Abstract
Background:
Critical limb-threatening ischemia (CLTI) is associated with an increased risk of major adverse limb events and mortality. High on-treatment platelet reactivity (HPR) is associated with an increased risk of ischemic events, while low on-treatment platelet reactivity (LPR) is associated with an increased risk of bleeding. This study investigates the frequency with which patients with CLTI on clopidogrel or ticagrelor achieve a “therapeutic window” (TW) of platelet inhibition.
Methods:
Data from the “Switch To Ticagrelor in Critical Limb Ischemia Anti-Platelet Study” were assessed retrospectively to determine the incidence of TW of on-treatment platelet reactivity in 50 consecutive patients with CLTI (mean age: 65.2 ± 10.5 years, 54% male). The data included 4 measurements of patients’ platelet reactivity using the VerifyNow P2Y12 Assay: baseline and steady state platelet reactivity on clopidogrel 75 mg daily and on ticagrelor 90 mg twice daily.
Results:
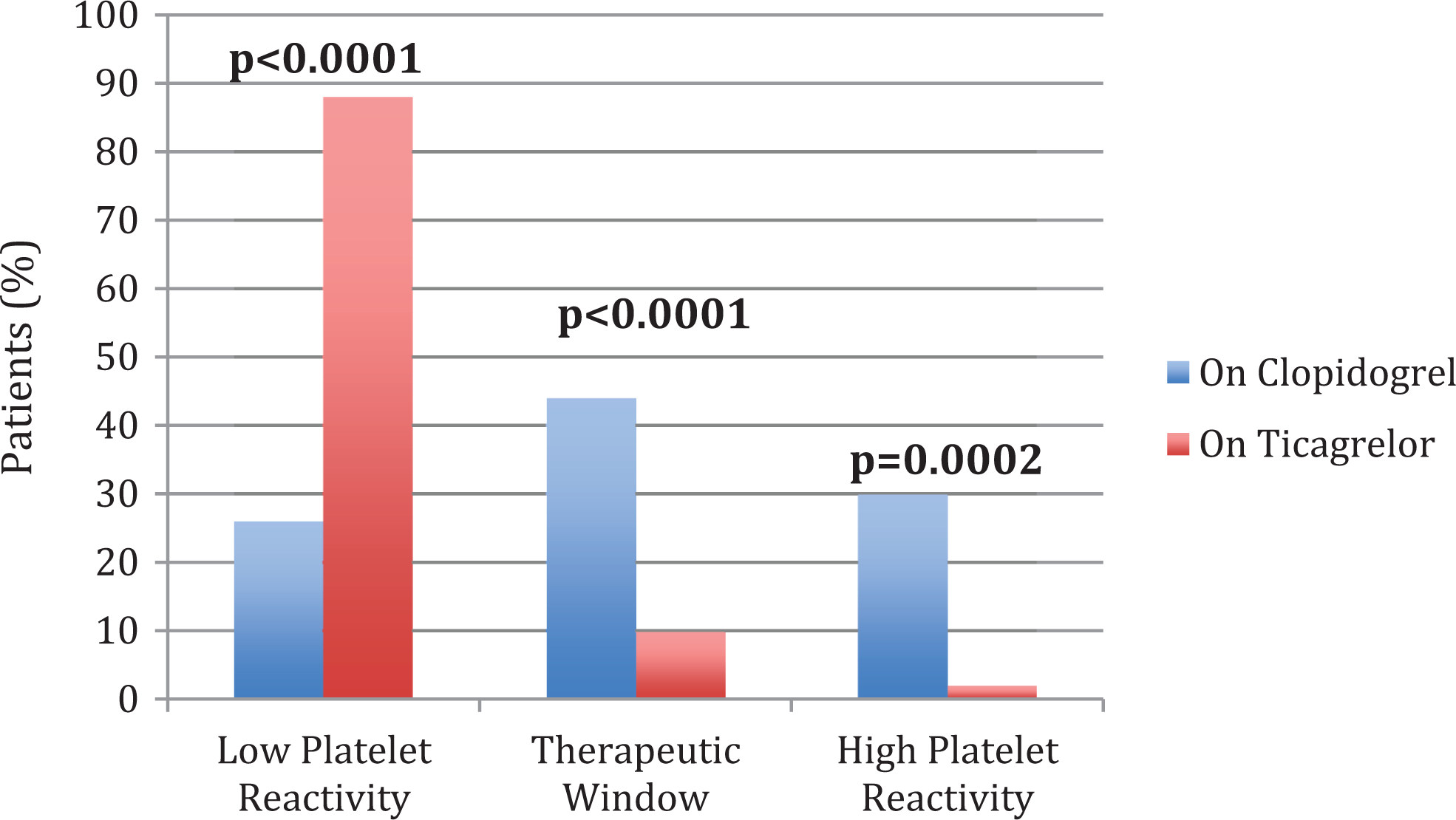
At baseline, 46% of patients on clopidogrel were within TW of on-treatment platelet reactivity compared to 10% of patients on ticagrelor (P < .0001). At steady state, 42% of patients on clopidogrel were within the TW compared to 10% of patients on ticagrelor (P < .0001). Patients on ticagrelor exhibited higher rates of LPR compared to those on clopidogrel at baseline as well as at steady state (baseline 88% vs 18%, steady state 88% vs 28%; P < .0001).
Conclusion:
Although ticagrelor has been proposed as an alternative for patients with HPR on clopidogrel, the current study observes an excess of platelet inhibition with ticagrelor in most patients with CLTI at a dose of 90 mg twice daily.
Keywords
Introduction
Critical limb-threatening ischemia (CLTI) is a complex disease in which impeded blood flow to an affected extremity may result in rest pain and/or tissue loss; it is associated with an increased risk of major adverse limb events (MALE), myocardial infarction (MI), major adverse cardiovascular events (MACE), and mortality rates that approach 22% and 50% at 1 and 5 years, respectively. 1 –3 Although revascularization is the primary mode of treatment for critical limb ischemia (CLI), medical therapy is crucial to maintain arterial patency and prevent MALE. 1 High on-treatment platelet reactivity (HPR) has been associated with an increased risk of MACEs such as MI, sudden cardiac death, and stroke. 2,3 In a study of 100 consecutive patients with CLI, Clavijo et al measured levels of platelet reactivity at baseline and subsequently at steady state plasma concentration (6 hours after administering clopidogrel, aspirin, or both). The study found that 25% of patients with CLI had HPR to aspirin, 26% had HPR to clopidogrel, and 8% had HPR to both aspirin and clopidogrel. 2 The rate of HPR in patients with CLI measured with the VerifyNow P2Y12 assay has been reported to be as high as 60% for clopidogrel, while in patients undergoing peripheral endovascular procedures, an HPR of 20.7% for aspirin and 11.7% for both has been reported. 4 High on-treatment platelet reactivity (platelet reactivity units >234) has been also related to increased adverse clinical events in patients undergoing femoropopliteal endovascular interventions. 5
In terms of antiplatelet therapy, ticagrelor is an alternative option to clopidogrel as an irreversible P2Y12 receptor antagonist. 3,6 With an increasing range of antiplatelet medications available, there is a focus on quantitative parameters of determining drug safety and efficacy. While most research into antiplatelet therapy has largely focused on reducing complications of ischemic events, bleeding complications have largely been considered an expected complication of antiplatelet therapy. Emerging data suggesting low platelet reactivity (LPR) is associated with an increased risk of bleeding complications, particularly with P2Y12 receptor antagonists, and this has warranted further investigation into refining the therapeutic index of antiplatelet medications. 7
The Switch To Ticagrelor in Critical Limb Ischemia Anti-Platelet Study (STT-CLIPS) used a within-subject design to assess the prevalence of HPR to clopidogrel and ticagrelor in 50 patients, both at baseline and at steady state for both medications. 8 Among patients on clopidogrel, 36% had HPR at baseline and 30% at steady state, compared to 2% of patients on ticagrelor at baseline and steady state. Another interesting finding was that 94% of patients with HPR on clopidogrel demonstrated appropriate platelet inhibition after switching to ticagrelor. 8
The current study aimed to further dissect the data of the STT-CLIPS study to investigate the therapeutic window (TW) of clopidogrel and ticagrelor, defined by platelet reactivity units (PRU) values between 85 and 208 as determined by the VerifyNow P2Y12 assay. 7
Methods
The STT-CLIPS study enrolled patients from Keck Hospital of the University of Southern California (USC) and the Los Angeles County + USC Medical Center. 8 The study was institutional review board approved, and all participants gave written and informed consent. Inclusion criteria for patients enrolled in the study were a diagnosis of CLI (Rutherford Category IV, V, and VI) on continuous dual antiplatelet therapy with clopidogrel 75 mg daily and aspirin 81 mg daily for at least 14 days. P2Y12 platelet receptor inhibition studies (vasodilator-stimulated phosphoprotein phosphorylation [VASP] and VerifyNow assays) were performed in 50 patients with CLTI. Tests were performed before daily clopidogrel dose (baseline) and 6 ± 1 hours after daily 75 mg clopidogrel dose (steady state). Patients were then switched to ticagrelor 90 mg twice daily for 2 weeks, and platelet assays were repeated at the end of the 2 weeks (baseline and steady state). Participants had venous blood samples collected at 4 distinct time points; platelet function assays were performed on these samples using the VerifyNow P2Y12 assay (Figure 1). The TW of clopidogrel and ticagrelor was defined by PRU values between 85 and 208 as determined by the VerifyNow P2Y12 assay in accordance with standards set by the International Working Group on on-treatment platelet reactivity. 7 The purpose of the STT-CLIPS study was to assess on-treatment platelet reactivity in patients with CLTI and compare the incidence of HPR between clopidogrel and ticagrelor. The study found that in patients with CLTI, ticagrelor achieved greater platelet inhibition than clopidogrel during maintenance treatment and at 6 hours after daily dosing and concluded that HPR to clopidogrel in patients with CLI can be overcome by switching to ticagrelor. 8
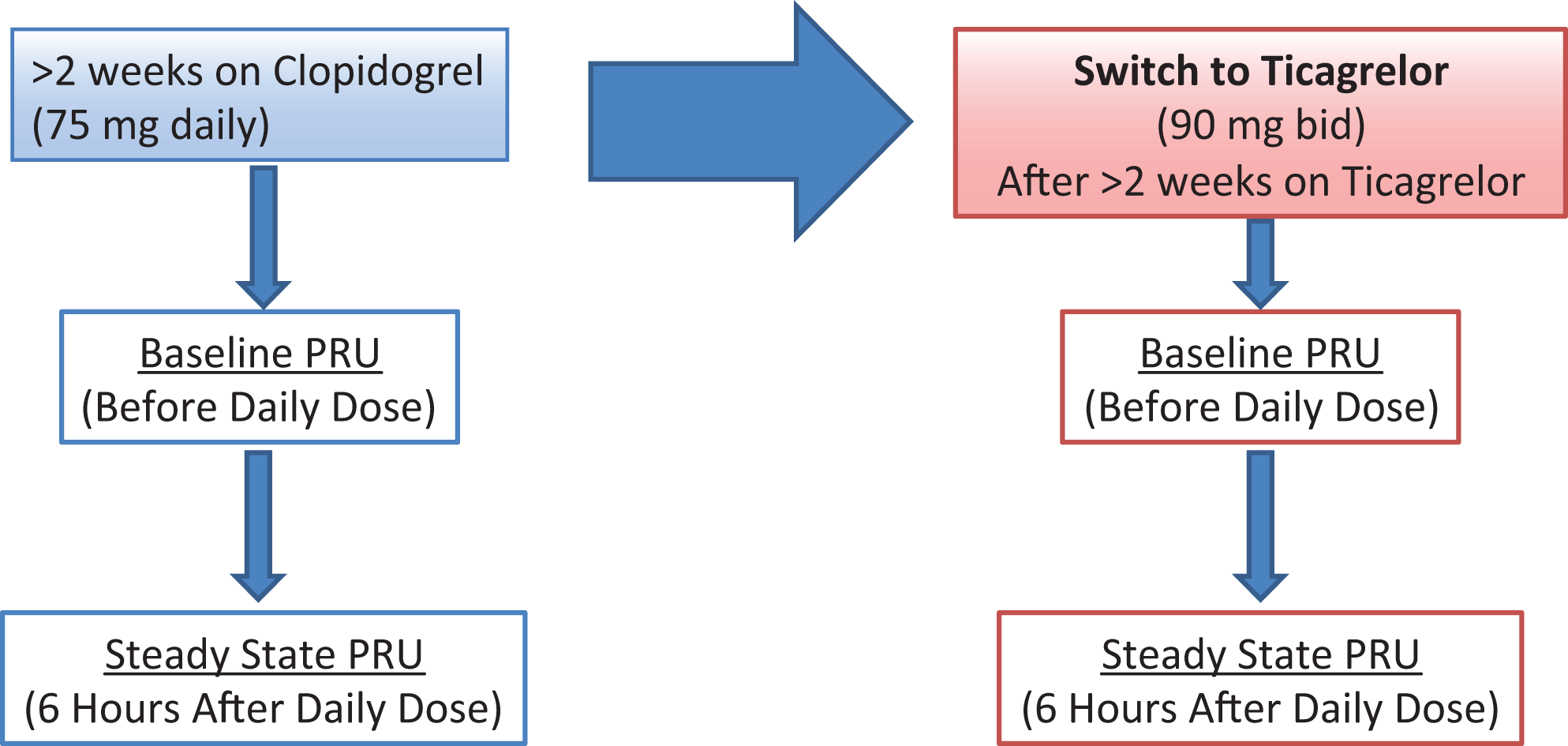
Study Design.
For the current study, data from the STT-CLIPS study were retrospectively studied to assess the TW in a sample of 50 patients with CLI. The data included 4 measurements of patients’ platelet reactivity using the VerifyNow P2Y12 assay: the baseline and steady state (6 hours after first daily dose) while taking clopidogrel 75 mg daily and the baseline and steady state 2 weeks after switching to ticagrelor 90 mg twice daily (Figure 1). A χ2 analysis was used to assess the 3 different categories of PRU values (LPR, therapeutic range, and HPR) of clopidogrel and ticagrelor at both baseline and steady state (Supplementary Table).
Results
Fifty consecutive patients (mean age: 65.3 ±10.6 years, 54% male) with CLTI were analyzed retrospectively for this study. Baseline clinical characteristics are presented in Table 1.
Baseline Clinical Characteristics.a
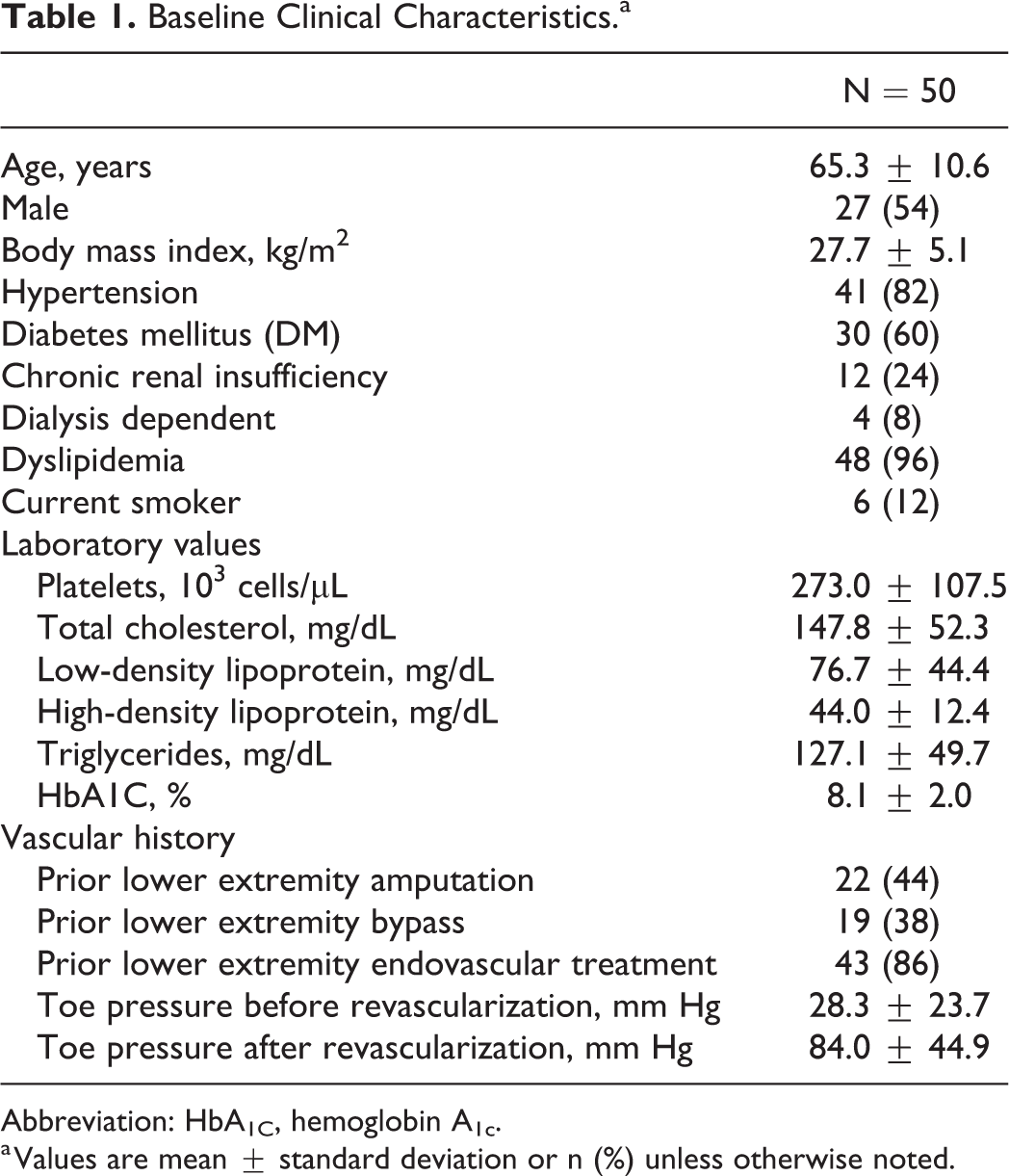
Abbreviation: HbA1C, hemoglobin A1c.
a Values are mean ± standard deviation or n (%) unless otherwise noted.
Baseline On-Treatment Platelet Reactivity
Results of the PRU values at baseline on clopidogrel compared to ticagrelor are presented in Figure 2. At baseline, 46% of patients on clopidogrel were within TW of on-treatment platelet reactivity compared to 10% of patients on ticagrelor (P < .0001). Thirty-six percent of patients on clopidogrel demonstrated HPR compared to 2% of patients on ticagrelor (P = .0001). The LPR was more common in ticagrelor compared to clopidogrel at steady state (88% vs 18%, P < .0001; see Table 2).

Baseline (before daily morning dose) On-Treatment Platelet Reactivity.
Platelet Reactivity at Baseline and Steady State on Clopidogrel and Ticagrelor.a

a Values are n (%).
P < 0.05 considered significant are bold faced.
Steady State On-Treatment Platelet Reactivity
Results of the PRU values at steady state on clopidogrel compared to ticagrelor are presented in Figure 3. At steady state, 42% of patients on clopidogrel were within the TW compared to 10% of patients on ticagrelor (P < .0001). The HPR was more common in patients on clopidogrel compared to ticagrelor at steady state (30% vs 2%; P = .0002). The LPR was more common in ticagrelor compared to clopidogrel at steady state (88% vs 28%, P < .0001; see Table 2, Figures 4 and 5).
Steady-state (6 hours after daily dose) On-Treatment Platelet Reactivity.
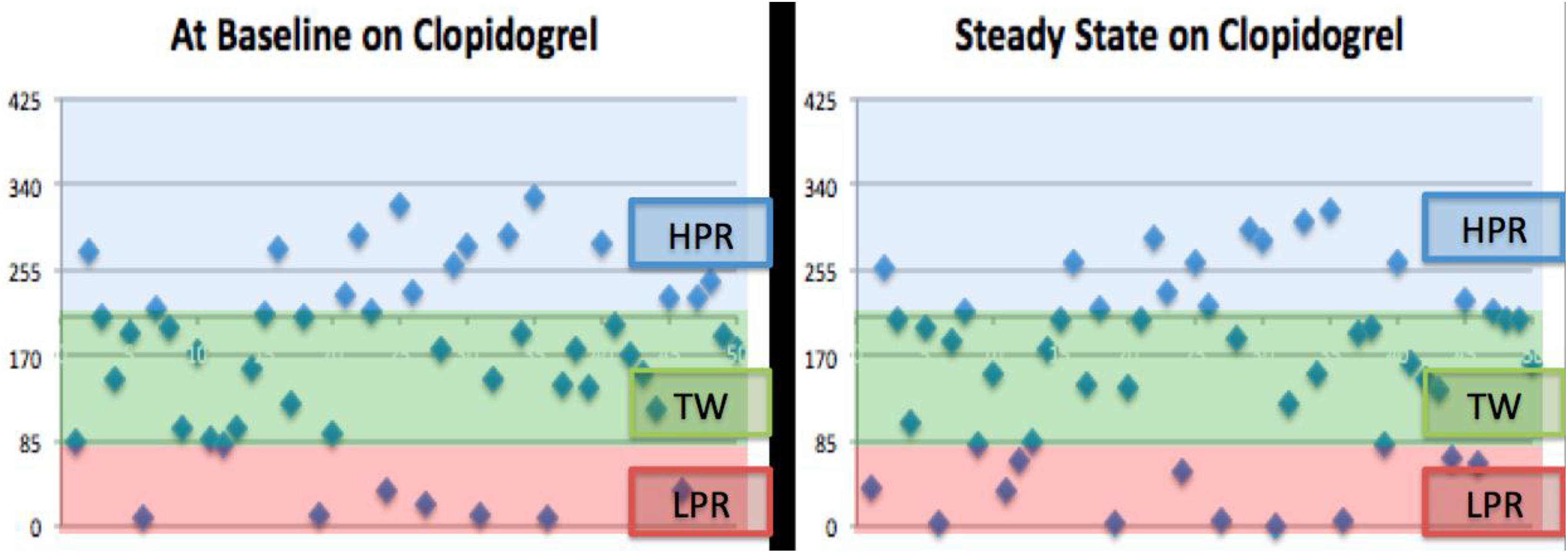
Individual On-Treatment Platelet Reactivity Units on Clopidogrel. Y-axis denotes individual platelet reactivity units of patients (N = 50). HPR indicates high on-treatment platelet reactivity (PRU > 208); LPR, low on-treatment platelet reactivity (PRU < 85); TW, therapeutic window of platelet reactivity (PRU = 85-208).

Individual On-Treatment Platelet Reactivity Units on Ticagrelor. Y-axis denotes individual platelet reactivity units of patients (N = 50). HPR indicates high on-treatment platelet reactivity (PRU > 208); LPR, low on-treatment platelet reactivity (PRU < 85); TW, therapeutic window of platelet reactivity (PRU = 85-208).
Discussion
In our analysis of patients with CLTI, we found that at baseline (prior to daily dose of antiplatelet agent) 46% of patients on clopidogrel and 10% of patients on ticagrelor were within the TW of on-treatment platelet reactivity. A majority of patients outside the TW on clopidogrel were found to have HPR, while the vast majority of patients outside the TW on ticagrelor were found to have LPR (Figures 4 and 5). Ticagrelor has previously been proposed as an alternative for patients who demonstrate HPR while on clopidogrel. 3,8 –11 Although highly efficacious in terms of platelet inhibition, we found a significantly higher rate of excessive platelet inhibition in a majority of patients on ticagrelor, warranting a valid concern for bleeding complications. This finding is supported by a prior study of patients with acute coronary syndrome in which bleeding events were found to be significantly increased in the ticagrelor arm (dose 90 mg twice daily) compared to prasugrel. 9 A prior study of patients with CLTI by Spiliopoulos et al found that mean PRU during clopidogrel therapy was 308.4 ± 41.8, and when switched to ticagrelor, the mean PRU was significantly lower at 67.0 ± 52.8. 11 However, the correlation of on-treatment platelet reactivity with clinical outcomes in patients with CLTI has not been definitively demonstrated. 12 Even so, there continues to be increasing data that on-treatment platelet reactivity is significantly lower with ticagrelor when compared to clopidogrel. 2,3,8,11 Furthermore, recent data suggest that ticagrelor at a lower dose 60 mg twice daily may be as effective as the standard dose (90 mg twice daily) for preventing cardiovascular death, MI, or stroke in patients with prior MI, while having a lower rate of major bleeding. 13 Given the correlation of excessive platelet inhibition and bleeding events, it may be worthwhile to consider a lower dose of ticagrelor (60 mg twice daily) in patients with CLTI who receive long-term antiplatelet therapy. 14 –19
Even within the spectrum of patients with known cardiovascular disease, those with CLTI are at an increased risk of adverse ischemic outcomes. A subgroup analysis of the patients with CLI within the Examining Use of Ticagrelor in Peripheral Artery Disease (EUCLID) trial found that the primary end point of cardiovascular death, MI, or stroke was approximately twice as high among patients with CLTI compared to those without CLTI with a rate of 8.8 versus 4.3/100 patient-years. 3 As such, an appropriate antiplatelet regimen is of paramount importance in this patient population. Even so, side effects and bleeding complications of long-term antiplatelet therapy must be kept in mind when treating patients with CLTI. In the EUCLID trial, there were significantly more bleeding events that caused discontinuation of antiplatelet therapy with ticagrelor than with clopidogrel. It is worth noting that the dose of ticagrelor in the EUCLID trial was 90 mg twice daily. 6
Our study sought to elucidate the incidence of an appropriate TW of on-treatment platelet reactivity in patients with CLTI. Our findings may guide future research into correlating on-treatment platelet reactivity with clinical outcomes such as MACE and MALE, as well as safety outcomes in terms of bleeding events. Previous studies have focused on decreasing ischemic events in patients on antiplatelet therapy and have considered bleeding events an “inevitable and acceptable complication.” 7 Due to poor outcomes associated with major bleeding and ischemic complications, the importance of balancing an appropriate anti-ischemic effect with the bleeding risks associated with long-term antiplatelet therapy in patients with CLTI cannot be overemphasized.
Limitations
Since this was a retrospective analysis, it has selection bias. Confounding factors that were not assessed cannot be ruled out. Our sample size was small and limited to 2 medical centers. Despite these limitations, we believe our findings can guide prospective studies to assess clinical outcomes in patients with CLTI based on choice of antiplatelet therapy.
Conclusions
In our analysis of patients with CLTI, we found that at baseline (prior to daily dose of antiplatelet agent) 46% of patients on clopidogrel and 10% of patients on ticagrelor were within the TW of on-treatment platelet reactivity. A majority of patients outside the TW on clopidogrel were found to have HPR, while the vast majority of patients outside the TW on ticagrelor were found to have LPR. Although ticagrelor has been proposed as an alternative for patients with HPR on clopidogrel, the current study observes an excess of platelet inhibition with ticagrelor in most patients with CLTI at a dose of 90 mg twice daily. Going forward, long-term prospective studies are needed to assess clinical end points to validate the “TW” of on-treatment platelet reactivity in patients with CLTI who are subjected to long-term antiplatelet therapy.
Supplemental Material
Suplementary_Table - Therapeutic Window of Clopidogrel and Ticagrelor in Patients With Critical Limb-Threatening Ischemia
Suplementary_Table for Therapeutic Window of Clopidogrel and Ticagrelor in Patients With Critical Limb-Threatening Ischemia by Ashwat S. Dhillon, Jorge Caro, Han Tun, David G. Armstrong, Vincent Rowe, David M. Shavelle and Leonardo C. Clavijo in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
ASD and JC helped in data collection, data analysis, prepared and edited the manuscript. DGA, VR, DMS and LCC helped to design the study, analyze the data, and prepare and edit the manuscript. HT provided statistical analysis, prepared and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
