Abstract
Hyperlipidemia and hypercoagulability states are linked with the increased risks of myocardial infarction (MI). Levosimendan has vasorelaxant and anti-aggregatory properties. The present study evaluated the anti-aggregatory and cardioprotective effects of levosimendan versus cilostazol in high-fat diet (HFD)-fed rats subjected to isoproterenol-induced MI. Rats were assigned to normal, HFD, HFD + isoproterenol, HFD + isoproterenol + cilostazol, and HFD + isoproterenol + levosimendan. The present study investigated the anti-aggregatory effect of both levosimendan and cilostazol and revealed that both drugs attenuated the severity of platelet aggregation. Moreover, both levosimendan and cilostazol revealed effectiveness in attenuating the severity of HFD/isoproterenol-induced myocardial injury as revealed by electrocardiogram signs, apoptotic markers, and histopathological score via counteracting the oxidative stress burden, increments in the expression of inflammatory mediators, and modulating nuclear factor kappa-B (NF-κB) and phosphatidylinositide 3-kinases (PI3K)/protein kinase B (Akt)/ mechanistic target of rapamycin (mTOR) pathway. It was obvious that levosimendan offered more cardioprotective properties than cilostazol. The study showed the relations between hyperlipedemia, hyperaggregability state, and myocardial injury with the modulation of NF-κB and PI3K/Akt/mTOR pathway.
Introduction
Myocardial infarction (MI) is a central presentation of ischemic heart disease (IHD), a clinical disorder arising from the sudden and persistent restriction of the myocardial blood supply and resulting in myocardial necrosis. 1 -3 During ischemia, cardiomyocytes lose their capacity to modify cell homeostasis causing sarcolemmal disruption and death. 4 This process stimulates leukocyte infiltration, which releases the oxygen free radicals. 3,4 Isoproterenol 4-[1-hydroxy-2-(isopropylamino) ethyl]benzene-1, 2-diol hydrochloride is a β-adrenergic agonist reported to create myocardial stress leading to MI if administered in high dosages. 5,6 Isoproterenol rodent model is characterized by oxidative stress, inflammatory cytokines release, and myocardial cell death. 3,6
High-fat diet (HFD)-induced hyperlipidemia is associated with increased platelet hyperaggregability and oxidative stress, leading to IHD. 7 Hyperlipidemia initiates vascular inflammatory reaction, 8 enhances plaque stability, 9 and increases cardiac oxidative stress burden and inflammation, which aggravates cardiomyocyte apoptosis. 10 Cardiomyocyte apoptotic processes are regulated by signaling pathway of phosphatidylinositide 3-kinases (PI3K), protein kinase B (Akt), and mechanistic target of rapamycin (mTOR). 11 Phosphatidylinositide 3-kinases/Akt/mTOR signaling pathway regulates cardiomyocyte apoptosis, preserves, and enhances cardiac function. 12 -15
Cilostazol,6-[4-(1-cyclohexyl-1H-tetrazole-5-yl)butoxy]-3, 4-dihydro-2-(1H) quinolinone is phosphodiesterase type 3 (PDE3) inhibitor and increases cardiac cyclic adenosine monophosphate (cAMP)-dependent protein kinase A. 16 Platelet aggregation induced by collagen and adenosine diphosphate can be inhibited by cilostazol. 17 Also, cilostazol reduces hyperlipidemia and triglyceride content in the aortic atherosclerotic plaques. 18 Cilostazol is also reported to inhibit inflammatory response molecules as nuclear factor kappa-B (NF-κB) and attenuate, in aortic rat tissue, inducible nitric oxide synthase (iNOS) gene expression. 17 Moreover, in patients with ischemia, cilostazol inhibited intercellular adhesion molecule. 19
Levosimendan ({4-[(4R)-4-methyl-6-oxo-1,4,5,6-tetrahydro-3 pyridazinyl] phenyl} hydrazone) is another PDE3 inhibitor, a calcium sensitizer, that does not increase cytosolic calcium, and therefore, it has a fewer arrhythmogenic consequence than other PDE3 inhibitors. The drug has anti-ischemic and vasodilating effects through opening Adenosine Tri Phosphate (ATP)-dependent potassium channel. Therefore, levosimendan can be used during myocardial ischemia. 20 -22 Moreover, levosimendan has anti-inflammatory and antioxidant effects and reduces the circulating inflammatory cytokines and apoptotic mediators. 23
Reports concerning a new strategy for MI treatment by the PDE3 inhibitors such as cilostazol 24 and levosimendan to reduce infarct size 1 are evolving. However, there is less information in the literature regarding the usefulness of levosimendan as antiplatelet in MI. 25,26 The MI-induced apoptosis is regulated by PI3K/Akt/mTOR signaling pathway. 11 The present study explored the mechanistic protective effects of cilostazol and levosimendan on acute post-MI in HFD rat model via promoting PI3K/Akt/mTOR phosphorylation. This pathway may be a potential target to understand the pathophysiology and treatment of IHD.
Materials and Methods
Experimental Animals
Fifty adult albino rats weighing 170 to 182 g were left for acclimatization for 1 week before the start of the study. The animal’s care and handling were done in agreement with the guidelines of the National Institutes of Health (NIH), Maryland, USA and the Suez Canal University, faculty of medicine animal care committee. Rats received HFD, prepared by mixing 2% cholesterol, 0.3% bile salts, and 10% lard with the basal diet for 12 weeks 7 followed by 1-week therapeutic period. Control rats were fed with normal palatable diet (NPD).
Drugs
Isoproterenol hydrochloride, cilostazol, and levosimendan (Sigma-Aldrich Chemical Co, Cairo, Egypt) were supplied as a white crystalline powder, off-white powder, and crystalline powder, respectively, and dissolved in saline. Cilostazol was given twice a day orally by gastric tube at a dose of 10 mg/kg, 27 whereas levosimendan was given intrapretioneal (IP) injection once daily at a dose of 24 μg/kg. 14
Experimental Design and MI Rat Model
The rats (n = 50) were randomly divided into 5 groups, 10 rats each.
Group I: the normal group, rats fed with NPD.
Experimental acute MI was induced by subcutaneous (SC) injection of isoproterenol hydrochloride (85 mg/ kg) in the last 2 successive days. 5 The HFD-fed animals were divided into 4 groups:
Group 2 (HFD control group) received IP injection of normal saline + SC injection with saline 2 mL/kg/d on 2 successive days (isoproterenol vehicle).
Group 3 (isoproterenol control group) received IP injection of normal saline + SC isoproterenol injection on 2 successive days.
Group 4 (cilostazol treated group) received cilostazol orally twice a day + SC isoproterenol injection on 2 successive days.
Group 5 (levosimendan-treated group) received IP injection of levosimendan + SC isoproterenol injection on 2 successive days.
Electrocardiogram and Hemodynamic Monitoring
Rats’ body weights throughout the experiment were measured (Supplemental Figure 1). Rats were monitored by electrocardiogram (ECG), tail noninvasive blood pressure (BP), and pulse monitoring (Supplemental Tables S1 and S2) after 24 hours from the last isoproterenol injection. Research Biopac data acquisition system MP150 (BIOPAC Systems, Inc, CA, USA) was used. Rats were anesthetized using thiopental sodium (40 mg/kg IP). 28 Blood pressure and pulse were measured by a built-in pump that automatically inflates and slowly deflates to detect the BP and pulse. The EL 405 needle electrodes were placed SC, and ECG amplifier was connected and recorded from lead II. Lead II was recorded in which V+ and V− electrodes were connected to the left leg and right arm, respectively, and the ground electrode was connected to the right leg of the rat. The ECG signs of MI were monitored. 29,30
Blood and Tissue Sampling
Blood samples were collected from the anesthetized animals with a cardiac puncture, and then animal killed by exsanguination at the end of the procedure for tissue collection. Blood samples were divided for platelet function assay and serum collection. Serum collection was done from blood samples by lifting it for 30 minutes, then centrifuged (3000g for 15 minutes), and kept at −80°C until use for various biochemical analyses.
Hearts and aorta were removed and washed with phosphate-buffered, ice-cold saline. Left ventricles were divided into 2 parts. The first one was immediately frozen at −80°C for the various biochemical analyses, whereas the other part was processed to perform the histopathological and immunohistochemical assays. Cardiac specimens were dissected, fixed in 10% paraformaldehyde, and embedded in paraffin for histopathological and immunohistochemical examination.
Platelet Function
The aggregation assay was processed as described by Tawfik. 7 Briefly, to prepare platelet-rich plasma (PRP), gathered blood was mixed with 3.8% sodium citrate and centrifuged (160g for 15 minutes) at room temperature. Platelet-rich plasma was collected into plastic tubes, and the remaining blood was centrifuged for 10 minutes to get the platelet-poor plasma (PPP). Platelet count in PRP was acclimated to 5 × 108/mL utilizing PPP. Platelet aggregation was performed after adding 5 µg/mL collagen (Chrono-Log corp, Pennsylvania, USA) utilizing a double-channel aggregometer (Clot 2, SEAC-Radim Company, Florence, Italy) and was assessed by the change in transmitted light. Aggregation was calculated as a percentage of the total aggregate.
Biochemical Analysis
Measurement of serum lipid profile
Serum cholesterol, low-density lipoproteins (LDLs), high-density lipoproteins (HDLs), and triglycerides (TGs) levels were enzymatically measured (Bio Diagnostics kit, Egypt), using spectrophotometer (UV-1601PC, Shimadzu, Kyoto, Japan).
Estimation of cardiac biomarkers
Serum lactate dehydrogenase (LDH) 31 and creatine phosphokinase 32 activities were estimated using spectrophotometer (UV-1601PC, Shimadzu, Japan). Serum creatine kinase-MB (CK-MB) isoenzyme activity was measured using immune-inhibition method. 33
Assessment of aortic and cardiac inflammatory biomarkers and cardiac oxidative stress markers
In aortic homogenates, determination of iNOS, intracellular adhesion molecule (ICAM), and vascular endothelial growth factor (VEGF) levels were assessed by enzyme-linked immunosorbent assay kits, which were supplied by BioSource Europe S.A (Brussels, Belgium).
Tumor necrosis factor α (TNF-α) and interleukin 6 (IL-6) levels were measured in the cardiac homogenates by enzyme-linked immunosorbent assay kits (supplied by R&Dsystems, Inc, Minnesota). Cardiac tissue lipid peroxides malondialdehyde (MDA) 34 and reduced glutathione (GSH) levels 35 were expressed per gram tissue weight.
Determination of cardiac NF-κB and mTOR messenger RNA expression by real-time polymerase chain reaction
RNA was extracted from cardiac tissue homogenate using SV total RNA isolation System (Promega, Madison, Wisconsin) according to manufacturer’s instruction. RNA concentration and purity were measured by an ultraviolet spectrophotometer. The complementary DNA was made from 1 μg RNA using SuperScript III First-Strand Synthesis System as described in the manufacturer’s procedure (#K1621, Fermentas, Waltham, Massachusetts). Then, real-time polymerase chain reaction (RT-PCR) amplification step and analysis were completed by Applied Biosystems software version 3.1 (StepOne). The PCR reaction contained SYBR Green Master Mix (Applied Biosystems, Foster, USA). The gene-specific primer pairs (NF-κB), forward primer 5′-CATTGAGGTGTATTTCACGG-3, reverse primer:5′-GGCAAGTGGCCATTGTGTTC-3. ThemTORforward primer:5′-TTGAGGTTGCTATGACCAGAGAGAA-3′, and reverse primer:5′-TTACCAGAAAGGACACCAGCCAATG-3′). Primers design was done using Gene Runner Software (Hasting Software, Inc, Hasting, New York) from RNA sequences gene bank. Amplification cycles were 2 minutes at 50°, 10 minutes at 95°, and 15 seconds with 40 cycles of denaturation and 10 minutes annealing/extension at 60°. Data from RT-PCR were computed with the v1·7 sequencing program (PE Biosystems, Foster City, California). Relative expression of studied gene messenger RNA (mRNA) was estimated by the comparative (Ct) method. Results were normalized to the β-actin which was used as the control housekeeping gene and reported as fold change over background levels detected in the studied groups.
Determination of PI3K and pAKt gene expression by Western blot
The PI3K and the phosphorylated form of AKt (pAKt) proteins were extracted from tissue homogenates using ice-cold radioimmunoprecipitation assay, usingV3 Western Workflow Complete System (Bio-Rad Hercules, California). To detect the phosphorylated proteins, phosphatase and protease inhibitors were mixed with cardiac tissue homogenates (buffer composition, 0.5 mmol/L phenylmethylsulphonyl fluoride, 50 mmol/L sodium vanadate, 2 mg/mL aprotinin, and 0.5 mg/mL leupeptin). The mixture of each sample was then centrifugated (4000g for 20 minutes), and the protein concentration was determined using Bradford assay. Equal amounts of protein (20-30 µg of total protein) were separated by sodium dodecyl sulfate/polyacrylamide gel electrophoresis (10% acrylamide gel) using a Bio-Rad Mini-Protein II system. The protein was transferred to polyvinylidene difluoride membranes (Pierce, Rockford, Illinois). After transfer, the membranes were washed with PBS and were blocked for 1 hour at room temperature with 5% (w/v) skimmed milk powder in PBS. Following blocking, the blots were developed using antibodies for PI3K, pAKt, and β-actin supplied by Thermoscientific (Rockford, Illinois) incubated overnight at pH 7.6 at 4°C with gentle shaking. After washing, peroxidase-labeled secondary antibodies were added, and the membranes were incubated at 37°C for 1 hour. Band intensity was captured and analyzed using ChemiDoc imaging and Image LabTM software version 5.1 (Bio-Rad Laboratories Inc). The results were normalized to β-actin protein expression and expressed as arbitrary units.
Heart Histopathological Examination
Tissues were examined using a light microscope for the existence of edema, inflammatory cells infiltration and necrosis (degenerated muscle fibers presented as shrinkage and loss of myofibrils and loss of striations), and degenerated myocytes (appearing as small cells with large nuclei and scant shrunken cytoplasm with disorganized arrangement). The result was categorized into 5 different grades ranging from grade 0 to 4. Grade 0 indicates no change and 1+, 2+, 3+, and 4+ indicate slight, mild, moderate, and severe histopathological changes, respectively. 36
Immunohistochemistry of Caspase 3 and bcl-2 Expression
For immunohistochemical staining, the heart specimens were deparaffinized and rehydrated. Caspase 3 and B-cell lymphoma 2 (bcel-2)expression were assessed by rat monoclonal antibodies (Abcam, Cambridge, United Kingdom) and examined by light microscope (Olympus cx21, Japan). The percentage of the immunopositive area was estimated using Image J 1.45 F (National Institute of Health).
Statistical Analysis
Data are expressed as mean ± standard deviation (SD). Difference between groups was analyzed using one-way analysis of variance and Kruskal-Wallis test followed by Tukey post hoc test for multiple comparisons. Difference between group means was considered statistically significant when P value was < .05. Data were analyzed using the statistical package for the social sciences (SPSS), version 17.
Results
Effect of Cilostazol or Levosimendan on Serum Lipid Profile in HFD-Myocardial Infarct Rats
Table 1 showed that HFD-fed rats exhibited hyperlipidemia as is evident by the elevation in serum total cholesterol, TG, and LDL levels and decline in HDL level compared to the NPD control group (P < .05; Table 1). Treatment with cilostazol reduced the elevated cholesterol, TG, and LDL levels but enhanced HDL in comparison to HFD group (P < .05). Notably, levosimendan-treated group exhibited a nonsignificant improvement in the severity of elevated LDL and declined HDL levels compared to the HFD control group (P > .05, Table 1).
Effect of Treatment With Cilostazol and Levosimendan on Serum Total Cholesterol, Triglycerides, HDL-C, and LDL-C in HFD-Myocardial Infarct Rats.a,b

Abbreviations: ANOVA, analysis of variance; HFD, high-fat diet; HDL-C, high density lipoprotein-cholesterol; LDL-C, low density lipoprotein-cholesterol; NPD, normal palatable diet; SD, standard deviation.
aRats were fed with a HFD for 12 weeks.
bResults are expressed as mean ± SD and analyzed using one-way ANOVA followed by Tukey post-hoc test at P < .05.
cCompared to NPD group.
dCompared to HFD group.
ecompared to isoproterenol control group.
fCompared to cilostazol, n = 10.
Effect of Cilostazol or Levosimendan on the Percentage of Platelet Aggregation in HFD-Myocardial Infarct Rats
It was evident that the percentage of platelet aggregation was higher (P < .05) in the HFD-fed rats, and HFD + isoproterenol group, when compared to the NPD control group (83.0 ± 5.2 and 86.0 ± 4.8, respectively, versus 58.0 ± 4.1). Both cilostazol and levosimendan revealed effectiveness in attenuating platelet aggregation percentage in comparison to either HFD or HFD + isoproterenol groups (P < .05), without a significant difference between the two treated groups (P > 0.05, Figure 1A).

Effect of cilostazol or levosimendan on (A) platelet aggregation “%,” (B) aortic iNOS “μ/mg protein,” (C) aortic ICAM “pg/mg protein,” and (D) aortic VEGF “pg/mg protein”) in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared with NPD control group, *compared with HFD control group, #compared with isoproterenol control group, and $compared with cilostazol-treated group. ANOVA indicates analysis of variance; HFD, high-fat diet; ICAM, intracellular adhesion molecule; iNOS, inducible nitric oxide synthase; NPD, normal palatable diet; VEGF, vascular endothelial growth factor; SD, standard deviation.
Effect of Cilostazol or Levosimendan on Aortic iNOS, ICAM, and VEGF in HFD-Myocardial Infarct Rats
Figure 1B-D highlighted that the implemented pharmacological agents revealed effectiveness (P < .05) in ameliorating HFD/isoproterenol-induced elevations in aortic iNOS, ICAM, and VEGF levels in comparison to both HFD and isoproterenol control groups. Levosimendan-treated group displayed a significant decline in the higher ICAM marker level when compared to the cilostazol-treated group (P < .05, Figure 1C).
Effect of Cilostazol or Levosimendan on ECG pattern in HFD-Myocardial Infarct Rats
The current results revealed that HFD/isoproterenol injection induced greater ST interval, ST-segment elevation, and QTc duration compared to both NPD and HFD control groups (P < .05, Figure 2). These deleterious effects associated with HFD/isoproterenol injection were ameliorated (P < .05) by treatment with either cilostazol or levosimendan in comparison to isoproterenol control group, with a significant difference between the two treated groups in ST-segment elevation (Figure 2B).

(A) Electrocardiograms in HFD-fed rats subjected to isoproterenol-induced MI. (B) Effect of cilostazol or levosimendan on ECG pattern (a, ST interval” milliseconds,” b, ST-segment elevation “milliseconds,” and c, QTc duration “milliseconds”) in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared with NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. ANOVA indicates analysis of variance; HFD, high-fat diet; MI, myocardial infarction; NPD, normal palatable diet; SD, standard deviation.
Effect of Cilostazol or Levosimendan on Cardiac Enzymes in HFD-Myocardial Infarct Rats
Figure 3 highlighted the effect of HFD/isoproterenol injection on the elevation (P < .05) in cardiac enzymes markers such as LDH, CK, and CK-MB in comparison with either NPD or HFD groups. Cilostazol- and levosimendan-treated groups exhibited amelioration (P < .05) in these elevated parameters in comparison to isoproterenol control group. Levosimendan-treated group displayed a significant (P < .05) decline in the higher cardio-specific serum markers levels when compared to the cilostazol-treated group (P < .05; Figure 3). Effect of cilostazol or levosimendan on cardiac enzymes (A) LDH “U/L,” (B) CK “U/L,” and (C) CK-MB “U/L” in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. ANOVA indicates analysis of variance; CK-MB, creatine kinase-MB; HFD, high-fat diet; LDH, lactate dehydrogenase; NPD, normal palatable diet; SD, standard deviation.
Effect of Cilostazol or Levosimendan on NF-κB Expression and Cardiac TNF-α and IL-6 Content in HFD-Myocardial Infarct Rats
Figure 4 shows that HFD-induced mRNA expression of NF-κB (P < .05) associated with increments in TNF-α and IL-6 content in cardiac tissue homogenates in comparison to NPD control group (P < .05, Figure 4). Isoproterenol injection was accompanied by further elevation (P < .05) in these markers compared to the HFD control group (Figure 4). These elevated markers associated with HFD and isoproterenol injection were ameliorated (P < .05) by treatment with either cilostazol or levosimendan as evidenced by the inhibitory effects on NF-κB, TNF-α, and IL-6 compared to the isoproterenol control group. Notably, levosimendan administration induced a significant improvement in these markers compared to cilostazol-treated group (P < .05, Figure 4).

Effect of cilostazol or levosimendan on inflammatory markers (A) relative NF-κB expression, (B) TNF-α “pg/mg tissue,” and (C) IL-6 “pg/mg tissue” in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. ANOVA indicates analysis of variance; HFD, high-fat diet; IL-6, interleukin-6; NF-κB, nuclear factor kappa-B; NPD, normal palatable diet; SD, standard deviation; TNF-α, tumor necrosis factor α.
Effect of Cilostazol or Levosimendan on Cardiac Oxidative Stress Markers in HFD-Myocardial Infarct Rats
High-fat diet-induced oxidative stress in cardiac tissue homogenates as revealed by the elevated MDA concentrations and the reduced GSH activities compared to the NPD group (P < .05; Figure 5). Isoproterenol-injected rats exhibited aggravation of oxidative stress burden compared to the HFD control group. These deleterious effects associated with HFD/isoproterenol injection were ameliorated (P < .05) by treatment with either cilostazol or levosimendan in comparison to either HFD or HFD/isoproterenol groups. Levosimendan-treated group exhibited significant improvement in oxidative stress burden compared to cilostazol-treated group (P < .05; Figure 5).

Effect of cilostazol or levosimendan on oxidative stress markers (A) MDA “nmol/mg protein” and (B) GSH “nmol/mg protein” in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. ANOVA indicates analysis of variance; GSH, glutathione; HFD, high-fat diet; MDA, malondialdehyde; NPD, normal palatable diet; SD, standard deviation.
Effect of Cilostazol or Levosimendan on Expression of PI3K, pAKt, and mTOR in HFD-Myocardial Infarct Rats
Figure 6 shows that the implemented pharmacological agents revealed effectiveness in enhancing the phosphorylation of PI3K/Akt/mTOR signaling pathway (P < .05) in comparison to either HFD or HFD/isoproterenol groups. It was evident that levosimendan strengthens the activation of PI3K/Akt/mTOR signaling pathway compared to cilostazol--treated group (P < .05; Figure 6).

(A) Effect of cilostazol or levosimendan on the relative expression of (a, PI3K, b, pAKt, and c, mTOR) in HFD-myocardial infarct rats. (B) Representative Western blots for PI3K, pAKt, and their internal standard (β-actin). Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. Akt indicates protein kinase B; ANOVA analysis of variance; HFD, high-fat diet; mTOR, mechanistic target of rapamycin; NPD, normal palatable diet; PI3K, phosphatidylinositide 3-kinases; SD, standard deviation.
Effect of Cilostazol or Levosimendan on the Cardiac Histopathological Picture in HFD-Myocardial Infarct Rats
The histopathological examination of the cardiac tissues revealed that the untreated control group feeding on NPD showed preservation of the normal arrangement of muscle fibers (Figure 7A(a)). However, cardiac tissues in HF-fed rats exhibited degenerated myocytes with loss of cytoplasmic myofibrils (Figure 7A(b)), which were aggravated further by isoproterenol injection (Figure 7A(c)). Both HF-fed and isoproterenol-injected rats were associated with an increase in the mean histopathological score in comparison with the NPD group (P < .05, Figure 7B).

(A) Photomicrographs of heart muscle tissue in control-untreated group feeding on NPD (a) showed the normal arrangement of muscle fibers into fascicles formed of myocytes and abundant eosinophilic cytoplasm rich in myofibrils, showing cytoplasmic striations (black arrow) with oval vesicular nuclei (red arrow). Interstitial tissue showed scanty loose connective tissue with thin walled vessels (green arrow). Rats’ heart muscle tissue of HFD control group (b) showed moderate degenerated myocytes (red arrow) with mild loss of cytoplasmic myofibrils (black arrow). However, rats’ heart muscle tissue of isoproterenol control group (c) showed wide loss of muscle cells replaced by degenerated tissue appear as small cells with large nuclei (red arrow), with few peripheral foci showing remnants muscle fibers (black arrow), and increased cellularity due to infiltration by inflammatory cells (green arrow) including neutrophils and lymphocytes with marked edema (blue arrow). On the other hand, rats’ heart muscle tissue of cilostazol-treated group (d) showed moderate improvement of tissue, focally restored arrangement of muscle fibers into fascicles (black arrow), with few foci showing residual loss of myocytes and degenerated myocytes (red arrow) infiltrated by inflammatory cells (green arrow). There is also residual interstitial edema (blue arrow). Although rats’ heart muscle tissue of levosimendan-treated group (e) showed preserved myocardial architecture with marked improvement of cardiac tissue and normal arrangement of most muscle fibers into fascicles formed of myocytes (red arrow). Very few cells showed residual focal interstitial edema (blue arrow) and degeneration (black arrow; H & E ×400). (B) The histopathological scores in HFD-myocardial infarct rats. Values are mean ± SD (n = 10), analyzed by one-way ANOVA followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol-treated group. ANOVA indicates analysis of variance; HFD, high-fat diet; NPD, normal palatable diet; SD, standard deviation; H & E, hemtoxylin and eosin.
These deleterious effects associated with HFD/isoproterenol injection were ameliorated by administration of either cilostazol or levosimendan (Figure 7A(d and e)). It was obvious that treatment with either cilostazol or levosimendan was associated with a reduction (P < .05) in the mean histopathological score in comparison to the isoproterenol control group (2.16 ± 0.3 and 1.16 ± 0.25, respectively, versus 3.8 ± 0.4; Figure 7B). However, treatment with levosimendan was more beneficial in attenuating the severity of fibrosis and improving the histopathological score in comparison to the cilostazol-treated group (P < .05, Figure 7B).
Effect of Cilostazol or Levosimendan on Apoptotic Markers (Caspase-3 and bcl-2) Immunostaining in HFD-Myocardial Infarct Rats
Figures 8 and 9 highlighted that isoproterenol injection was associated with enhancement of apoptosis (P < .05) as shown by the increment in the percentage of proapoptotic activators “caspase-3” and reduction in antiapoptotic protein bcl-2 immunopositive nuclei when compared to both NPD and HFD (P < .05, Figures 8 and 9). The implemented pharmacological agents reduced apoptosis (P < .05) as shown by the reduction in the proapoptotic activators “caspase-3” percentage and the elevation (P < .05) in the antiapoptotic protein bcl-2 immunopositive nuclei when compared to isoproterenol control group (Figures 8 and 9). However, the levosimendan administration produced a decline in the proapoptotic activators and elevation in the antiapoptotic protein compared to cilostazol-treated group (P < .05; Figures 8 and 9B).
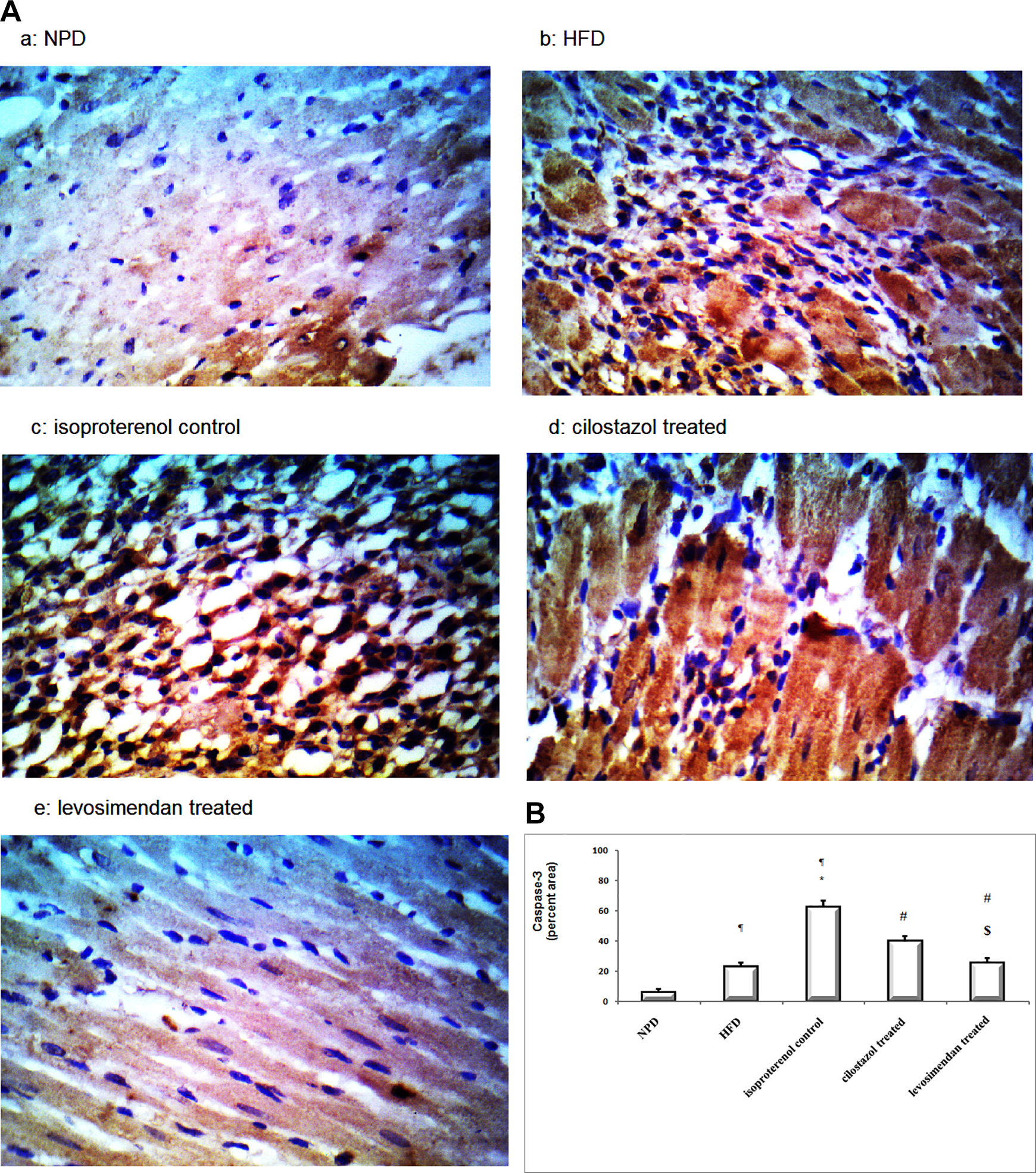
(A) Photomicrographs sections in heart muscle tissue represent the intensity of caspase-3 expression in different groups a, NPD; b, HFD; c, isoproterenolcontrol; d, cilostazol treated; and e, levosimendan-treated (×400). (B): Effect of cilostazol or levosimendan on the expression percentage of the caspase-3 protein in HFD-myocardial infarct rats. Results are expressed as mean ± SD (n = 10), analyzed by Kruskal-Wallis test followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, $compared to cilostazol treated group. HFD indicates high-fat diet; NPD, normal palatable diet; SD, standard deviation.
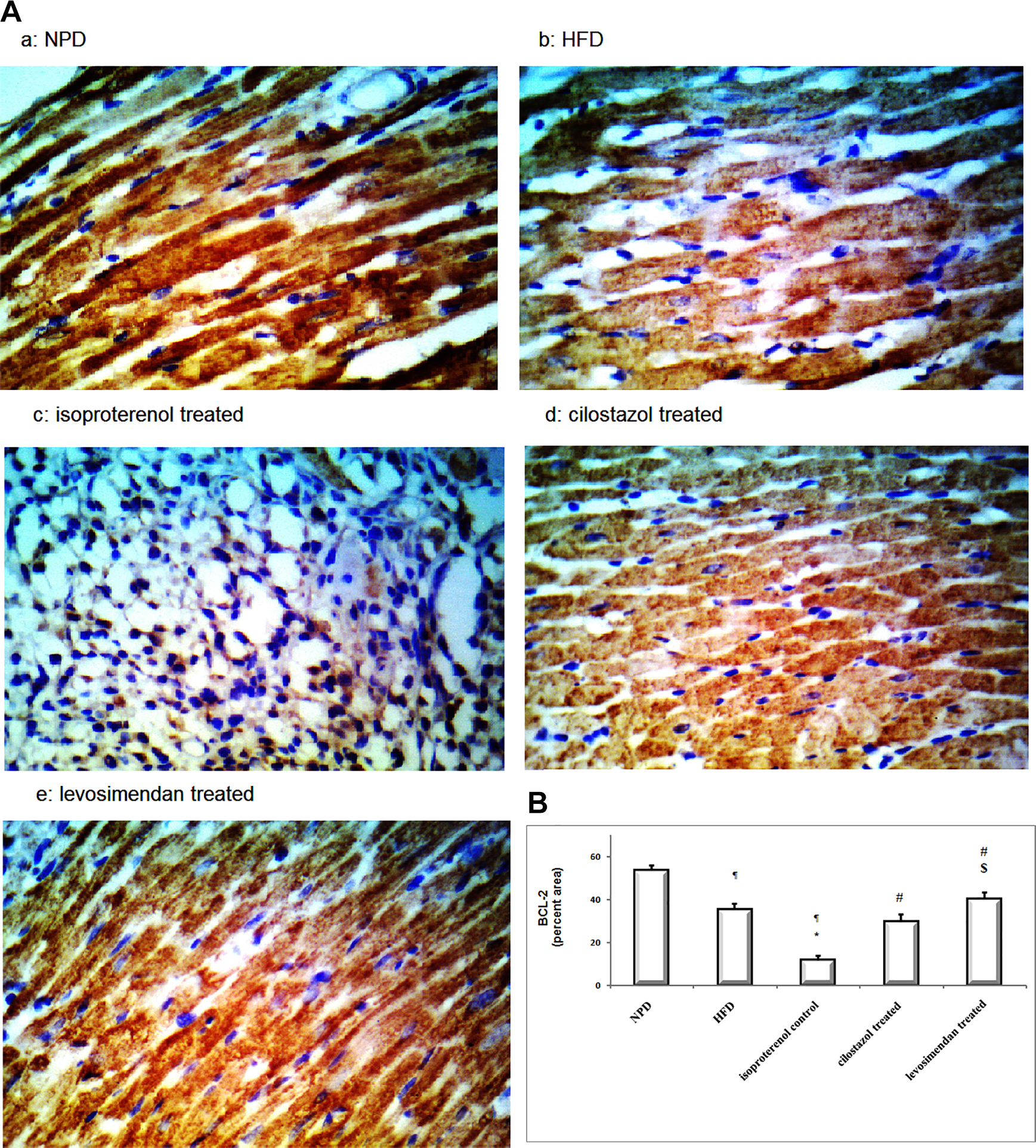
(A) Photomicrographs sections in heart muscle tissue represent the intensity of bcl-2 expression in different groups a, NPD; b, HFD; c, isoproterenolcontrol; d, cilostazol treated; e, levosimendan-treated (×400). (B): Effect of cilostazol or levosimendan on the expression percentage of the bcl-2 protein in HFD- myocardial infarct rats. Results are expressed as mean ± SD (n = 10), analyzed by Kruskal-Wallis test followed by Tukey multiple comparisons test. ¶,*, #, $P < .05; ¶compared to NPD control group, *compared to HFD control group, #compared to isoproterenol control group, and $compared to cilostazol treated group. HFD denotes high-fat diet; NPD, normal palatable diet; SD, standard deviation.
Discussion
High-fat diet is one of the main dietary factors promoting platelet aggregation and involved in the pathophysiology of coronary and other blood vessels’ thrombotic complications. Platelet hyperaggregability in HFD-fed rats is linked to blood vessel inflammation 7 -9 and enhanced Reactive Oxygen Species (ROS) production, 37 which subsequently led to MI. 10 The present study was aimed to evaluate vascular and cardiac protective factors provided by the PDE3 inhibitors, cilostazol and levosimendan, during MI. As PI3K/Akt/mTOR axis is involved in regulating transcription genes associated with myocardial injury, 38 in this context, the present animal study evaluated the modulating role of cilostazol and levosimendan on the PI3K-Akt-mTOR pathways in HFD/isoproterenol-induced myocardial ischemia and hypercoagulable state rat model.
In HFD hypercoagulable state, levosimendan and cilostazol showed reduction in isoproterenol-induced ischemic changes detected by ECG changes and cardio-specific enzyme markers through inhibiting the inflammatory markers NF-κB, TNF-α, and IL-6 expression and oxidative stress burden. Subsequently, levosimendan and cilostazol treatment inhibited apoptotic cell markers via modulation of PI3K/Akt/mTOR pathway. The present study indicates that acute administration of the new calcium sensitizer levosimendan is superior to the commonly used cilostazol regarding decreasing MI, and pro-inflammatory cytokine levels, as well as oxidative stress in isoproterenol-induced MI in the HFD-fed rat model. The schematic representation of the effect of the implemented pharmacological drugs on the isoproterenol-induced myocardial injury in HFD-fed rats is summarized in Figure 10.

The schematic representation of the effect of cilostazol or levosimendan on isoproterenol-induced myocardial injury in high-fat fed rats.
The pathogenesis of vascular inflammation is linked to platelet hyperaggregability in HFD-fed rats, 7,8 in which aortic tissue inflammation enhances iNOS, ICAM, and VEGF activities. 9,39 The present results revealed a possible correlation between hyperlipidemia, hypercoagulability, and inflammation as a risk for atherosclerosis. 7 Previous studies reported the anti-inflammatory effect of cilostazol, as a PDE3 inhibitor, in vascular smooth muscle cells and renal ischemia–reperfusion injury, which is associated with increased cytokines and adhesive molecules levels. 17,40 Additionally, Krychtiuk et al 22 documented the anti-inflammatory effects of levosimendan on human heart microvascular endothelial cells and adult cardiac myocytes. In agreement with previous studies, the current results revealed the inhibitory effect of both cilostazol and levosimendan on HFD/isoproterenol-induced elevations in aortic iNOS, ICAM, and VEGF levels, which are predisposing to vascular injury 41 -43 and subsequent atherosclerosis. 44 The present results are in agreement with other antithrombotic with antioxidative and antiplatelet potentials in preventing the endothelial dysfunction, which is an essential process in the pathophysiology of cardiovascular disease 39,45 besides its anti-atherosclerotic and antiplatelet effects. 18 Levosimendan is more effiecient than cilistazol due to its vasodilator anti-ischemic effects 46 mediated by increasing the blood vesseles smooth muscle cells cAMP level and opening ATP-sensitive K+ channels. 19 -21,47 In addition to levosimendan vasorelaxing effect, it has antiaggregatory effects that accumulate in the vascular tissue and platelets. 26
High-fat diet induces endothelial injury and increases platelet aggregation, 48 offering an excellent background for vascular blockage and consequent myocardial ischemia. In aggrement with other lines of evidence 18,49 -52 which suggested HFD-accelerated platelet aggregation, the present results revealed that HFD-fed rats exhibited higher platelet hyperaggregability and raised serum lipid level. Together, both results were reflected on myocardial ischemia indices including myocardial-specific enzymes and ECG signs.
The current results demonstrated that HFD/isoproterenol-injected rats developed ST-segment elevation refers to acute injury phase for MI. 53,54 A prolonged QT or QTc interval suggests impairment in ventricular recovery and depleted myocardial energy. 55,56 Additionally, ventricular ischemia is reflected by widening QRS interval which reveals disturbance in ventricular conduction. 54 The PR interval prolongation reflects the atrioventricular conduction defect that is reduced in ischemic tissues. 57 In acute myocardial ischemia, hypercholesterolemia increases myocardial ROS and inflammation, which attenuates cell survival and induces myocardial necrosis. 10 Additionally, isoproterenol, as a sympathomimetic β-adrenergic receptor agonist, causes severe myocardial stress and consequently cardiomyocyte necrosis, 58,59 through coronary insufficiency, cellular hypoxia, and oxidative stress. 4
In the present study, the improving anti-ischemic properties of levosimendan on ECG signs was obvious, which is attributed to the opening of K+ATP-sensitive channels in vascular smooth muscle and its subsequent vasodilating effect. 46 Clinical studies suggested levosimendan has short- and long-term improvements in mortality through its effect on coronary, pulmonary, and peripheral circulations. 46 Both cilostazol, through its anti-aggregatory effects, 26 and levosimendan through its anti-atherosclerotic, antiplatelet, and peripheral vasodilatory effects, 17 protect the myocardium against ischemia in HFD experimental models. 18
This improving effect of both cilostazol and levosimendan may in part be related to their antioxidant capabilities. The present results showed that both drugs through their antioxidant effects alter the oxidant–antioxidant balance and attenuate oxidative stress burden in ischemic myocardium. The balance between free radical and scavenger enzymes has been found in ischemic tissue injury. Therefore, oxidative stress burden has been implicated in the development of isoproterenol-induced changes in mitochondria, alterating the enzyme activity and transport systems and causing disturbances in cellular homeostasis. 60 -62 The present results agree with some published data that highlight the critical link between HFD/isoproterenol-induced cardiotoxicity and the development of oxidative stress burden as evidenced by the ROS generation and depression in the endogenous antioxidant system. 3,4,59 Additionally, and in harmony with other studies 17,22,23,43,63 the ischemic ameliorating effect of levosimendan and to a lesser extent cilostazol on increments in the inflammatory markers was evident. The increased cytokine production has been involved in the deleterious effects associated with HFD and isoproterenol injection, providing a connection between inflammation and the progression of myocardial injury. 10 Hypercholesterolemia has been shown to be associated with higher myocardial ischemia-induced injury, in which inflammation-mediated necrosis plays a vital role. 64 In line with this hypothesis, isoproterenol-induced MI in HF-fed rats was associated with the augmented production of the inflammatory mediators, IL-6 and TNF-α, accompanied by NF-κB activation/transcription. 3 The present results are consistent with reports by others supporting the link between the proinflammatory state, oxidative stress burden, NF-κ B activation, and enhancing PI3K/Akt/mTOR signaling pathway. 65,66
Previous reports 11,65,67 documented the positive correlation between activated PI3K/AKT/mTOR pathway and amelioration of ischemic changes in the myocardium. The present results revealed that both levosimendan and cilostazol, through activating PI3K/AKT/mTOR axis, counteract HFD/isoproterenol-induced apoptosis revealed by their inhibitory effects on the apoptotic markers and the histopathological assessment. Zhao et al 65 emphasized that the PI3K/AkT/mTOR pathway is important in regulating the cell cycle and that the serine/threonine kinase Akt is activated downstream of PI3K signaling. Akt plays a critically important role in cellular survival and proliferation, in which mTOR mediate AkT phosphorylation and full activation is a limiting step. 68 Isoproterenol-induced myocardial ischemic hypoxia stimulate hypoxia-inducible factor, 69 which is considered the master of angiogenesis (de novo vessel formation), especially in IHD 70 with potential myocardial salvage capability at early stages after MI. 71 This was confirmed by diminishing the angiogenesis with rapamycin, mTOR inhibitor, 72 and consequently the protective effect on the myocardium. 73 The in vitro cardiac fibroblasts rat model 15 results are compatible with the current in vivo rat model on the activation effect of levosimendan on the PI3K/Akt pathway. Levosimendan reduction in infarct size in the MI rat model was shown to involve phosphorylation of PI3K/Akt pathway and the activation of mitochondrial KATP channels, which is independent of PDE 3 inhibition. 14 Notably, the present results revealed the effectiveness of levosimendan in ameliorating isoproterenol-induced myocardial injury compared to cilostazol due to its antioxidant, anti-inflammatory effects, and decreasing cellular apoptosis. 5,19,24,62 The interest in finding new drugs with anti-inflammatory properties is increasing. One potential treatment of levosimendan, in MI associated with hyperlipidemia, is to reduce the inflammatory mediator’s release, which has shown protective effects on cardiomyocytes in the in vitro study. 22
Significance and future implication
Hyperlipidemia is a risk factor for IHD and MI. The current study can be useful in treating patients in the acute setting of MI, especially in hypercoagulable and hyperlipidemic conditions, where it reduces MI-associated inflammation and subsequent remodeling. Levosimendan and cilostazol would be beneficial in treating chronic atherosclerotic changes and cardiovascular diseases complicated with diabetes. 12,16,18,54 Targeted gene research is needed to determine the clinical importance of levosimendan and cilostazol treatments on neovascularization and modification of cell signaling downstream mTOR pathway on cardiomyocytes viability.
Conclusion
The present results confirmed the antiaggregatory effect of both levosimendan and cilostazol and their effectiveness in counteracting HFD/isoproterenol-induced myocardial injury via counteracting the oxidative stress burden, increments in the expression of inflammatory mediators, and modulating the PI3K/AKT/mTOR axis. To the best our knowledge, the effects of these two pharmacological drugs were compared for the first time in this study, and the protective properties of levosimendan were comparable, but higher, to those of cilostazol. Moreover, the protective effect of levosimendan on aortic injury in hyperlipidemic isoproterenol rat model was evident. These protective effects prove to be useful for improving the protection of the heart after transient coronaries occlusion. Therefore, levosimendan treatment may encourage its consideration as a potential adjuvant to existing drugs, which are utilized to treat patients with platelet hyperaggregability.
Supplemental Material
Supplementry - Cardioprotective and Anti-Aggregatory Effects of Levosimendan on Isoproterenol-Induced Myocardial Injury in High-Fat-Fed Rats Involves Modulation of PI3K/Akt/mTOR Signaling Pathway and Inhibition of Apoptosis: Comparison to Cilostazol
Supplementry for Cardioprotective and Anti-Aggregatory Effects of Levosimendan on Isoproterenol-Induced Myocardial Injury in High-Fat-Fed Rats Involves Modulation of PI3K/Akt/mTOR Signaling Pathway and Inhibition of Apoptosis: Comparison to Cilostazol by Mona K. Tawfik, Mohamed K. El-Kherbetawy, and Samy Makary in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
The authors thank Prof Laila Rashed, Biochemistry Department, Faculty of Medicine, Kasr Aini University.
Authors’ Contribution
Mona K. Tawfik and Samy Makary contributed to conception, design, acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Mohamed K. El-Kherbetawy contributed to conception, acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
