Abstract
Ticagrelor is a P2Y12 receptor antagonist with proven clinical benefit in patients with acute coronary syndrome. Apart from its principal antiplatelet action, pleiotropic effects have been implicated in the clinical profile of ticagrelor, including a potentially beneficial impact on endothelial function. In light of the common presence and prognostic value of endothelial dysfunction in patients with coronary artery disease, several clinical studies have investigated the postulated effect of ticagrelor on endothelial function, yielding conflicting results. Limitations of the relevant studies as well as substantial differences in patient population, study design, and methods may account for these controversial findings. Most of these studies, however, support a beneficial impact of ticagrelor on endothelial function, which seems to be significant in the higher risk patients. In order to elucidate this effect, further research efforts should aim to clarify how quickly does endothelial function respond to ticagrelor, how sustained this response is during the dosing intervals and in the long term, which mechanisms are implicated, and whether this pleiotropic action is clinically significant. Future studies should include larger and diverse populations of patients, assess endothelial function at several time points after treatment initiation, and use multiple methods of endothelial function measurement, while implementing strict methodology. Nevertheless, the extent of the clinical benefit of ticagrelor attributable to actions beyond its potent and consistent antiplatelet effect remains uncertain.
Introduction
Ticagrelor (AZD6140) is a cyclopentyl-triazolopyrimidine, which, in contrast to thienopyridines clopidogrel and prasugrel, directly and reversibly inhibits the platelet adenosine diphosphate (ADP) P2Y12 receptor, providing rapid and potent antiplatelet effect after oral administration. Ticagrelor has proven clinical benefit in patients with acute coronary syndrome (ACS) and constitutes an established antiplatelet agent in this population. 1 Apart from its principal antiplatelet action, pleiotropic effects have been implicated in the clinical profile of ticagrelor, including a potentially beneficial—though controversial—impact on endothelial function, which is discussed in the present article.
Pleiotropic Effects of Ticagrelor
The remarkable clinical benefit of ticagrelor compared with clopidogrel in patients with ACS (including significant reduction of vascular and total mortality) observed in the PLATelet Inhibition and Patient Outcomes (PLATO) study, 1 along with its distinctive adverse effects (increased incidence of dyspnea and ventricular pauses), triggered speculations that off-target effects may contribute to the clinical profile of ticagrelor.
Substantial evidence has emerged during the recent years that—apart from their primary antiplatelet effect—P2Y12 receptor inhibitors exhibit several pleiotropic actions. 2 Regarding ticagrelor in particular, recent research efforts have focused on the hypothesis of an adenosine-mediated mode of action. 3 Adenosine is a naturally occurring endogenous purine nucleoside, released by endothelial cells and myocytes in response to ischemia, oxidative stress, and cell injury (as happens during an ACS). Adenosine interacts with the adenosine receptor subtypes A1, A2A, A2B, and A3, exerting a range of beneficial cardiovascular effects. 4 Ticagrelor has been demonstrated to inhibit adenosine uptake by human erythrocytes 5,6 through inhibition of the sodium-independent equilibrative nucleoside transporter 1. 6 Moreover, ticagrelor induces adenosine triphosphate (ATP) release from human erythrocytes, which is subsequently degraded to adenosine. 7 Importantly and in line with the above preclinical evidence, significantly higher adenosine plasma concentration has been confirmed in patients with ACS treated with ticagrelor compared with clopidogrel. 8 Furthermore, in animal models of ischemia/reperfusion, P2Y12 blockade conferred cardioprotective effect (such as reduction in infarct size), 9,10 and adenosine-mediated mechanisms were implicated in this pleiotropic action for ticagrelor. 10 This effect might account for much of the benefit observed with ticagrelor in patients with ACS treated with percutaneous coronary intervention (PCI). Additionally, in healthy volunteers and in patients with ACS, ticagrelor has been shown to enhance the coronary blood flow velocity increases in response to incremental doses of adenosine infusion. 11,12
It has been proposed that the adenosine-mediated effects of ticagrelor might ameliorate endothelial dysfunction (ED), which is common and of prognostic significance in patients with coronary artery disease (CAD). 13 Endothelial dysfunction refers to the impairment of endothelium-dependent vasodilation featured by an imbalance between vasodilators (mainly nitric oxide [NO]) and endothelium-derived vasoconstrictors. Moreover, ED is characterized by a state of increased pro-inflammatory, proliferative, and prothrombotic activity, often referred to as “endothelial activation.” 13 It is not confined to the coronary vessels but is systemic in nature and constitutes a well-recognized contributor to the initiation of atherosclerosis, progression of atherosclerotic lesions, and occurrence of atherothrombotic complications. 13,14 Several techniques are available for evaluation of endothelial function, including coronary epicardial vasoreactivity, coronary microvascular function assessment with Doppler wires, flow-mediated dilation (FMD), venous occlusion plethysmography, and peripheral arterial tonometry (PAT). These methods examine diverse vascular beds and are characterized by different advantages and limitations 15 (Table 1). More specifically, FMD refers to the dilation of the brachial artery as measured by ultrasound following a transient cessation of its blood flow by a pressure cuff, while venous occlusion plethysmography assesses the rate at which forearm volume increases in response to occlusion of its venous outflow. In PAT, reactive hyperemia is measured by a fingertip tonometer following a transient cessation of arterial blood flow by a pressure cuff on the upper arm. In animal models, ticagrelor has been shown to dose-dependently augment reactive hyperemia after left anterior descending artery transient occlusion 5 and mitigate significant vasoconstrictions in response to maximal intracoronary acetylcholine infusion. 16 However, in healthy volunteers, results have been contradictory. In a double-blind, placebo-controlled, crossover trial of 14 healthy individuals, a ticagrelor loading dose (180 mg) did not affect forearm blood flow (FBF; measured with venous occlusion plethysmography) during the infusion of incremental adenosine doses into the brachial artery. 17 However, in a recent study of healthy males undergoing forearm ischemia, ticagrelor (oral dose and maintenance dose), compared with clopidogrel, attenuated ED assessed by FBF in response to the endothelium-dependent vasodilator acetylcholine. 18
Advantages and Disadvantages of the Most Commonly Used Techniques to Assess Endothelial Function.
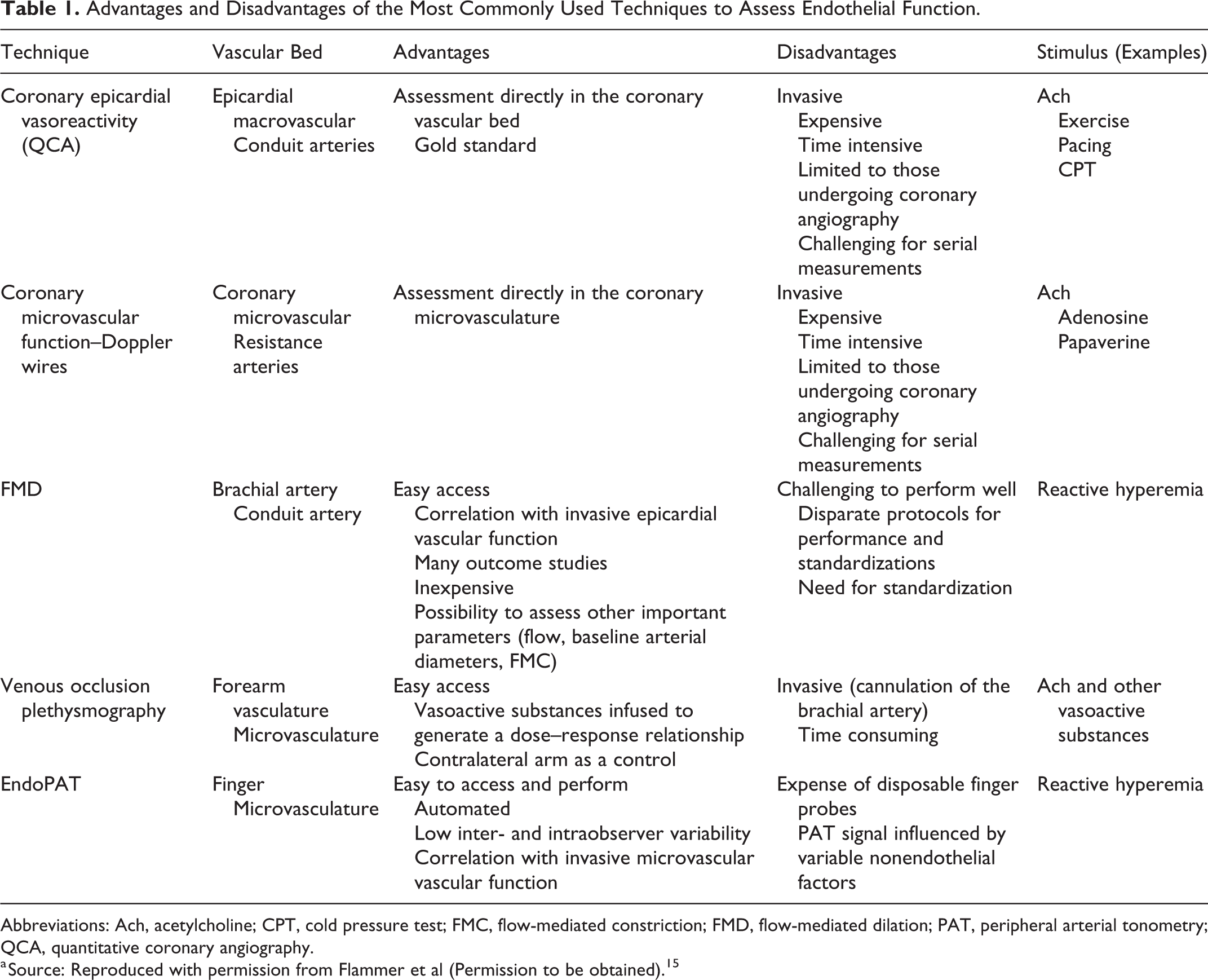
Abbreviations: Ach, acetylcholine; CPT, cold pressure test; FMC, flow-mediated constriction; FMD, flow-mediated dilation; PAT, peripheral arterial tonometry; QCA, quantitative coronary angiography.
a Source: Reproduced with permission from Flammer et al (Permission to be obtained). 15
Mechanisms potentially involved in this effect (Table 2) 3,19 might primarily relate to the aforementioned increase in plasma adenosine levels and release of ATP triggered by ticagrelor. Both mediators stimulate vasodilation, adenosine mainly via a direct effect on vascular smooth muscle cells, and ATP by promoting release from the endothelium of vasodilatory factors such as NO and endothelial hyperpolarizing factor. 20 Moreover, vascular smooth muscle cells express P2Y12 receptors and ticagrelor has been shown to inhibit the ADP-induced vasoconstriction. 21 An increase in number of circulating endothelial progenitor cells observed with ticagrelor may also be implicated. 22 Finally, ticagrelor might improve endothelial function by the reduction of circulating epidermal growth factor and the consequent promotion of the activation of endothelial NO synthase in the vascular endothelium. 23
Mechanisms Potentially Involved in the Interaction Between Ticagrelor and Endothelium.

Abbreviations: ADP, adenosine diphosphate; ATP, adenosine triphosphate; EGF, epidermal growth factor; eNOS, endothelial nitric oxide synthase; NO, nitric oxide; ROS, reactive oxygen species; VSMCs, vascular smooth muscle cells.
Clinical Studies Assessing the Effect of Ticagrelor on Endothelial Function in Patients With CAD
In light of the common presence and prognostic value of ED in patients with CAD along with the existing preclinical evidence, several clinical studies have investigated the postulated impact of ticagrelor on endothelial function. These studies differ in several aspects, including design, patient population studied, and method applied for endothelial function assessment. They are characterized by different strengths and limitations and have yielded conflicting results (Table 3).
Clinical Studies Evaluating the Effect of Ticagrelor on the Endothelial Function in Patients With CAD.

Abbreviations: ACS, acute coronary syndrome; CAD, coronary artery disease; CI, confidence interval; COPD, chronic obstructive pulmonary disease; DES, drug eluting stent; FMD, flow-mediated dilation; HUVECs, human umbilical vein endothelial cells; PAT, peripheral arterial tonometry (assessed by EndoPAT 2000); PCI, percutaneous coronary intervention; NSTE, non-ST-elevation; RHI, reactive hyperemia index; SD, standard deviation.
a The rate of apoptosis in human umbilical vein endothelial cells (HUVECs) was assessed after 48-hour incubation with 20% serum from patients.
Initially, in a study limited by its nonrandomized design, chronic ticagrelor treatment compared with prasugrel and clopidogrel was shown to improve peripheral endothelial function assessed by PAT (EndoPAT 2000 System; Itamar Medical, Caesarea, Israel) in a population of 127 stable patients with prior ACS (>3 months to <3years). The rate of ED (defined as reactive hyperemia index [RHI] <1.67) was also lower with ticagrelor. 24 A beneficial effect of standard ticagrelor maintenance dose compared to high-dose clopidogrel (150 mg) has also been described in a randomized, crossover study having enrolled 42 patients with type II diabetes mellitus and stable CAD treated with PCI at least 1 month before randomization. The 14-day treatment period of ticagrelor led to improvement in endothelial function assessed by FMD in comparison with high-dose clopidogrel. 25 In a prospective, randomized study of 60 primary patients with ACS, ticagrelor greatly increased RHI after 30 days of treatment compared with clopidogrel, which had only a slight effect. Importantly, all the patients had ED (RHΙ < 1.67) at baseline and the observed effect of ticagrelor in endothelial function correlated with a significant increase in adenosine plasma level. 26 A positive impact of ticagrelor on endothelial function has also been described by Alemayehu et al in a prospective, randomized, crossover study of 50 patients with stable CAD having at least 2 cardiovascular risk factors at baseline along with ED (RHΙ < 2.0). Both clopidogrel and ticagrelor improved vascular reactivity compared with baseline after a treatment period of 3 weeks, with the effect being more pronounced with ticagrelor. 27 The improved endothelial function was observed during peak ticagrelor levels (at 2 hours after administration), but not at trough (12 hours after last ticagrelor dose). 28 In a prospective randomized study focusing on patients with chronic obstructive pulmonary disease (COPD) with PCI treated stable CAD, the rate of apoptosis in human umbilical vein endothelial cells after incubation with serum from patients was lower in those treated for 1 month with ticagrelor compared with clopidogrel, suggesting a beneficial impact of ticagrelor in this specific subgroup of patients, 19 which is characterized by high rates of ED. 33 Furthermore, in a prospective, randomized, crossover study by Jeong et al, the effect of ticagrelor compared with prasugrel in endothelial function was investigated in 62 patients with non-ST-segment elevation ACS with successful PCI and history of type II diabetes, using FMD. After a treatment period of 5 weeks with each of the studied regiments, authors reported a significantly greater improvement with ticagrelor compared with prasugrel. Moreover, ticagrelor significantly decreased levels of inflammatory cytokines and increased absolute numbers of circulating endothelial progenitor cells, findings potentially contributing to the observed improvement in endothelial function. 29
Contrary to the above results, in a prospective, observational study of 30 patients with stable CAD with prior PCI for ACS 1 year earlier, the cessation of ticagrelor treatment did not result in deterioration of endothelial function (RHI measured by EndoPAT) at day 5 following ticagrelor discontinuation. The rate of ED was also not affected. 30 Our group also failed to find evidence of alteration of endothelial function evaluated by EndoPAT, following ticagrelor compared to prasugrel treatment in a prospective, randomized, crossover study of 24 patients with stable CAD. 31 Finally, the most recent research work on this topic by Ariotti et al also challenges the hypothesis of a beneficial effect of ticagrelor on endothelial function, adding to the controversy. In a prospective, randomized, crossover, multicenter study of stabilized patients with prior ACS at least 30 days prior to enrollment (ranging greatly from 38 to 1023 days), RHI did not differ after ticagrelor as compared with prasugrel or clopidogrel 4-week treatment period. 32
Potential Explanations of Controversial Findings
The contradictory results of the relevant clinical studies make ticagrelor’s impact on endothelial function of patients with CAD quite a controversial issue. It should be mentioned that most of the relevant studies have several limitations, such as relatively small sample size, open-label design, prior treatment with P2Y12 receptor inhibitor before enrollment, and the absence of washout period in most of those with crossover design. In our view, the substantial differences in study design, methods, and patient population account for these discrepancies but also offer important insights into this effect.
More specifically, patient populations among the various studies on this issue differ regarding CAD presentation (stable CAD vs ACS), time of enrollment after the index ACS event, presence of diabetes, rate and severity of preexisting ED, and duration of treatment. Overall, a positive effect of ticagrelor—if present—is more probable to be obvious in the higher risk patients with preexisting ED.
The timing of endothelial function assessment in relation to the administration of ticagrelor’s maintenance dose seems to play an important role, since this effect is more prominent at peak drug levels. 27 Most of the studies have not taken this parameter into account, and this may have affected the results. In addition, concomitant medication, mainly statins and angiotensin-converting enzyme inhibitors, 34 have the potential to improve ED and might have modified the effect of ticagrelor. Furthermore, reliable endothelial function assessment greatly depends on strict methodology, with elimination of factors which might interfere with measurements (strenuous physical activity, caffeine, smoking, high-fat food, etc). 15 In published studies, it is not clear to which extent these restrictions have been applied.
Finally, FMD and—most commonly—PAT have been used to evaluate endothelial function. Both of these noninvasive methods have been extensively utilized as research tools for this purpose and have been shown to predict future adverse events in patients with CAD. 35 The FMD is the most established and commonly used method; however, it is technically challenging, and there is variability among studies regarding the protocol and the method of analysis applied. The PAT has the advantages of more standardized methodology and results are operator independent. However, the two methods assess different levels of vascular function, as FMD measures conduit artery vasodilation (macrovascular response of the brachial artery), while PAT measures microvascular response (Table 2). The degree to which these two methods correlate is also a matter of debate. 36
Future Directions
Elucidation of the effect of ticagrelor on the endothelial function is important in order to better understand the clinical profile of this agent. Such an action—if present—could have long-term beneficial effects in patients with CAD, where ED often exists and affects prognosis. Therefore, and in view of the contradictory results of performed studies, further research is warranted on this issue.
Future studies attempting to investigate the effect of ticagrelor on the endothelial function should aim to clarify how quickly does endothelial function respond to ticagrelor, how sustained this response is during the dosing intervals and in the long term, which mechanisms are implicated, and whether this pleiotropic action is clinically significant. Future research efforts should include larger and diverse populations of patients, assess endothelial function at several time points after treatment initiation, and use multiple methods of endothelial function measurement (eg, both RHI and FMD), while implementing strict methodology. Finally, although emphasis has been given in clinical research on this topic, perhaps more studies in animal models are needed in an attempt to clarify the underlying mechanisms and shed light on the role of platelets and adenosine receptors. 37
Conclusion
Several lines of evidence support a beneficial effect of ticagrelor on endothelial function, which seems to be more prominent among subgroups of high-risk patients with CAD (recent ACS, diabetes, several cardiovascular risk factors, preexisting ED, COPD). However, this issue is still controversial, and future research is necessary to confirm and better describe this effect. It should though be emphasized that the extent of the clinical benefit of ticagrelor attributable to actions beyond its potent and consistent antiplatelet effect remains uncertain.
Footnotes
Author Contributions
Moulias A, Xanthopoulou I, and Alexopoulos, D contributed to conception and design, drafted the manuscript, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Alexopoulos reports lecturing honoraria from Astrazeneca, Bayer, Boehringer Ingelheim, and advisory board fees from Astrazeneca, Bayer, Boehringer Ingelheim, and Medtronic.
