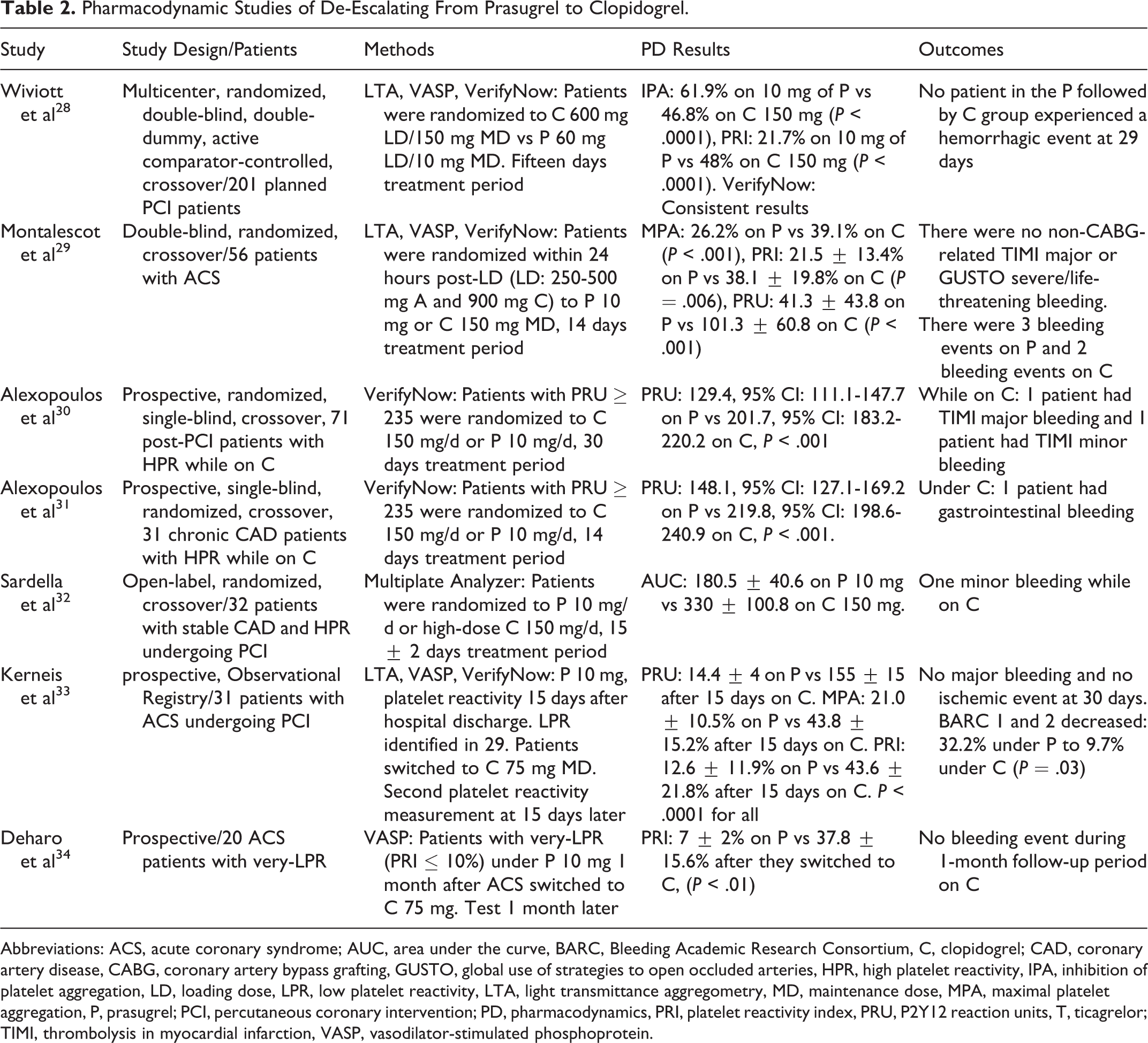
Abbreviations: ACS, acute coronary syndrome; AUC, area under the curve, BARC, Bleeding Academic Research Consortium, C, clopidogrel; CAD, coronary artery disease, CABG, coronary artery bypass grafting, GUSTO, global use of strategies to open occluded arteries, HPR, high platelet reactivity, IPA, inhibition of platelet aggregation, LD, loading dose, LPR, low platelet reactivity, LTA, light transmittance aggregometry, MD, maintenance dose, MPA, maximal platelet aggregation, P, prasugrel; PCI, percutaneous coronary intervention; PD, pharmacodynamics, PRI, platelet reactivity index, PRU, P2Y12 reaction units, T, ticagrelor; TIMI, thrombolysis in myocardial infarction, VASP, vasodilator-stimulated phosphoprotein.
Pharmacodynamic Studies of De-Escalating From Ticagrelor to Clopidogrel.
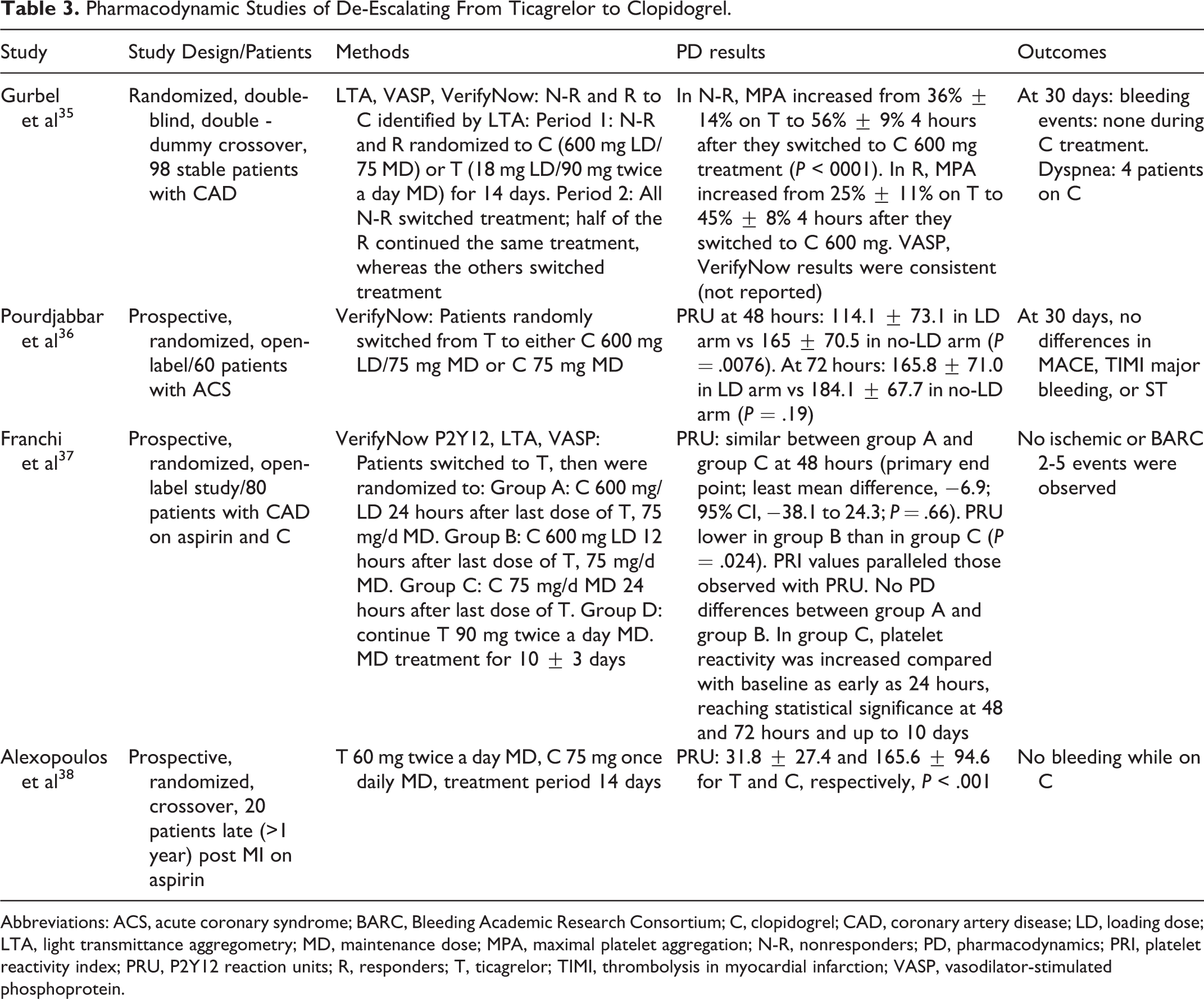
Abbreviations: ACS, acute coronary syndrome; BARC, Bleeding Academic Research Consortium; C, clopidogrel; CAD, coronary artery disease; LD, loading dose; LTA, light transmittance aggregometry; MD, maintenance dose; MPA, maximal platelet aggregation; N-R, nonresponders; PD, pharmacodynamics; PRI, platelet reactivity index; PRU, P2Y12 reaction units; R, responders; T, ticagrelor; TIMI, thrombolysis in myocardial infarction; VASP, vasodilator-stimulated phosphoprotein.
De-Escalation and Clinical Outcome
Studies involving de-escalation strategies and clinical outcome are shown in Table 4. In the Timing Of Platelet Inhibition after ACS (TOPIC) trial, de-escalation from ticagrelor or prasugrel to clopidogrel 1 month after ACS with PCI resulted in net clinical benefit (a combination of ischemic and Bleeding Academic Research Consortium [BARC] ≥ 2 bleeding events), compared to the unchanged DAPT over 1-year follow-up. This was driven by significantly fewer bleeding events, while ischemic events did not differ between groups. 39 Further assessment of low on-treatment platelet reactivity at 1 month—which was observed in 47% of cases—could identify those patients who experienced benefit in both bleeding and ischemic end points by de-escalation treatment. 40 Importantly, in the Testing Responsiveness To Platelet Inhibition On Chronic Antiplatelet Treatment For Acute Coronary Syndromes (TROPICAL-ACS) trial in biomarker-positive patients with ACS undergoing PCI, a strategy involving de-escalation based on a Multiplate analyzer (Roche Diagnostics, Rotkreuz, Switzerland) results while on clopidogrel 14 days post-PCI was proved to be noninferior to prasugrel treatment, in regards of the composite of death from cardiovascular causes, MI, stroke, and BARC ≥2 throughout the 1-year period. 41 The TROPICAL-ACS did not show superiority with de-escalation. Moreover, whether TROPICAL-ACS results involving prasugrel can be extrapolated to ticagrelor-treated patients is unclear. Furthermore, within the PRAGUE-18 study in 1230 patients with MI undergoing primary PCI and randomized to prasugrel or ticagrelor, 481 (39.1%) patients were economically motivated to de-escalate treatment to clopidogrel. Over the 1-year follow-up, patients who de-escalated treatment had lower ischemic risk and presented fewer major adverse cardiovascular events (MACEs) compared to those who continued initial treatment (2.5% vs 8.5%, hazard ratio, HR [95% confidence interval, [CI]: 0.433 [0.210-0.894], P = .024). De-escalation also resulted in a significant decrease in the bleeding risk in comparison with continued treatment with the initial regimen, 7.3% vs 13.4%, HR (95% CI) 0.416 (0.246-0.701), P = .001. 42 A recent meta-analysis of 6 studies with 574 patients who de-escalated treatment from ticagrelor to clopidogrel showed a 2.1% (95% CI: 0.9-3.3), 1.4% (95% CI: 0-2.9), and 2.0% (95% CI: 0.3-3.8) rate of MACE, cardiovascular mortality, and MI, respectively. All bleeding events’ (major and minor) rate was 3.3% (95% CI: 1.7 to 4.9), while major bleeding rate was 1.1% (95% CI: −0.3 to 2.5). 43 Overall, it should be stressed that clinical studies on de-escalation are undersized and no solid evidences support de-escalation.
De-Escalation Therapy and Clinical Outcome.

Abbreviations: DAPT, dual antiplatelet treatment; HTPR, high platelet reactivity; LTPR, low on-treatment platelet reactivity; MACE, major adverse cardiovascular events; NACE, net adverse clinical events; OR, odds ratio; PFT, platelet function testing; SCOPE, Switching from Clopidogrel to New Oral Antiplatelet Agents during PErcutaneous Coronary Intervention; TOPIC, Timing Of Platelet Inhibition after ACS; TRANSLATE-ACS, TReatment with ADP receptor iNhibitorS: Longitudinal Assessment of Treatment Patterns and Events after Acute Coronary Syndrome; TROPICALACS, Testing Responsiveness To Platelet Inhibition On Chronic Antiplatelet Treatment For Acute Coronary Syndromes.a NACE, combination of MACE and any bleeding event.
Role of Platelet Function Testing and Genotyping
It has been suggested that maintenance of platelet reactivity within certain limits may improve outcome, and therefore, the concept of a therapeutic window has been proposed. 44,45 It is not known, however, whether a single therapeutic window applies for all clinical scenarios, for example, ST-segment elevation MI versus moderate risk ACS. De-escalation in cases of low on-treatment platelet reactivity is expected to increase the percentage of patients within the therapeutic window. In the aforementioned TROPICAL-ACS platelet function, testing was used to confirm that prior de-escalation led to within-therapeutic range platelet reactivity and to escalate treatment in those with high on-clopidogrel platelet reactivity. De-escalation guided by platelet function testing was also the most common way of switching in order to achieve platelet reactivity levels within the therapeutic range in the Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged >75 Years to Reduce the Composite of Bleeding, Stent Thrombosis and Ischemic Complications trial. 46 In this trial, elderly patients with ACS were randomized to prasugrel 5 mg once daily either with dose or drug adjustment, or without monitoring or treatment adjustments. Platelet function testing was performed at 14 days after randomization and at 14 days after treatment adjustment in the monitoring group. Overall, 171 (39%) patients had been switched from prasugrel to clopidogrel for low on-treatment platelet reactivity. This, along with 16 cases of escalation to prasugrel 10 mg, resulted in an increase in patients within the therapeutic range from 42% to 66% in the monitoring group. However, this was not mirrored into improvement in the clinical outcome. Overall, platelet function testing in patients on novel agents for the purpose of de-escalating treatment resulted in a better outcome only in the small TOPIC-VASP cohort (Table 4), indicating the need for further studies in this field. 40
The role of genotyping in de-escalation strategies is even more underexplored. Studies have proved the feasibility of genotype-guided antiplatelet therapy after PCI usually in the form of treatment escalation in patients with a CYP2C19 loss-of-function allele. 47,48 In a study of 48 East Asian patients with MI undergoing PCI treated with prasugrel who were genotyped for CYP2C19, those (n = 38) without 2 copies of either CYP2C19 *2 or *3 loss-of-function alleles were de-escalated to clopidogrel 75 mg once daily. This resulted in an increase in the proportion of patients within the platelet reactivity therapeutic range according to East Asian criteria. 49
What the Guidelines/Experts Suggest?
In the latest 2016 American College of Cardiology/American Heart Association Guideline Focused Update on Duration of Dual Antiplatelet Therapy in patients with coronary artery disease, no specific comment regarding de-escalation is made apart from the lack of randomized data on the safety or efficacy of a switching strategy. 3 In the 2017 European Society of Cardiology (ESC) focused update on DAPT guidelines, switching oral P2Y12 receptor inhibitors was considered as reasonable in cases of side effects or intolerance (class IIb; Table 5). 50 De-escalation was generally discouraged due to a lack of safety/efficacy data at that time. Nevertheless, switching algorithms based on pharmacodynamic studies were provided. Clopidogrel 600 mg LD is suggested in all cases of de-escalation, apart from prasugrel to clopidogrel in chronic (postdischarge) phase, when clopidogrel 75 mg MD is considered as adequate. Similar algorithms have been provided by an international expert consensus document. 5 In the latter, acute/subacute phase was defined up to 30 days postintervention and the option of de-escalation with clopidogrel 75 mg MD (24 hours after last prasugrel or ticagrelor dose) in patients with bleeding or bleeding concerns was offered. The more recent 2018 Canadian Cardiovascular Society/Canadian Association of Interventional Cardiology Focused Update of the Guidelines for the use of antiplatelet therapy was published following the publication of TOPIC and TROPICAL-ACS results. Accordingly, in patients following a major bleeding complication that has resolved, with concomitant high-risk features for thrombotic events, de-escalation to clopidogrel instead of novel antiplatelet discontinuation may be considered. Treatment intolerance or side effects to potent agents and a new indication for treatment with an oral anticoagulant are emphasized as additional scenarios for de-escalation therapy. 9 Importantly, in the very recently released ESC/European Association for Cardio-Thoracic Surgery guidelines on myocardial revascularization, de-escalation guided by platelet function testing is given consideration as an alternative DAPT strategy, especially for patients with ACS who were deemed unsuitable for 12-month potent platelet inhibition, with class IIb recommendation, level of evidence B. 51 This recommendation was driven mainly by the TROPICAL-ACS results.
Guidelines/Expert Consensus Recommendations for De-Escalation of P2Y12 Receptor Inhibitor Therapy.

Abbreviations: C, clopidogrel; EACTS, European Association of Cardiothoracic Surgery; ESC, European Society of Cardiology; LD, loading dose; MD, maintenance dose; P, prasugrel; T, ticagrelor.
a Consider de-escalation with clopidogrel 75 mg MD (24 hours after last prasugrel or ticagrelor dose) in patients with bleeding or bleeding concerns.
Perspectives
Great variations in the prevalence of de-escalation as applied in real-life practice make the exact assessment of the magnitude of this practice difficult (Table 1). Differences in methodologies, populations, and clinical settings, reasoning of de-escalation, type of antiplatelet, socioeconomic conditions, and health insurance policies are among factors potentially involved. Undoubtedly, a de-escalation strategy appears as an attractive solution at least in certain populations, for example, those with high bleeding risk, side effects, or cost constraints, especially after the positive results of the 2 randomized studies (TOPIC and TROPICAL ACS). 39,41 Nevertheless, significant safety concerns remain about early de-escalation, which may lead to inadequate platelet inhibition, particularly in clopidogrel poor metabolizers, associated with an increased risk of thrombotic events. 27 As there is no large randomized trial with de-escalation and definitive clinical outcomes data to support such a strategy, its application cannot be recommended routinely and should be restricted and be considered only on an individual basis and following very careful risk/benefit assessment (Figure 1). Particularly de-escalation because of side effects or twice-daily administration—both factors with negative impact on adherence—appears as a reasonable practice, if this is going to improve patient adherence. Patient and physician preferences are important, although the mortality benefit obtained with enhanced platelet inhibition up to 1 year cannot be ignored and this should be clearly understandable. 2

Rationale and concerns for de-escalation balances. In the early phase, post-ACS/PCI concerns regarding de-escalation outweigh rationale. In the late phase, rationale of de-escalation may outweigh concerns. ACS indicates acute coronary syndrome; PCI, percutaneous coronary intervention.
An interesting alternative de-escalation strategy in patients with MI has been suggested with the use of ticagrelor standard 90 mg twice a day dose for 1 month, followed by de-escalation to ticagrelor 60 mg twice a day. 52 The pharmacokinetic/pharmacodynamics results of this approach are expected shortly. Several pharmacodynamic studies have mostly clarified the expected changes in platelet reactivity using different de-escalation strategies. Despite concerns for excessive or inadequate platelet inhibition during de-escalation and an increase in bleeding or ischemic events during this transition, no such sign has been observed so far (Tables 2 and 3). A simplified scheme of a clopidogrel 600 mg LD 24 hours after the last potent agent dose for in-hospital or during the first 30 days occurring de-escalation is accepted by most experts. 5,9,50 Similarly, an LD of clopidogrel is suggested for de-escalation from ticagrelor to clopidogrel occurring 1 to 12 months postindex event. Clopidogrel LD can be omitted in de-escalation reasoned by bleeding or bleeding concerns.
Regarding the role of platelet function tests and CYP2C19 polymorphism analysis as decision-making tools to de-escalate, although promising, is not definitely established. This is further hampered by the fact that these tests are not yet widely available. An extensive review of genotyping-guided escalation studies is beyond the scope of this review and can be found elsewhere. 53 A genotyping approach to the selection of P2Y12 receptor inhibitors for patients with ACS was recently reported to lead into a reduction in ischemic and bleeding events. 54 Further insights into the role of genotyping are expected to be offered by 2 ongoing randomized trials, the Tailored Antiplatelet Therapy following PCI (NCT01742117) and the Cost effectiveness of Genotype Guided Treatment With Antiplatelet Drugs in STEMI Patients: Optimization of Treatment (POPular Genetics, NCT01761786). Furthermore, the results of the Genotyping Infarct Patients to Adjust and Normalize Thienopyridine Treatment (NCT 01134380) are expected. This is an observational case–control study, including STEMI patients treated with primary PCI. The genetic information (CYP2C19*2 allele) of the patients tested after primary PCI is communicated to the treating physician, who can subsequently change or adjust the initial clopidogrel or prasugrel regimen. Of note, a cost-effectiveness analysis of platelet function testing and/or genotyping-guided de-escalation is also lacking.
Following the DAPT and the Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin-Thrombolysis in Myocardial Infarction 54 trials results, 55,56 the concept of extending beyond 1 year the treatment with a P2Y12 receptor inhibitor, on top of aspirin, has gained interest; this practice is suggested with a IIb recommendation for cases with high ischemic and low bleeding risk. 3,50 Although ticagrelor 60 mg twice a day has a relevant indication, which has been approved by regulatory authorities, clopidogrel is also a viable option, especially when availability or cost concern coexists. 19 De-escalation may therefore occur in the very late phase (>12 months) from acute event/PCI. Nevertheless, the issue of de-escalating in the very late phase remains practically unexplored, with only very limited pharmacodynamic data being available. 38
Conclusions
De-escalation of a potent P2Y12 receptor inhibitor to clopidogrel occurs with a variable frequency in real-life practice. Under certain circumstances, de-escalation appears to be unavoidable. Despite positive messages from recent trials and registries, lack of definitive efficacy or safety results of such a strategy remains an obstacle to suggest de-escalation in a routine basis. Carefully designed studies are likely to improve our understanding of the impact of de-escalation and help to better define its position in current pharmacotherapy.
Footnotes
Authors’ Note
D. Alexopoulos contributed to conception or design and critically revised the manuscript. I. Lianos, P. Vlachakis, D. Sfantou, V. M. Dragona, C. Varlamos contributed to acquisition, analysis, or interpretation. All authors drafted the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Alexopoulos disclosed lecturing honoraria from Astrazeneca, Bayer, Boehringer Ingelheim, and Biotronik and Advisory Board fees from Astrazeneca, Bayer, Boehringer Ingelheim, and Medtronic Moderate.
