Abstract
Background:
Dihydropyridine calcium channel blockers are a heterogeneous group of antihypertensive drugs. Long-acting dihydropyridine agent amlodipine is widely used for monotherapy and combination therapy for hypertension in clinical practice, while intermediate-acting dihydropyridine agents have shown inconsistent results in randomized clinical trials (RCTs).
Methods and Results:
A meta-analysis of 18 RCTs enrolling a total of 80,483 patients with hypertension followed for a mean of 51.4 months was performed. Amlodipine therapy was associated with 25% higher risk of heart failure (relative risk [RR]: 1.25, 95% confidence interval [CI], 1.05-1.49, P = .019) but 17% lower risk of stroke (RR: 0.83, [95% CI, 0.72-0.97], P = .009) without statistically significant effect on acute myocardial infarction (AMI) compared to major alternative antihypertensive therapy (MAAT), including β-blocker, diuretic, angiotensin-converting enzyme inhibitor, or angiotensin-receptor blocker. Intermediate-acting dihydropyridine calcium channel blocker therapy was associated with 25% higher risk of heart failure (RR: 1.25, [95% CI, 1.06-1.47], 0.005, P = .005) and 26% higher risk of AMI (RR: 1.26, [95% CI, 1.05-1.51], 0.019, P = .019) compared to MAAT. Results of the subgroup analysis suggested that the intermediate-acting dihydropyridine calcium channel blocker was associated with higher risk of heart failure (RR: 1.30, [95% CI, 1.08-1.56], P = .005) and AMI (RR: 1.50, [95% CI, 1.01-2.22], P = .043) compared to renin–angiotensin system blockers and a trend toward higher risk of AMI (RR: 1.17, [95% CI, 0.99-1.38], P = .064) compared to conventional therapy, including β-blockers and diuretics. Meta-regression analyses suggested that long-acting dihydropyridine calcium channel blocker is associated with lower risk of AMI (B: −0.327, [95% CI, −0.530 to −0.123], P = .002) with a trend toward lower risk of stroke (B: −0.203, [95% CI, −0.410 to 0.003] P = .054).
Conclusions:
This study suggests that Amlodipine offers greater protection against major complications of hypertension compared to intermediate-acting dihydropyridine calcium channel blockers.
Keywords
Introduction
Calcium channel blockers (CCBs) and renin–angiotensin system (RAS) blockers, including angiotensin-converting enzyme inhibitor (ACE I) and angiotensin-receptor blocker (ARB), are newer antihypertensive agents widely applied in clinical practice and extensively investigated in recent randomized controlled trials (RCTs) as monotherapy and combination therapy for hypertension, while β-blockers and diuretics represent the older conventional antihypertensive therapy. There is growing evidence that major antihypertensive drug classes, including CCBs, exert blood pressure (BP)–independent effects that may contribute to interclass variations against cause-specific clinical outcomes. 1 -10 For example, previous studies have suggested that ARB may be associated with higher risk of acute myocardial infarction (AMI) in patients with hypertension, 11 and CCBs may be associated with a higher risk of AMI and heart failure. 12,13
The CCBs are classified into 3 subclasses, dihydropyridines, diphenylalkylamines, and benzothiazepines, based on their vascular and cardiac selectivity. Dihydropyridine calcium channel blocker (dCCB) represents a subclass of CCBs with high vascular selectivity that is used primarily as antihypertensive drug. The 3 subclasses of CCBs are further classified into 3 generations based on pharmacotherapeutic properties, such as structure, rate of onset, and duration of action, which are primarily determined by their half-life. Drugs with shorter half-lives have rapid onset of action that may provoke neurohumoral activation, shorter duration of action that may result in inadequate control of 24-hour BP, and some degree of negative dromotropic and inotropic effects. Consistent with that the short-acting, first-generation dCCB was not approved for hypertension because of safety concerns. 14 -18 Subsequent studies suggested the need to critically examine the complexity of evidence regarding the safety and therapeutic efficacy before implicating the entire class of CCBs, the subclasses, or individual drugs within these subclasses. 14 -16 The inherently long-acting, third-generation dihydropyridine CCB (LA-dCCB) Amlodipine (elimination half-life 35-50 hours) 17 has become one of the most widely used antihypertensive drug, 18 while the results of RCTs investigating the potential application of intermediate-acting, second-generation dihydropyridine CCBs (IA-dCCB; defined as drugs with half-life ranging between 6 and 24 hours) in hypertension have been inconsistent. 19 -21
Previous meta-analytic studies have suggested CCBs are superior to placebo in preventing major complications of hypertension, but the results have been divergent against other antihypertensive drugs on AMI, stroke, and heart failure. 12,22 -24 This could reflect differences between dCCB and non-dCCB or between IA-dCCB and LA-dCCB or differences in study design such as inclusion of placebo-controlled trials, nonhypertension trials, or non-dCCBs. The objective of the current study was to investigate potential therapeutic differences between the inherently LA-dCCB agent amlodipine and IA-dCCB agents in patients with hypertension against cause-specific cardiovascular protection based on prospective, randomized, actively controlled trials.
Methods
Data Sources and Search Strategy
Electronic database PubMed was searched using the key words “calcium channel blocker,” “calcium blocker,” “dihydropyridine,” “dihydropyridine calcium channel blocker,” “individual names in these drug classes,” and “hypertension.” Additionally, references of retrieved articles and previous meta-analysis were manually searched to identify studies not missed by our primary search strategy. No restrictions were imposed on dates of publication, but the search was restricted to abstracts of clinical trials in humans published in English. The last search update was performed on March 13, 2018.
Study Selection
Inclusion criteria were: Prospective RCTs. Trials enrolling male or female participants aged ≥18 years with systolic BP (SBP) ≥140 and/or diastolic BP (DBP) ≥90 mm Hg or taking antihypertensive therapy. Trials comparing IA-dCCB or LA-dCCB against major alternative antihypertensive therapy (MAAT), including β-blockers, diuretics, ACEI, or ARB.
Exclusion criteria were: Trials using short-acting dCCB or non-dCCB. Placebo-controlled trials. Combination antihypertensive therapy as first-line drug. Follow-up duration <1 year. Trials not reporting outcomes of interest. Animal studies, reviews, case reports, and conference abstracts.
End Points
Endpoints of the meta-analysis included all-cause mortality, cardiovascular mortality, heart failure, AMI, and stroke.
Data Extraction and Quality Assessment
Data were extracted by 2 independent reviewers (SC and LYS), and disagreements were resolved by consensus. The included trials were evaluated for risk of bias using standard criteria: random sequence generation, allocation concealment, blinding of participants, personnel, and outcome assessor, incomplete outcome ascertainment, selective reporting, and other potential sources of bias as recommended by the Cochrane Collaboration (Supplementary Table 1).
Statistical Analysis
Measurement data were pooled across studies and analyzed using Mantel-Haenszel method with random effects meta-analysis models with inverse variance weighting. The magnitude of the between-studies heterogeneity was estimated using the I2 statistic, where a value over 50% requires further examination and is of concern. 25 The results are presented as relative risk (RR) with 95% confidence interval (CI). Univariate meta-regression models with inverse variance weighting (inverse of the sum of the within-trial variance and the residual between-trial variance) were performed to identify potential effect modifiers. Additive (between-study) component of the variance τ2 was estimated using restricted maximum likelihood method to account for residual heterogeneity that is not explained by the potential effect modifier. We hypothesized dCCB drug class (LA-dCCB vs IA-dCCB), difference in mean SBP reduction between cases and controls, and baseline presence of diabetes may affect the outcomes. Publication bias was explored by performing Begg and Mazumdar rank correlation test, Egger regression test, 26,27 and visual inspection of funnel plots. All P values are 2 tailed with the statistical significance set at P ≤ .05. Statistical analyses were performed using STATA software 12.0, or the comprehensive meta-analysis (version 2.0) 28 for confirmation when appropriate.
Results
Study Selection and Characteristics
The flow diagram of study selection is shown in Figure 1. Of the initial 10 505 studies identified, 138 articles were retrieved for detailed evaluation for potential inclusion. Eighteen randomized, actively controlled trials (N = 80 483) were included in the meta-analysis. Tables 1 and 2 summarize the trial design and baseline characteristics of the participants.

Flow diagram of study selection.
Basic Characteristics of Included Trials.

Abbreviations: ABCD, Appropriate Blood Pressure Control in Diabetes; ACE I, angiotensin-converting enzyme inhibitors; AF, atrial fibrillation; ALLHAT, Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial; ASSK, African American Study of Kidney Disease and Hypertension Trial; BB, β-blockers; CAD, coronary artery disease; CVE, cardiovascular events; D, diuretics; DBP, diastolic blood pressure; DM2, type 2 diabetes mellitus; ELSA, European Lacidipine Study on Atherosclerosis; EU, European Union; FACET, Fosinopril versus Amlodipine Cardiovascular Events Randomized Trial; HCTZ, hydrochlorothiazide; IDNT, Irbesartan Diabetic Nephropathy Trial; IGT, impaired glucose tolerance; IMT, intima-media thickness; INSIGHT, Intervention as a Goal in Hypertension Treatment; JIMC-B, Japan Multicenter Investigation for Cardiovascular Diseases-B; J-RHYTHM II, Japanese Rhythm Management Trial II for Atrial Fibrillation; MIDAS, Multicenter Isradipine Diuretic Atherosclerosis Study; NICS-EH, National Intervention Cooperative Study in Elderly Hypertensives; NIDDM, Noninsulin-dependent diabetes mellitus; PROBE, prospective, randomized, open-labeled, blinded endpoint; SBP, systolic blood pressure; STOP-2, Swedish Trial in Old Patients with Hypertension-2; VALUE, Valsartan Antihypertensive Long-term Use Evaluation Trial.
Baseline Characteristics of Trial Participants.

Abbreviations: A, Asian; AA, African American; ABCD, Appropriate Blood Pressure Control in Diabetes; ALLHAT, Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial; ASSK, African American Study of Kidney Disease and Hypertension Trial; C, Caucasian; CHD, coronary heart disease; DBP, diastolic blood pressure; ELSA, European Lacidipine Study on Atherosclerosis; FACET, Fosinopril versus Amlodipine Cardiovascular Events Randomized Trial; H, Hispanic; IDNT, Irbesartan Diabetic Nephropathy Trial; INSIGHT, Intervention as a Goal in Hypertension Treatment; JIMC-B, Japan Multicenter Investigation for Cardiovascular Diseases-B; J-RHYTHM II, Japanese Rhythm Management Trial II for Atrial Fibrillation; MIDAS, Multicenter Isradipine Diuretic Atherosclerosis Study; NICS-EH, National Intervention Cooperative Study in Elderly Hypertensives; NR, not reported; SBP, systolic blood pressure; STOP-2, Swedish Trial in Old Patients with Hypertension-2; VALUE, Valsartan Antihypertensive Long-term Use Evaluation Trial.
The IA-dCCB included felodipine (Swedish Trial in Old Patients with Hypertension-2 [STOP-2]), 29 isradipine (STOP-2 29 and Multicenter Isradipine Diuretic Atherosclerosis Study [MIDAS] 30 ), nicardipine SR (National Intervention Cooperative Study in Elderly Hypertensives [NICS-EH]), 31 nisoldipine (Appropriate Blood Pressure Control in Diabetes Trail [ABCD]), 19 nitrenidipine (Morbidity and Mortality After Stroke, Eprosartan Compared With Nitrendipine for Secondary Prevention [MOSES], 32 nifedipine retard (Japan Multicenter Investigation for Cardiovascular Diseases-B [JMIC-B] 33 )/nifedipine GITS (Intervention as a Goal in Hypertension Treatment [INSIGHT]) 34 and Nifedipine versus telmisatan on prevention of atrial fibrillation recurrence [NTP-AF] 35 , and lacidipine (European Lacidipine Study on Atherosclerosis [ELSA]) 20 and Systolic Hypetension in the Elderly [SHELL] 21 , and LA-dCCB included amlodipine (Fosinopril versus Amlodipine Cardiovascular Events Randomized Trial [FACET]), 36 (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial [ALLHAT]), 37 (African American Study of Kidney Disease and Hypertension Trial [AASK]), 38 Irbesartan Diabetic Nephropathy Trial [IDNT], 39 (Valsartan Antihypertensive Long-term Use Evaluation [VALUE]), 40 (Candesartan Antihypertensive Survival Evaluation in Japan [CASE-J]), (Japanese Rhythm Management Trial II for Atrial Fibrillation [J-RHYTHM II]), 41 and NAGOYA HEART Study (NHS) 42 ). MIDAS, FACET, SHELL, and MOSES did not report data on cardiovascular mortality; and FACET and ELSA did not provide data on heart failure.
Mean age of participants was 55.2 years, 66.2% were males, and mean follow-up duration was 51.4 months. Table 3 summarizes the main results of the meta-analysis.
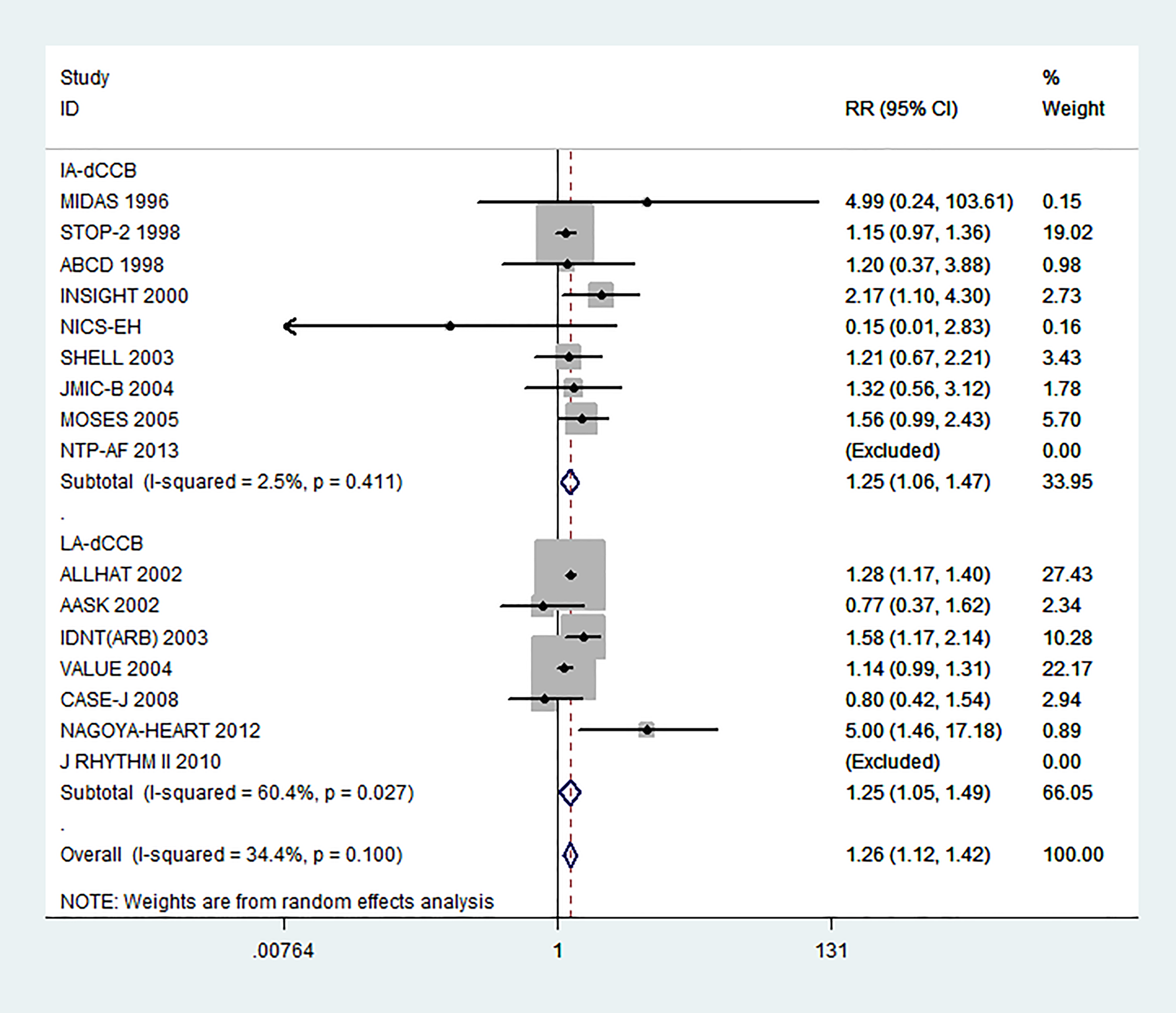
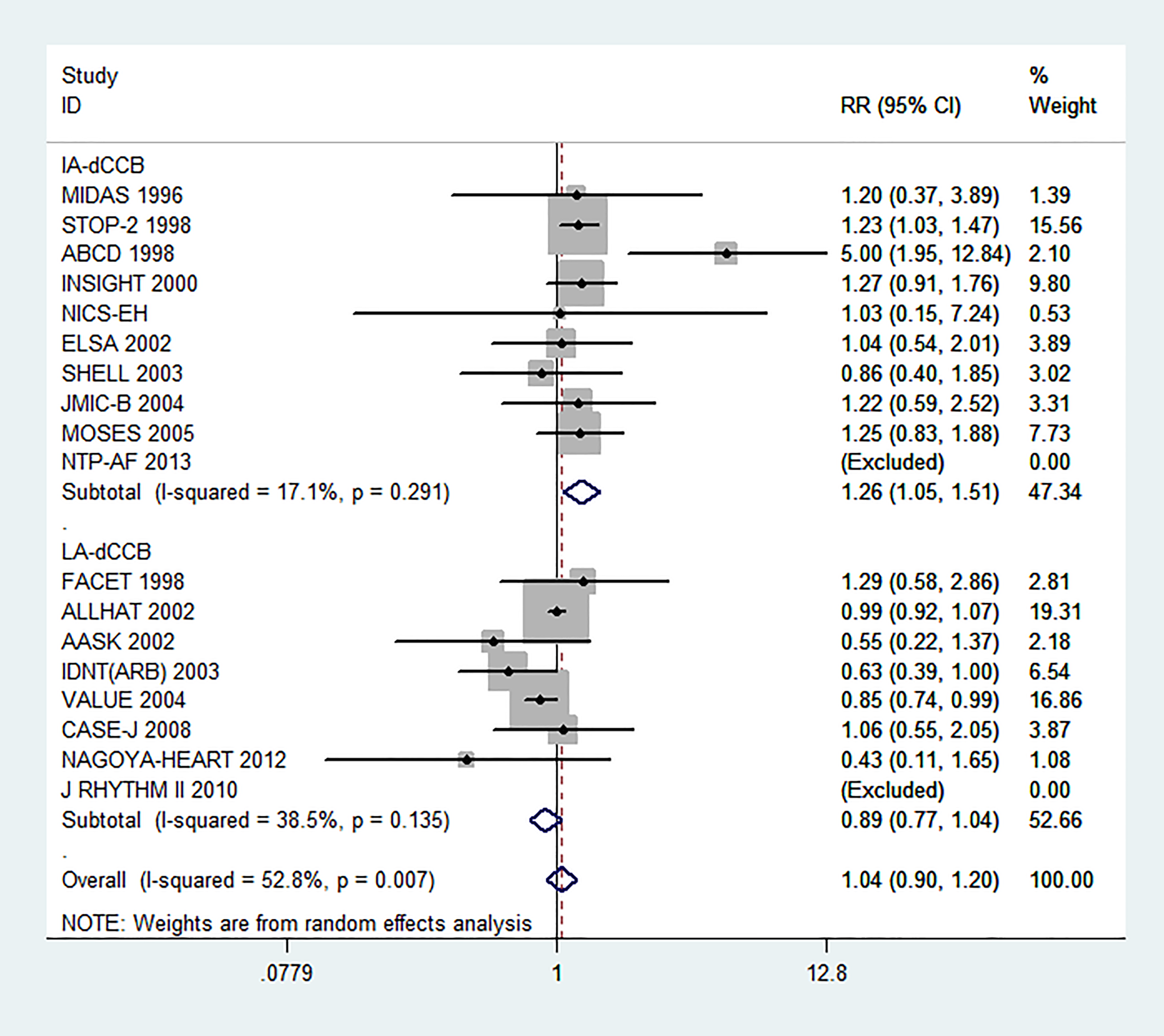
Effect of IA-dCCB and LA-dCCB on Cardiovascular Outcomes in Hypertensive Patients.

Abbreviations: CI, confidence interval; IA-dCCB, intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk.
All-cause mortality: No significant effect was observed on all-cause mortality for LA-dCCB (RR: 0.96, [95% CI, 0.91-1.01], P = .10) or IA-dCCB (RR: 1.00, [95% CI, 0.92-1.09], P = .94) compared to MAAT (Figure 2). Cardiovascular mortality: No significant effect was observed on cardiovascular mortality for LA-dCCB (RR: 0.99, [95% CI, 0.92-1.07], P = .800) or IA-dCCB (RR: 0.99, [95% CI, 0.87-1.14], P = .92) compared to MAAT (Figure 3). Heart failure: LA-dCCB therapy was associated with a 24% higher risk of heart failure (RR: 1.25, [95% CI, 1.05-1.49], P = .019), and IA-dCCB was associated with a 24% higher risk of heart failure (RR: 1.25, [95% CI, 1.06-1.47], P = .005) with significant heterogeneity observed for LA-dCCB (I2: 60.4% and P = .027) compared to MAAT (Figure 4). Stroke: LA-dCCB therapy was associated with a statistically significant protection (RR: 0.83, [95% CI, 0.72-0.97], P = .009), but no effect of IA-dCCB was seen (RR: 1.04, [95% CI, 0.89-1.22], P = .58) compared to MAAT (Figure 5). AMI: LA-dCCB showed a trend toward protection (RR: 0.89, [95% CI, 0.77-1.04], P = .13), but IA-dCCB was associated with significantly higher risk of MI (RR: 1.26, [95% CI, 1.05-1.51], P = .019) compared to MAAT (Figure 6). Subgroup analysis by comparator drug class: β-Blockers and diuretics were grouped as conventional therapy, while ACE I and ARB were grouped as RAS blockers for the subgroup analysis. Results of the subgroup analysis were similar in direction to the main analysis and are shown in Table 4. The IA-dCCB showed higher risk of heart failure (RR: 1.30, [95% CI, 1.08-1.56], P = .005) and AMI (RR: 1.50, [95% CI, 1.01-2.22], P = .043) compared to RAS blockers. Compared to conventional therapy, IA-dCCB therapy showed a nonsignificant trend toward higher risk of heart failure (RR: 1.27, [95% CI, 0.84-1.92], P = .25), while LA-dCCB analysis was inadequately powered. Publication bias, sensitivity analysis, and meta-regression: No evidence of publication bias was detected for any of the outcomes examined (data not shown). Effect of IA-dCCB and LA-dCCB on all-cause mortality. IA-dCCB indicates intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk; CI, confidence interval. Effect of IA-dCCB and LA-dCCB on cardiovascular mortality. IA-dCCB indicates intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk; CI, confidence interval. Effect of IA-dCCB and LA-dCCB on heart failure. IA-dCCB indicates intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk; CI, confidence interval. Effect of IA-dCCB and LA-dCCB on stroke. IA-dCCB indicates intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk; CI, confidence interval. Effect of IA-dCCB and LA-dCCB on acute myocardial infarction. IA-dCCB indicates intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RR, relative risk; CI, confidence interval. Results of Subgroup Analysis by Comparator Drug Class. Abbreviations: CI, confidence interval; IA-dCCB, intermediate-acting dihydropyridine calcium channel blocker; LA-dCCB, long-acting dihydropyridine calcium channel blocker; RAS blockers, renin–angiotenin system blockers; RR, relative risk.




Sensitivity analysis performed (1) by excluding 1 study at a time to estimate the effect size for remainder studies for heart failure, stroke, and AMI and (2) by restricting the analysis to double-blind trials or trials designed to prevent cardiovascular events suggesting the results to be robust (data not shown).
Table 5 summarizes the results of meta-regression analyses. Univariate meta-regression analyses were performed to evaluate the effect of dCCB drug class (IA-dCCB as control), difference in mean SBP reduction between cases and controls, and baseline presence of diabetes on heart failure, stroke, and AMI. Univariate meta-regression analysis suggested that LA-dCCB therapy was associated with lower risk of AMI (B: −0.327, [95% CI, −0.530 to −0.123], P = .002) with a trend toward lower risk of stroke (B: −0.203, [95% CI, −0.410 to 0.003], P = .054) compared to MAAT, and baseline presence of diabetes was associated with a modestly elevated risk of heart failure (B: 0.004, [95% CI, 0.001-0.008], P = .025; Figure 7).
Classic Univariate Variable Meta-Regression Using the REML.a
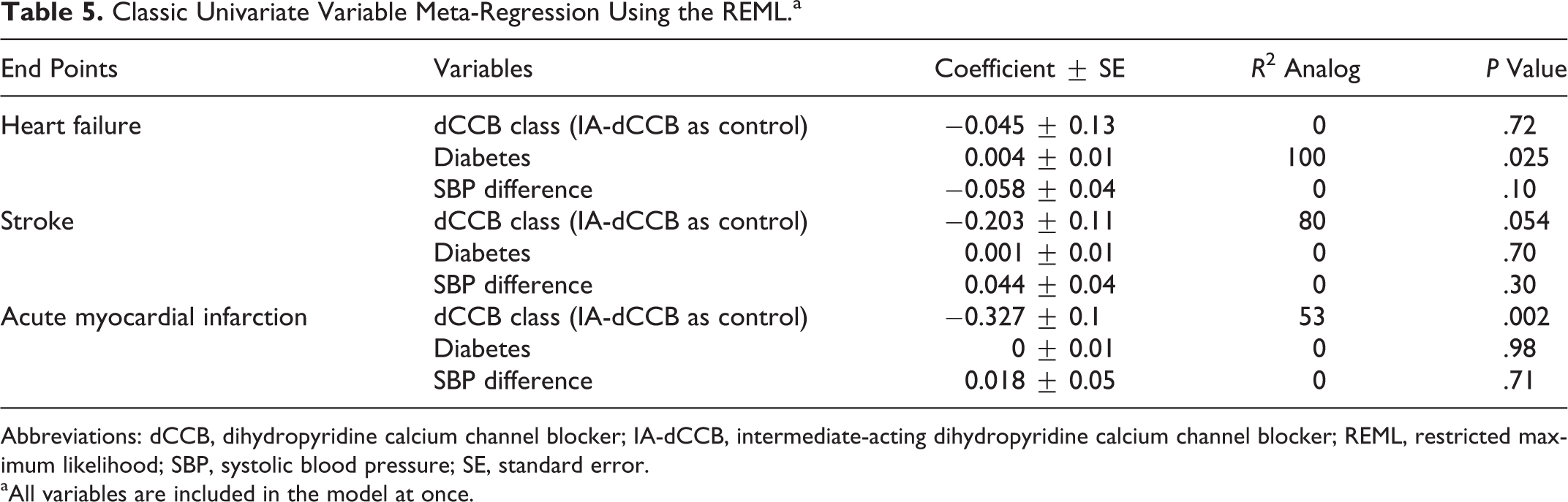
Abbreviations: dCCB, dihydropyridine calcium channel blocker; IA-dCCB, intermediate-acting dihydropyridine calcium channel blocker; REML, restricted maximum likelihood; SBP, systolic blood pressure; SE, standard error.
aAll variables are included in the model at once.

Effect of baseline presence of diabetes on the risk of heart failure (univariate random-effects meta-regression). RR indicates relative risk; SE of RR, standard error of relative risk.
Discussion
The current meta-analysis of 18 prospective, randomized actively controlled trials enrolling 80,83 patients with hypertension compared the therapeutic efficacy of IA-dCCB and LA-dCCB against MAAT. No significant difference in all-cause or cardiovascular mortality was found. Our major finding is that IA-dCCB therapy is associated with higher risk of AMI and lacks protection against stroke which is generally thought to be a feature of dCCB. 22 -24,43 Further, we confirmed previous observations of higher risk of heart failure with dCCBs and extend it further by showing both IA-dCCBs and LA-dCCBs are associated with higher risk of heart failure and that the risk may be slightly higher in diabetics.
Both BP-dependent and BP-independent mechanisms may account for differences in therapeutic efficacy between major antihypertensive drug classes, despite similar lowering of office SBP. Recent studies have shown that 24-hour BP, including nighttime BP, BP variability, and central BP may predict cardiovascular outcomes. 44 This could explain why drugs with comparable effects on office SBP in clinical trials differ in cardiovascular protection. There is accumulating evidence that amlodipine may offer better control of BP over 24 hours, including early morning and nighttime BP, 45 BP variability, 46,47 and central BP measured at aorta. 48,49 In a recent head-to-head trial comparing equivalent doses of Amlodipine to extended-release Felodipine, amlodipine offered better control of nighttime BP despite similar effect on daytime BP. Additionally, greater proportion of patients achieved target BP. 50
The dCCBs are vasculoselective agents that elicit compensatory responses including sympathetic nervous system and RAS activation that drives the risk of adverse clinical outcomes. 2 These responses are comparatively lower for agents with longer elimination half-life, such as amlodipine, compared to short- or intermediate-acting agents. 51,52 Further, IA-dCCBs may have additional limitations, such as significant variations in 24-hour BP control, 53 sudden reduction in activity causing a rapid loss of antihypertensive efficacy, 54 potential intermittent sympathetic activation, 55 and incomplete release of drugs in delayed-release formulations. 54 This could partly contribute to the observed differences in therapeutic efficacy between LA-dCCB and IA-dCCB.
Previous studies have suggested that major antihypertensive drug classes do not differ in all-cause or cardiovascular mortality prevention. 22,56 α-Blockers showing a higher risk of adverse cardiovascular events in ALLHAT was not associated with higher risk of all-cause or cardiovascular mortality. 57 Our data on all-cause and cardiovascular mortality are in line with previous reports. Differential therapeutic efficacy against cause-specific cardiovascular morbidity without significant differences in all-cause or cardiovascular mortality suggests that major antihypertensive drug classes may differ primarily on their effect on nonfatal cardiovascular events.
Both IA-dCCB and LA-dCCB showed comparable degree of risk against heart failure. This risk appeared particularly exaggerated in trials enrolling diabetic population. 39,42 Large-scale studies have failed to show lowering of the risk of heart failure, despite lowering of mortality in patients with hypertension. 37,40,58 Application of calcium-antagonist therapy in concert with ACE-I 58 or β-blockers 37 doesn’t appear to mitigate the risk of heart failure. Placebo-controlled trials of IA-dCCBs have failed to show lowering of the risk of heart failure, 39,49,59,60 suggesting the effect on heart failure to be a BP-independent class effect resulting primarily from cardiomyocyte apoptosis and inhibition of calcium flux within cells. 61,62
Reduction in carotid intima–media thickness, 3 -5 left ventricular hypertrophy regression, 7 differential efficacy on central BP reduction and BP variability, 48,49 and antiinflammatory and antioxidant effects 6 could explain differential effects of antihypertensive drug classes on stroke protection beyond that 6 of office SBP reduction. The CCBs and ARBs offer greater protection against stroke compared to alternative antihypertensives. 22 Our results provide further confirmation of this and also suggest that amlodipine may be superior to ARBs (RR: 0.86, [95% CI, 0.75-0.99], P = .035) against stroke protection which may be attributed, at least partly, to its superior antihypertensive effects. 45
Rapid dropping of BP provokes reflex sympathetic activation that increases heart rate and left ventricular contractility, while coronary vasodilation diverts blood flow from the coronary bed. The ensuing mismatch of coronary demand and supply may drive the risk of myocardial ischemic events. Initial concern regarding the risk of AMI associated with CCBs 63 was addressed by Opie et al who suggested that nifedipine increased the risk of fatal and nonfatal MI in patients with old MI, while non-dCCBs verapamil and diltiazem were associated with reduced risk of MI 64 highlighting the heterogeneous nature of CCBs. Our results suggest similar conclusions, where IA-dCCBs are associated with higher risk of AMI, while Amlodipine appears comparable to alternative antihypertensive agents.
Some of the older placebo-controlled studies suggested that IA-dCCBs may lower the risk of major adverse cardiovascular outcomes except for heart failure. 39 Interpretation of the results of these studies requires caution as the large interarm differences in SBP reduction favoring IA-dCCBs in these trials would be expected to significantly affect the cardiovascular outcomes. These studies argue against a higher risk of major adverse cardiovascular outcomes with IA-dCCBs as opposed to a lower risk with alternative antihypertensive drugs. This suggests the possibility that IA-dCCBs may be used as an add-on antihypertensive agent in patients whose BP remains inadequately controlled, despite adequate doses of major alternative antihypertensive drugs, namely, diuretics, β-blockers, ACE Is, and ARBs.
Study Limitations
As a meta-analysis based on published summary statistics, the degree of standardization achieved was lower than quantitative overviews based on individual patient data. So the participants’ characteristics may have influenced the estimates of effect size and the definition and validation of end points in individual trials. The specific cardiovascular end points definition varied slightly across the individual trials. This is of greater concern for heart failure, since peripheral edema, which is a side effect of dCCBs, may be mistaken for heart failure, while β-blockers and diuretics may mask the symptoms of heart failure. Drawbacks pertinent to the current study include (1) the results are based on indirect comparisons and hence should be interpreted with caution; (2) due to the paucity of published data, precise effect size against the individual comparator antihypertensive drug classes and in different patient subpopulations, such as diabetics and chronic kidney disease patients, could not be estimated; (3) some of the sensitivity analysis and drug group comparisons were inadequately powered; and (4) the effect of dCCBs on SBP and DBP reduction could not be estimated, since most trials did not report standard deviation for BP values. (5) although multiple studies were included in the analysis, 3 studies contributed heavily to the analysis (ALLHAT and VALUE for LA-dCCB and STOP-2 for IA-dCCB). Limitations of these studies should be considered while interpreting our results. There was a significant difference in SBP favoring amlodipine in VALUE. STOP-2 was an older, open-label trial that was not designed to compare individual drugs. In ALLHAT, African American patients were prescribed ACE-I particularly in combination with β-blockers, and heart failure was a secondary end point that was not well validated. 65
Conclusions
Collectively, our results suggest that IA-dCCB is inferior to alternative antihypertensive drugs as first-line agents in reducing the risks of major complications of hypertension. Application of dCCB therapy in patients at high risk of heart failure should require caution. Our data support preferential use of LA-dCCB amlodipine over IA-dCCBs when dCCBs are to be applied for hypertension therapy.
Supplemental Material
Supplemental Material, Suppl_table_1 - Effects of Long- and Intermediate-Acting Dihydropyridine Calcium Channel Blockers in Hypertension: A Systematic Review and Meta-Analysis of 18 Prospective, Randomized, Actively Controlled Trials
Supplemental Material, Suppl_table_1 for Effects of Long- and Intermediate-Acting Dihydropyridine Calcium Channel Blockers in Hypertension: A Systematic Review and Meta-Analysis of 18 Prospective, Randomized, Actively Controlled Trials by Sandip Chaugai, Lhamo Yangchen Sherpa, Amir Ali Sepehry, Scott Reza Jafarian Kerman, and Hisatomi Arima in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
Chaugai, Sandip and Arima, Hisatomi contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Sherpa, Lhamo Yangchen contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy, Kerman, Scott Rezza Jafarian contributed to acquisition, analysis, and interpretation, drafted manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Sepehry, Amir contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Novelty
Higher risk of AMI and lack of protection against stroke with IA-dCCB. Higher risk of heart failure with amlodipine and IA-dCCB. Diabetes modestly increases the risk heart failure in hypertensives taking dCCB.
Relevance
Amlodipine is one of the most widely used antihypertensive medication. There is a growing interest in IA-dCCB drug class, especially the delayed-release formulations although the results of clinical studies remain conflicting. Our data suggest preferential application of amlodipine may provide clinical benefit comparable to alternative drug classes that greatly exceeds the therapeutic efficacy of IA-dCCB.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
