Abstract
Background:
Impact of atrial fibrillation on clinical outcomes is well recognized, and application of renin–angiotensin–aldosterone system (RAAS) blockers for the prevention of atrial fibrillation (AF) is a theoretically appealing concept. However, clinical trials have yielded inconsistent results.
Methods:
A pooled study of 26 randomized controlled trials (RCTs) assessing the efficacy of RAAS blockers on AF prophylaxis was performed.
Results:
A total of 28 reports from 26 randomized controlled trials enrolled 165 387 patients, with an overall 24% reduction in the incidence of AF (odds ratio [OR]: 0.76, 95% confidence interval [CI]: 0.68-0.85], P = .000). Forty-nine percent reduction in the incidence of AF (OR: 0.51, 95% CI: 0.30-0.85, P = .010) in systolic heart failure was observed, whereas no significant effect was observed in patients with diastolic heart failure, postmyocardial infarction, and high cardiovascular disease risk. There was a 19% (OR: 0.81, 95% CI: 0.67-1.00, P = .037) reduction in new-onset and 54% (OR: 0.46, 95% CI: 0.33-0.62, P = .000) reduction in recurrent AF in hypertensive patients with 39% (OR: 0.61, 95% CI: 0.44-0.84, P = .003) risk reduction against calcium blockers and 41% (OR: 0.59, 95% CI: 0.44-0.80, P = .001) risk reduction against β blockers. Angiotensin-receptor blocker appeared marginally superior to angiotensin-converting enzyme inhibitor in primary and secondary prevention.
Conclusion:
This study suggests that RAAS blockade effectively suppresses AF in systolic heart failure, and hypertensives derive greater benefit against new-onset and recurrent AF compared to β blockers, calcium channel blockers, and diuretics.
Keywords
Background
Impact of atrial fibrillation (AF) on major adverse clinical outcomes including mortality, heart failure, and stroke in participants with heart failure, myocardial infarction (MI), hypertension, or high cardiovascular disease (CVD) risk factors is well recognized, and prevention of AF in these patient populations is highly desirable. 1 –3 Ability of antihypertensive drugs such as renin–angiotensin–aldosterone system (RAAS) blockers, β blockers (BBs), and calcium channel blockers (CCBs) to suppress AF has been an area of high interest. Among these, RAAS blockers appear to be the most appealing therapeutic agents based on our current understanding of the proarrhythmic effects of RAAS on AF onset, progression, and persistence. 4 Clinical trials have yielded inconsistent results with some showing impressive effects of RAAS blockade (Losartan Intervention For End Point Reduction in Hypertension [LIFE]), whereas others failing to show any benefit (Atrial Fibrillation Clopidogrel Trial With Irbesartan for Prevention of Vascular Events [ACTIVE-I] and Gruppo Italiano per lo Studio della Sopravvivenza nellʹInfarto Miocardico, Atrial Fibrillation [GISSI-AF]) on AF prevention. Major antihypertensive drug classes, especially RAAS blockers and BBs, remain the cornerstone of management of heart failure and MI, irrespective of their role in primary prevention of AF. From clinical perspective, knowledge of comparative efficacy of these drugs against AF prophylaxis in hypertensives is essential to curb the excess morbidity and mortality incited by AF before the onset of heart failure or MI. This is especially important in hypertensives with AF as hypertension is present in almost two-third of patients with AF and is associated with adverse clinical outcomes. 5 Further, a distinction between systolic and diastolic heart failure is essential considering their differential therapeutic response to RAAS blockers. These remain unexplored in previous meta-analysis. Further, disappointing results of recent a priori designed secondary prevention trials challenge our understanding of secondary AF prevention by RAAS blockade, especially in the absence of concomitant antiarrhythmic drug (AAD) therapy.
Methods
Data Sources and Search Strategy
A systematic review and meta-analysis was performed in accordance with the standards set forth by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 6,7 Electronic databases PubMed, EMBASE, and Cochrane Collaboration of Controlled Clinical Trials registery were searched using the key words “beta blockers,” “calcium channel blockers,” “angiotensin,” “angiotensin converting enzyme inhibitors,” “angiotensin receptor blockers,” “individual names in these drug classes,” “atrial fibrillation,” “ atrial arrhythmia,” “hypertension,” “heart failure,” and “myocardial infarction.” Additionally, references of retrieved articles and previous meta-analysis were manually searched to identify studies not captured by our primary search strategy. No restrictions were imposed on dates of publication, but the search was restricted to trials published in English. The final search was run on December 1, 2014.
Study Selection
Randomized controlled trials (RCTs) of RAAS blockers against AF prophylaxis in patients with heart failure, MI, hypertension, or CVD risk factors were screened. Exclusion criteria were uncontrolled studies, application of AAD or electrical cardioversion as part of the treatment protocol in the trial, enrollment of specialized patient populations such as postcardiac surgery, case reports, reviews, studies not reporting the outcome of interest, and no access to full text for quality assessment and data extraction. Studies were categorized into “heart failure” enrolling participants with systolic and/or diastolic heart failure, “post MI” enrolling patients after acute MI, “high CVD risk” enrolling participants with traditional CVD risk factors, and “hypertension” restricted to trials enrolling all participants with blood pressure (BP) >140/90 or on antihypertensive therapy.
Data Extraction and Quality Assessment
Data were extracted in duplicate by 2 independent reviewers (Drs Chaugai and Meng), and disagreements were resolved by consensus. The methodological quality of each trial was evaluated for risk of bias using standard criteria: random sequence generation; allocation concealment; blinding of participants, personnel, and outcome assessor; incomplete outcome ascertainment; selective reporting; and other potential sources of bias as recommended by the Cochrane Collaboration (Table 1).
Risk of Bias Across Studies Included in the Meta-Analysis.
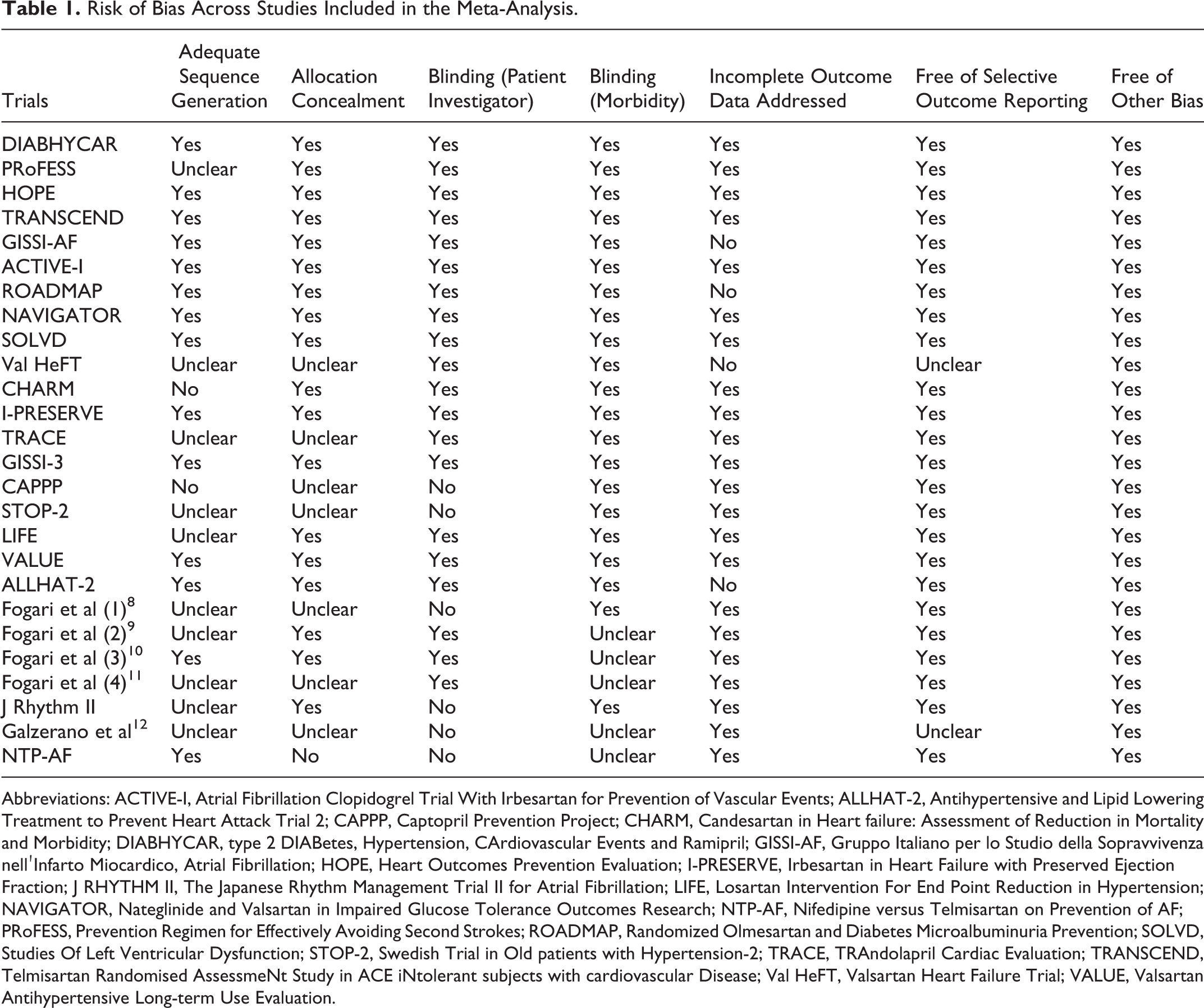
Abbreviations: ACTIVE-I, Atrial Fibrillation Clopidogrel Trial With Irbesartan for Prevention of Vascular Events; ALLHAT-2, Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial 2; CAPPP, Captopril Prevention Project; CHARM, Candesartan in Heart failure: Assessment of Reduction in Mortality and Morbidity; DIABHYCAR, type 2 DIABetes, Hypertension, CArdiovascular Events and Ramipril; GISSI-AF, Gruppo Italiano per lo Studio della Sopravvivenza nellʹInfarto Miocardico, Atrial Fibrillation; HOPE, Heart Outcomes Prevention Evaluation; I-PRESERVE, Irbesartan in Heart Failure with Preserved Ejection Fraction; J RHYTHM II, The Japanese Rhythm Management Trial II for Atrial Fibrillation; LIFE, Losartan Intervention For End Point Reduction in Hypertension; NAVIGATOR, Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research; NTP-AF, Nifedipine versus Telmisartan on Prevention of AF; PRoFESS, Prevention Regimen for Effectively Avoiding Second Strokes; ROADMAP, Randomized Olmesartan and Diabetes Microalbuminuria Prevention; SOLVD, Studies Of Left Ventricular Dysfunction; STOP-2, Swedish Trial in Old patients with Hypertension-2; TRACE, TRAndolapril Cardiac Evaluation; TRANSCEND, Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease; Val HeFT, Valsartan Heart Failure Trial; VALUE, Valsartan Antihypertensive Long-term Use Evaluation.
Data Synthesis and Statistical Analysis
The measurement data were pooled across studies and analyzed using random-effects meta-analysis model with inverse variance weighting. These are presented as odds ratios (ORs) with 95% confidence intervals (CIs). The magnitude of heterogeneity present was estimated using the I 2 statistic, an estimate of the proportion of the total observed variance that is attributed to the “between-study variance”. Random-effects meta-regression analysis was performed to identify potential effect modifiers. All P values are 2 tailed with the statistical significance set at .05. All statistical analyses were performed using STATA software 12.0.
Results
Study Selection
The flow diagram of study selection is shown in Figure 1. Of the initial 4739 hits, 113 articles were retrieved for detailed evaluation for potential inclusion. Tables 2 and 3 summarize the design and baseline characteristics of the included studies. Twenty-eight reports from 26 RCTs (2 reports from LIFE and Candesartan in Heart failure: Assessment of Reduction in Mortality and Morbidity [CHARM] trials each) enrolled a total of 165 387 patients with 75 899 in the RAAS blockers arm and 89 488 in the control arm followed up for an average of 1 to 6 years. Fourteen placebo controlled trials, 4 heart failure, 21 –24 2 post-MI, 25,26 and 8 high CVD risk, 13 –20 and 13 reports from 12 actively controlled trials in hypertensive participants 3,8 –12,27 –33 were included in the meta-analysis. Eight trials used angiotensin-converting enzyme inhibitor (ACE I), 13,15,21,25 –28,31 17 trials used angiotensin-receptor blocker (ARB), 3,8,11,12,14,16 –20,22 –24,29,30,32,33 and 2 trials were designed with parallel ACE I and ARB arms. 9,10 Table 4 summarizes the results of the meta-analysis.
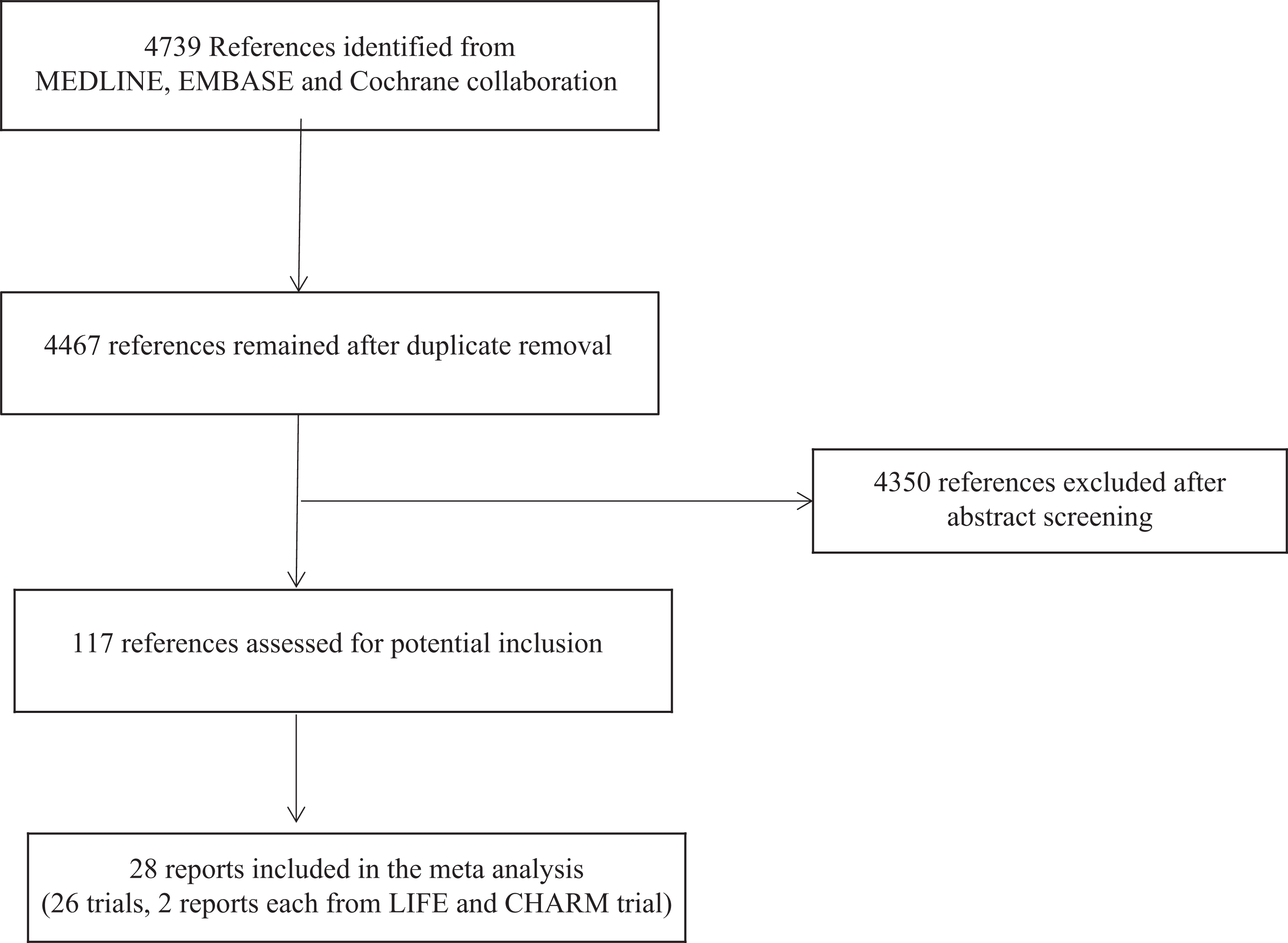
Search flow diagram for studies included.
Basic Characteristics of the Included Studies.a

Abbreviations: ACTIVE-I, Atrial Fibrillation Clopidogrel Trial With Irbesartan for Prevention of Vascular Events; AF, atrial fibrillation; ALLHAT-2, Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial 2; BB, β blocker; CAPPP, Captopril Prevention Project; CCB, calcium channel blocker; CHARM, Candesartan in Heart failure: Assessment of Reduction in Mortality and Morbidity; CVD, cardiovascular disease; DD, diastolic dysfunction; DIABHYCAR, type 2 DIABetes, Hypertension, CArdiovascular Events and Ramipril; DM, diabetes mellitus; ECG, electrocardiogram; GISSI-AF, Gruppo Italiano per lo Studio della Sopravvivenza nellʹInfarto Miocardico, Atrial Fibrillation; HM, Holter monitoring; HOPE, Heart Outcomes Prevention Evaluation; HTN, hypertension; IGT, impaired glucose tolerance; I-PRESERVE, Irbesartan in Heart Failure with Preserved Ejection Fraction; J RHYTHM II, The Japanese Rhythm Management Trial II for Atrial Fibrillation; LIFE, Losartan Intervention For End Point Reduction in Hypertension; LVDD, left ventricular diastolic dysfunction; LVH, left ventricle hypertrophy; LVSD, left ventricular systolic dysfunction; MI, myocardial infarction; MS, multiple sclerosis; NA, not available; NAVIGATOR, Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research; NTP-AF, Nifedipine versus Telmisartan on Prevention of AF; PRoFESS, Prevention Regimen for Effectively Avoiding Second Strokes; ROADMAP, Randomized Olmesartan and Diabetes Microalbuminuria Prevention; SOLVD, Studies Of Left Ventricular Dysfunction; STOP-2: Swedish Trial in Old Patients with hypertension-2; TRACE, TRAndolapril Cardiac Evaluation; TRANSCEND, Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease; TTM, transtelephonic monitoring; Val HeFT, Valsartan Heart Failure Trial; VALUE, Valsartan Antihypertensive Long-term Use Evaluation.
a Values are mean or percentage.
Baseline Characteristics of Trial Participants.a

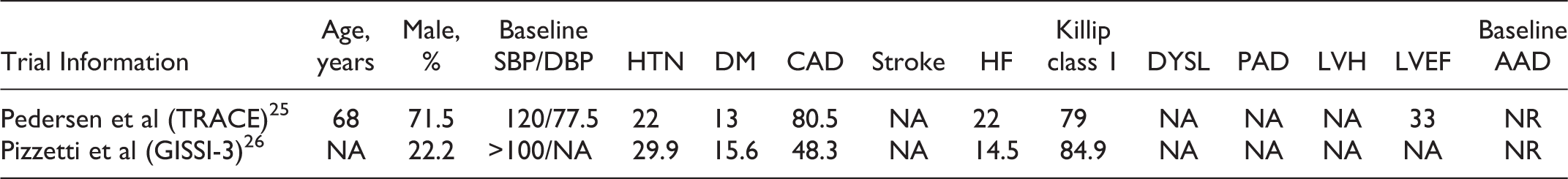

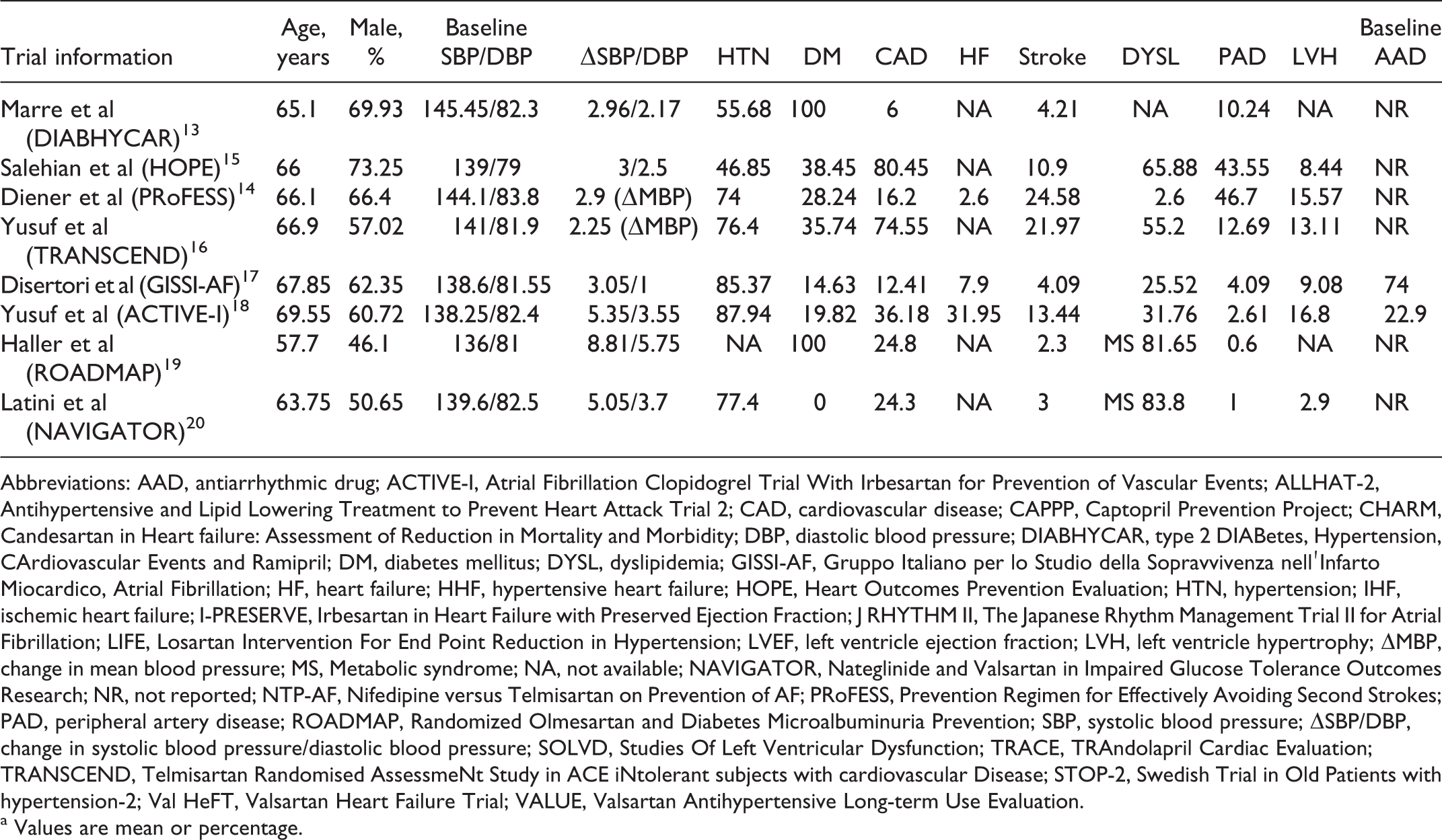
Abbreviations: AAD, antiarrhythmic drug; ACTIVE-I, Atrial Fibrillation Clopidogrel Trial With Irbesartan for Prevention of Vascular Events; ALLHAT-2, Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial 2; CAD, cardiovascular disease; CAPPP, Captopril Prevention Project; CHARM, Candesartan in Heart failure: Assessment of Reduction in Mortality and Morbidity; DBP, diastolic blood pressure; DIABHYCAR, type 2 DIABetes, Hypertension, CArdiovascular Events and Ramipril; DM, diabetes mellitus; DYSL, dyslipidemia; GISSI-AF, Gruppo Italiano per lo Studio della Sopravvivenza nellʹInfarto Miocardico, Atrial Fibrillation; HF, heart failure; HHF, hypertensive heart failure; HOPE, Heart Outcomes Prevention Evaluation; HTN, hypertension; IHF, ischemic heart failure; I-PRESERVE, Irbesartan in Heart Failure with Preserved Ejection Fraction; J RHYTHM II, The Japanese Rhythm Management Trial II for Atrial Fibrillation; LIFE, Losartan Intervention For End Point Reduction in Hypertension; LVEF, left ventricle ejection fraction; LVH, left ventricle hypertrophy; ΔMBP, change in mean blood pressure; MS, Metabolic syndrome; NA, not available; NAVIGATOR, Nateglinide and Valsartan in Impaired Glucose Tolerance Outcomes Research; NR, not reported; NTP-AF, Nifedipine versus Telmisartan on Prevention of AF; PRoFESS, Prevention Regimen for Effectively Avoiding Second Strokes; PAD, peripheral artery disease; ROADMAP, Randomized Olmesartan and Diabetes Microalbuminuria Prevention; SBP, systolic blood pressure; ΔSBP/DBP, change in systolic blood pressure/diastolic blood pressure; SOLVD, Studies Of Left Ventricular Dysfunction; TRACE, TRAndolapril Cardiac Evaluation; TRANSCEND, Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease; STOP-2, Swedish Trial in Old Patients with hypertension-2; Val HeFT, Valsartan Heart Failure Trial; VALUE, Valsartan Antihypertensive Long-term Use Evaluation.
a Values are mean or percentage.
Summary of the Results of Meta-Analysis.
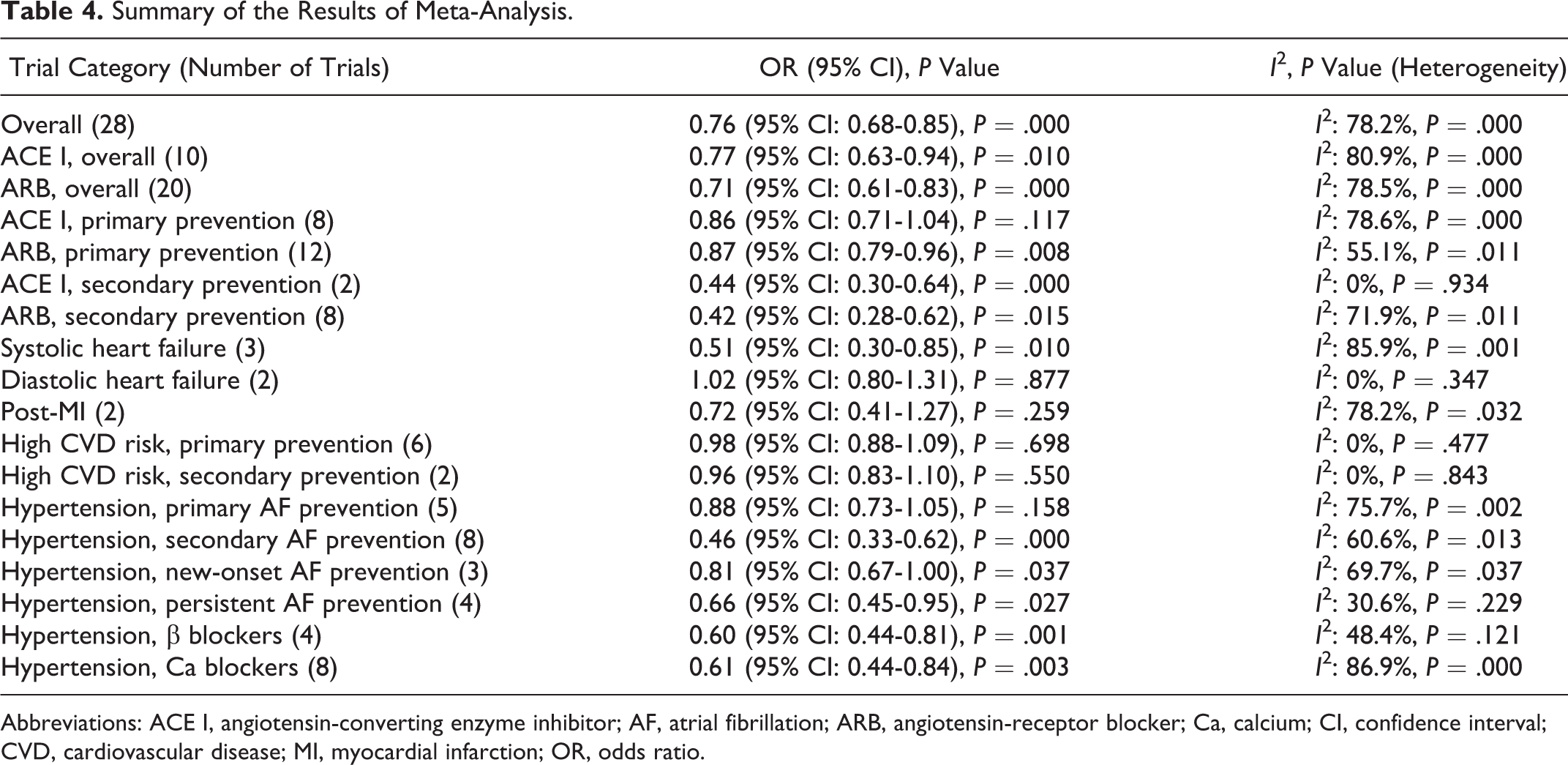
Abbreviations: ACE I, angiotensin-converting enzyme inhibitor; AF, atrial fibrillation; ARB, angiotensin-receptor blocker; Ca, calcium; CI, confidence interval; CVD, cardiovascular disease; MI, myocardial infarction; OR, odds ratio.
Incidence of AF
Overall, there was a 24% reduction in the incidence of AF (OR: 0.76, 95% CI: 0.68-0.85, P = .000; Figure 2). The degree of heterogeneity in the treatment effect was I 2 = 78.2% and P = .000. Post hoc exclusion of all the trials with a sample size less than 500 decreased the observed treatment effect to 10% (OR: 0.90, 95% CI: 0.83-0.97, P = .008; Supplementary Figure 1). The degree of heterogeneity in the treatment effect also declined to I 2 = 55.8% and P = .002. Further subgroup analysis was performed considering the significant variation in effect size between trial groups, primary and secondary AF prevention, and the experimental therapy used, that is, ACE I and ARB.

Effect of renin–angiotensin–aldosterone system (RAAS) inhibition on atrial fibrillation (AF) prevention.
Comparison between ACE I and ARB
Overall, ACE I therapy was associated with 23% reduction in the incidence of AF (OR: 0.77, 95% CI: 0.63-0.94, P = .010) and ARB therapy was associated with a 29% reduction in the incidence of AF (OR: 0.71, 95% CI: 0.61-0.83, P = .000; Supplementary Figure 2). In primary prevention, ACE I therapy showed a modest trend toward fewer AF (OR: 0.86, 95% CI: 0.71-1.04, P = .117), whereas ARB therapy was associated with a 13% reduction in the incidence of AF (OR: 0.87, 95% CI: 0.79-0.96, P = .015; Figure 3). In secondary prevention, ACE I caused 56% risk reduction (OR: 0.44, 95% CI: 0.30-0.64, P = .000), whereas ARB caused 58% reduction in the incidence of recurrent AF (OR: 0.42, 95% CI: 0.28-0.62, P = .015; Figure 3).

Comparison of angiotensin-converting enzyme inhibitor (ACE I) and angiotensin-receptor blocker (ARB) on primary and secondary atrial fibrillation (AF) prevention.
Placebo-Controlled Trials
Systolic heart failure
Renin–angiotensin–aldosterone system blocker therapy was associated with a 49% reduction in the incidence of AF (OR: 0.51, 95% CI: 0.30-0.85, P = .010; Figure 2). The degree of heterogeneity in the treatment effect was I 2 = 85.9% and P = .001. Post hoc exclusion of the smallest trial Studies Of Left Ventricular Dysfunction (SOLVD) that enrolled participants with lowest mean left ventricle ejection fraction (LVEF) and showed the largest risk reduction decreased the heterogeneity to 30.1% with an accompanying decline in observed treatment effect to 31% (Figure 2).
High CVD risk, diastolic heart failure, and post-MI
No significant effect of RAAS blocker therapy was observed in patients with high CVD risk (OR: 0.97, 95% CI: 0.89-1.06, P = .505), diastolic heart failure (OR: 1.02, 95% CI: 0.80-1.31, P = .877), or post-MI (OR: 0.72, 95% CI: 0.41-1.27, P = .259), albeit only 2 trials were available in each of the last 2 categories.
Actively Controlled Trials
A total of 13 reports from 12 trials were included in the analysis, with 8 reporting AF recurrence, 3 new-onset AF, and 2 total AF. Overall, RAAS blocker therapy was associated with a 37% reduction in the incidence of AF (OR: 0.63, 95% CI: 0.50-0.78, P = .000; Figure 2). The degree of heterogeneity in the treatment effect was I 2 = 83.7% and P = .000. In fixed-effects model, RAAS blocker therapy was associated with a 23% reduction in the incidence of AF (OR: 0.77, 95% CI: 0.72-0.84, P = .000).
Subgroup Analysis
Drug class
Beta-blocker trials
Renin–angiotensin–aldosterone system blocker therapy was associated with a 40% reduction in the incidence of AF (OR: 0.60, 95% CI: 0.44-0.81, P = .001; Figure 4). No significant heterogeneity was observed (I 2 = 48.4% and P = .121).

Effect of renin–angiotensin–aldosterone system (RAAS) blockers against calcium blockers and β blockers on atrial fibrillation (AF) prevention in hypertension.
Calcium channel blocker trials
Overall, RAAS blocker therapy was associated with a 39% reduction in the incidence of AF (OR: 0.61, 95% CI: 0.44-0.84, P = .003; Figure 4). The degree of heterogeneity in the treatment effect was I 2 = 86.9% and P = .000. Post hoc exclusion of The Japanese Rhythm Management Trial II for Atrial Fibrillation (J RHYTHM II), which reported only the incidence rate of persistent AF, did not affect the results.
Primary versus secondary (recurrent) AF prevention in hypertension
Renin–angiotensin–aldosterone system blocker therapy showed a trend toward protection in primary prevention trials (OR: 0.88, 95% CI: 0.73-1.05, P = .158; Figure 2) with significant heterogeneity (I 2 = 75.7% and P = .002). Limiting the analysis to trials reporting new-onset AF alone, 19% reduction in the incidence of new-onset AF (OR: 0.81, 95% CI: 0.67-1.00, P = .037; Figure 5) without significant heterogeneity (I 2 = 69.7% and P = .037) was noted. Renin–angiotensin–aldosterone system blocker therapy was associated with a 54% reduction in the incidence of recurrent AF in secondary prevention trials (OR: 0.46, 95% CI: 0.33-0.62, P = .000). The degree of heterogeneity in the treatment effect was I 2 = 60.6% and P = .013.

Effect of renin–angiotensin–aldosterone system (RAAS) blockade on new onset and recurrent atrial fibrillation (AF) in hypertension.
Persistent AF in hypertension
Renin–angiotensin–aldosterone system blocker therapy was associated with a 34% reduction in the incidence of persistent AF (OR: 0.66, 95% CI: 0.45-0.95, P = .027). No significant heterogeneity was observed (I 2 = 30.6% and P = .229; Figure 6).

Effect of renin–angiotensin–aldosterone system (RAAS) blockade on progression to persistent atrial fibrillation (AF) in hypertension.
Discussion
The results of the current meta-analysis based on 165 387 patients suggested that RAAS blockade results in a statistically significant 24% reduction in the risk of AF. Participants with systolic dysfunction appear to derive the greatest benefit with 49% reduction in the risk of AF, whereas the pooled estimates from high CVD risk, diastolic heart failure, and post-MI trials failed to show evidence of therapeutic benefit. Overall, 37% reduction in the risk of AF was noted in hypertension with 54% risk reduction in recurrent AF prevention and 19% risk reduction against new-onset AF. Overall, RAAS blockers were associated with 40% lower risk of AF against BB and 39% against CCB in hypertensive patients. Further, 34% lower risk of progression to persistent AF was noted in hypertensive patients.
Placebo-Controlled Trials
Heart failure
Significant protection against AF was consistently observed in systolic heart failure in all the trials, whereas CHARM-PRESERVED and Irbesartan in Heart Failure with Preserved Ejection Fraction (I-PRESERVED) yielded inconclusive results in participants with diastolic dysfunction. Trend toward lower AF recurrence and lower hospitalization rates for heart failure was observed in GISSI-AF and ACTIVE-I, respectively. Similarly, in a placebo-controlled study of 30 participants with congestive heart failure having chronic AF, lisinopril produced greater maintenance of sinus rhythm and improved mean peak oxygen consumption after 6 weeks of therapy. 34
Post-MI
Both the placebo-controlled trials enrolling post-MI patients favored RAAS blockers over placebo. The TRAndolapril Cardiac Evaluation (TRACE) was the smaller study with a longer follow-up (2-4 years) showing a significant protection in participants with left ventricular systolic dysfunction (LVSD), whereas GISSI-3 was the larger study with 6 weeks of follow-up that failed to detect a statistically significant effect. Interestingly, nitrates were associated with a greater reduction in AF compared to lisinopril, and lisinopril and nitrate combination was associated with the greatest risk reduction of 26% compared to control. This likely represents AF protection resulting predominately from hemodynamic restoration rather than anticardiac remodeling effects in the early post-MI phase.
High CVD risk
Both the a priori designed secondary prevention trials enrolling participants with AF and CVD risk factors, ACTIVE-I and GISSI-AF, failed to detect any significant effect of RAAS blockade. It is noteworthy that baseline ACE I and AAD therapy was very high, and mean BP was <138/80, despite the enrollment of a very a high number of hypertensive participants (88% in ACTIVE-I and 81% in GISSI-AF). Further, the majority of participants had advanced AF (persistent and permanent AF) where RAAS blockade may no longer be effective. Similarly, angiotensin II-antagonist in paroxysmal atrial fibrillation (ANTIPAF), another a priori designed placebo-controlled secondary prevention trial in participants without structural heart disease, failed to detect a beneficial effect of RAAS blockade except for an earlier prescription of AAD in placebo arm. 36 The Randomized Olmesartan and Diabetes Microalbuminuria Prevention (ROADMAP) was the only primary prevention trial reporting a composite of new-onset AF or transient ischemic attack as the primary end point but failed to detect a statistically significant difference probably due to low event rates. It is noteworthy that the mean BP in all these trials was <140/90, despite a high prevalence of hypertension indicating the participants enrolled had well-controlled hypertension, and the low event rates in these trials further limited their power to detect modest differences in effect size.
Actively Controlled Trials
Overall, RAAS blocker therapy was associated with a significant reduction in the risk of AF against any active comparator that included BB, CCB, diuretics, or a combination of BB and diuretics collectively classified as conventional treatment. This effect was evident despite a statistically lower BP reduction with RAAS blockers in Captopril Prevention Project (CAPPP), Valsartan Antihypertensive Long-term Use Evaluation (VALUE), Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), J RHYTHM II, and Nifedipine versus Telmisartan on Prevention of AF (NTP-AF). These results are further supported by several recent nested case–control studies that have consistently reported a lower risk of AF in hypertensives receiving RAAS blockers compared to alternative antihypertensives. 35,37 –39 These findings lend further support to the notion that RAAS blockers exert BP-independent effects against AF prophylaxis. Angiotensin-receptor blocker displayed more consistent effect on AF suppression than ACE I, albeit the number of trails was limited.
High CVD risk trials failed to show a significant incremental benefit of RAAS blockade on AF risk in normotensives or uncomplicated and well-controlled hypertension, and the discrepancy between the results in hypertension and high CVD risk trials is surprising. Several factors could account for this such as intertrial variations in inclusion criteria among high CVD risk trials with inclusion of very low number of hypertensive patients in some trials, differences in trial design, and lack of AF prespecification. Further, hypertension trials, unlike high CVD risk trials, required discontinuation of previous antihypertensive medications followed by a washout period with subsequential addition of prespecified antihypertensives as needed for BP control. The only exception to this was J RHYTHM II that allowed continuation of previous antihypertensive medications and ALLHAT II that lacked a washout period. This raises the possibility that the expected modest risk reduction was masked by the background therapy with multiple antihypertensives. Finally, AF was not a prespecified end point in these trials.
Primary Prevention
Pooled estimate from all the published trials showed a modest trend toward protection with significant heterogeneity observed between trials. Two of the more recent ARB trials with AF prespecified as an end point, LIFE and VALUE, demonstrating significant effect of RAAS blockade had more stringent BP control unlike the older trials using ACE I, CAPPP, and Swedish Trial in Old Patients with Hypertension-2 (STOP-2), whereas the inadequately powered post hoc analysis of ALLHAT II failed to detect significant effect of RAAS blockade. All the trials favored RAAS blockers, with the single exception of the smallest trial STOP-2 that enrolled an elderly cohort (>75 years) of predominately females (67%) with a lower prevalence of CVD risk factors. Of note, the baseline BP was very high (194/98) and BP reduction was inadequate, at least by current standards, with systolic blood pressure ≥ 160 mm Hg throughout follow-up until the end of the study. Systolic blood pressure of 160 mm Hg or greater is associated with significant risk of AF with progressive lowering of the risk with BP lowering. 39 Further, baseline AF was high and unequally distributed (RAAS blocker 5.3% vs CCB 4.1% vs conventional therapy 4.7%), and the analysis did not exclude participants with baseline AF. Conventional therapy was favorable over both RAAS blocker and CCB for AF prophylaxis, although this did not reach statistical significance. This contradicts with other studies. In the post hoc analysis of Systolic Hypertension in the Elderly Program, Pilot Study (SHEP-PS), conventional therapy failed to reduce AF onset against placebo, whereas CCB and ACE I showed trends toward protection against conventional therapy in NORdic DILtiazem Study (NORDIL) and CAPPP, respectively. 40,41 This poses question to the generalization of these results. The VALUE was the largest trial demonstrating 24% and 16% risk reduction during the first 3 years and at 6 years, respectively. Interestingly, pooled estimate from 3 recent large trials enrolling 47 943 patients suggested a statistically significant 20% reduction in new-onset AF. These data strongly suggest that application of RAAS blockers in hypertensive patients could provide a greater protection against AF.
Secondary Prevention
Five trials reported a significantly lower recurrence rate against BB 3,8,12 and CCB 9 –11 in hypertensives with paroxysmal AF without overt cardiovascular disease. However, 2 recent studies, NTP-AF and J RHYTHM II, showed modest trends toward protection, although RAAS arm had a statistically significant lesser BP reduction in these 2 trials. Still, NTP-AF noted a significant reduction of 33% in progression to persistent AF and a modest trend toward lower total AF recurrence, and J Rhythm II observed a trend toward lower persistent AF. However, in J Rhythm II, RAAS arm had significantly higher LVEF and diuretic use at baseline and allowed the use of AAD with a trend toward higher use of AAD in CCB arm at baseline and throughout the follow-up. Further, it was the only trial under this category that allowed continuation of previous antihypertensive therapy (>75%) and observed lowest BP reduction. However, failure of ACTIVE-I, GISSI-AF, ANTIPAF, J RHYTHM II, and modest risk reduction in NTP-AF suggests the need to exercise caution while interpreting the pooled estimates for secondary prevention, and it appears that the presence of comorbidities such as left ventricle hypertrophy (LVH) could be an important determinant of effect size as suggested by LIFE trial.
These studies also provide evidence of benefit of RAAS blockade beyond AF suppression. Renin–angiotensin–aldosterone system blockers are superior anticardiac remodeling agents with the evidence base being stronger for ARB compared to ACE I, whereas BBs are particularly inefficient with CCBs following RAAS blockers. 42 A recent RCT reported a greater regression of LVH in hypertensives, despite lower BP lowering and similar clinical outcomes, with RAAS blocker in direct comparison to CCB. 43 NTP-AF and Galzerano et al reproduced these findings in participants with AF. Our results on AF prevention appear to parallel these findings. Further, RAAS blockade significantly decreased progression to persistent AF with a pooled estimate of 34% from VALUE, NTP-AF, J RHYTHM II, and Galzerano et al, enrolling a total of 14 359 patients. Interestingly, ANTIPAF and J RHYTHM II reported significantly lower use of AAD in RAAS arm.
Mechanistic Implications
Precise mechanisms underlying effects of RAAS blockers on AF suppression remain incompletely understood but may include a combination of its effect on various compensatory mechanisms such as neurohormonal activation, left ventricular (LV) remodeling, left atrial stretch, atrial fibrosis, and direct electrophysiological effects that interact to provide a pathophysiologic substrate for AF. 21 Beneficial effects of RAAS blockade on structural and electrical remodeling have been demonstrated in animal 44 –47 as well as human studies. 48,49 Neurohumoral antagonism and inhibition of structural remodeling suggested by greater regression of LV hypertrophy and left atrial enlargement compared to alternative antihypertensive agents, despite lower BP reduction, is an important BP-independent mechanism. 42,43,50 Renin–angiotensin–aldosterone system blockade attenuates atrial fibrosis with protection against AF in experimental heart failure induced by rapid ventricular pacing. 44 Clinical studies have provided direct biochemical evidence supporting important roles of RAAS blockers in collagen metabolism such as lowering of serum procollagen type I carboxy-terminal peptide levels, a marker of increased collagen type I synthesis and myocardial fibrosis, 9,10 and increased serum carboxy-terminal telopeptide of collagen type I, a marker of extracellular collagen type I degradation. 10 Direct electrophysiological effects such as attenuation of shortening of the atrial refractory period during rapid atrial pacing, 45 reduction in P-wave dispersion, a marker of inhomogeneous atrial propagation of sinus impulses, 9,10 and angiotensin II-mediated inhibition of outward potassium currents thought to be involved in AF generation 51 through angiotensin II type 1 receptors located in proximity to potassium channels within the membrane 52 may also add to protective effects of RAAS blockers. Further, left atrial stretch induced by increased atrial pressure may contribute to proarrhythmic effects by shortening of refractory period and lengthening of intra-atrial conduction time 46,47 that can be antagonized by RAAS blockers. 53 Blood pressure optimization especially in hypertensive patients and hemodynamic restoration such as afterload reduction by central BP lowering, 49 lowering of left atrial and ventricular end-diastolic pressures, 53,54 may add to AF protection by attenuation of left atrial enlargement. The fact that RAAS blockade resulted in greater protection against AF despite lower BP reduction (compared to CCBs, BB, and diuretics) in hypertensive patients supports the notion that RAAS blockade offers some BP-independent effects such as its effect on structural and electrical remodeling. Contrasting results in high CVD risk participants suggest that the therapeutic benefit against AF prophylaxis in participants without structural changes such as LVSD and/or LVH results predominately from BP lowering.
Our data on the efficacy of RAAS blockade against AF prophylaxis appear to parallel the effect of RAAS blockade on mortality and cardiovascular morbidity where strong evidence of benefit is observed in patients with systolic heart failure and high-risk MI as compared to patients with diastolic heart failure or low-risk MI. 55 The benefit of RAAS blockade on AF prophylaxis may be expected to depend on the presence of structural and/or functional abnormality, the degree of compensatory RAAS activation, and the underlying mechanisms of arrhythmia generation. Significant lowering of ejection fraction and greater degree of RAAS activation in patients with systolic heart failure and high-risk post-MI could account for superior efficacy of RAAS blockade compared to patients with diastolic heart failure or low-risk post-MI. Additionally, differences in underlying mechanisms of arrhythmia generation could contribute to differential efficacy of RAAS blockade on AF prevention in systolic and diastolic heart failure. Superior efficacy of RAAS blockers in recurrent AF prevention in hypertensive population likely reflects greater efficacy in antagonizing proarrhythmic effects of RAAS once RAAS activation has set in.
Publication Bias
There was no evidence of publication bias among placebo-controlled trials (Figure 7A and B). However, asymmetry of funnel plot and evidence from Egger (P = .018) and Begg (P = .018) tests (Figure 8A and B) suggested the possibility of publication bias among actively controlled trials. This was further explored post hoc. The big difference in effect size between pure hypertension trials (or new-onset AF prevention) and trials designed for recurrent AF prevention, despite comparable BP reduction, likely suggests publication bias. However, additional factors need to be considered as well. First, true difference in effect size between new-onset and recurrent AF prevention between the trials included in the current analysis is likely to contribute to the observed difference, especially considering the difference in precision between the 2 groups of studies. 56 Second, the degree of symmetry found in a funnel plot may depend on the statistic used to measure effect. Odds ratios overestimate the relative reduction, or increase, in risk if the event rate is high. This can lead to funnel plot asymmetry if the smaller trials were consistently conducted in patients at higher risk. 57,58 Substitution of OR with relative risk resulted in a decline of the observed risk reduction from 59% to 46% for smaller studies without significant effect on larger studies. Third, the a priori design with regular and intensive AF monitoring and higher event rate in smaller trials enhances power to detect modest risk reduction. In particular, symptom-based AF monitoring or reporting AF as adverse effect may underestimate true AF burden. This may be greater in BB and CCB arms, which by virtue of their rate-controlling properties may obscure the symptoms. The encouraging results seen in VALUE would have been missed if the analyses were based on adverse effect reporting. 30 Fourth, smaller trials are associated with lower patient dropout and unplanned crossing over between treatment arms that may enhance power. 55

A and B, Begg and Egger test for placebo-controlled trials.

A and B, Begg and Egger test for actively controlled trials.
Meta-Regression
Meta-regression analysis suggested the effect size to be independent of age and sex of the participants. Meta-regression analysis on heart failure and post-MI trials suggested an association between baseline LVEF and effect size. Among hypertensive population, meta-regression analysis suggested an association between effect size and sample size (12 trials) but failed to show an association between proportion of participants with LVH (4 trials: LIVE, VALUE, ALLHAT, and Fogari et al (2)) or left atrial diameter (4 trials: Fogari et al (1-4). 8 –11 Galzerano et al, NTP-AF, and J-Rhythm II) and effect size. However, these analyses are based on aggregate rather than individual patient data, and limited number of trials providing data on LVH and left atrial diameter could be included.
Sources of Heterogeneity
No heterogeneity was observed in high CVD risk trials (primary or secondary AF prevention) and diastolic heart failure trials. Meta-regression suggested LVEF to be a source of heterogeneity among trials enrolling patients with heart failure and post-MI. In primary AF prevention in hypertension, post hoc exclusion of STOP-2 eliminated heterogeneity (I 2 = 59.9, P = .058). In secondary AF prevention and drug group comparisons (BB and CCB) in hypertension, small study effects contributed to heterogeneity.
Limitations and Strengths
Drawbacks pertinent to this study include publication bias, inclusion of post hoc data, and intertrial variations in the mode and frequency of AF detection. A comparison between post hoc reports and trials with prespecified AF failed to detect any difference in effect size (Supplementary Figure 3). Effect of different modes of AF detection on effect size could not be assessed reliably. Current analysis included almost double the number of participants compared with the previous meta-analysis, 59 11 trials accounting for a total of 79 183 new patients were included, and 15 smaller trials with larger effect size deploying cardioversion or AAD therapy as part of treatment protocol were excluded. This narrows the topic and likely minimized the small study effects suggested by an overall 12% decrease in risk reduction. Effect of RAAS inhibition on recurrent AF in the absence of concurrent AAD therapy was analyzed. Most importantly, comparative efficacy of the major antihypertensive drug classes on new-onset and recurrent AF in hypertensive patients was compared, which may help in guiding optimal antihypertensive therapy. Further, LVEF was identified to be an effect modifier through meta-regression studies.
Conclusion
Renin–angiotensin–aldosterone system blocker therapy is associated with a modest but statistically significant reduction in AF onset and/or recurrence, with the greatest benefit consistently observed in heart failure with systolic dysfunction. Evidence from hypertensive patients suggests that RAAS blocker therapy is associated with a lower risk of new-onset and recurrent AF in direct comparison to alternative antihypertensives, specially BBs, and may be considered as the preferential therapeutic option for hypertensives with AF or at high risk of AF. Reduction in AF recurrence, especially progression to persistent AF, reversal of atrial and ventricular remodeling, and potential AAD sparing effects and translation of these into actual clinical benefit seen in the smaller a priori designed trials require further confirmation in large-scale studies. This is important considering the increasing prevalence of AF with the aging of population and the modern pandemic of hypertension and other CVD risk factors. Renin–angiotensin–aldosterone system blockade resulted in improved clinical outcomes in participants with AF in LIFE, ADVANCED, and CHARM 50 trials and in participants with heart failure in ACTIVE-I and GISSI AF. Additional well-designed studies are needed to identify post-MI subpopulations that could benefit from RAAS blockade against AF prevention and translation of AF suppression into actual clinical benefit in patients with hypertension, systolic heart failure, and high risk post-MI. This is suggested by a recent meta-analysis of placebo-controlled trial of BBs failing to detect reduction in mortality or hospitalization for patients with heart failure having AF, despite significant suppression of AF by BBs in patients with heart failure. 60,61 Future studies should be designed taking into consideration the stage and etiology of AF and background medical therapy.
Footnotes
Author Contributions
S. Chaugai contributed to conception and design, acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. W. Yeng Meng contributed to conception and design, analysis, and drafted the manuscript. A. Sepehry contributed to design, analysis, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
The authors acknowledge the assistance of Prajwol Pathak, Department of Internal Medicine, Nassau University Medical Center, NY, USA, in editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
