Abstract
Aims:
There are limited data on aspirin (ASA) desensitization for patients with coronary disease. We present our experience with a rapid nurse-led oral desensitization regimen in patients with aspirin sensitivity undergoing coronary angiography.
Methods:
This single-center retrospective observational study includes patients with a history of ASA sensitivity undergoing coronary angiography with intent to perform percutaneous coronary intervention (PCI).
Results:
Between January 2012 and January 2017, 24 patients undergoing coronary angiography for stable coronary disease (7 cases) or acute coronary syndromes (non-ST-segment myocardial infarction [NSTEMI; 8 cases], STEMI [9 cases]) underwent aspirin desensitization having reported previous reactions to aspirin. At initial presentation, previous sensitivity reactions were reported as: mucocutaneous reactions in 17 patients (urticaria in 3 [13%], nonurticarial rash in 6 [25%], angio-oedema in 8 [33%]), respiratory sensitivity in 4 (17%), and systemic anaphylactoid reactions in 3 (13%). Seventeen (71%) patients underwent PCI. Desensitization was acutely successful in 22 (92%) patients and unsuccessful in 2 (8%) patients who both had a single short-lived episode of acute bronchospasm treated successfully with nebulized salbutamol. Fifteen successfully desensitized patients completed 12 months of aspirin; no patient had recurrent hypersensitivity reaction. Aspirin was stopped prior to 12 months in 7 patients (replaced by warfarin [1 case], no antiplatelet or single antiplatelet clinically indicated and clopidogrel chosen [4 cases], patient choice without evidence of recurrent hypersensitivity [1 case], and death due to cardiogenic shock following STEMI [1 case]).
Conclusion:
A rapid aspirin desensitization protocol is safe and effective across a broad spectrum of hypersensitivity reactions and clinical presentations.
Introduction
Aspirin plays a vital role in both the medical management of coronary artery disease and following revascularization, whether percutaneously or surgically. However, aspirin intolerance occurs in 6% to 20% of the general population with “true” aspirin hypersensitivity occurring in between 0.6% and 2.4%. 1,2 In patients requiring antiplatelet monotherapy who are intolerant of aspirin, guidelines recommend an alternative agent such as clopidogrel 3 ; however, following acute coronary syndromes and in particularly after percutaneous coronary intervention (PCI), dual antiplatelet therapy is recommended. 3,4 Aspirin desensitization represents an option for these patients; however, many “standard” desensitization protocols require several days making them impractical in the acute setting. 5 –8
We report our single-center real-life experience of a rapid nurse-led aspirin desensitization protocol in patients with a wide range of hypersensitivity reactions and both stable and unstable coronary artery disease.
Methods
Patient Selection
The James Cook University Hospital is an interventional cardiology center providing PCI (including 24/7 primary PCI) for over 1.5 million people in the Teesside, County Durham and Darlington and North Yorkshire regions. We perform approximately 1650 coronary interventions per year.
Patients were identified by searching our electronic patient record database of clinic letters, discharge summaries and procedural reports for the phrase “desensiti” (this search includes “desensitization,” “desensitization,” “desensitized,” and “desensitized”) and “de-sensiti” for hyphenated spellings.
All patients who presented to our center for either elective or urgent/emergency coronary angiography ± revascularization between June 2012 (introduction date of the protocol) and June 2017 were included.
Desensitization Protocol
Aspirin desensitization was performed prior to angiography in all patients except those presenting with ST-segment myocardial infarction (STEMI). In patients presenting with STEMI, an alternative antiplatelet (clopidogrel or ticagrelor) was administered along with unfractionated heparin and intravenous glycoprotein inhibitors with desensitization taking place postprocedure.
Our protocol is based on existing rapid aspirin desensitization protocols described in the current literature. 9 –11 Desensitization involved the oral administration of 5 sequential doses of aspirin solution (Table 1) over a period of 2 hours to a target dose of 75 mg, the standard United Kingdom treatment dose. All patients had blood pressure, oxygen saturation, and peak flow measurements every 30 minutes during the drug administration and for 3 hours after the final dose. The protocol was nurse-led and a doctor was not required to be present during the administration; however, a nominated doctor was available at short notice if required. A crash trolley with full resuscitation equipment including defibrillator was nearby. All desensitizations were performed in a standard ward setting and continuous electrocardiogram (ECG) monitoring was not routinely performed unless indicated on other clinical grounds.
Aspirin Desensitization Protocol.
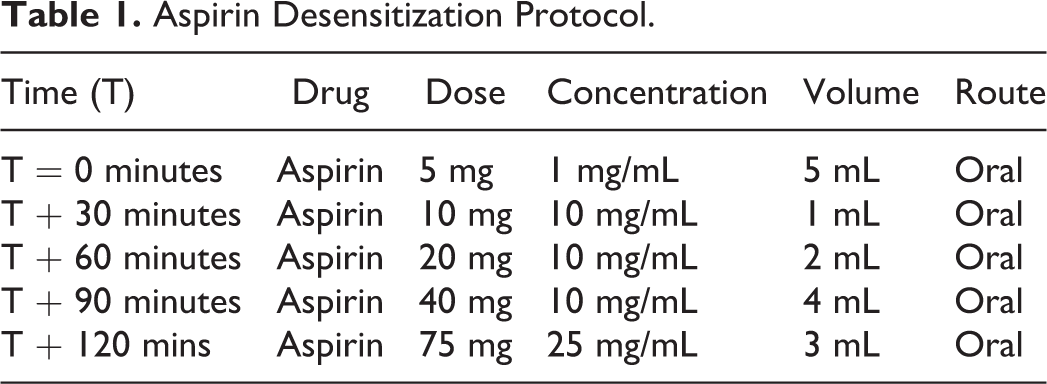
Pretreatment with steroids, antihistamines or antileukotrienes was not routinely administered.
Outcomes
The primary outcome was discontinuation of aspirin due to a hypersensitivity reaction prior to completion of 12-months treatment following angioplasty.
Statistical Analysis
Continuous variables were presented as mean (standard deviation, min-max). Comparisons between proportions were performed using a χ2 test.
Results
From January 2012 to January 2017, 24 patients underwent aspirin desensitization using the rapid desensitization protocol. Prior reactions were: mucocutaneous reactions in 17 patients (urticaria in 3 [13%], nonurticarial rash in 6 [25%], angio-oedema in 8 [33%]), respiratory sensitivity in 4 (17%), and systemic anaphylactoid reactions in 3 (13%). Baseline demographics are shown in Table 2; patients were aged 67.6 ± 9.1 years (48-86) and 14 (58%) were male. Eleven (41%) patients were taking inhaled bronchodilators and/or corticosteroids due to concurrent respiratory disease and 2 (7.1%) patients were taking nasal corticosteroid sprays to manage nasal polyps.
Demographics.

Abbreviations: CABG, coronary artery bypass grafting; NOAC, novel oral anticoagulation (direct factor X inhibitors); PCI, percutaneous coronary intervention; STEMI, ST-segment myocardial infarction; VKA, vitamin K antagonist.
Two (8.3%) had previously undergone coronary artery bypass grafting (CABG). None had undergone previous PCI. Nineteen (79%) were treated with β blockers at the time of desensitization. Only one patient was premedicated; this gentleman was immunosuppressed following a renal transplant and after consultation with both renal and immunology specialists was premedicated with antihistamines (fexofenadine), antileukotrienes (montelukast), and steroids.
Acute coronary syndrome was the initial diagnosis in 17 (71%) cases (NSTEMI [8 cases] or STEMI [9 cases]). All 9 patients presenting with STEMI underwent primary angioplasty and were treated periprocedurally with a single antiplatelet agent (clopidogrel [2 cases], ticagrelor [6 cases], prasugrel [1 case]); intravenous glycoprotein inhibitors (Tirofiban) administered in 5 (63%) cases. These patients were then desensitized as an inpatient during the first 24 hours following their procedure. Patients presenting with NSTEMI were desensitized prior to undergoing coronary angiography.
The desensitization procedure was acutely successful in 22 (92%) cases. Two cases developed bronchospasm after completion of the protocol; both had undergone primary PCI for STEMI. One patient reported a previous anaphylactoid reaction and had acute bronchospasm following completion of the protocol. One patient previously had angio-oedema and had an exacerbation of their poorly controlled asthma following completion of the protocol without evidence of angio-oedema. Both patients were successfully treated with nebulized salbutamol and were discharged on a single antiplatelet agent (prasugrel and clopidogrel, respectively) at standard doses.
There was one death in the cohort following successful desensitization. This male patient had a large anterior STEMI, presenting in cardiogenic shock and undergoing primary PCI as an emergency. He underwent successful desensitization without evidence of recurrent hypersensitivity the day following his procedure but subsequently deteriorated over the following 4 days, developing cardiogenic shock secondary to his severe left ventricular dysfunction refractory to medical therapy. There was no clinical evidence to suggest a further hypersensitivity reaction contributing to the death.
Following successful desensitization, 15 patients successfully completed their prescribed 12-month course of aspirin (68%). Aspirin was discontinued in 7 (32%) cases; for medical reasons in 6 (27%) cases (replaced by warfarin [1 case], no antiplatelet or single antiplatelet clinically indicated and clopidogrel chosen [4 cases], death due to cardiogenic shock [unrelated to the desensitization protocol] following STEMI [1 case]) and patient choice for personal reasons without evidence of recurrent hypersensitivity (1 case; 4.5%). No patients had a recurrent hypersensitivity reaction following acutely successful desensitization.
Revascularization was performed in 18 patients (PCI [17 cases], CABG [1 case]). Of the 17 patients who underwent PCI, drug-eluting stents were used in 15 (88%) patients, drug coated stents in 1 (5.9%) patient, and balloon angioplasty alone in 1 (5.9%) patient. Additional antiplatelet therapy is documented in Table 2.
Discussion
The key findings of our study are:
The rapid aspirin desensitization protocol was effective in 92% of patients with known/suspected coronary artery disease presenting with stable and unstable cardiac presentation, including STEMI. The protocol was safe with no serious adverse effects from its implementation. Adverse reactions were uncommon and responded quickly to treatment. There has been no recurrence of hypersensitivity reactions in the medium term.
Dual antiplatelet therapy with a combination of aspirin and P2Y12 inhibitor (such as clopidogrel, prasugrel, or ticagrelor) is the gold standard treatment following acute coronary syndromes and PCIs. 3,4 Aspirin hypersensitivity may prevent patients from receiving optimal treatment following these events. However, aspirin desensitization protocols are not commonly used in real-world practice as they can be time consuming. 7,8,11 –14
The Scripps clinic protocol is the most widely reported protocol for aspirin desensitization (7). Used for the diagnosis and management of aspirin-exacerbated respiratory disease (AERD), graded doses of oral aspirin are administered over 2 to 3 days ending with a final dose of 650 mg; patients are simultaneously treated with antileukotriene agents to reduce bronchospastic reactions. Over 1400 patient have been successfully treated using this protocol without death or complication; however, the prolonged nature of the regime and the recommendation that it should not be used in patients taking concomitant β blockers (79% of our cohort were taking β blockers at the time of their desensitization) or following a recent myocardial infarction (71% of our cohort) limit the usefulness of this protocol in the setting of coronary artery disease.
A number of small studies have described the use of rapid aspirin desensitization protocols in the setting of coronary artery disease requiring PCI. Wong et al described 11 patients with cutaneous hypersensitivity, 9 of whom had known stable coronary artery disease, who underwent graded oral administration of aspirin every 20 minutes in the following doses; 0.1 mg, 0.3 mg, 1 mg, 3 mg, 10 mg, 30 mg, 40 mg, 81 mg, 162 mg, and 325 mg. 9 Patients were pretreated with antihistamines and/or corticosteroids prior to undergoing desensitization. Long-term aspirin was tolerated in 9 patients, although 2 patients also continued long-term antihistamines. Only one patient underwent subsequent angioplasty.
Silberman et al described 16 patients with a mixture of respiratory or cutaneous reactions who needed urgent PCI. 10 Patients received increasing doses of oral aspirin, either commencing at 1 mg with a doubling of the dose every 30 minutes to 100 mg (duration 3.5 hours) or commencing at 5 mg with a doubling of the dose every 30 minutes to 75 mg (duration 2.5 hours). There was no pretreatment with antihistamines or steroids, although β blockade was withheld predesensitization. Two patients had reactions, although one was successfully rechallenged and 11 of the 16 patients underwent successful PCI, tolerating aspirin and clopidogrel for the full 12-month course.
Similar protocols were used by Dalmau and Hernandez on a further 5 and 12 patients respectively; all patients were successfully desensitized and 9 in total underwent PCI. 11,15 Of note, all desensitization procedures took place in the hospital setting and no patients with anaphylactoid reactions were included in any of these small studies. De Luca et al encountered 43 patients with aspirin hypersensitivity and desensitized 42 patients over 4.5 hours with 8 gradually increasing doses to 250 mg (1, 2, 4, 8, 16, 32 64, 128, 250 mg) although the reason for such a high final dose is not clear. 16
Cortellini et al investigated a more pragmatic approach by tailoring the protocol depending on the degree of reaction, evaluating 61 patients and dividing them into low-risk mild reactions (30 patients) and high-risk severe reactions (31 patients). 17 Desensitization was then performed using a cumulative dose of 150 mg over 220 minutes, administered in 5 steps to patients deemed to be low risk and 12 steps to high-risk patients. This was successful in 29 of 30 low-risk patients and 28 of 31 high-risk patients. In a unique outpatient approach, Fajt et al desensitized 9 patients using 10 doses over 2 days or 12 days over 3 and a half days; this was successful in 8 patients but clearly not appropriate for unstable patients requiring urgent revascularization. 18
The largest evidence base currently is the ADAPTED registry which enrolled 330 patients including those with mucocutaneous (74.5%), respiratory (19.7%), and anaphylactoid (5.8%) aspirin hypersensitivity reactions and those with unstable coronary disease including NSTEMI (33.6%) and STEMIs (23.6%) 19 ; as in our study, patients with STEMI underwent desensitization following their procedure and were treated with glycoprotein inhibitors periprocedurally at the operator’s discretion. Using a rapid desensitization protocol (1 mg at 0 hour, 5 mg at 30 minutes, 10 mg at 60 minutes, 20 mg at 90 minutes, 40 mg at 210 minutes, and 100 mg at 330 minutes), successful desensitization was achieved in 95.4% of patients including those with anaphylactoid reactions and 71% underwent PCI. While all desensitization was performed in the hospital setting, monitoring was less than in previous studies with blood pressure, pulse, and oxygen saturations checked every 30 minutes.
Our experience reflects that of the ADAPTED registry; patients with all forms of hypersensitivity reactions and both stable and unstable coronary disease were studied and similar rates of acutely successful desensitization demonstrated (92% vs 95.4%, P = .412). Our protocol is significantly quicker than that used in the ADAPTED registry (120 vs 330 minutes) and more rapid than all previously described rapid desensitization protocols. Similarly, we administered a lower total dose than used in the ADAPTED registry (150 mg vs 176 mg) and comparable to Silbermann. All rapid desensitization protocols are significantly faster and use lower total doses than the protocols prescribed to trigger respiratory and cutaneous reactions aiding diagnosis of AERD (over 1 g total administered over 3 days in the Scripps protocol) and the lower target aspirin dose used in cardiac patients (75 mg) may therefore allow smaller doses to be administered more frequently without compromising success rates.
Compared to the ADAPTED study, similar levels of monitoring were used (routine clinical observations and peak flow measurements); continuous ECG monitoring during the desensitization protocol was only used for other clinical indications in patients post-STEMI. We propose that aspirin desensitization can be safely performed in an outpatient or day unit setting, although patients with anaphylactoid reactions may still be desensitized in a more monitored environment.
Two patients in our cohort presented with STEMI and underwent primary PCI with a single-drug-eluting stent; however, neither could be successfully desensitized postprocedure. Both patients were discharged on a single-antiplatelet agent (prasugrel [1 case], clopidogrel [1 case]) and have had no adverse events in the 12-month follow-up period. Despite evidence suggesting worse outcomes following PCI without aspirin and using clopidogrel alone, 20 trials evaluating antiplatelet monotherapy using more potent agents (ticagrelor, prasugrel) are ongoing. 21,22 Alternatively, patients who failed initial attempts at desensitization could be rechallenged following pretreatment with antihistamines, antileukotrienes, and steroids; however, we did not attempt this in either patient in our cohort. In patients with atrial fibrillation, the WOEST study suggested that outcomes from clopidogrel and warfarin were no worse than triple therapy with aspirin with reduced incidences of bleeding. 23 Whether the issue of aspirin hypersensitivity remains clinically relevant to patients undergoing PCI remains to be seen.
Strengths
All patients planned for coronary revascularization during the study period were included.
Limitations
The small study cohort and retrospective single-center nature of the study limits generalization of the results. Further study with a larger patient cohort is needed to confirm our findings.
Hypersensitivity reactions were patient-reported and oral challenge or immunological testing was not performed. It is possible that some cases may either not have true hypersensitivity or that some cases did not report their allergy and took aspirin without incident.
Aspirin and nonsteroidal anti-inflammatory drug (NSAID) reactions are now divided into crossreactive nonallergic nonimmunologically mediated reactions (NSAIDs exacerbated respiratory disease [NERD], NSAIDs exacerbated cutaneous disease [NECD], and NSAIDs-induced urticaria-angioedema [NIUA]) and selective-allergic immunologically mediated reactions (single NSAID-induced urticaria, angioedema, or anaphylaxis [SNIUAA] or NSAID-induced delayed hypersensitivity reactions). 24 By these criteria, 3 patients had NIUA reactions (reacting to both aspirin and a different class of NSAID), 5 had NERD reactions and 1 had a NECD reaction. Unfortunately, we did not have sufficient information on prior NSAID use and/or reactions to differentiate between NIUA and SNIUAA reactions in 18 patients. Also, patients were often unable to recall the time frame of their reaction sufficiently to identify delayed reactions.
The background history of asthma or nasal polyps is not known in all patients.
Conclusion
Our single center experience of rapid aspirin desensitization using a faster protocol than those in the published literature demonstrates high rates of success and includes all types of hypersensitivity reaction and patients with stable coronary disease and acute coronary syndromes including STEMI. Aspirin desensitization should continue to be used in all patients with aspirin hypersensitivity until trials of single potent antiplatelet agents following PCI are completed. 25
Footnotes
Authors Contribution
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation and gave final approval. M. J. contributed to conception, design, acquisition, analysis, and interpretation and drafted and critically revised the manuscript. S. C., S. B., and D. A. contributed to acquisition, analysis, and interpretation and critically revised the manuscript. J. S. and M. B. contributed to conception and design and critically revised the manuscript. D. M., A. S., P. D. W., J. H., J. C., and R. W. contributed to acquisition and critically revised the manuscript. N. S. contributed to conception, design, analysis, and interpretation and critically revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
