Abstract
Introduction and Objectives:
Atherosclerosis is recognized as a chronic inflammatory disease. The aim of this study was to examine the role of urinary trypsin inhibitor (UTI) in inflammation response induced by hyperlipidemia in rabbits.
Methods:
Thirty rabbits after injury of the right iliac artery endothelium were randomly divided into 3 groups: control group, model group, and UTI group. Iliac arteries were isolated and histology was performed on arterial regions that were injured by balloon after 8 weeks. Neointimal thickness (NT) and neointimal to media radio (N/M) were measured. Blood lipids, interleukin 6, and tumor necrosis factor-α were evaluated. Macrophages were evaluated by immunohistochemical analysis. MicroRNA-181b (miR-181b) was measured by reverse transcriptase-polymerase chain reaction.
Results:
Urinary trypsin inhibitor therapy decreased serum inflammatory factor levels without significant changes in blood lipids. Compared with model group, UTI reduced macrophage infiltration of iliac artery (13.91 ± 2.03% vs 24.21 ± 8.94%, P < .01). Hyperlipidemia reduced the expression of miR-181b and increased NT and N/M ratio. Systemic administration of UTI rescued miR-181b expression and inhibited neointimal formation.
Conclusions:
Urinary trypsin inhibitor could reduce neointimal hyperplasia by inhibiting inflammatory response induced by hyperlipidemia and may become a potential antiatherosclerosis supplement.
Introduction
Atherosclerosis is recognized as a chronic inflammatory disease of the arterial wall. 1,2 Inflammatory processes play a pivotal role in the pathogenesis of atherosclerosis and mediate many of the stages of atheroma development from initial leukocyte recruitment to eventual rupture of the unstable atherosclerotic plaque. MicroRNAs (miRNAs) are a class of small noncoding RNAs approximately 22 nucleotides in length that regulate gene expression by accelerating messenger RNA degradation or inhibiting translation at the posttranscriptional level. 3 In recent years, some researchers found that miRNAs were involved in the regulation of the development, differentiation, and the executive function of inflammatory cells (including endothelial cells and monocyte/macrophage) of atherosclerosis. 4 –10 A recent study has found that systemic delivery of miRNA-181b (miR-181b) inhibits necrosis factor κB (NF-κB) activation, vascular inflammation, and atherosclerosis in ApoE−/− mice. Regulation of miR-181b expression may become a viable strategy to treat patients with advanced atherosclerosis. 11
Urinary trypsin inhibitor (UTI) is also known as “Ulinastatin,” and it can inhibit the production and cascade of inflammatory cytokines and improve the inflammatory symptom and has been widely used to treat nonspecific inflammatory response diseases in clinical setting. 12 –17 We found that UTI could also reduce neointimal hyperplasia by inhibiting the nonspecific systemic inflammatory response induced by lipopolysaccharide after balloon injury in rabbits. 18 However, the role of UTI in chronic inflammatory disease states such as atherosclerosis has not been examined. The present study is designed to examine the role and the possible mechanisms of UTI on the neointimal formation and the inflammatory response induced by hyperlipidemia in rabbits.
Materials and Methods
Animal Model
Thirty male New Zealand white rabbits about 12 weeks old and weighing about 3 kg were provided by the Laboratory Animal Research Center of the 2nd Affiliated Hospital of Harbin Medical University. All studies were performed under the approval of the hospital scientific affairs committee on animal research and ethics. They were individually housed in air-conditioned room equipped with a laminar flow. Rabbits were anaesthetized with ketamine (35 mg/kg intramuscularly [IM]) and xylazine (8 mg/kg IM). Anesthesia was maintained during the procedure with isoflurane inhalation via mask. The right femoral arteries were exposed and ligated by artery clamp, and the right iliac arteries were denuded of endothelium by the intraluminal passage of a balloon catheter (Cordis, 3 × 20 mm2) passed to the the common iliac artery and withdrawn in the inflated state 3 times (1 minute inflation at 8 atm). The catheter was then removed, and the incision was sutured closed. Heparin (150 U/kg intravenously [IV]) and atropine (0.10 mg/rabbit IV) were administered immediately after arteriotomy, and animals were treated with penicillin (80 000 U/kg/d IM) for 3 days. Animals were randomly assigned to the control group (fed with standard rabbit chow for 8 weeks), the model group 19 (fed with high-fat diet (50 g/L lard, 10 g/L cholesterol, 70 g/L egg yolk powder and began with injection of saline of the same amount as that of UTI group after balloon injury, twice a week for 8 weeks), and the UTI group (fed with high-fat diet after balloon injury and began with intravenous injection of 20 000 U/kg/d of UTI [Techpool Biochemical Pharmaceutical Co Ltd, China], twice a week for 8 weeks).
Measurement of Blood Parameters
Plasma concentrations of total cholesterol (TC), low-density lipoprotein C (LDL-C), and triglyceride (TG) were measured by enzymatic method. Interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α) were measured by enzyme-linked immunosorbent assay. All the measurements were performed according to the manufacturer’s instructions, and each sample was assayed in duplicate.
Arterial Harvest and Morphometric Analysis
Rabbits were killed using an overdose of intravenous sodium pentobarbital (100 mg/kg) and iliac arteries were perfusion fixed with 4% paraformaldehyde solution (pH 7.4) at physiological pressure. Balloon-injured arteries were embedded in paraffin, and 6 sections, 5-µm thick, were cut from 3 equally spaced locations, staining with hematoxylin and eosin. Histologic sections were examined microscopically by an investigator blinded to treatment. All sites were analyzed by computerized morphometry (NIH Image), and the results were averaged. The intimal thickness, media thickness (MT), the ratio of neointimal thickness (NT) and MT were measured.
Immunohistochemistry for Macrophages
After deparaffinization and hydration of specimens, endogenous peroxidase activity was blocked, and the specimens were fixed by immersion in 0.3% hydrogen peroxide in methanol for 20 minutes. Immunohistochemical staining was performed with a rat monoclonal antibody against rabbit macrophage antibody 11 (working dilution 1:1200, Sigma company, USA) using the labeled streptavidin biotin complex method (simple-stain MAXPO kit; Nichirei, Tokyo, Japan). After blocking with 10% goat serum, the slides were incubated overnight with a primary antibody at 4°C in a moisture chamber. The slides were washed with Tris-buffered saline (TBS) and incubated with a biotinylated secondary antibody at room temperature for 30 minutes. After washing with TBS, the slides were incubated with streptavidin at room temperature for 30 minutes and visualized with 3,3′′-diaminobenzidine. The positively immunostained area of macrophages, identified by Brown staining in the cytosol, was quantified using the image analysis system.
Reverse Transcriptase Polymerase Chain Reaction
Expression of MiR-181b in the vessels were detected using transcriptase-polymerase chain reaction. Total RNA (5 µg) was isolated using TRIzol reagent (Invitrogen, Carlsbad, California) from homogenized tissues. QuantiTect Reverse Transcription Kit (QIAGEN, The Netherlands) was used to generate complementary DNA, and QuantiFast SYBR Green PCR Kit was used for quantitative real-time PCR with the Mx3000P Real-time PCR system (Stratagene, La Jolla, California) following the manufacturer’s instructions. The specifc miR-181b reverse transcriptional primer was 5′-GTCGTATCCAGTGCAGGGTCCGAGGTGCACTGGATACGACCACCCACC-3′. The PCR primers were 5′-TGCGGAACATTCATTGCTGTC-3′ (forward) and 5′-CCAGTGCAGGGTCCGAGGT-3′ (reverse). Under amplification conditions, a denaturation step was carried out at 94°C for 5 minutes, followed by 40 cycles at 94°C for 30 seconds, 50°C for 30 seconds, and 72°C for 30 seconds. After the reactions, the threshold cycle (Ct) value of the PCR curve was analyzed. U6 ribosomal RNA (the specific reverse transcriptional primer was 5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGTACGACAAAA
TATGGAAC-3′, the PCR primers were U6—forward: 5′-TGCGGGTGCTCGCTTCGGCAGC-3′; reverse: 5′-CCAGTGCAGGGTCCGAGGT-3′) was used as the positive internal reference to correct cell copy number on the PCR template and eradicate error in the sample application amount, which was based on the following formula: ΔCt value of the target gene = Ct value of the target gene − U6 Ct value of the same sample. The procedure was repeated 3 times. The relative cyclic value of the target gene (ΔΔCt) was calculated based on the following formula: ΔCt value = average ΔCt value of the treated group − average ΔCt of the control group. The ΔΔCt value was calculated using the 2−ΔΔCt method.
Statistics
All data were presented as mean ± standard deviation. Numeric values were analyzed for the presence of normal distribution. Comparisons among groups were performed by 1-way analysis of variance analysis. A value of P ≤ .05 was considered significant. All statistics were calculated with SPSS v18.0 software.
Results
There were 30 NZW male rabbits in this study and the average body weight was 3.14 ± 0.08 kg. All animals survived during the experiment.
Effects of UTI on Blood Lipids, IL-6, and TNF-α in Hyperlipidemic Rabbits
Data in Table 1 indicated that blood lipids (TC, LDL-C, and TG) and inflammatory marker (IL-6 and TNF-α) remarkably increased after 8 weeks of high-fat diet (P < .01). The UTI therapy had no significant impact on blood lipids but decreased the serum level of IL-6 and TNF-α (P < .01).
The Results of Blood Lipids and IL-6 of Each Group.a

Abbreviations: IL-6, interleukin 6; LDL-C, low-density lipoprotein C; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor α; SD, standard deviation; UTI, urinary trypsin inhibitor.
aThe data are expressed as mean ± SD. P value <.05 indicates a significant difference.
b P < .01, compared with control group.
c P < .01, compared with model group.
Effect of UTI on Intimal Hyperplasia Induced by Hyperlipidemia
Morphometric analysis of hyperlipidemia-injured arteries at 8 weeks revealed neointimal hyperplasia. The NT in the control group was 32.10 ± 0.02 µm, MT was 151.83 ± 5.38 µm, and neointima-to-media (N/M) ratio was 0.21 ± 0.03. Hyperlipidemia significantly increased NT and N/M to 484.16 ± 40.22 µm and 3.29 ± 0.30, respectively (P < .01). Upon administration of UTI to hyperlipidemic rabbits, the neointimal growth was decreased significantly compared with the model group (P < .01; Figure 1A-E).

Intimal hyperplasia of each group at 8 weeks. Forty magnification photomicrographs (hematoxylin and eosin [HE] staining) of arterial sections from control group (A), model group (B), and urinary trypsin inhibitor (UTI) group (C). Note thicker intima in model group. D and E, Bar graph shows increased neointima formation and neointima to media ratio in arteries of model animals (n = 10) versus control rabbits (*P < .01). Treatment with UTI blocked these effects induced by hyperlipemia (# P < .01).
Expression of miR-181b
As shown in Figure 2, expression of miR-181b was reduced by 62% in the iliac intima of hyperlipidemic rabbits, and it was increased significantly in UTI therapy group compared with model group (3.78 ± 0.54 vs 1.63 ± 0.33, P < .01). In the model group, the levels of IL-6 and TNF-α were negatively correlated with the expression of miR-181b after being fed with high-fat diet for 8 weeks (R 2 = .991 and .989, respectively; Figure 3A and B).

Expression of microRNA-181b (MiR-181b) in each group. Compared with model group, urinary trypsin inhibitor (UTI) rescued the expression of miR-181b. *P < .01, versus control group; # P < .01 versus model group.
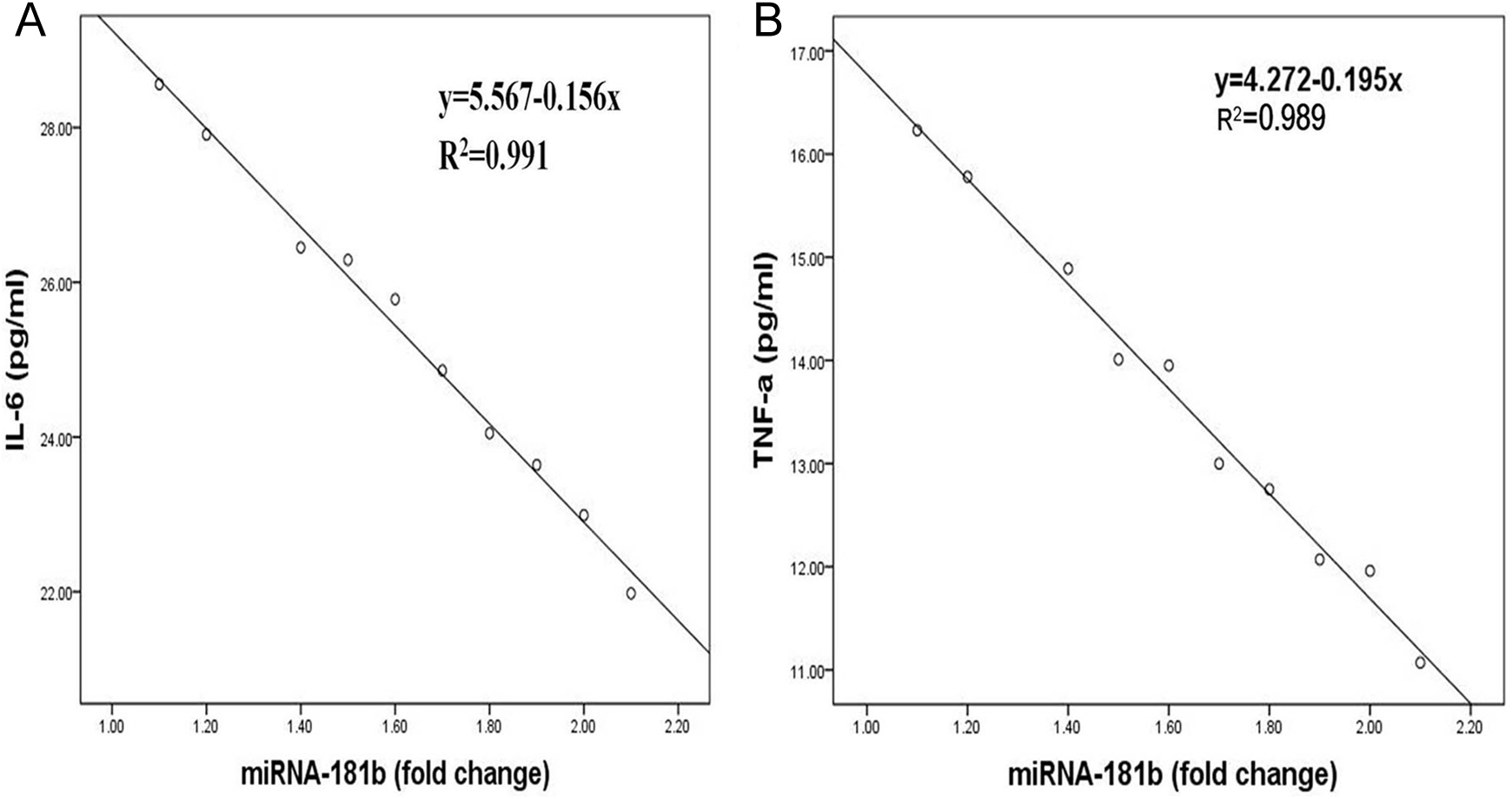
A, Bar graph shows a strong negative correlation between the level of interleukin 6 (IL-6) and microRNA-181b (miR-181b) expression after fed with high-fat diet for 8 weeks (R 2 = .991). B, Bar graph showing a strong negative correlation between the level of tumor necrosis factor α (TNF-α) and miR-181b expression after fed with high-fat diet for 8 weeks (R 2 = .989).
Immunohistochemical Staining for Macrophage Infiltration in the Iliac Artery
Figure 4A to D shows the results of immunohistochemical staining of each group. The lack of expression of macrophages in normal vascular tissues but the positively immunostained area of macrophages in the iliac artery of hyperlipidemic rabbits was significantly increased. Compared with the model group, UTI could significantly reduce macrophage infiltration (13.91 ± 2.03% vs 24.21 ± 8.94%, P < .01). In the model group, the macrophage positive area was negatively correlated with expression of miR-181b after being fed with high-fat diet for 8 weeks (R 2 = .966, Figure 4E).
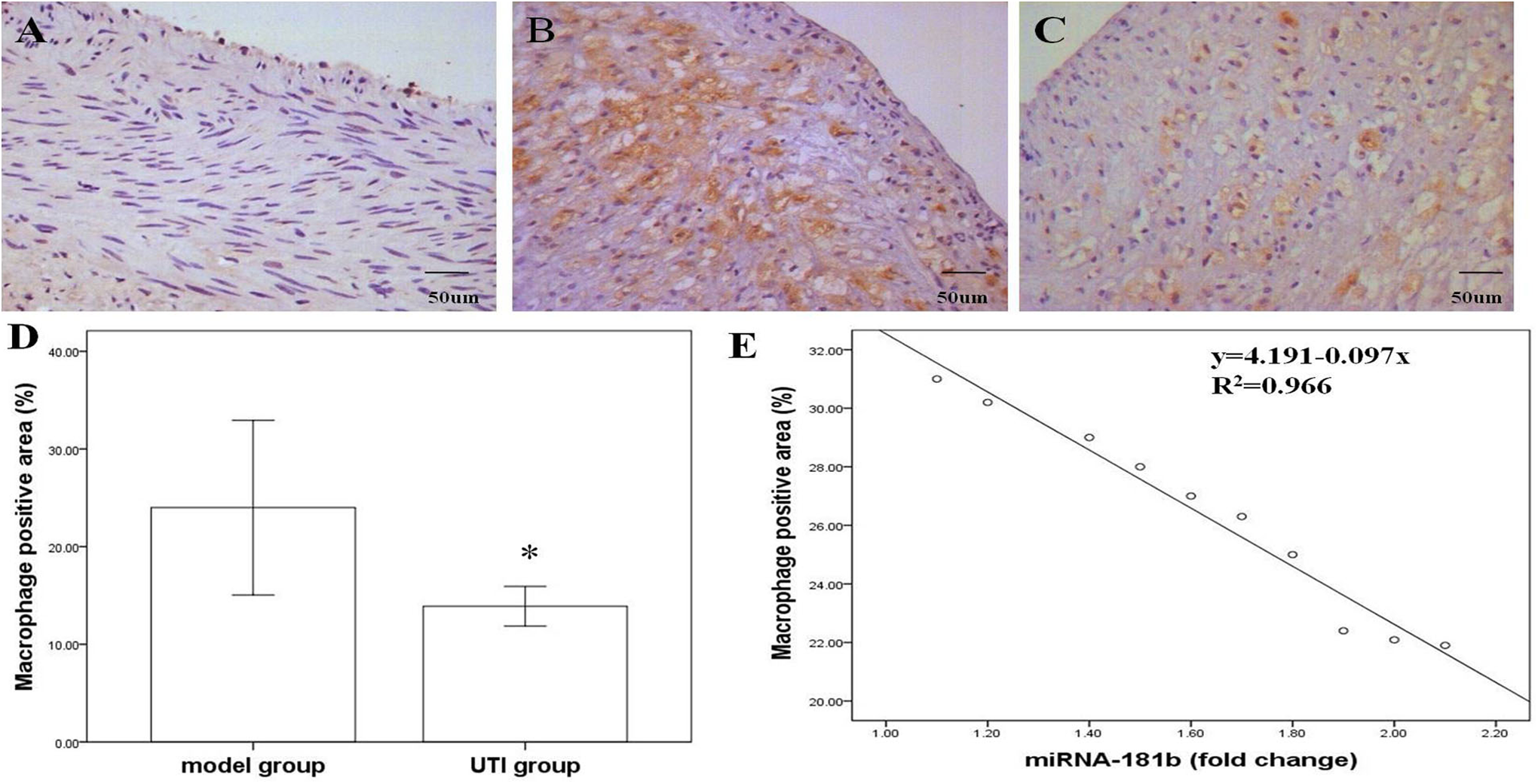
Macrophage infiltration of each group. A, Control group, (B) model group, and (C) urinary trypsin inhibitor (UTI) group. D, Bar graph showing decreased macrophage infiltration in arteries of UTI group compared with model group (*P < .01). E, Bar graph showing a strong negative correlation between the macrophage positive area and microRNA-181b (miR-181b) expression after fed with high-fat diet for 8 weeks (R 2 = .966).
Discussion
The principal findings in the present study are as follows: (1) hyperlipidemia can remarkably increase the levels of blood lipids and induce inflammatory response and significant neointimal hyperplasia. (2) The UTI could reduce neointimal hyperplasia induced by hyperlipidemia by inhibiting the local and the systemic inflammatory response.
The UTI is a kind of broad-spectrumed proteases inhibitor, taken from healthy adult urine, which has a variety of pharmacological effects: (1) activity inhibition of neutrophil elastase and other proteases; (2) suppression of protease secretion from neutrophils via stabilization of the lysosomal membrane; (3) an inhibitory effect on the production of cytokines and adhesion molecules, such as ILs 1, 6, and 8, TNF-α, intercellular adhesion molecule-1 and endothelial leukocyte adhesion molecule-1; (4) anti-inflammation; (5) antioxidation, and (6) reversing the imbalance of matrix metalloproteinase activation. 20 –26 Therefore, UTI is clinically used for the treatment of inflammatory disease states, such as sepsis, pancreatitis, and acute respiratory distress syndrome. From animal studies, we also find that UTI can reduce neointimal formation after stenting or lipopolysaccharide injection by inhibiting the local and the systemic inflammatory response. 18,27 Inflammation is a major force in the pathophysiology of atherothrombosis, and it takes part in the formation, progression, and destabilization of the atherosclerotic lesion. Several anti-inflammatory strategies have successfully attenuated atherosclerosis, such as Xuemaitong granules, 28 AnxA5, 29 IL-10, 30 and so on. We conclude that UTI may attenuate vascular injury caused by atherosclerosis by its anti-inflammatory effect.
The miR-181b is emerging as a new regulator of inflammatory and immune responses. Recent studies show that expression of miR-181b was reduced in plasma from patients with sepsis or in aortic intima and plasma of ApoE−/− mice or in human plasma from patients with coronary artery disease, and the effect is associated with a poor prognosis. 11,31 The miR-181b level is decreased dramatically by chronic inflammatory stimuli in the vascular endothelium and plasma of mice, suggesting it may be involved in the early pathogenesis of atherosclerosis, such as endothelial cell dysfunction and damage, smooth muscle cell migration and proliferation, and increased macrophages and T cells. 11 This study also finds that systemic delivery of miR-181b can significantly decrease arterial NF-κB activation and NF-κB-regulated gene expression associated with inflammatory disease states, such as adhesion molecules, chemokines and chemokine receptors, and other key inflammatory mediators in the vascular endothelium resulting in reduced leukocyte accumulation and atherosclerosis. 11 The researchers offer that strategies aimed at restoring expression of miR-181b may provide a novel therapeutic approach for acute or chronic inflammatory disease states. Previously, we found that UTI could inhibit inflammatory response after lipopolysaccharide injection in rabbits by inhibiting NF-κB signaling and suppressing the expression of downstream inflammatory target genes. 18 Therefore, we assume that UTI inhibiting inflammatory response might be through targeting of upstream gene miR-181b of NF-κB inflammatory signal pathway. In this study, we find that expression of miR-181b in hyperlipidemia rabbits was reduced, and miR-181b can negatively regulate the expression of IL-6, TNF-α, and macrophage, and systemic delivery of UTI can significantly increase expression of miR-181b and relieve the blood vessel damage ensues caused by hyperlipidemia. These findings are consistent with our premise. However, overexpression or inhibition of miR-181b studies are needed to draw this conclusion and it is one of the limitations of this study. This study is also limited to observe in the animal models and whose relevance to human clinical condition was uncertain. Therefore, a prospective clinical study should be performed in order to evaluate the effect of UTI on human atherosclerotic lesions.
Conclusions
In conclusion, UTI may manipulate the expression of miR-181b and its downstream inflammatory target genes to inhibit intimal hyperplasia and inflammatory response induced by hyperlipidemia. It is suggested that inhibition of inflammatory pathway can effectively prevent advanced atherosclerosis, and UTI may become a potential antiatherosclerosis supplement.
Footnotes
Authors’ Contribution
Ying Deng contributed to conception and design, drafted the article, and gave final approval. Junying Kong contributed to acquisition, analysis, and interpretation; critically revised the article; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
