Abstract
Aims:
Patients with acute coronary syndrome undergoing percutaneous coronary intervention (PCI) are recommended to be placed on potent P2Y12 blockade. However, the long-term bleeding risk is high. Therefore, despite no definitive evidence, switching to clopidogrel beyond the acute phase is common. We aimed to evaluate the clinical outcomes of antiplatelet de-escalation compared with continuation in patients treated with PCI.
Methods:
We searched databases for randomized clinical trials (RCTs) that evaluated the safety and efficacy of antiplatelet de-escalation compared with continuation in patients treated with PCI. Pooled summary estimates were calculated.
Results:
We included 3 RCTs with 3391 patients (median follow-up: 12 months). Compared with the continued group, the net clinical outcome (composite of bleeding or thrombotic events) was significantly reduced in the group switched to clopidogrel (8.7% vs 12.1%; risk ratio [RR]: 0.64; 95% confidence interval [CI]: 0.43-0.97;
Conclusions:
Our results suggest a net clinical benefit of de-escalation therapy shortly after PCI, without increased risk of MACE. Larger randomized trials will be necessary to confirm these findings.
Introduction
Potent P2Y12-receptor inhibitors are recommended in the management of acute coronary syndrome (ACS) treated with percutaneous coronary intervention (PCI). 1 As the risk of thrombotic complications from an ACS event is highest in the early “ischemic phase,” the greatest benefit of potent P2Y12 blockade occurs early. 1 However, the risk of bleeding from such therapy tends to occur with chronic treatment during the “maintenance phase.” Therefore, a stage-adapted strategy with the early use of potent P2Y12 blockade in acute treatment, followed by switching to clopidogrel during the maintenance phase is common. 2 Nevertheless, clinical outcomes supporting this strategy are lacking. Therefore, we conducted a meta-analysis of all randomized clinical trials (RCTs) to evaluate the safety and efficacy of antiplatelet de-escalation compared to continuation.
Methods and Materials
Data Source
The study protocol of our meta-analysis was prespecified and registered with the International Prospective Register of Systematic Reviews (PROSPERO; ID: CRD42018092362). We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols (PRISMA-P) Statement 2015 3 to guide our performance of the meta-analysis. A systematic search of PubMed, Cochrane Collaboration, and Embase was conducted from inception to March 2018. The search was performed independently by M.O. and A.A., and any disagreements were resolved by B.K.
Selection Criteria
Per our prespecified protocol, we included only RCTs that evaluated the role of antiplatelet de-escalation compared to continuation therapy. The search terms were as follows: “de-escalation,” “switching,” “antiplatelet,” “clopidogrel,” “ticagrelor,” “prasugrel,” “acute coronary syndrome,” “ACS,” “percutaneous coronary intervention,” and “PCI.” S.A. and K.O. extracted the data independently, and B.K. was consulted for any discrepancies.
Quality Assessment
B.K. and S.A. assessed the quality of each RCT independently, and this assessment was based on the following factors: the study design, whether it was a single or multicenter study, the blinding to intervention and/or outcomes, the treatment assignment generation, and the follow-up completion rates.
Clinical Outcomes
Our primary outcome was the net clinical outcome, defined as combined thrombotic or bleeding events. Secondary outcomes were major adverse cardiovascular events (MACEs) as defined per each RCT (myocardial infarction, cardiovascular mortality, stroke, and/or revascularization) and Bleeding Academic Research Consortium (BARC) bleeding as the only bleeding definition used in most of the included RCTs. 4
Statistical Analysis
We calculated the pooled risk ratios (RRs) with the Mantel-Haenszel method using a random-effects model to account for in-between study heterogeneity. Analysis was performed using RevMan v5.3 Windows. We assessed publication bias by visually inspecting the funnel plot. Furthermore, sensitivity analysis was conducted for the net clinical outcome by excluding each RCT and identifying any differences in the summary estimates. We also performed subgroup analyses to identify any treatment interactions in relation to baseline covariates.
Results
We included 3 RCTs with 3391 total patients (median follow-up: 12 months). 5 -7 Figure 1 shows the PRISMA flow diagram. The characteristics and design of the included RCTs are shown in Table 1. There were differences among the included trials. The timing of de-escalation varied (≤2 weeks vs >2 weeks). Both the The Testing Responsiveness To Platelet Inhibition On Chronic Antiplatelet Treatment For Acute Coronary Syndromes (TROPICAL-ACS) and Timing Of Platelet Inhibition after Acute Coronary Syndrome (TOPIC) trials included patients who presented exclusively with ACS. However, TROPICAL-ACS incorporated platelet function testing (PFT) to aid in the decision for de-escalation. To ensure consistency among the switched group, we only included those who were switched to clopidogrel. Table 2 shows the baseline patient-level characteristics. The mean age was 59, and 79% were males. The major risk factor for coronary artery disease was hypertension. All patients presented with ACS and/or were confirmed to have myocardial ischemia and underwent PCI.
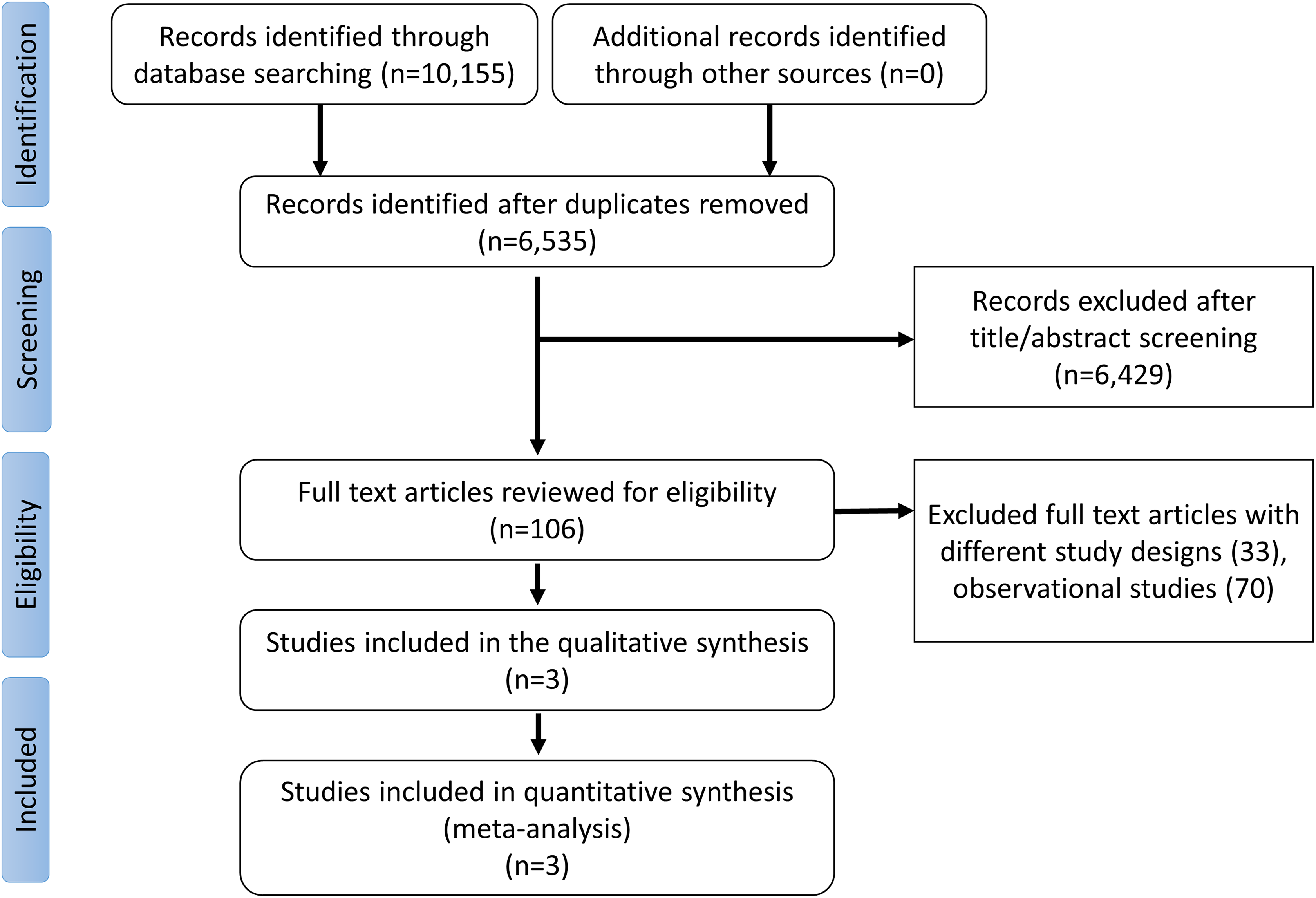
Study selection process.
Details of the Included RCTs.

Abbreviations: BARC: Bleeding Academic Research Consortium; RCT, randomized controlled trial; TOPIC: Timing of Platelet Inhibition after Acute Coronary Syndrome; TROPICAL-ACS: The Testing Responsiveness to Platelet Inhibition on Chronic Antiplatelet Treatment for Acute Coronary Syndromes.
a Mean values reported as appeared in the original manuscript.
Baseline Patient-Level Characteristics.a
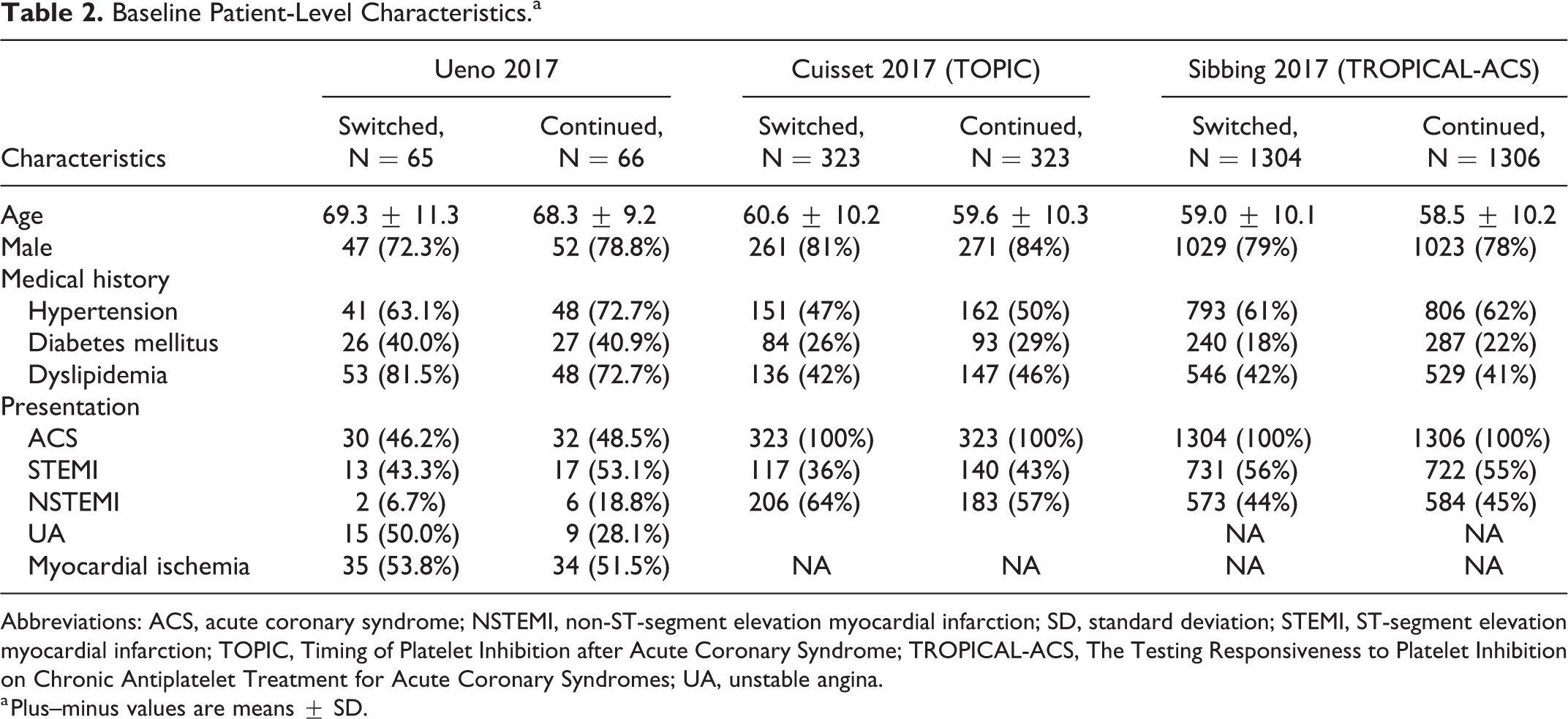
Abbreviations: ACS, acute coronary syndrome; NSTEMI, non-ST-segment elevation myocardial infarction; SD, standard deviation; STEMI, ST-segment elevation myocardial infarction; TOPIC, Timing of Platelet Inhibition after Acute Coronary Syndrome; TROPICAL-ACS, The Testing Responsiveness to Platelet Inhibition on Chronic Antiplatelet Treatment for Acute Coronary Syndromes; UA, unstable angina.
a Plus–minus values are means ± SD.
The net clinical outcome was significantly reduced in the switched group compared to the continued group (8.7% vs 12.1%; RR: 0.64; 95% CI: 0.43-0.97;

Forest plots for the clinical outcomes.

Funnel plot of the primary outcome.
The results of subgroup analyses showed an interaction based on the timing of de-escalation (
Subgroup Analyses of the Net Clinical Outcomes in Relation to Baseline Covariates.

Abbreviations: TOPIC, Timing of Platelet Inhibition After Acute Coronary Syndrome; TROPICAL-ACS, The Testing Responsiveness to Platelet Inhibition on Chronic Antiplatelet Treatment for Acute Coronary Syndromes.
Discussion
In the present meta-analysis, there are several noteworthy findings. First, de-escalation of dual antiplatelet intensity after the early ischemic phase post-PCI compared to the standard continuation of therapy was associated with reduced adverse clinical outcomes in regard to the net thrombotic and bleeding events. Second, the benefit of de-escalation was not dependent on the initial potent P2Y12, and switching from either ticagrelor or prasugrel maintained a net clinical benefit. Third, the benefit of de-escalation was more pronounced beyond the early ischemic phase from the cardiac event.
Potent P2Y12 blockade is the cornerstone in the management of ACS due to a significant reduction in ischemic complications. The clinical superiority of ticagrelor and prasugrel over clopidogrel in patients treated with ACS has been demonstrated in the Platelet Inhibition and Patient Outcomes and Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction 38 trials, respectively. 8,9 The increased major bleeding risk involved with such therapy, especially beyond 30 days of cardiac events, has limited even wider usage. 10 Therefore, in routine clinical practice, de-escalation from a novel P2Y12 inhibitor to clopidogrel is common due to cost considerations as well. 2 Nevertheless, prior studies of the pharmacodynamic effects of potent P2Y12 inhibitors showed de-escalation to clopidogrel was associated with high platelet reactivity. 11 -14 In the Switching Antiplatelet Therapy-4 Study, de-escalation from ticagrelor to clopidogrel was associated with an increase in platelet reactivity, suggestive of a drug–drug interaction that could translate clinically into a higher ischemic complication risk. However, administration of a loading clopidogrel dose was found to mitigate these observations. 11 In our study, we found that de-escalation therapy beyond the early ischemic phase was associated with a significant reduction in combined ischemic and bleeding events. The benefits of such a strategy were more apparent when de-escalation occurred at least 1 month after the event. However, the results of the subgroup analysis should be taken as hypotheses generating, and further adequately powered trials are needed to examine such effects.
The TROPICAL-ACS trial (n = 2610) was the first trial that incorporated PFT to guide prasugrel de-escalation, accounting for clopidogrel’s substantial response variability.
7
The 1-year net clinical benefit (combined ischemic and bleeding end point) was reduced in the PFT-guided group (7% vs 9%;
Limitations of this analysis include the lack of patient-level data, differences in clinical outcomes, varied follow-up duration, and inconsistency in the timing of switching. There were also a limited number of trials and relatively few events. Therefore, unless de-escalation is necessary due to clinical bleeding or cost considerations, further adequately powered RCTs are first necessary to evaluate de-escalation (timing and dosing) and the role of genetic and PFT prior to applying such a strategy in routine practice.
Conclusion
De-escalation of dual antiplatelet therapy beyond the early ischemic phase poststent implantation compared to the standard continuation of therapy was associated with improved clinical outcomes with regard to net thrombotic and bleeding events, although further confirmatory randomized clinical trials are needed.
Footnotes
Acknowledgments
The authors would like to thank Katherine Negele, Hurley Medical Center, Research Department, for assistance with manuscript editing.
Author Contributions
B. Kheiri and M. Osman contributed to conception and design; acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Abdalla, A. Chahine, S. Ahmed, and K. Osman contributed to conception and design; acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. G. Bachuwa, M. Hassan, and D. L. Bhatt contributed to conception and design; acquisition, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.L.B. discloses the following relationships—Advisory Board: Cardax, Elsevier Practice Update Cardiology, Medscape Cardiology, Regado Biosciences; Board of Directors: Boston VA Research Institute, Society of Cardiovascular Patient Care, TobeSoft; Chair: American Heart Association Quality Oversight Committee; Data Monitoring Committees: Baim Institute for Clinical Research (formerly Harvard Clinical Research Institute, for the PORTICO trial, funded by St Jude Medical, now Abbott), Cleveland Clinic, Duke Clinical Research Institute, Mayo Clinic, Mount Sinai School of Medicine (for the ENVISAGE trial, funded by Daiichi Sankyo), Population Health Research Institute; Honoraria: American College of Cardiology (Senior Associate Editor, Clinical Trials and News, ![]() ; Vice-Chair, ACC Accreditation Committee), Baim Institute for Clinical Research (formerly Harvard Clinical Research Institute; RE-DUAL PCI clinical trial steering committee funded by Boehringer Ingelheim), Belvoir Publications (Editor in Chief, Harvard Heart Letter), Duke Clinical Research Institute (clinical trial steering committees), HMP Global (Editor in Chief, Journal of Invasive Cardiology),
; Vice-Chair, ACC Accreditation Committee), Baim Institute for Clinical Research (formerly Harvard Clinical Research Institute; RE-DUAL PCI clinical trial steering committee funded by Boehringer Ingelheim), Belvoir Publications (Editor in Chief, Harvard Heart Letter), Duke Clinical Research Institute (clinical trial steering committees), HMP Global (Editor in Chief, Journal of Invasive Cardiology),
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.S. received a research grant (Abbott).
