Abstract
Objectives:
We explored the impact of rosiglitazone on endothelial function in normal volunteers and its interaction with glyceryl trinitrate (GTN)-induced abnormalities in endothelial function. We hypothesized that rosiglitazone would have a neutral effect on endothelial function in normal volunteers and would favorably modify endothelial dysfunction induced by GTN.
Methods:
In this double-blind, randomized, placebo-controlled study, 44 participants were randomized to placebo, rosiglitazone (4 mg twice daily), transdermal GTN (0.6 mg/h), or both GTN and rosiglitazone. After 7 days of treatment, participants underwent measures of forearm blood flow during brachial artery infusion of acetylcholine (Ach). Serum glucose concentrations and insulin sensitivity were assessed.
Results:
Unexpectedly, rosiglitazone-treated participants experienced blunted responses to endothelium-dependent responses to Ach (P < .05 vs placebo). Sustained GTN administration caused similar abnormalities in endothelial function (P < .05 vs placebo) and rosiglitazone + GTN (P < .05 vs placebo; P = ns vs rosiglitazone). Interestingly, co-infusion of the antioxidant vitamin C improved endothelial responses in those randomized to rosiglitazone and GTN alone (P = not significant [ns] compared with placebo), but it did not improve endothelial function in those treated with rosiglitazone + GTN. Neither rosiglitazone nor GTN treatment modified the measures of glucose metabolism.
Conclusions:
Unexpectedly, therapy with rosiglitazone caused abnormalities in endothelial function in normal volunteers. These findings have important implications with respect to drug development and surveillance.
Introduction
Prior to its removal from the European market and the increased restrictions on its use in the United States over concerns of cardiovascular risk, rosiglitazone held promise as an insulin-sensitizing agent capable of improving endothelial function in patients with diabetes mellitus and the metabolic syndrome. 1 –3 Rosiglitazone improves glycemic control by modulating transcription factors involved in the regulation of glucose homeostasis and is widely used in the management of type 2 diabetes mellitus. 4 –6 Beyond its effects on glucose metabolism, rosiglitazone has also been proposed to have antioxidant and anti-inflammatory properties. 7 –9 Several observations have demonstrated that rosiglitazone can reduce vascular oxygen radical bioavailability. 7,9,10
Glyceryl trinitrate (GTN) and other organic nitrates are commonly used in the management of cardiovascular disease. 11 However, the clinical utility of nitrates rapidly diminishes with continuous dosing, a phenomenon termed “nitrate tolerance.” An increased production of reactive oxygen species during sustained nitrate therapy has been associated with both the development of tolerance and endothelial dysfunction. 12 –14 Since oxidative stress appears to play a central role in the development of nitrate-induced endothelial dysfunction, 14 we hypothesized that rosiglitazone might have favorable effects on the development of endothelial dysfunction induced by GTN therapy. Beyond exploring this potential mechanistic interaction, this investigation was also undertaken to further explore reports of an increased risk of adverse cardiovascular events in patients taking both rosiglitazone and organic nitrates. 15
Methods
The Mount Sinai Hospital Research Ethics Board approved the study protocol and informed written consent was obtained from all participants.
Participants
Forty-four healthy, nonsmoking male volunteers, age 18 to 30, were recruited. Prior to enrollment, participants underwent a review of their medical history and had a physical examination. Participants abstained from alcohol or any drugs, including supplemental vitamins, for the duration of the study. All studies were done in the fasting state.
Study protocol
Forearm blood flow (FBF) was measured in both arms by venous occlusion strain-gauge plethysmography (DE Hokanson Inc, Bellevue, Washington) with calibrated mercury-in-silastic strain gauges. 16,17 Upper arm cuffs were inflated to 40 mm Hg and deflated at 10-second intervals (Hokanson rapid cuff inflator, DE Hokanson Inc). Circulation of the hand was excluded by inflating wrist cuffs to 200 mm Hg during measurement periods. With venous outflow occluded, continued arterial inflow leads to an increase in forearm diameter that is linearly related to arterial inflow. Baseline FBF and changes induced in FBF by pharmacologic interventions were recorded as the average of 5 consecutive measurements. A blinded investigator performed all FBF measurements and final calculations. On visit 2, FBF was measured in response to brachial infusions of normal saline and vasoactive agents as described below.
Study day 1
After screening for admission into the study, a venous blood sample was obtained. Subsequently, standing blood pressure and heart rate measurements (mean of 3 measurements) were obtained using an automatic, calibrated, sphygmomanometer (Critikon Company LLC, Tampa, Florida). Participants were randomized to receive placebo (twice daily [bid]; n = 12), rosiglitazone alone (4 mg, bid; n = 11, a dose shown to improve endothelial function 18 ), transdermal GTN (0.6 mg/h; n = 11, a dose known to cause endothelial dysfunction 16,19 ), or a combination of the same dosages of the 2 drugs (rosiglitazone + GTN; n = 10). Placebo and rosiglitazone were given in a double-blind fashion. Placebo transdermal preparations were not available, therefore participants randomized to GTN alone or GTN + rosiglitazone received transdermal GTN in a fashion with the investigator blind to the treatment, that is a nurse coordinator not involved in any study measurements performed the randomizations. Participants were instructed to wear the GTN preparations under clothing during study visits after their randomization and to direct any questions about their study treatment to that coordinator. All participants were asked to return 3 hours after the first dose of the study medication to repeat standing blood pressure and heart rate measurements.
Study day 2
Participants within each of the 4 groups returned to the laboratory after 7 days of study therapy. A second venous blood sample was obtained and standing blood pressure and heart rate measurements were repeated. The brachial artery of the nondominant arm was cannulated, and FBF was measured at baseline (normal saline infusion). Blood flow was also measured in the contralateral arm to control for any systemic effects caused by the brachial artery infusions in the nondominant arm. The endothelium-dependent vasodilator acetylcholine (Ach; Novartis Pharmaceuticals, Ontario, Canada) was infused at 7.5, 15, and 30 μg/min along with normal saline. Thirty minutes after the completion of Ach infusions, FBF measurements (during saline infusions alone) were repeated to ensure the responses had returned to baseline. At this point, Ach infusions were repeated during co-infusion with vitamin C at 24 mg/min. This concentration of vitamin C is expected to achieve a local plasma concentration of ~10 mmol/L (ie, approximately 50 times the normal concentration). This infusion rate of vitamin C has been used in previous studies to demonstrate a role of reactive oxygen species in the pathogenesis of endothelial dysfunction in patients with cardiovascular disease. 20 Each concentration of Ach was infused for 6 minutes at a constant rate of 0.4 mL/min (Harvard apparatus, South Natick, Massachusetts), and FBF measurements were taken during the last 3 minutes of infusion. All responses were evaluated as changes from control values (normal saline infusion) immediately before each drug infusion. Intra-arterial blood pressure was recorded after each infusion (Horizon 2000; Mennen Medical Inc, Clarence, New York) using the average of at least 15 cardiac cycles. Between drug infusions, normal saline was infused until the flow returned to baseline values. At the end of the study, the arterial line was removed, study medications discontinued, and participants were discharged from the laboratory. All analyses of blood flow, heart rate, and blood pressure measurements were performed in a blinded fashion.
Measurement of glucose and insulin concentrations
Serum glucose concentrations were measured by the glucose oxidase technique on an automated analyzer, with samples processed by the Mount Sinai Hospital central laboratory. Specific insulin was measured using the Roche Elecsys 1010 immunoassay analyzer and a chemiluminescence immunoassay kit (Roche Diagnostics, Laval, Quebec, Canada). This assay has 0.05% cross-reactivity to intact human proinsulin and the primary circulating split form des-31, 32 proinsulin.
An estimate of insulin sensitivity, the homeostasis model assessment–insulin resistance (HOMA-IR) 21 was derived from the fasting insulin and glucose values obtained before and after each treatment period.
HOMA-IR = [fasting insulin (mU/L) × fasting glucose (mmol/L)]/22.5
Statistical Analysis
All data are expressed as mean ± standard error. A value of P < .05 was set as the threshold for significance. Heart rate and blood pressure responses were evaluated using a repeated measures analysis of variance (ANOVA). Differences in blood flow responses were tested using a 2-period, multigroup, repeated-measures ANOVA on the changes induced by increasing infusion rates of Ach compared with the corresponding baseline infusion (saline). The periods were defined as the responses to Ach in the presence and absence of vitamin C. If the overall group effect was significant, intratreatment comparisons were made by a Student Newman-Keuls post hoc test. Statistical analysis was performed using Statview 4.0 (SAS Institute, Cary, North Carolina).
Results
Blood Pressure and Heart Rate Responses
Baseline assessment of standing heart rate and systolic blood pressure did not differ between treatment groups (Table 1). When compared with the baseline values, a significant decrease in standing systolic blood pressure was observed 3 hours after the administration of the first transdermal GTN preparation in the participants randomized to GTN alone and rosiglitazone + GTN group (P < .05, ANOVA; Table 1). A corresponding significant increase in heart rate was also observed in the same groups (P < .05, ANOVA; Table 1). After 7 days of continuous treatment, the hemodynamic effects of GTN were no longer evident in either group (P = not significant [ns] compared with placebo and with initial baseline measures). Blood pressure and heart rate in the placebo and rosiglitazone-alone groups were similar at all time points.
Blood Pressure and Heart Rate Responses a
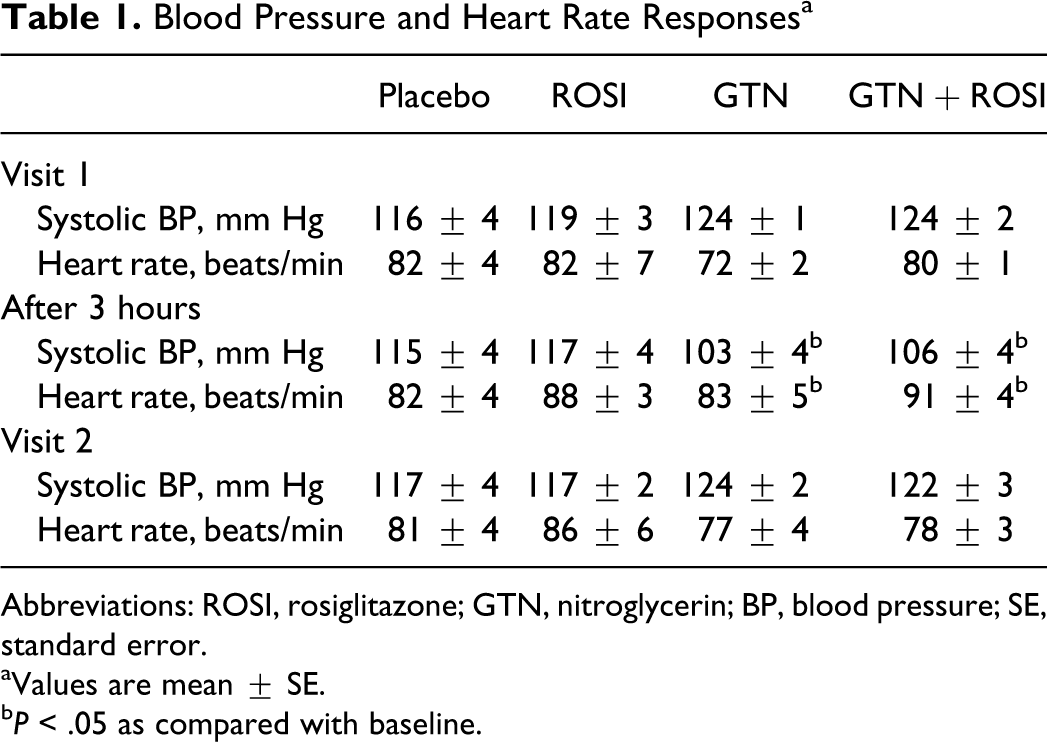
Abbreviations: ROSI, rosiglitazone; GTN, nitroglycerin; BP, blood pressure; SE, standard error.
aValues are mean ± SE.
b P < .05 as compared with baseline.
The Effect of GTN and Rosiglitazone on Endothelium-Dependent Vasodilation
The mean basal (saline infusion) FBFs were similar in all groups (Table 2). When normalized to the contralateral arm, the ratios were 1.1 ± 0.1, 1.1 ± 0.3, 1.2 ± 0.4, and 1.2 ± 0.2 for participants randomized to placebo, rosiglitazone, GTN, and GTN + rosiglitazone, respectively (P = ns).
Forearm Blood Flows (Infused to Noninfused Arm Ratios) a
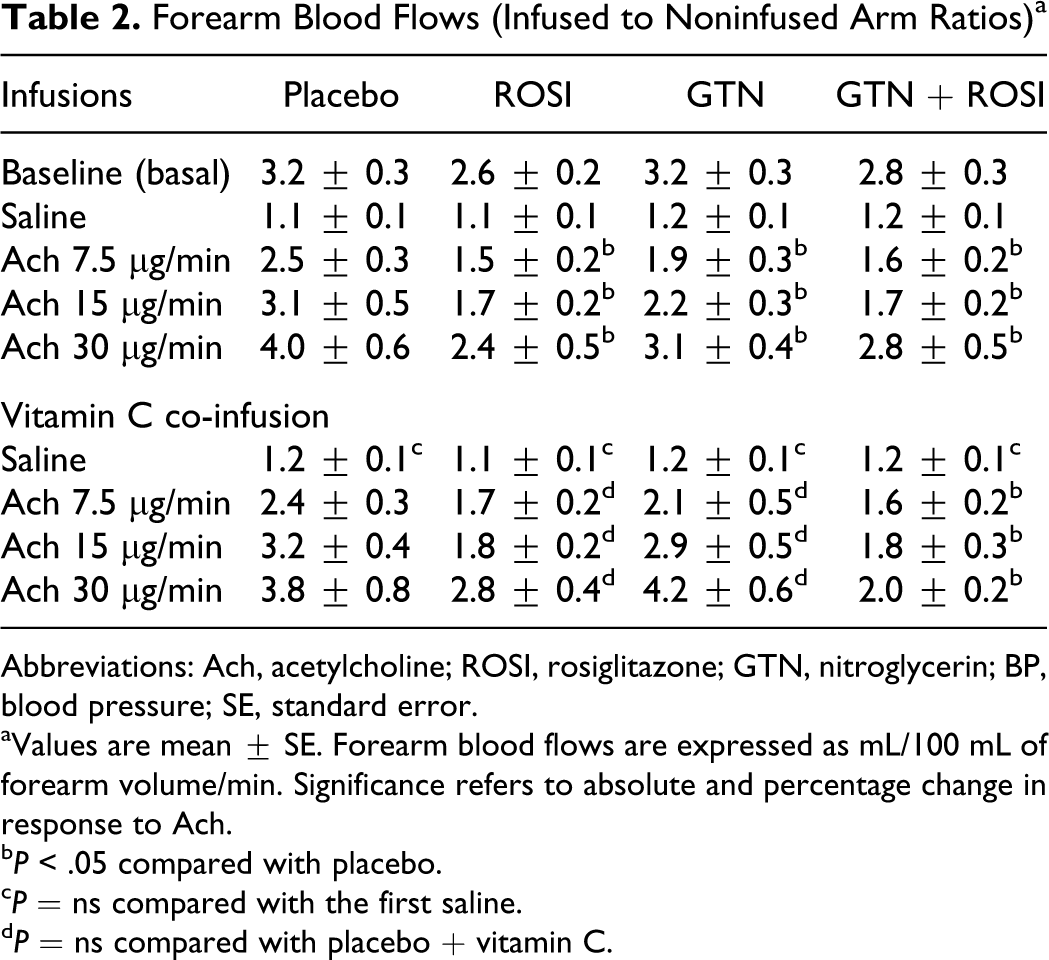
Abbreviations: Ach, acetylcholine; ROSI, rosiglitazone; GTN, nitroglycerin; BP, blood pressure; SE, standard error.
aValues are mean ± SE. Forearm blood flows are expressed as mL/100 mL of forearm volume/min. Significance refers to absolute and percentage change in response to Ach.
b P < .05 compared with placebo.
c P = ns compared with the first saline.
d P = ns compared with placebo + vitamin C.
Responses to intra-arterial infusions of Ach caused a significant dose-dependent increase in FBF in all groups (P < .05; Table 2 and Figure 1). The responses to Ach were significantly blunted in the GTN, rosiglitazone, and GTN + rosiglitazone groups compared with placebo (group effect, P < .05 2-way repeated-measures ANOVA), documenting that GTN, rosiglitazone, and their combination were associated with the development of endothelial dysfunction.
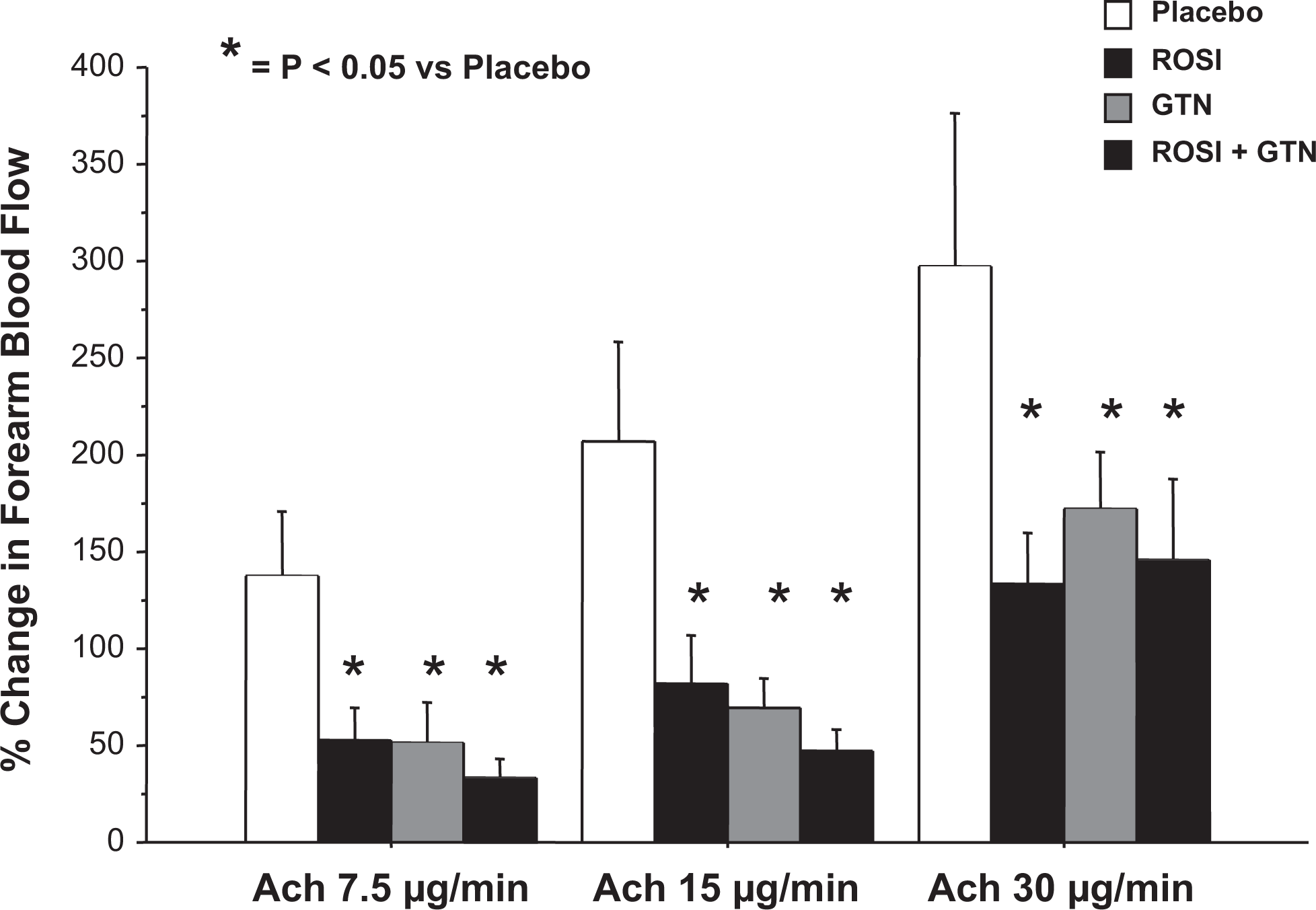
Responses of forearm blood flow (expressed as absolute percentage change from baseline) to Ach. Responses to Ach were significantly attenuated in participants randomized to GTN, ROSI, and GTN + ROSI compared to placebo. *P < .05 compared to placebo; effect of group in ANOVA. Ach indicates acetylcholine; ROSI, rosiglitazone; GTN, nitroglycerin; ANOVA, analysis of variance.
The Effect of Vitamin C on Endothelium-Dependent Vasodilation
Prior to infusions of vitamin C, saline was reinfused to ensure that FBFs were returned to levels comparable to baseline (P = ns; Table 2). Co-infusion of vitamin C did not modify the responses to Ach in the placebo group (P = ns). In the participants randomized to GTN, vitamin C co-infusion was associated with the normalization of the FBF responses to Ach (P = ns compared with placebo group). Similarly, in participants randomized to rosiglitazone alone, intra-arterial vitamin C improved responses to Ach infusions (P = ns compared with placebo group). In contrast, the responses to Ach during vitamin C co-infusion remained significantly blunted in the group randomized to GTN + rosiglitazone when compared with placebo and GTN (2-way ANOVA, effect of group, P < .02; Table 2 and Figure 2).
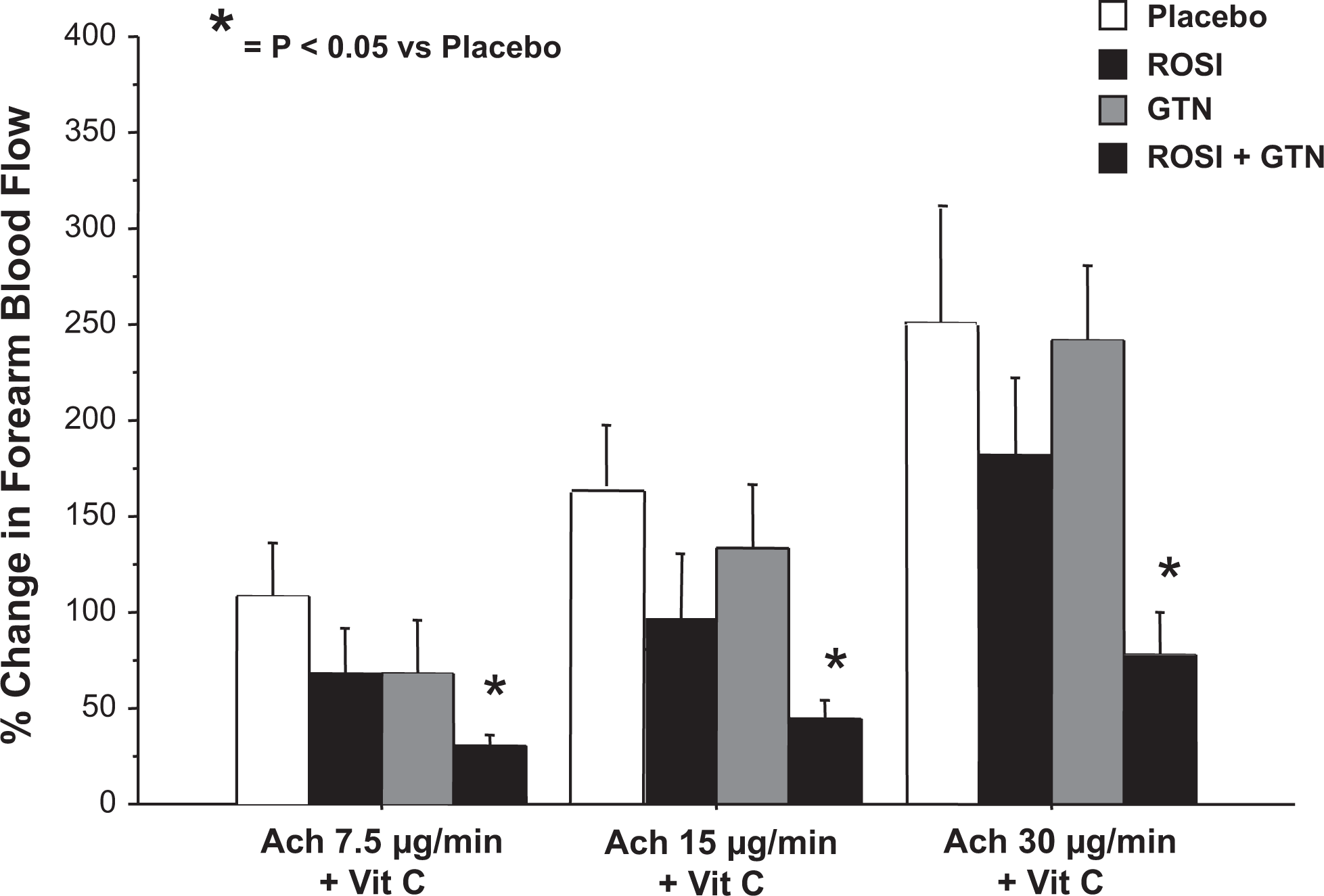
Forearm blood flow responses to vitamin C and acetylcholine. In the presence of vitamin C, endothelial responses remained significantly attenuated in participants randomized to ROSI + GTN compared to placebo. In contrast, forearm blood flow responses in the participants randomized to GTN and rosiglitazone were no longer significantly different from placebo. *P < .05 compared to placebo; effect of group in ANOVA. Ach indicates acetylcholine; ROSI, rosiglitazone; GTN, nitroglycerin; ANOVA, analysis of variance.
Indices of Insulin Sensitivity
As shown in Table 3, neither rosiglitazone nor GTN treatment modified fasting glucose, fasting insulin, or the HOMA-IR. Baseline median fasting glucose levels were 5.0, 4.8, 5.0, and 5.1 mmol/L for participants randomized to placebo, rosiglitazone, GTN, and GTN + rosiglitazone, respectively. None of the study medications had a significant effect on the HOMA-IR index (P = ns, ANOVA; Table 3).
Insulin Sensitivity Indexes a
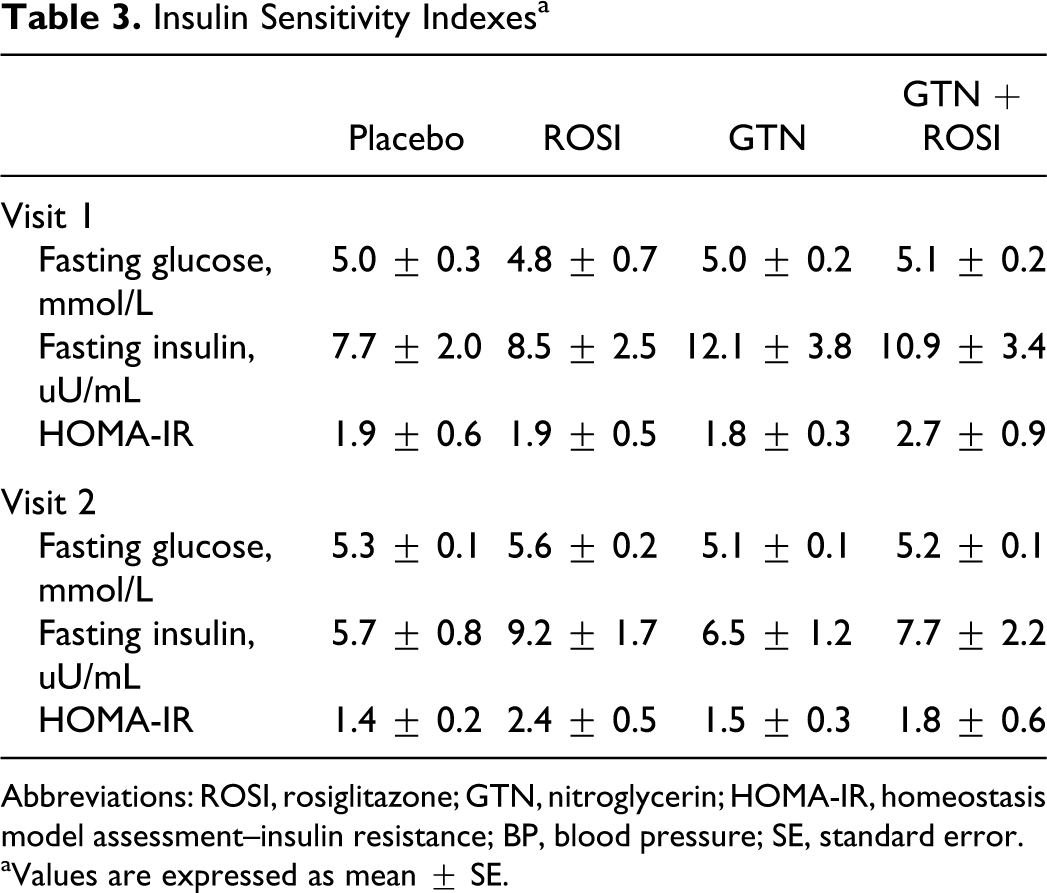
Abbreviations: ROSI, rosiglitazone; GTN, nitroglycerin; HOMA-IR, homeostasis model assessment–insulin resistance; BP, blood pressure; SE, standard error.
aValues are expressed as mean ± SE.
Discussion
Thiazolidinediones are a class of insulin-sensitizing drugs used in the treatment of diabetes mellitus. 4 Their effect on endothelial function has been evaluated in a number of clinical studies in nondiabetic patients with the metabolic syndrome. 1 –3 Wang et al 3 reported a significant improvement in both endothelium-dependent and nonendothelial independent vasomotor function after 8 weeks of treatment with rosiglitazone. Similarly, Campia et al observed an improvement in endothelial responses with pioglitazone in nondiabetic patients with major untreated cardiovascular risk factors. 22 In these reports, the improvements in endothelial function were also paralleled by an improvement in insulin sensitivity. Although the mechanism of these beneficial effects is uncertain, it seems likely that they are mediated, in part, by their ability to reduce free radical bioavailability caused by hyperglycemia-derived oxygen free radicals. 23 –26 Furthermore, thiazolidinediones also have favorable effects on free fatty acid and cholesterol profiles in patients with metabolic syndrome or frank diabetes which can also lead to a decrease in free radical bioavailability. 27,28 Therefore, the beneficial effects of thiazolidinediones on endothelial function may be manifested only in the presence of concurrent metabolic abnormalities. The finding that thiazolidinediones did not improve the endothelium-dependent vasomotor responses in nondiabetic individuals with coronary artery disease supports this hypothesis. 29 Therefore, we hypothesize that rosiglitazone may have heterogeneous effects on the endothelial function and, even in patients with diabetes, can have detrimental effects on endothelial function.
The important new finding of the current study is that short-term therapy with rosiglitazone alone caused abnormalities in endothelial function as assessed by FBF responses to Ach. The infusion of vitamin C improved endothelium-dependent vasodilator responses in the rosiglitazone group such that they were no longer significantly different from those observed in the placebo group. Co-treatment with rosiglitazone with continuous GTN therapy had a similar negative impact on endothelial function but, interestingly, coadministration with the antioxidant vitamin C did not modify this evidence of pharmacologically induced endothelial dysfunction. A possible explanation for this finding is that rosiglitazone-mediated effects on endothelial function are mediated through improvements in insulin sensitivity rather than changes in vascular nitric oxide (NO) bioavailability, as a previous study demonstrated rosiglitazone therapy did not alter the levels of oxidative stress or asymmetric dimethylarginine. 30
This study cannot address the exact mechanism of rosiglitazone-induced abnormalities in endothelial function. Its reversal with vitamin C demonstrates that an increase in free radical bioavailability is involved. However, the incompletely understood genome-level effects of thiazolidinediones suggest that a number of mechanisms could cause this change in vascular free radical bioavailability. Our data contrast with those of Hetzel et al 18 who found improvements in endothelial function in healthy volunteers randomized to treatment (21 days) with rosiglitazone. Differences in the techniques used to evaluate endothelial function (plethysmography vs flow-mediated dilation) may explain this conflict as the former examines resistance vessels and the latter conduit vessel responses. Importantly, assessment of conduit vessel endothelial function using flow-mediated dilation is not possible in studies using sustained therapy with GTN, as GTN persistently dilates the conduit arteries, making it difficult to assess the changes in NO bioavailability with various interventions. 31
After 7 days of treatment with GTN, participants randomized to GTN alone showed evidence of endothelial dysfunction. This finding, consistent with previous data from our laboratory, 16,19 was completely reversed by the administration of vitamin C, confirming that the modifications induced by continuous GTN therapy are mediated by a net increase in the bioavailability of reactive oxygen species. A number of studies, including those from our laboratory, have demonstrated that exposure to continuous GTN leads to an increase in the bioavailability of oxygen free radical species of which a number of sources have been proposed. 12 –14 Contrary to our hypothesis, rosiglitazone did not prevent the abnormality of endothelial function induced by nitrate therapy. In retrospect, this is not surprising, given the unexpected negative effect of the glitazone on endothelial function. Of note, other pharmacologic agents when given concurrently with GTN have been shown to completely prevent the development of such abnormalities via a mechanism that appears to be mediated by a prevention of GTN-induced increases in vascular free radical bioavailability. 19,32
The present findings have important clinical implications and provide a mechanistic background for observations questioning the safety of rosiglitazone, particularly in association with GTN, in patients with cardiovascular disease. Importantly, recent meta-analyses from the Food and Drug Administration 33 and from Nissen et al 34 found that rosiglitazone use might be associated with a higher risk of myocardial ischemic events. Interestingly, the risk appeared to be even greater in combination with nitrate therapy. 15,34 We think our results provide a potential mechanistic hypothesis for these observations. Is it possible that the positive effects of rosiglitazone mediated by an improved hyperglycemic control might, in certain cases, be counterbalanced by an increase in adverse vascular effects mediated by the development of worsening endothelial function? This possibility would be compatible with the observed negative interaction between rosiglitazone and organic nitrates, which also cause endothelial dysfunction. Concern about the potential for toxicity led to revised regulatory labels curtailing its use in patients with or at risk of heart failure. 15,35 –37 Most recently, based on the totality of evidence including prospective evidence of adverse invents in the Thiazolidinedione Intervention With Vitamin D Evaluation (TIDE) trial (ClinicalTrials.gov Identifier: NCT00879970), rosiglitazone has been withdrawn from many jurisdictions (www.ema.europa.eu). Finally, although it is quite likely that the use of rosiglitazone will soon disappear, our findings are important in that they suggest that early human testing of drugs that will be prescribed in patients with overt or major risk factors for cardiovascular disease should include a broader range of exploratory cardiovascular effect/effects testing during early (phase 1) stages of their development.
Footnotes
Acknowledgments
The authors recognize the support provided by the John H. Daniels Cardiac Research Centre and the Mecklinger and Posluns Cardiac Catheterization Research Laboratory of the Mount Sinai Hospital, Toronto.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant-in-aid of the Heart and Stroke Foundation of Ontario. Dr Parker holds a Career Investigator Award from the Heart and Stroke Foundation of Ontario, Canada.
