Abstract
Atrial fibrillation (AF) affects more than 3 million Americans and is expected to reach epidemic proportions as the US population ages. The presence of AF increases lifetime stroke risk nearly 5-fold. Conventionally, patients at moderate or high risk of stroke have been prescribed antiplatelet agents or vitamin K antagonists to reduce the risk, but each has significant limitations. Accordingly, the development of new oral anticoagulants (direct thrombin inhibitors [DTIs] and factor Xa inhibitors) has attracted significant interest. The DTI dabigatran etexilate was recently shown to provide superior risk reduction to warfarin for stroke and systemic embolism for patients with nonvalvular AF and recently gained US Food and Drug Administration approval for this indication. Dabigatran etexilate is the first new agent for this indication in the United States in more than 50 years. Herein, we outline the options for stroke prevention in AF in the new oral anticoagulant era. The efficacy and practical obstacles surrounding the use of warfarin are summarized. We then review the mechanism of action, efficacy, and safety of dabigatran—including clinically relevant pharmacokinetics. Practical issues of initiation, conversion of anticoagulant therapy, and recommendations for dabigatran use in patients at high risk of bleeding and other special populations are discussed. We conclude by proposing a role for dabigatran in the armamentarium of drugs available for the management of stroke risk in AF.
Introduction
Atrial Fibrillation and Stroke Risk
Atrial fibrillation (AF) is the most common serious arrhythmia encountered in clinical practice 1 and may affect over 12 million 2 Americans by 2050. In addition to providing effective control of rhythm and/or ventricular rate in AF, an important management goal in patients with AF is prevention of stroke and systemic thromboembolism (SSE). 3 –5
In contrast to the low stroke risk state of “lone AF” patients, 3 many others have increased risk; for example, those with prior stroke or transient ischemic attack have annual stroke rates of ≥10%, despite treatment with aspirin. 4 Multiple additional studies have identified several clinical variables that increase stroke risk in patients with AF, resulting in risk stratification schemes that classify patients at low, intermediate, and high risk of SSE. The CHADS2 score assigns 1 point for each of several risk factors (Congestive heart failure, Hypertension, Age greater than 75 years, and Diabetes mellitus), with an additional point for previous Stroke or transient ischemic attack, given the larger odds ratio associated with this clinical risk factor. 6 More recently, the CHA2DS2-VASc score was developed, which modified CHADS2 by assigning 2 points to age ≥75 years, reflecting the greater odds ratio associated with this factor, and 1 point each for Vascular disease, Age between 65 and 74 years, and female Sex. 7 Patients with a CHADS2 or CHA2DS2-VASc score of 0 are called low risk of SSE and may be managed with no therapy or aspirin. 3,4 Patients with a score of 1 are considered intermediate risk; their management is controversial. They may receive either aspirin or oral anticoagulation, 4,5 although the recent European guidelines recommend oral anticoagulation in favor of aspirin. 3 Scores of ≥2 identify patients at high risk of SSE, making them appropriate candidates for oral anticoagulation. 3 –5 Additional risk factors (eg, renal disease and hypercoagulable states) may also modulate the stroke risk in an individual patient.
Importantly, bleeding risk must also be assessed, for which there are also several risk stratification schemes, including the HAS-BLED score. 8 One point is assigned to each of the following components of the HAS-BLED score: Hypertension, Abnormal renal/liver function (1 potential point for each), previous Stroke, Bleeding history or predisposition, Labile coagulation tests, Elderly individuals (over age 65), and concomitant use of Drugs that increase bleeding risk (such as nonsteroidal anti-inflammatory drugs, aspirin, and/or clopidogrel) or alcohol abuse (1 potential point for any high bleed risk drugs and 1 potential point for alcohol). The CHADS2 or CHA2DS2-VASc score may be considered alongside the HAS-BLED score to estimate the risks and determine the optimal anticoagulation strategy for an individual patient. Although its role is evolving, percutaneous device occlusion of the left atrial appendage, or surgical occlusion or resection, may be considered for patients at unacceptably high bleeding risk. 9
Below we review the currently available options for oral anticoagulation to reduce SSE in AF, beginning with warfarin—the standard of care for the past 5 decades—and then focus on dabigatran etexilate (hereafter referred to as dabigatran), the first new agent available for this indication in the United States in the last 50 years.
Warfarin
Warfarin inhibits synthesis of multiple clotting factors necessary to form fibrin clots by inhibiting vitamin K epoxide reductase (gene VKORC1), 10 the enzyme that prevents regeneration (reduction) of vitamin K. An important cofactor, vitamin K, is oxidized during the carboxylation reaction, which activates factors II, VII, IX, X (major players in the extrinsic and intrinsic pathways of coagulation), as well as the anticoagulant proteins C and S (Figure 1). By inhibiting the activation of these factors, warfarin prevents the downstream activation of thrombin (or factor IIa). The coagulation cascade step involving thrombin is commonly termed the final common pathway of coagulation and is essential for the conversion of fibrinogen to fibrin and cross-linking of a fibrin clot, as well as homeostatic activation of upstream factors in the cascade (such as factors V, VIII, and protein C). 11 The degree of anticoagulation induced by warfarin is easily measured. The prothrombin time (PT) directly assays the degree of impairment of coagulation and is usually expressed as a ratio to control serum, the international normalized ratio (INR), with a normal 12 value of 1.0. Measurement of serum INR reliably assesses the degree of anticoagulation, 12 with an optimal target INR 4 for AF of 2.0 to 3.0. Vitamin K therapy may be used if necessary in cases of overdose. 13 Warfarin is also inexpensive and widely available since its introduction in 1954. 13
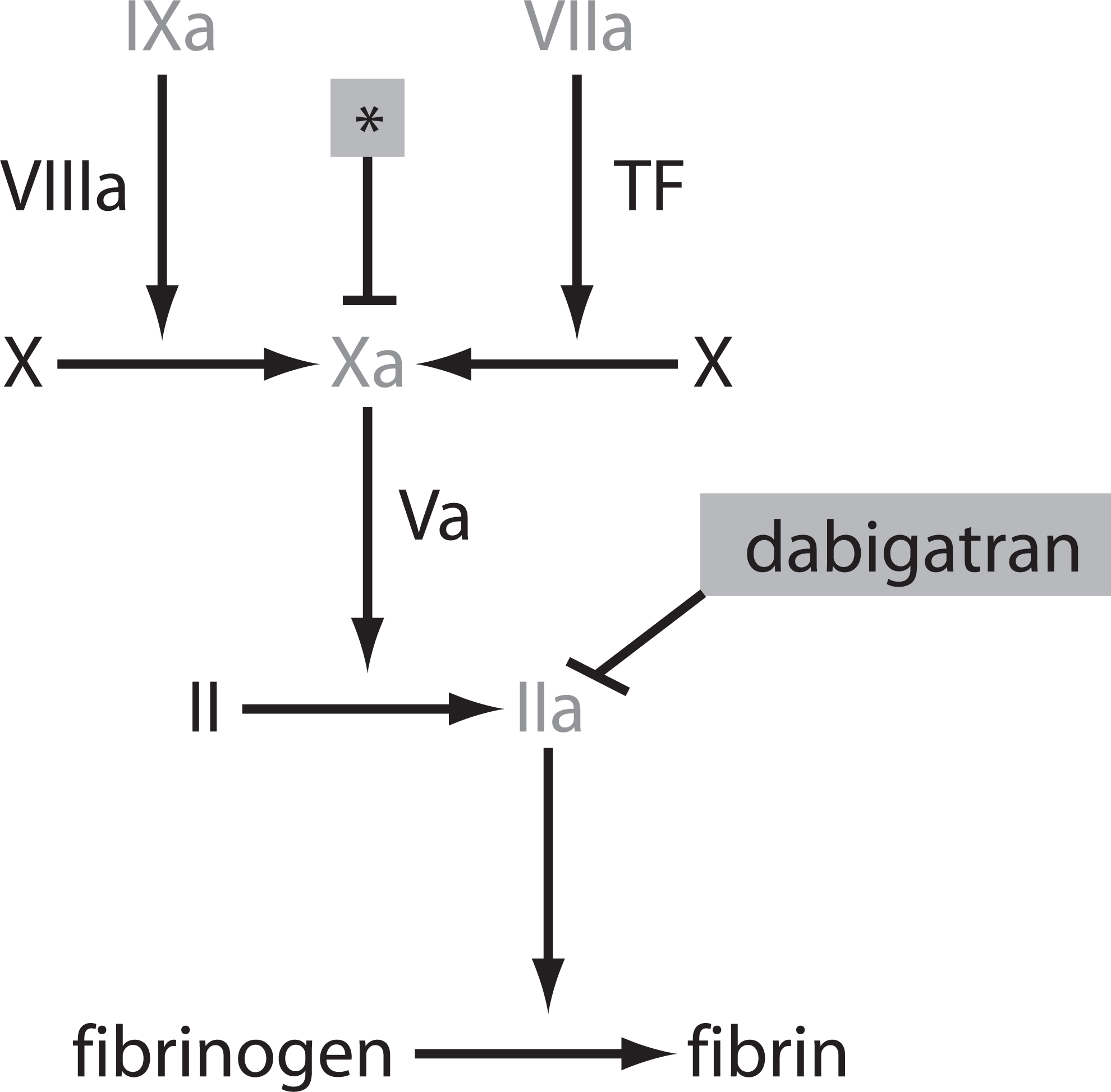
The coagulation cascade. Blood coagulation proceeds through a cascade of activation of humoral factors, culminating in the generation of thrombin, which catalyzes the conversion of fibrinogen to fibrin. Warfarin acts by inhibiting the activation of several of these factors (shaded in gray). Dabigatran, a direct thrombin inhibitor, reversibly inhibits factor IIa (thrombin). The site of action of factor Xa inhibitors (such as rivaroxaban and apixaban) is denoted by an asterisk (*). TF indicates tissue factor.
Foremost among the advantages of warfarin is its remarkable efficacy. A recent meta-analysis of 6 clinical trials in patients with AF demonstrated a substantial reduction in stroke incidence with adequately dosed warfarin, with a 62% risk reduction relative to no treatment. 14 These trials have firmly established warfarin as the standard of care to prevent SSE in patients with AF at elevated risk. In fact, since no direct comparative trial in patients with AF has ever shown aspirin to be as effective as warfarin, one could question why all consensus guidelines do not indicate a clear choice for warfarin in patients with a CHADS2 score of 1, as the most recent European stroke prevention guidelines have promoted. 3
Despite its efficacy, warfarin has multiple drawbacks, making clinical administration difficult and increasing the chances of undesirable side effects. Indeed, it has been estimated that only up to two thirds of eligible patients are actually prescribed warfarin. 15,16 Warfarin has a narrow therapeutic window, with supratherapeutic levels carrying a risk of hemorrhage, and subtherapeutic levels predisposing toward SSE. 12 One major difficulty with the drug is unpredictability of dosing. While warfarin’s clinical effect can be assessed via the INR, there is no reliable dose–response curve for the drug, there is significant genetic variability in its handling, and there is substantial inter- and intra-patient variability in dose needed to maintain a therapeutic INR. 12 Environmental modulation from diet, gut flora, and concurrent drug administration are also major problems. 12
Solutions tailored to each individual, including genetic testing to determine optimal initial dose, 17 appear promising but are costly and insufficient to directly guide subsequent dosing. 12 The consequent obligate follow-up for INRs is often impractical, cumbersome, or financially burdensome to the patient and to the health care system. Furthermore, despite frequent INRs to ensure acceptable anticoagulation, the time spent in therapeutic range (TTR) for warfarin-treated patients is often unacceptably low (often below 60%-70%, even in the clinical trial setting, 18,19 where adherence and monitoring may exceed what is observed in clinical practice). The Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE) W trial showed that when TTR is low, warfarin is no better than aspirin plus clopidogrel. 19 The above concerns are familiar to most clinicians who have prescribed warfarin. There is thus a need for new oral anticoagulants with similarly high efficacy but more favorable dosing scheme and side effect profile.
New Oral Anticoagulants
The first new oral anticoagulant, ximelagatran (briefly marketed in Europe 20 ), showed noninferiority to warfarin in preventing SSE in patients with nonvalvular AF (NVAF). 18 Despite considerable excitement in the field due to perceived advantages over warfarin, the detection of serious hepatotoxicity led to its withdrawal from the European market 20 and rejection by the Food and Drug Administration (FDA) for use in the United States. 21
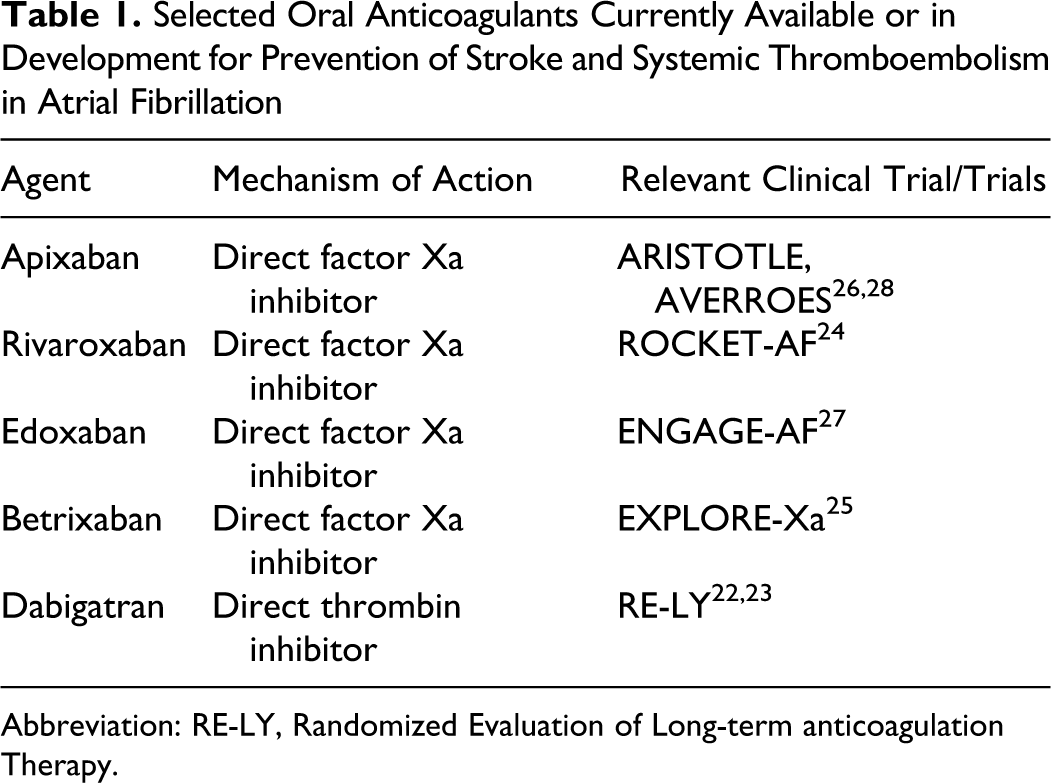
Other new anticoagulants are listed in the Table 1. 22,24 –27 Dabigatran has both published results from a phase 3 trial comparing it with warfarin 22,23 and gained FDA approval (in late 2010) for reducing the risk of SSE in patients with NVAF. 29 Rivaroxaban has also recently gained FDA approval for the same indication, while apixaban will soon be submitted for FDA approval.
Selected Oral Anticoagulants Currently Available or in Development for Prevention of Stroke and Systemic Thromboembolism in Atrial Fibrillation
Abbreviation: RE-LY, Randomized Evaluation of Long-term anticoagulation Therapy.
Dabigatran pharmacokinetics
Dabigatran (pronounced da-big’-∂-tran, trade name Pradaxa), dispensed as a prodrug dabigatran etexilate, is quickly absorbed (time to maximum concentration 1 hour fasting, 2 hours fed) and converted rapidly by ester hydrolysis in liver and plasma to the active drug dabigatran. 30 Dabigatran is a nonpeptide small molecule that works as a reversible inhibitor of factor IIa, with a half-life of 12 to 17 hours. 21,31 The drug is dispensed in capsules, with micropellets of the prodrug in a tartaric acid core. There is no metabolism in the liver via the cytochrome P450 system. Approximately 80% of its elimination is renal, with minimal excretion in feces. Absolute bioavailability ranges from 3% to 7%. 30 These features of the drug’s pharmacokinetics were found to be dose proportional in testing on healthy control individuals over a wide range of doses from 10 to 400 mg. 30
Dabigatran safety and efficacy data
The Randomized Evaluation of Long-term anticoagulation therapY (RE-LY) trial was the multinational pivotal phase 3 trial that compared dabigatran’s efficacy and safety profile with warfarin for the prevention of SSE in 18 113 patients with electrocardiography-documented NVAF. 22 This randomized, controlled, noninferiority trial was open label for dose-adjusted warfarin, but the dose of dabigatran, either 110 mg twice daily ([bid]; (D110) or 150 mg bid (D150), was blinded. The trial’s primary outcome was documented stroke (ischemic, hemorrhagic, or unspecified) or systemic embolism. 22 Major inclusion and exclusion criteria are summarized in Table 2, along with safety outcomes and secondary outcomes. Outcome events were adjudicated in a blinded fashion, and data were analyzed by intention to treat. 22
RE-LY Trial 22 Inclusion Criteria, Exclusion Criteria, and End Points

Abbreviations: ECG, electrocardiogram; NVAF, nonvalvular atrial fibrillation; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; Hb, hemoglobin; MI, myocardial infarction; TIA, transient ischemic attack; RE-LY, Randomized Evaluation of Long-term anticoagulation Therapy.
Study participants were randomized 1:1:1 to receive D110, D150, or warfarin dose adjusted to a target INR of 2.0 to 3.0. The 150-mg bid dose was derived from data collected in the phase 2 Prevention of Embolic and Thrombotic Events in Patients with Persistent AF (PETRO) trial. 32 All 3 study arms had similar patient baseline characteristics (Table 3). The overall risk profile of the study population for thromboembolic events was at least intermediate, with an average CHADS2 score of 2.1 and 32% with CHADS2 score of 3 or higher. Patients in the warfarin arm were reported to be in the therapeutic window 64% of the time, 22 which is comparable with other well-performed warfarin contemporary studies. 18,19
Patient Characteristics in the RE-LY Trial 22

Abbreviations: RE-LY, Randomized Evaluation of Long-term anticoagulation Therapy; AF, atrial fibrillation.
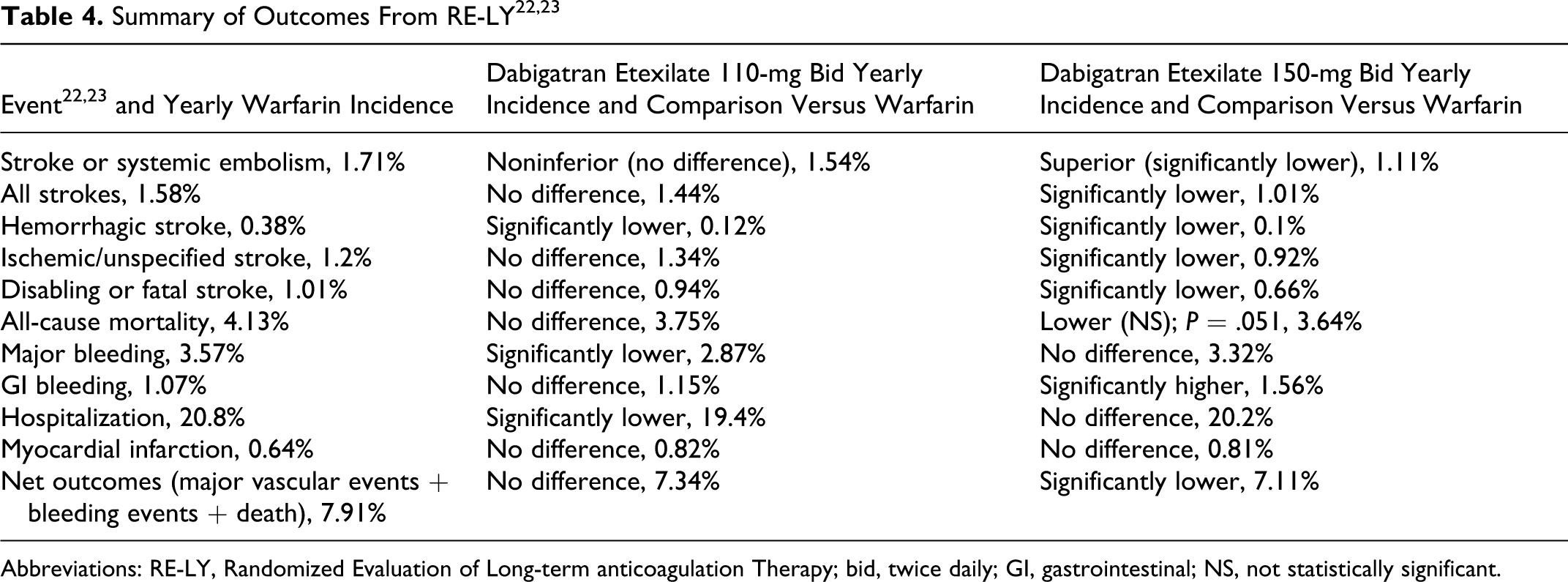
As demonstrated in Figure 2 from the RE-LY study, SSE occurred in 183 patients (1.5% per year) receiving D110, 134 patients (1.1% per year) receiving D150, and 202 patients (1.7% per year) receiving warfarin with a median follow-up of 2 years. 22,23 Both dabigatran doses were noninferior to warfarin for the primary outcome. The D150 was also superior to warfarin in reducing the risk of SSE (relative risk [RR] 0.65, 95% confidence interval [0.52-0.81]; P < .001) and also to D110. 22,23
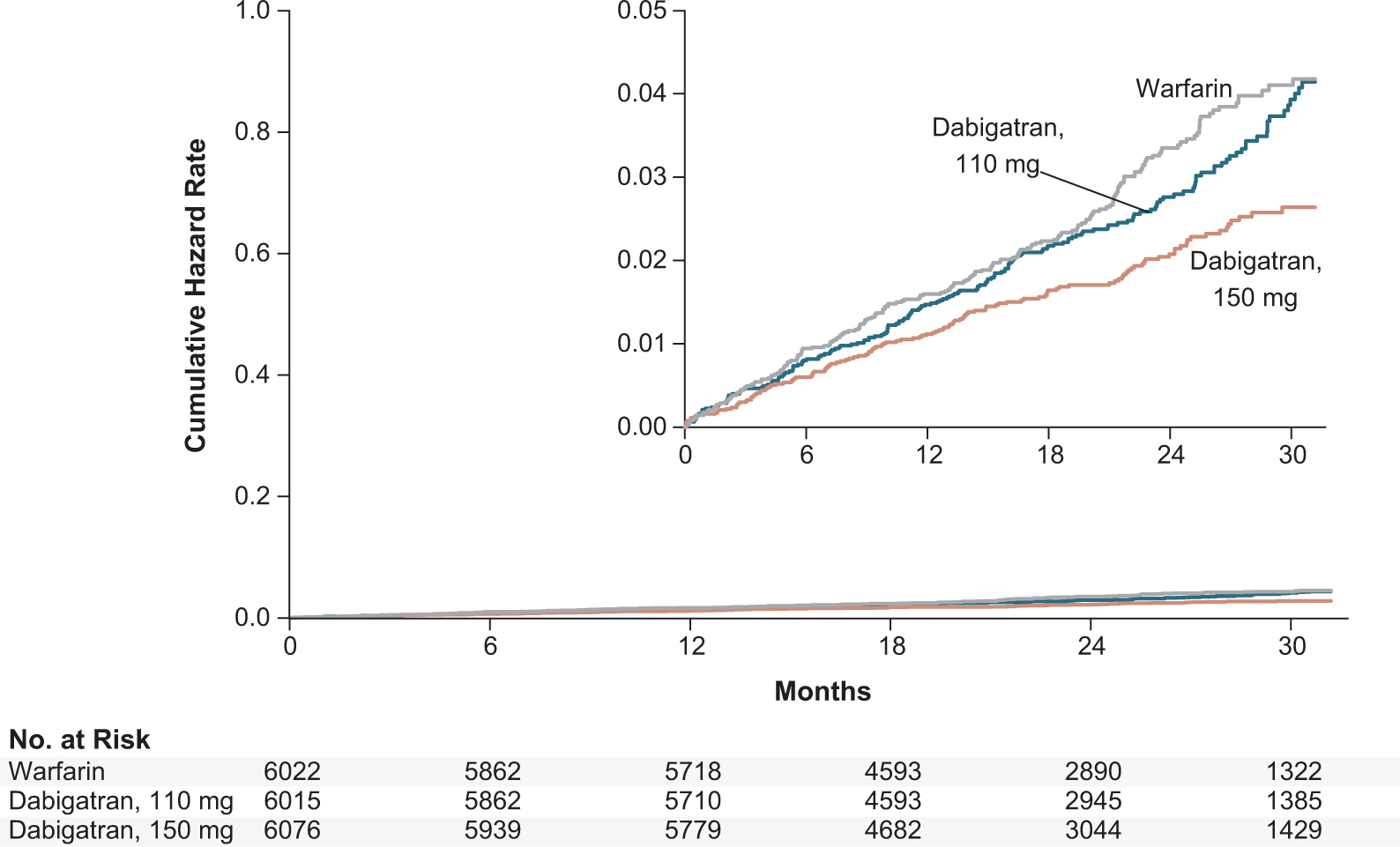
Dabigatran versus warfarin in patients with AF: the RE-LY trial. After median follow-up of 24 months, dabigatran administered at 150 mg bid was shown to be superior to warfarin (adjusted to INR 2.0-3.0) in preventing stroke and systemic embolism in nonvalvular atrial fibrillation, while the 110-mg bid dosing was shown to be noninferior. Taken from the RE-LY study. 22 Bid indicates twice daily; AF, atrial fibrillation; RE-LY, Randomized Evaluation of Long-term anticoagulation therapY; INR, international normalized ratio.
Table 4 provides a summary of the other major trial findings. 22,23 For the rate of ischemic strokes, D110 was noninferior to warfarin (P = .35) and D150 was superior to warfarin (P = .03). Both doses of dabigatran were superior to warfarin in preventing hemorrhagic strokes. Among the secondary outcome measures, there was no significant difference in the incidence of all-cause mortality among the 3 groups (although for mortality, D150 was lowest and almost achieved significance; P = .051).
Abbreviations: RE-LY, Randomized Evaluation of Long-term anticoagulation Therapy; bid, twice daily; GI, gastrointestinal; NS, not statistically significant.
Annual rates of major bleeding were similar between D150 and warfarin (P = .32) but less with D110 compared with warfarin (P = .003). Furthermore, there was a statistically significant decrease in both life-threatening bleeding (1.22%, 1.45%, and 1.80%, respectively, for D110, D150, and warfarin; P < .001 for D110; P = .04 for D150) and intracerebral bleeding (0.23%, 0.30%, and 0.74%, respectively; P < .001 for both dabigatran doses). Gastrointestinal (GI) bleeding rates were similar with D110 and warfarin but slightly higher with D150.
In RE-LY, there were higher 2-year discontinuation rates of both doses of dabigatran (21%) compared with warfarin (17%), 22 most commonly for patient’s decision, study outcome event, and adverse events. Importantly, there were no differences in the rate of liver function test abnormalities. 22 The only adverse effect significantly more common in the dabigatran arms was dyspepsia, being higher with both D110 and D150 compared with warfarin (11.8% and 11.3%, compared with 5.8%; P < .001). The net clinical benefit for the 3 trial arms, defined as the sum of all major vascular events, major bleeding, and death was 7.09%, 6.91%, and 7.64% for D110, D150, and warfarin, respectively. 22 The comparison was statistically significant for D150, with an RR of 0.91 (P = .04; 95% confidence interval 0.82-1.00).
Monitoring of Anticoagulation Tests
Consistent with its mechanism of action (Figure 1), dabigatran predictably alters commonly used serum tests of coagulation. 33 As a direct inhibitor of thrombin, it affects the PT and INR. However, as reaffirmed by recent labeling changes, the INR cannot be used as a reliable measure of anticoagulation effect as the effects are variable and have not been shown to correlate with dosing of medication under controlled settings. 12 Activated partial thromboplastin time (aPTT) has a direct but nonlinear relationship with plasma dabigatran concentrations, making it an attractive test for assessing compliance and drug elimination. 32 At steady-state trough plasma levels (ie, immediately preceding a scheduled dose), aPTT remains below 65 (2 times upper limit of normal) in greater than 90% of individuals taking a typical 150-mg bid dose, whereas peak serum levels result in higher values.31,32,34 Thrombin time assesses the final coagulation cascade step and is affected by dabigatran concentrations in a linear fashion. 31 Thrombin time (TT) ratios of 10 to 20 times baseline are usually observed in volunteers taking typical doses of dabigatran at therapeutic plasma levels, 30 though there can be substantial variability in laboratory testing methodology for TT. Since correlation between aPTT and TT and clinical bleeding or thromboembolic events in patients receiving dabigatran has not been determined, values of coagulant testing should be interpreted with caution and have no role in assessing the clinical efficacy and safety of an individual patient’s dosing regimen. They may, however, guide assessment of a patient’s compliance or the progress of drug washout. 30
Practical Use of Dabigatran Etexilate
Based upon superior efficacy to both warfarin and 110-mg bid dosing, lower intracranial bleeding and no increased overall bleeding versus warfarin, dabigatran 150-mg bid received FDA approval in the United States. In addition, based on the pharmacokinetic modeling, 75-mg bid was also approved, but only for patients with creatinine clearance (CrCl) between 15 and 30 mL/min (see special populations below). 30 The 110-mg bid dose was approved in Canada. 21 Splitting or chewing the capsule should be avoided as this can increase exposure to the drug and its bioavailability by 75%. 30 Capsules are dispensed in a bottle or in blister packs and should be kept away from moisture and extreme heat or cold. Bottled medication must be used within 60 days of opening. 30 Capsules may be taken with meals (although coadministration with a high-fat meal may delay attainment of peak plasma concentrations). 30 In the event of a missed dose, patients should take their medication as soon as possible; if more than 6 hours have elapsed since the missed dose, they should skip it and resume at the next scheduled time, without doubling the dose to compensate for the missed dose. 30
Initiation of dabigatran and conversion to/from warfarin is easily accomplished with the judicious use of INR testing and recognition of the basic pharmacokinetics of the drug. Table 5 summarizes the major recommendations for conversion between the agents. For patients undergoing planned invasive procedures or surgery while taking dabigatran treatment should, if possible, be discontinued at least 1 to 2 days ahead (for patients with CrCl of at least 50 mL/min). 30 Recommendations for patients with CrCl less than 50 mL/min are summarized in Table 5. Longer discontinuation may be considered for procedures with significant expected bleeding, instrumentation of the epidural space, or at other sites where hemostasis may be difficult to achieve. 30 Individual clinical estimation of relative bleeding risk and thromboembolic risk may modulate these guidelines. For emergent surgery in patients taking dabigatran, bleeding risk is increased. 30 An estimate of bleeding propensity may be obtained by measuring aPTT, TT, or ecarin coagulation time (see Monitoring of Coagulation Tests section); markedly elevated levels would suggest persistent drug effect. 30
Recommendations for Transition Between Commonly Used Oral Antithrombotic Agents and Temporary Cessation of Dabigatran
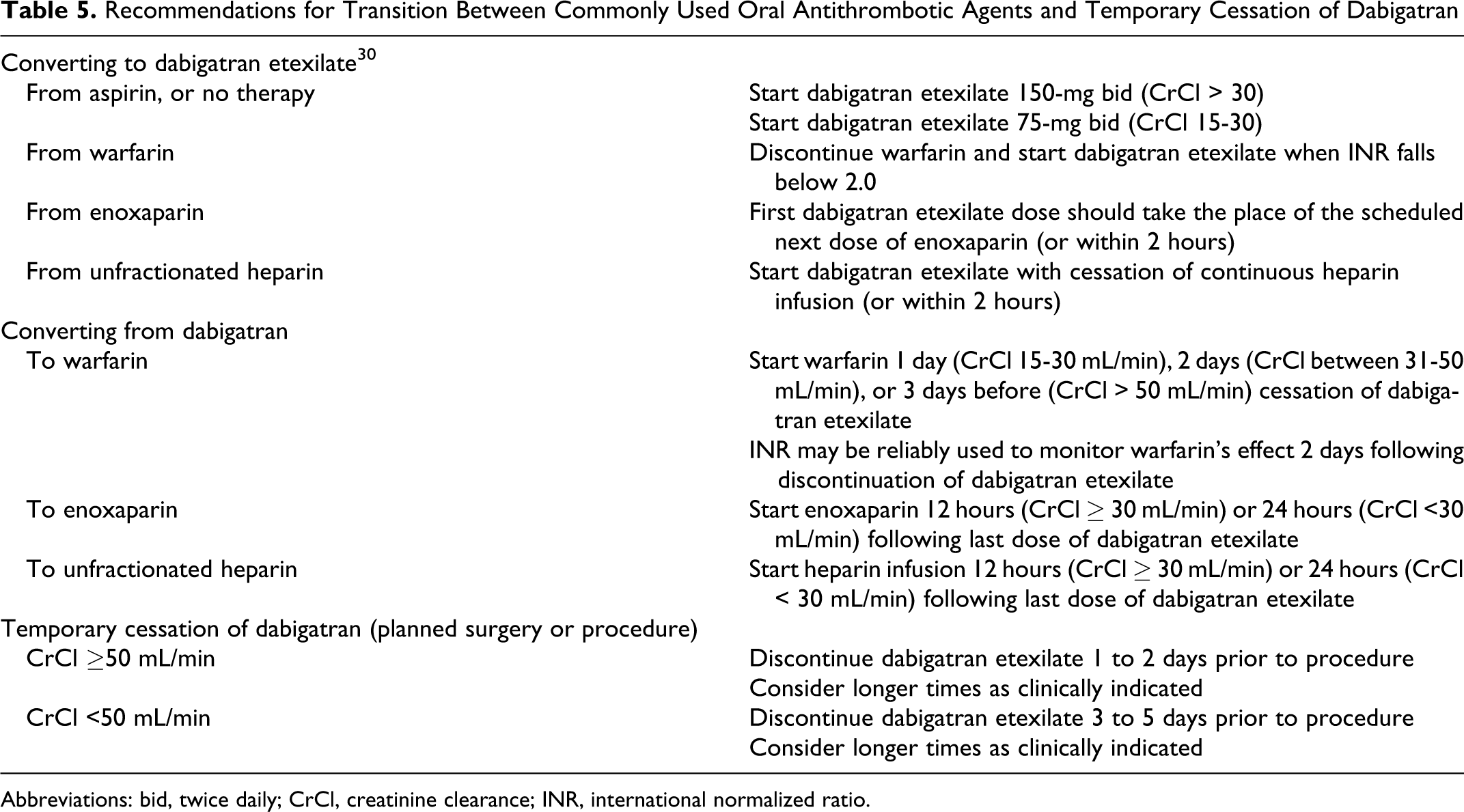
Abbreviations: bid, twice daily; CrCl, creatinine clearance; INR, international normalized ratio.
Overdose of dabigatran may precipitate hemorrhage. There is no specific antidote, and care is mostly supportive, with focus on control of bleeding sites (if any), transfusion of red blood cells, or transfusion of fresh frozen plasma, if appropriate. 30 Since the drug is renally excreted, promoting diuresis facilitates elimination. Several other approaches in the case of severe bleeding may be helpful, although supported by limited clinical evidence. Hemodialysis may be considered as approximately 60% of the drug can be dialyzed over 2 to 3 hours. Concentrates of protein factors may also be helpful. 30 Concurrent thrombocytopenia (either absolute or functional due to coadministration of antiplatelet agents) should be identified promptly and addressed through platelet transfusion, if necessary. Monitoring of serum coagulation tests may be helpful to guide therapy. 30
Special Populations
Several populations deserve special mention regarding the suitability of dabigatran use. Although these groups were not prospectively studied in the RE-LY trial, the use might be judiciously considered in a number of clinical situations that have been retrospectively analyzed.
Elective cardioversions
Current practice for patients with new onset or persistent AF requiring cardioversion dictates either pretreatment with warfarin for at least 3 weeks at target-range INR followed by cardioversion or early cardioversion guided by transesophageal echocardiogram to exclude left atrial thrombus. Anticoagulation is then continued for at least 4 weeks following the cardioversion. 35 Dabigatran should be a particularly attractive option for this setting, given its fixed dosing, rapidity of effect, and ease of initiation. The safety and efficacy of using dabigatran prior to cardioversion was examined in a RE-LY substudy in which close to 2000 cardioversions in 1270 patients were analyzed across the 3 treatment arms (D110, D150, and warfarin). 36 In these groups, the rates of SSE at 30 days were similar (0.8%, 0.3%, and 0.6%, respectively), as were the rates of major bleeding events (1.7%, 0.6%, and 0.6%, respectively). Conclusions drawn from this study are subject to the constraints of a retrospective study. However, while no randomized data are currently available regarding this important issue, the available data showing similar event rates in each treatment arm suggest that dabigatran is a safe and suitable alternative to warfarin prior to cardioversion. 36
Safety considerations
While rates of major bleeding in RE-LY were similar between warfarin and D150, rates of GI bleeding were higher in the D150 group. 22 Thus, it is reasonable to conclude that dabigatran should be prescribed with caution in patients perceived to be at increased risk of GI bleeding disorders. Consultation and discussion with a gastroenterologist may be warranted to appropriately risk stratify such patients through selected evaluation. More data are required regarding bleeding risk in patients who develop dyspepsia or other upper GI symptoms on the drug. There was also a slightly higher bleeding risk with dabigatran in RE-LY patients ≥ age 75 years.
A small increase in the rate of myocardial infarction was reported with both doses of dabigatran in the initial RE-LY report, raising concern whether the drug is appropriate for patients with known coronary disease. Two hypotheses were advanced; one that this finding represents a protective effect of warfarin in preventing acute coronary syndrome or conversely that dabigatran increases the risk of myocardial infarction, 37 the former appearing more likely. However, the increase in myocardial infarction was not confirmed when a more complete set of RE-LY data were subsequently reported in September 2010. 23
For patients with chronic kidney disease, the lower dabigatran dose of 75 mg was FDA approved for CrCl between 15 and 30 mL/min despite never being tested in a randomized fashion in such patients. Rather, it was selected based on pharmacokinetic modeling. 38 Dabigatran should thus be used cautiously in patients with fluctuating renal function or advanced kidney disease. Changes to the drug prescribing information now recommends testing renal function prior to starting therapy and annually thereafter in patients 75 years of age or older or patients with CrCl <50 mL/min.
Analysis of the results from D110 arm of RE-LY suggests similar efficacy compared with warfarin, with lower rates of bleeding compared with the D150 arm, although not statistically significant. 22 Given the concerns regarding bleeding in patients deemed at higher risk of this complication (such as those with modestly reduced CrCl taking 1 or more P-glycoprotein [P-gp] inhibitors—see below), it is reasonable to conclude that lower dosing schemes may mitigate this bleeding risk. However, the FDA approved only the 150-mg bid dose, noting that the 110-mg dose provided less protection from SSE. 38 It is worthwhile to note that while the factor Xa inhibitor rivaroxaban was shown to be noninferior to warfarin in preventing SSE (by intention to treat) and noninferior in bleeding rates, it did obtain FDA approval, unlike the dabigatran 110-mg bid dose.
Although the 110-mg capsules are not available for prescription in the United States, the 75-mg capsules are available. Administration of 3 times daily 75 mg dabigatran or 150/75 mg alternate dosing provides a comparable daily dose of the drug as the 110-mg bid dosing scheme (225 vs 220 mg). We have received anecdotal reports from practitioners utilizing such a dosing scheme in patients with significant GI side effects to the drug, concomitant P-gp inhibitor use (see Drug Interactions section), and/or modest reduction in CrCl. 38 At present, there is no clinical trial data to support this dosing in this manner, and, as such, it must be considered off-label administration, with uncertain efficacy.
Unfortunately, there is no reported data for dabigatran in many other commonly encountered and important clinical situations, including patients with mechanical prosthetic heart valves, coronary stent implantation requiring dual antiplatelet therapy, or during pregnancy.
Drug interactions
Emerging evidence has identified potential interactions between dabigatran and commonly used medications. Dabigatran is a substrate of the efflux transporter P-gp (which affects absorption of the prodrug); consequently, ingestion of an inhibitor of the efflux transporter can raise exposure and ingestion of P-gp inducers, such as rifampin, may reduce exposure. 30 Rifampin and dabigatran should not be used in combination. 30 In RE-LY, amiodarone and verapamil, both inhibitors of P-gp, were widely used; furthermore, no specific increase in bleeding was reported in patients taking these P-gp inhibitors and concurrent dabigatran compared with concurrent warfarin. 22,39 While amiodarone increased the area under the curve and maximal concentration of dabigatran by 58% and 50%, respectively, this effect was mitigated by some increased renal excretion of drug at steady state when taken with amiodarone. 30,39 Similar pharmacokinetic changes in serum concentrations have been noted with quinidine coadministration. 30 More recently, in the data made available to the FDA by Boehringer-Ingelheim and Sanofi-Aventis, dronedarone was shown to also increase the serum concentration of dabigatran (about 1.7- to 2.0-fold). 40 These findings led to a recent labeling change for dabigatran, suggesting a dose of 75 mg bid when dronedarone and dabigatran are prescribed together in patients with moderately reduced renal function. In addition, concomitant use of P-gp inhibitors in patients with severe renal dysfunction (CrCl 15-30 mL/min) is contraindicated. The consequences of these interactions have not been adequately studied, and their clinical significance is unknown. Subsequently, the FDA has requested that a more substantial in vitro study of the effect of amiodarone and of dronedarone on the active transport of dabigatran be performed during the next year.
Cost-Effectiveness
One of the advantages of warfarin is its low cost, approximately $1 for 1 day’s generic dose. Dabigatran is costlier, with median cost (from early 2011) of approximately $9 per day at the 150-mg bid dosing. 12 However, cost-effectiveness of the drug must be judged not only by the price of the medication but also by the added cost of serum coagulation testing, as well as any saved costs and productivity from loss of work, avoided strokes, avoided bleeds, and hospitalizations saved. Cost-effectiveness analyses for dabigatran ideally should show acceptable marginal cost if it is to be widely used as a replacement for warfarin, particularly given the increasing prevalence of the condition as our population ages. A recent study compared dabigatran with warfarin, studying quality-adjusted survival, costs, and cost-effectiveness in patients similar to those enrolled in RE-LY. 41 The model used was comprehensive and included costs of INR testing and drug pricing (which was estimated at $13 daily for 150-mg bid dosing in the study), as well as the costs and quality-of-life sequelae of adverse events, such as stroke and MI. Incremental cost-effectiveness ratios were calculated for the base model as $51 229 per quality-adjusted life year for the 110-mg dose and $45 372 for the 150-mg dose compared with the ,standard warfarin therapy. 41 Both doses, by conventionally accepted cutoffs, approach acceptable cost-effectiveness for new therapies. As might be expected, the model was very sensitive to the daily pricing of dabigatran; if pricing drops to $8 per day, the incremental cost-effectiveness ratio drops to a mere $10 000 for the 150-mg dose. 41 These findings support the contention that dabigatran, despite higher initial pricing, may be an acceptable cost-effective therapy in the prevention of stroke in AF. Similar conclusions were reached by more recent analyses using updated costs.1,2,42
The Role of Dabigatran in the Treatment of AF
A number of characteristics make dabigatran an attractive option for reducing the risk of SSE in patients with NVAF. Given its rapid onset of action, patients can easily and quickly be initiated on the drug. Dabigatran is administered at a fixed dose, with minimal variation in anticoagulant effect with dietary intake or when coadministered with other common drugs. The relatively short half-life makes washout easy when surgical procedures are planned. Lack of routine serum coagulation test monitoring makes it a much more convenient therapy than warfarin and, potentially, a more cost-effective one. Lack of dietary restrictions and fewer concerns regarding interactions with prescription drugs, over-the-counter medications, and herbal agents are appealing for the patient and physician. Perhaps most strikingly, at the 150-mg bid dose, dabigatran is more efficacious than warfarin, with similar rates of bleeding.
However, additional attributes to the drug must also be considered. Dabigatran’s short half-life may pose a problem in the setting of poor compliance, as the consequences of a missed dose may be more clinically significant. Lack of an effective antidote to the drug similarly makes consequences of major or life-threatening bleeding of greater concern. While the fixed dosing scheme obviates the need for routine coagulation testing, the lack of a reliable serum test (either to assess for treatment failure or noncompliance or to titrate the intensity of therapy) is a potential disadvantage.
The recently published American college of Cardiology Foundation/American Heart Association/Heart Rhythm Society guidelines on AF, focused update on dabigatran, provide general considerations on the appropriate use of the new drug, noting the demonstrated efficacy of the drug and designating it as a class I recommendation (level of evidence B), with the caveat of the above limitations. 5
Figure 3 illustrates a flow sheet that may assist clinicians in selecting the ideal antithrombotic therapy for a given patient with AF. Due to the increased efficacy of the drug over warfarin, practitioners should consider prescribing dabigatran as first-line therapy for SSE prevention in NVAF, provided cost to the patient, GI bleeding risk, and advanced renal dysfunction do not prove prohibitive.
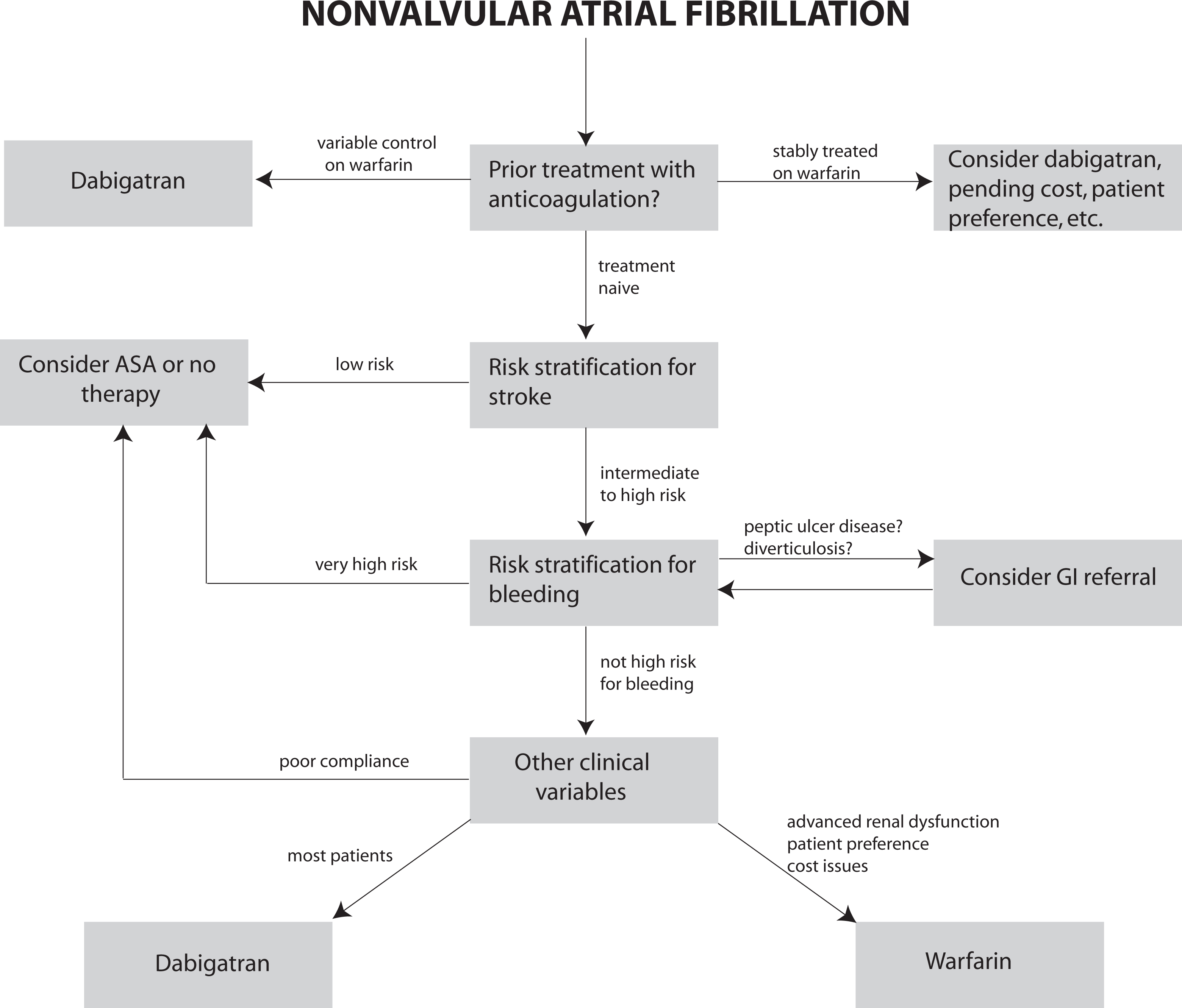
The role of dabigatran in the management of atrial fibrillation. Dabigatran etexilate can be considered a first-line agent for the prevention of stroke and systemic thromboembolism in AF, provided the prescriber accounts for several clinical considerations. AF indicates atrial fibrillation; ASA, aspirin; GI, gastrointestinal; INR, international normalized ratio.
An area of controversy is whether patients effectively and stably treated with warfarin are good candidates to be transitioned to dabigatran. On one hand, the 150-mg bid dose of dabigatran has demonstrated superiority in preventing strokes, with similar risk of bleeding. 22,23 However, the number of patients needed to treat with dabigatran to prevent one additional thromboembolic event compared with warfarin can be computed as 167 over 1 year. From this perspective, patients stable on warfarin with high TTR may have little to gain from transitioning to dabigatran. 15 However, cost, bleeding risk (especially intracranial hemorrhage), concomitant therapy, patient lifestyle, and patient preference may all factor into this decision.
Those at intermediate risk of SSE present another interesting category of patients for whom dabigatran therapy may be considered. Many clinicians may have been loath to initiate full anticoagulation with warfarin in this patient population due to patient preference and obligate lifestyle change, instead opting for aspirin, which confers less protection against SSE. 4 Dabigatran (perhaps even at a 110-mg bid dose, if and when available) may prove to be a sensible option in these patients.
Finally, if/when additional new anticoagulants for NVAF become available (Table 1), choosing among them should include consideration of relative efficacy versus warfarin, concomitant disorders that could affect dosing, potential interactions, and other factors. None of the candidate oral anticoagulants appear to have dosing intervals (thrice daily or more) that can seriously affect compliance. Rivaroxaban is now FDA approved for patients with NVAF, and at present apixaban will be submitted to the FDA shortly. Table 6 assumes that each of these agents will reach the US marketplace for AF and provides some comparative considerations versus dabigatran. Figure 4 attempts to put the selection issues into an algorithmic format.
Factors Affecting the Selection Among the New and Emerging Oral Anticoagulants
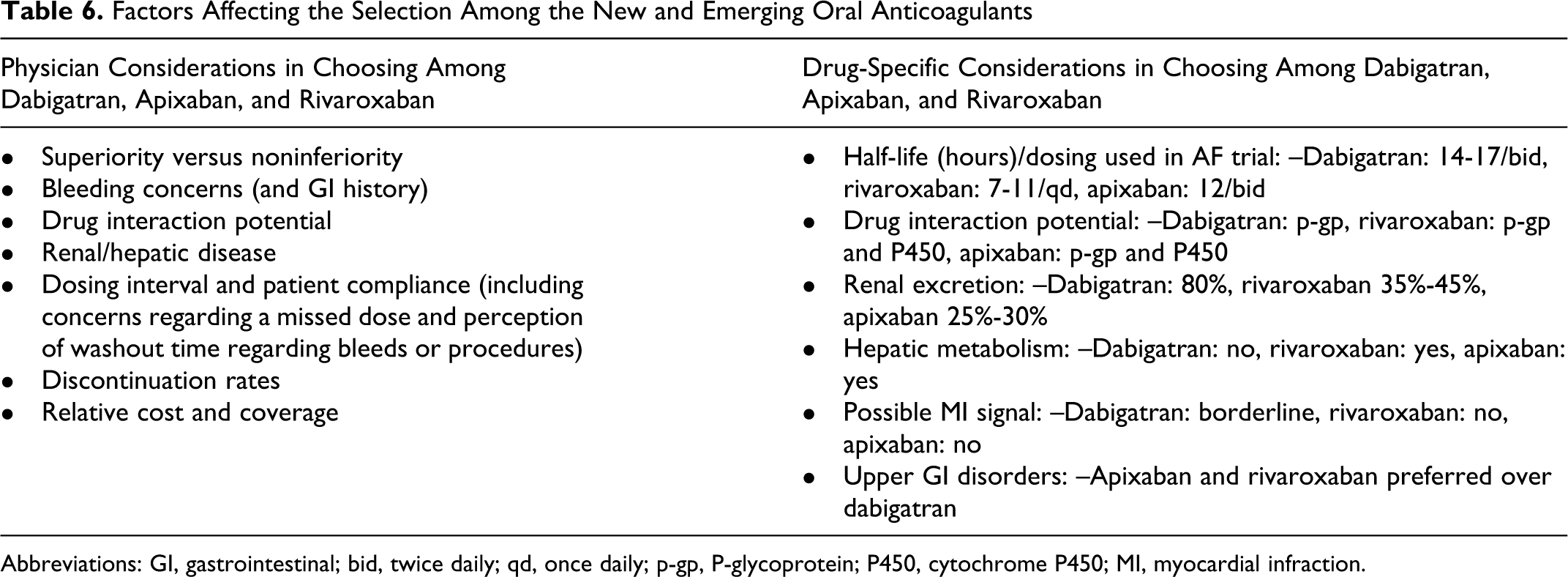
Abbreviations: GI, gastrointestinal; bid, twice daily; qd, once daily; p-gp, P-glycoprotein; P450, cytochrome P450; MI, myocardial infraction.
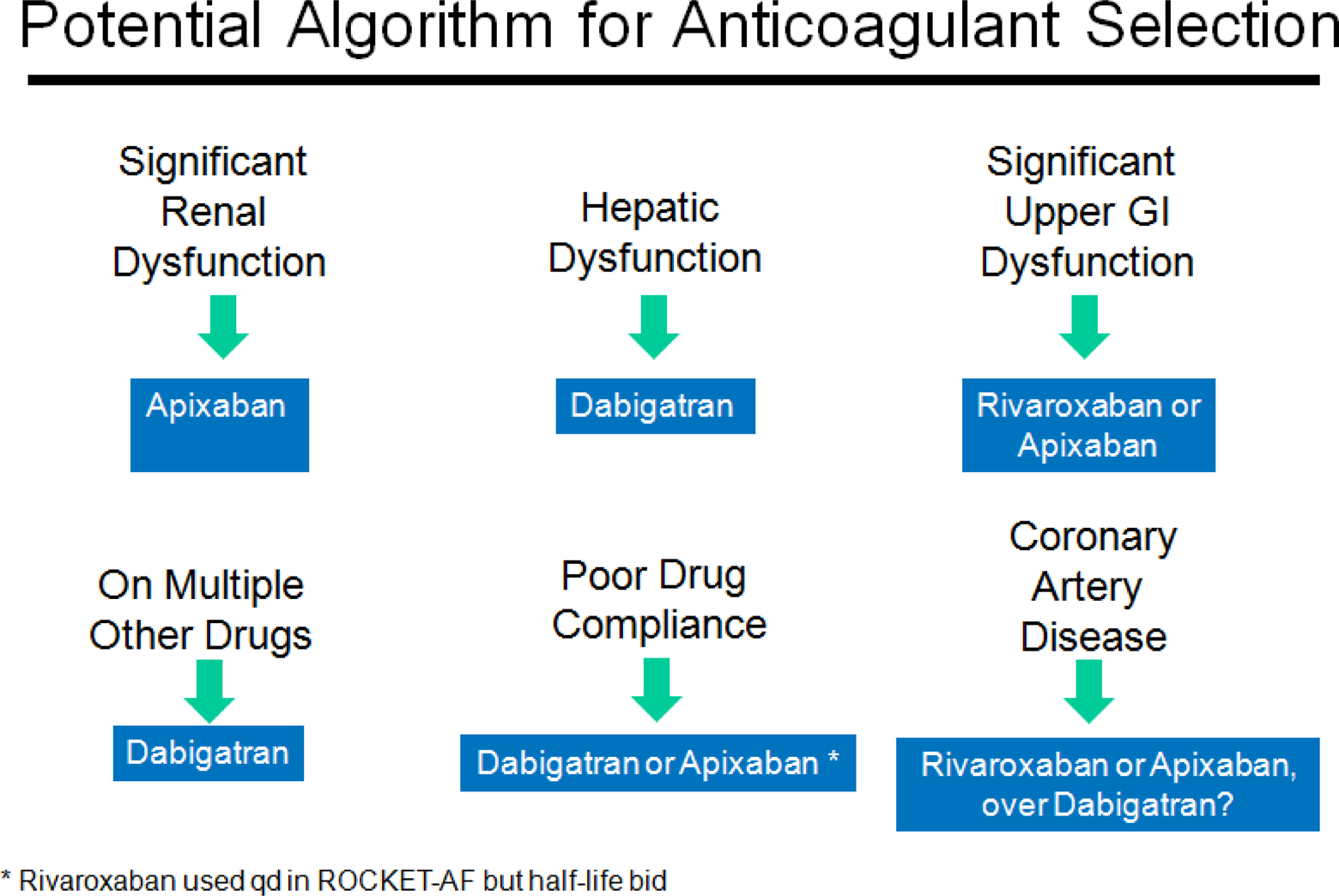
Potential algorithm for selection among new oral anticoagulants (ROCKET-AF). 24 The selection process among dabigatran, apixaban, and rivaroxaban, in an algorithmic format, based upon concomitant conditions. qd indicates once daily; bid, twice daily
In summary, the clinical trial outcomes and FDA approval of dabigatran for use in risk reduction of SSE is the most exciting development in the field in the last 50 years. With several important considerations, dabigatran should change how clinicians approach the management of AF, ushering in the dawn of the new oral anticoagulant era.
Footnotes
Acknowledgments
The authors would like to thank the staff at Medicus International for editorial review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.A.R. has served as a RE-LY investigator and consultant for Boehringer-Ingleheim, and sits on their speakers’ bureau.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.