Abstract
Introduction:
Pulmonary hypertension (PH) is a potentially deadly disease for infants and adults with few existing medical interventions and no cure. In PH, increased blood pressure in the pulmonary artery eventually leads to heart failure. Fasudil, an antagonist of Rho-kinase, causes vasodilation leading to decreased systemic artery pressure and pulmonary artery pressure (PAP). This study compared the effects of fasudil administered as either an intravenous infusion or inhaled aerosol in newborn lambs.
Hypothesis:
Inhaled aerosol delivery of fasudil will provide selective pulmonary vasodilation when compared with intravenous administration.
Methods:
Newborn lambs (∼11 days) were surgically instrumented and mechanically ventilated under anesthesia. A pulmonary artery catheter and ultrasonic flow probe were inserted to measure hemodynamics. Acute PH was pharmaceutically induced via continuous intravenous infusion of thromboxane. After achieving a 2- to 3-fold elevation of PAP, fasudil was administered either as intravenous infusion (2.5 mg/kg) or inhaled aerosol (100 mg of fasudil in 2 mL of saline). Changes in PAP, mean systemic arterial pressure (MABP), pulmonary vascular resistance (PVR), systemic vascular resistance (SVR), cardiac output, and heart rate were assessed. In addition, plasma concentrations of fasudil were measured.
Results:
Both routes of fasudil delivery produced significant decreases in PAP and PVR but also produced similar decreases in MABP and SVR. The Cmax for intravenous fasudil was greater than that for inhaled fasudil.
Conclusions:
These results suggest inhaled fasudil lacks pulmonary selectivity when compared with intravenous fasudil.
Background
Pulmonary hypertension (PH) is a devastating disease that afflicts patients of all ages, from neonates to adults. Pulmonary hypertension is characterized by increased pressure in the pulmonary arteries (resting mean pulmonary artery pressure [PAP] ≥25 mm Hg) and increased pulmonary vascular resistance (PVR ≥ 3 Wood units) and has many underlying etiologies. 1
In adults, the incidence and prevalence of pulmonary arterial hypertension World Health Organization [WHO] group 1 is 2.4 cases per million and 15.0 cases per million population per year, respectively, with a 5-year mortality of approximately 40%. 2 In children, the incidence and prevalence of pulmonary arterial hypertension World Health Organization (WHO group 1) is 0.48 cases per million and 2.1 cases per million children, respectively. 3 The death rate for PH as any contributing cause of death has increased from 5.5 per 100 000 in 2001 to 6.5 per 100 000 in 2010, with hospitalizations related to PH increasing by 44% during the same period despite modern therapy. 2
The biological mechanisms leading to PH are complex and not yet completely elucidated but include sustained vasoconstriction, progressive vascular remodeling, metabolic alterations, inflammation, and ultimately right heart dysfunction. 4 In short, PH has poorly understood etiology, ineffective therapy, and a high mortality, pointing to the need for further studies for novel therapies with alternative mechanisms of action.
Rho-kinase is a GTPase important to many myocyte functions including smooth muscle tone, cell growth, and proliferation 5 -7 and has been proposed by the Fifth World Symposium on Pulmonary Hypertension to be a potential therapeutic target in the future. 8 Recent evidence from our group and others show Rho-kinase inhibition lowers elevated pulmonary vascular pressures in various clinical and experimental models of PH. 6,9 -13 Rho-kinase regulates vascular contraction by modulating myosin phosphorylation, and it influences cell growth and proliferation by regulating transcription. 14 We and others have demonstrated that treatment with Rho-kinase inhibitors in animal models results in rapid and profound pulmonary vasodilation. 7,9,15 -17 In addition, others have demonstrated that chronic treatment reduces pulmonary vascular tone, arterial wall thickening, and right ventricular hypertrophy. 11,18,19 In newborn rats, Rho-kinase inhibition improves lung vascularization and alveolar arborization. 18 Early-phase clinical trials show that intravenous or oral fasudil treatment for PH resulted in significant reductions in PAP. 12,20 Fasudil has been approved for clinical use in Japan to prevent cerebral vasospasm secondary to subarachnoid hemorrhage. 21,22 However, Rho-kinase is important to the contraction of systemic as well as pulmonary arteries, and thus, its clinical usefulness can be limited by systemic hypotension. 9,23 Inhaled fasudil has been studied in adult humans where selective pulmonary vasodilation was observed. 12,15 However, other human studies show significant systemic hypotensive and cardiodepressive effects of fasudil. 8 Additional studies with nanoliposome-encapsulated fasudil in adult rats have also demonstrated selective pulmonary vasodilation. 24 -26 However, the effects of pulmonary-targeted fasudil administration have not been studied in newborns or in large animal models where direct measurement of pulmonary pressure and flow is possible. Thus, we tested the hypothesis that inhaled fasudil selectively reduces pulmonary vascular pressures without causing systemic hypotension in a newborn sheep model of acute PH.
Methods
All animal protocols and handling procedures were approved by the Institutional Animal Care and Use Committee of Loma Linda University and were in accordance with the National Institutes of Health guidelines for use of experimental animals. Lambs were obtained from Nebeker Ranch (Lancaster, California). Unless otherwise noted, all chemicals and supplies were obtained from Sigma-Aldrich (St Louis, Missouri).
Animal Preparation
The study was performed in a newborn lamb model of thromboxane-induced acute PH as previously described.
27
After induction of anesthesia with intravenous ketamine (10 mg/kg; Vet One, Boise, Idaho) and midazolam (0.5 mg/kg; Akorn Pharmaceuticals, Lake Forest, Illinois), 7- to 14-day-old lambs were intubated and mechanically ventilated with 1% to 2% isoflurane in O2 during surgical instrumentation. Both femoral veins were catheterized, one with a Swan-Ganz balloon wedge catheter (AI-07123, 5F × 60 cm; Arrow International Inc, Reading, Pennsylvania) advanced to the pulmonary artery and the other for infusion of the thromboxane analog U46619 (Cayman Chemical, Ann Arbor, Michigan) to induce PH as described below. A femoral artery was catheterized for blood gas sampling and collection of plasma samples for fasudil analysis. A carotid artery catheter was inserted for measurement of arterial blood pressure. The jugular vein was catheterized for administration of fasudil, ketamine, and vecuronium. An esophageal temperature probe and a pulse oximeter placed on the tongue were used to monitor body temperature and oxyhemoglobin saturations, respectively. Via thoracotomy, a transonic pulmonary artery flow probe (serial #: 10PAA175; Transonics, Ithaca, New York) was placed on the pulmonary artery to measure pulmonary artery blood flow. Following surgical instrumentation, isoflurane was discontinued and the lambs received intravenous ketamine (1 mg·kg−1·h−1) and vecuronium (0.1 mg·kg−1·hr−1; Sun Pharmaceutical Industries Inc, Cranbury, New Jersey) for the remainder of the experiment. Mechanical ventilation and inspired O2 were adjusted to achieve stable baseline arterial blood gases (P
Study Protocol
Following surgical instrumentation and a stable baseline period, U46619 was infused intravenously at a rate titrated to increase mean pulmonary pressures to 35 to 40 mm Hg (Figure 1). After 10 minutes of U46619 infusion, lambs received 1 of 3 treatments: (1) intravenous fasudil (SelleckChem, Houston, Texas; 2.5 mg/mL dissolved in saline) infused via syringe pump (model 22; Harvard Apparatus, Holliston, Massachusetts) at a rate of 2.5 mg/kg over 15 minutes, (2) inhaled fasudil (100 mg in 2 mL saline solution) delivered via nebulizer (Aeroneb Pro; Aerogen Ltd, Galway, Ireland) over a period of 2 to 5 minutes depending on the rate of fluid nebulization, or (3) inhaled saline (2 mL) as negative controls. The intravenous fasudil dose was the same as used previously in lamb studies 9 and similar to that used in early-phase clinical studies in Japan. 12,20,21 The inhaled dose was based on a predicted delivery of 10% of the nominal dose to the lung, with 90% deposited in the endotracheal tube adaptor, endotracheal tube, or exhaled. 28,29 There are newer methods of targeting drug delivery to the lung, such as nanoparticle inhalation, 25 which we plan to incorporate in the future. U46619 infusion was continued until 30 minutes after the start of fasudil or saline administration, and lambs were monitored for an additional 50 minutes until euthanization with intravenous euthasol (100 mg/kg; Virbac, Ft Worth, Texas).
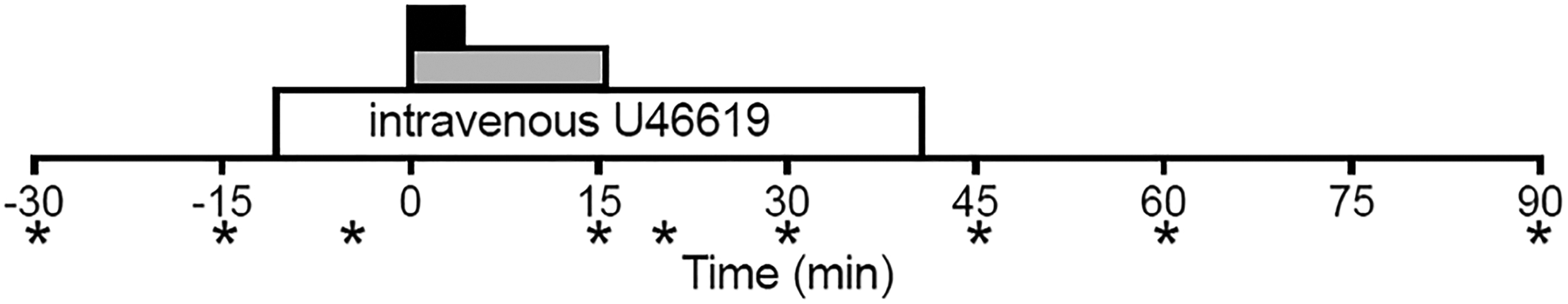
Black bar represents period of up to 5 minutes (T = 0 to 5 minutes) of inhaled fasudil. Gray bar represents 15-minute period (T = 0 to 15 minutes) of intravenous fasudil. White bar represents 50-minute period (T = −10 to 40 minutes) of intravenous thromboxane analog (U46619). Arterial blood samples (indicated by *), taken at time points −30, −15, −5, 15, 20, 30, 45, 60, and 90 minutes.
Plasma Fasudil Assay
Arterial blood samples (2 mL) for blood gases and fasudil analysis were collected in heparinized syringes at −30, −15, −5, 15, 20, 30, 45, 60, and 90 minutes relative to the start of fasudil administration. Plasma for fasudil assay was isolated by centrifugation of blood immediately following collection. Samples were snap-frozen in liquid nitrogen and stored at −80°C until analysis.
Fasudil concentrations were quantified by tandem mass spectrometer (AB QTRAP 5500; SCIEX, Framingham, Massachusetts) attached to an Ultra Performance Liquid Chromatography (UPLC) system (Shimadzu; Agilent Technology, Santa Clara, California) via an electrospray ionization (ESI) interface. Using ranitidine (GlaxoSmithKline, Philadelphia, Pennsylvania) as an internal standard, calibration curves were prepared in sheep plasma after extraction with methanol. For drug extraction, 500 μL of methanol was added to 100 μL of plasma, vortexed for 30 seconds, centrifuged at 11 000 rpm for 5 minutes, then transferred 500 μL of the supernatant to glass vials, and evaporated to dryness under nitrogen at 40°C. Later, the residue was reconstituted in 100 μL of 50% methanol, vortexed again, and 20 μL of reconstituted solution injected into LC–MS/MS machine.
For chromatographic separation, a 100 mm × 2.1 mm XB C18 column (Phenomenex, Torrance, California) attached to a 12.5 mm × 2.1 mm guard column was used with a gradient elution (2 mM ammonium formate in water, modified with 0.1% formic acid as eluent A and methanol as eluent B) at a flow rate of 0.4 mL/min. The drug was quantified via multiple reaction monitoring method, keeping the ESI source at a positive ionization mode, the transitions m/z 292.2 → 99.2 and m/z 315.3 → 176.2 for fasudil and internal standard ranitidine, respectively, with a dwell time of 200 milliseconds per transition. The amount of drug was read from a calibration curve prepared by plotting m/z area for fasudil against fasudil concentration.
Data Handling and Analysis
Blood pressure was measured using pressure transducers calibrated prior to each experiment (Cobe, Lakewood, Colorado). Pressure and flow signals were collected at 200 Hz via an analog-to-digital converter (Powerlab 16; ADInstruments Inc, Colorado Springs, Colorado) and recorded on computer via LabChart software v.6.1.3 for Macintosh (ADInstruments). One-minute averages of continuously recorded parameters were exported to Microsoft Excel for determination of calculated parameters. Pulmonary vascular resistance (PVR) and systemic vascular resistance (SVR) were calculated as: (mPAP − PAWP)/CO and (MAP − CVP)/CO, respectively, where mPAP = mean PAP, PAWP = pulmonary artery wedge pressure, CO = cardiac output, MAP = mean arterial pressure, and CVP = central venous pressure. 9,27,28 Cardiac output was taken to be equivalent to measured pulmonary artery flow, and central venous pressure was assumed to be constant throughout the study.
Statistical Analysis
Power analysis determined the number of animals necessary in each group to be ≥5 with an α = .05 and β = .8 and effect size of >0.4 (G*Power v3.1; University of Dusseldorf, Germany). Graphs were prepared and statistical analyses were conducted in Prism for Mac OS X (version 7.0a; GraphPad Software, Inc, La Jolla, California). The data are expressed as means ± standard error. After determining the data were normally distributed using Shapiro-Wilk and Kolmogorov-Smirnov tests, 1-way analysis of variance (ANOVA) was utilized to determine significant changes over time. Two-way ANOVA was used to detect significant differences between study groups. When appropriate, Tukey or Dunnett post hoc analyses were used for multiple comparisons. Unless noted otherwise, P value <.05 was accepted for statistical significance.
Results
A total of 18 lambs were studied (7 males and 11 females), with 7 in the control group (3 males and 4 females), 5 in the intravenous fasudil group (3 males and 2 females), and 6 in the inhaled fasudil group (1 male and 5 females). The mean age was 11.1 ± 0.3 days and mean weight was 5.7 ± 0.3 kg.
There were no significant differences between study groups in arterial pH, P
Cardiovascular Responses
U46619 infusion resulted in a rapid acute PH that was successfully titrated to result in elevated mean PAP that was comparable between the 3 study groups at 1 minute prior to the start of saline (controls), intravenous, or inhaled fasudil (35.3 ± 2.5 mm Hg, 36.9 ± 1.2 mm Hg, and 41.6 ± 1.0 mm Hg, respectively, P = .2; Table 1). Likewise there were no significant differences between groups in the mean values of PVR, cardiac output, mean arterial blood pressure, SVR, heart rate, or stroke volume.
Hemodynamic Values at Selected Protocol Time Points.
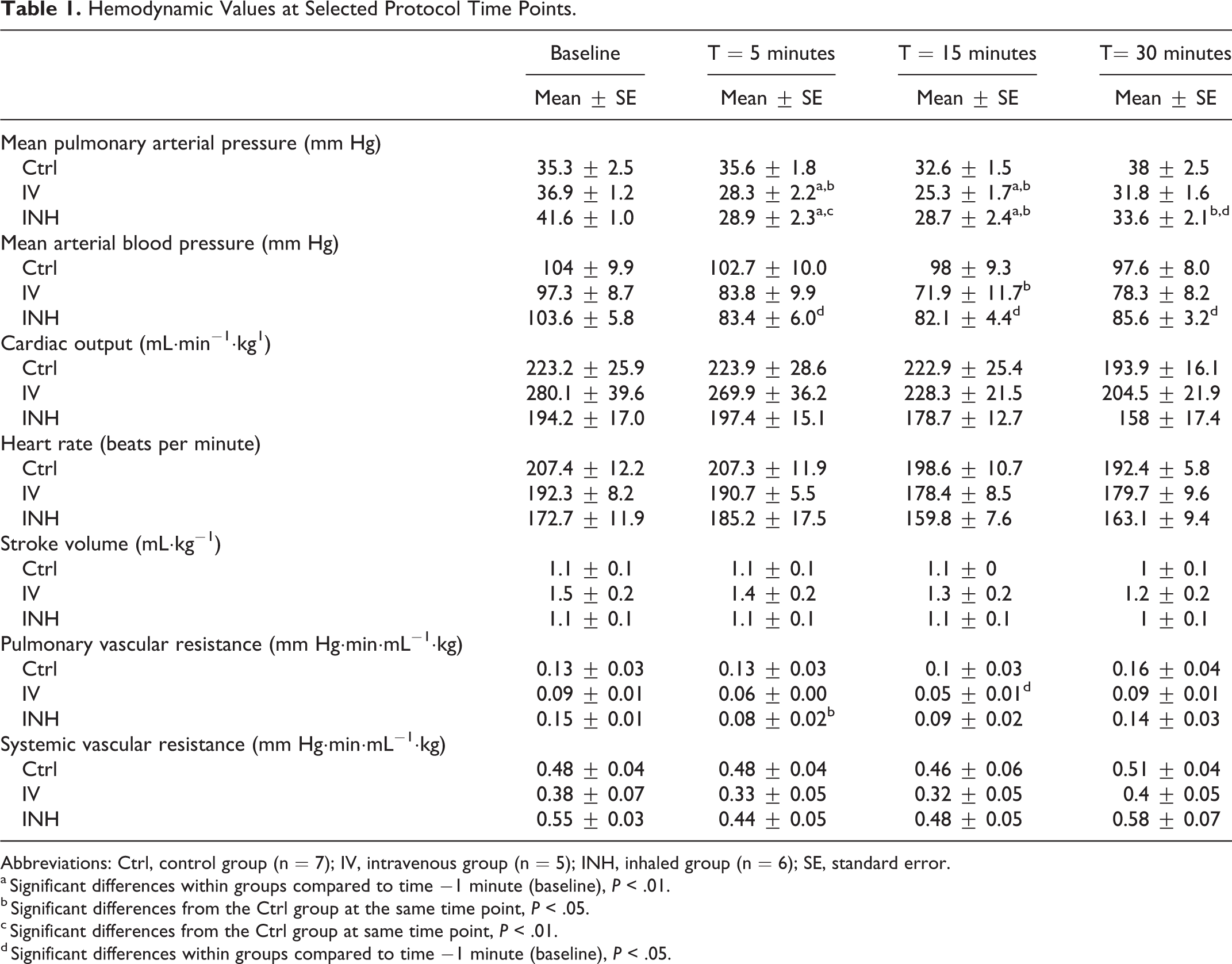
Abbreviations: Ctrl, control group (n = 7); IV, intravenous group (n = 5); INH, inhaled group (n = 6); SE, standard error.
a Significant differences within groups compared to time −1 minute (baseline), P < .01.
b Significant differences from the Ctrl group at the same time point, P < .05.
c Significant differences from the Ctrl group at same time point, P < .01.
d Significant differences within groups compared to time −1 minute (baseline), P < .05.
There were no significant changes in mean PAP in response to inhaled saline in the control animals (Figure 2A). In contrast, PAP was significantly decreased at 5 and 15 minutes after the start of intravenous fasudil and at 5, 15, and 30 minutes after the start of inhaled fasudil (P < .05; Figure 2A). While both intravenous and inhaled fasudil resulted in significantly decreased PAP compared to controls, there were no significant differences between the 2 fasudil groups. In addition, pulmonary artery wedge pressures did not differ significantly between the 3 study groups throughout the study.

Hemodynamic responses of animals treated with U-46619-induced pulmonary hypertension to inhaled saline (control, black bar), intravenous fasudil (infused, white bar), or inhaled fasudil (inhaled, gray bar). A-G, Data displayed as percentage change from baseline. *Significant differences from baseline. †Significant differences from control. */†P value <.05. **/††P value <.01. A, Mean pulmonary artery pressure (PAP) dropped significantly compared to baseline and control at 5 and 15 minutes after the start of intravenous fasudil and at 5, 15, and 30 minutes after the start of inhaled fasudil. B, Mean arterial blood pressure dropped significantly in the inhaled group at all time points compared to baseline and in the infused group at 15 minutes compared to control. C-E, No significant changes in cardiac output, heart rate, and stroke volume. F, Pulmonary vascular resistance was lower in both fasudil groups, significant from control at 5 minutes for inhaled and from baseline at 15 minutes for the infused group. G, No significant changes in systemic vascular resistance (SVR).
Similar to PAP, mean systemic blood pressure did not change significantly from baseline in the control group (Figure 2B). Mean arterial pressures decreased significantly at 15 minutes in the intravenous fasudil group when compared to saline controls but were not different when compared to baseline values or to the inhaled group. Mean arterial pressures were significantly decreased in the inhaled fasudil group when compared to baseline values, with post hoc analysis indicating significance at 5, 15, and 30 minutes (P < .05, Figure 2B). Important to the hypothesis that inhaled fasudil would achieve more selective pulmonary vasodilation with minimal systemic effects, no significant differences in mean arterial blood pressure changes were observed between the intravenous and inhaled groups.
Cardiac output tended to decrease relative to baseline levels in all study groups, particularly the infused fasudil groups by the 30-minute time point; however; this change was not statistically significant (Figure 2C). There were also no significant differences in cardiac output between the 3 study groups. Likewise, although heart rate and stroke volume tended to decrease from baseline values by the end of the study protocol, these changes were not statistically significant nor were differences between study groups (Figure 2D and E).
Pulmonary and systemic vascular resistances are shown in Figure 2F and G, respectively. Pulmonary vascular resistance did not change significantly from baseline in the control animals. Both intravenous and inhaled fasudil groups had lower PVRs, significant from control at 5 minutes for inhaled and from baseline at 15 minutes for infused groups. However, there were no significant differences between the intravenous and inhaled groups. Changes in SVR did not reach statistical significance for any group over time, and there were also no differences between groups.
Fasudil Pharmacokinetics
The time course of plasma fasudil concentrations for the intravenous and inhaled groups is shown in Figure 3A. The average maximum plasma fasudil concentration (Cmax) was 809.5 ± 75.3 (ng/mL) for the intravenous group and 133.9 ± 49.4 (ng/mL) for the inhaled group (P < .01; Figure 3B). The mean time of peak plasma fasudil concentrations was similar between the intravenous and inhaled groups (20.8 vs 20.0 minutes, respectively, P = .8). Plasma fasudil concentrations had fallen to less than 10% of peak concentrations by collection of the 90-minute sample in both inhaled and intravenous groups.

Plasma concentrations of fasudil following intravenous or inhaled administration. A, Time course of plasma concentrations measured in samples collected over the 90 minutes following dosing (*P < .05 infused vs inhaled, 2-way analysis of variance [ANOVA]). B, Average of peak plasma concentrations measured in the intravenous group was significantly greater than that of the inhaled group (t test).
Discussion
The results of this study show that fasudil decreases pulmonary and systemic arterial pressures and vascular resistances, suggesting that Rho-kinase-mediated Ca2+ sensitization has a constitutive role in regulating vascular tone in the pulmonary and systemic vascular beds in newborn sheep with acute PH caused by thromboxane (U46619). The novel finding of this study is that inhaled fasudil, when compared to intravenous fasudil, does not provide selective pulmonary vasodilation.
Pharmacological Considerations
The inhaled fasudil in our study was delivered as 100 mg dissolved in saline and nebulized into the inhaled air over a period of 2 to 5 minutes, depending on the performance of the nebulizer. This dose, based on a predicted delivery of approximately 10% of the fasudil to the distal airways, 29,30 would result in a fasudil dose of approximately 1.75 mg/kg, which is comparable to the intravenous dose of 2.5 mg/kg. In contrast, peak plasma concentrations in the intravenous animals were 4-fold greater than those of the inhaled animals. This finding suggests that much less than 10% of the nebulized fasudil may have been delivered to the animals. However, this would be unexpected given that the animals were receiving mechanical ventilation with tidal volumes and a circuit configuration within the specifications of the nebulizer and Afectair adapter which optimizes drug delivery. 30 The unexpectedly lower peak plasma fasudil concentrations in the inhaled animals could also be explained by poor uptake of fasudil from the lungs into the circulation. However, both intravenous and inhaled animals reached peak plasma concentrations at ∼20 minutes and had largely disappeared by 90 minutes, suggesting inhaled fasudil is absorbed rapidly and thus not likely to persist at high concentrations in the lung. A third possible explanation for the discrepancy in plasma concentrations could be that fasudil is efficiently metabolized in the lung, thus preventing a large portion of the inhaled dose from reaching systemic circulation. Further studies are needed to assess this possibility.
Inhaled Versus Intravenous Fasudil
Consistent with previous reports, the current study demonstrated that fasudil effectively lowers pulmonary vascular tone following both intravenous and inhaled administration. However, contrary to our main hypothesis and unlike other studies showing pulmonary selective effects of inhaled fasudil, 12,15 we did not observe selective pulmonary vasodilation in the inhaled group compared to the intravenous group. This unexpected finding is contrary to previous findings in adult humans, where an inhaled dose of 30 mg over 10 minutes (∼0.5 mg/kg based on an estimated 65 kg body weight) resulted in a significant decrease in PAP but with no effects on systemic arterial pressure. 12 However, consistent with our finding, intratracheal administration of 10 mg/kg to adult rats was found to have both pulmonary and systemic vasodilatory effects. 25 On a weight-adjusted basis, the dose of inhaled fasudil in the human study is notably lower than that of the current study and previous rat study. Thus, it is possible that pulmonary selectivity is achievable at lower inhaled doses. However, given that plasma concentrations were ∼4-fold lower in the inhaled compared to the intravenous group, with no difference between the systemic responses in the 2 groups, this possibility would also imply that the plasma concentrations achieved with the intravenous dose were >4-fold higher than levels needed to reach a maximal response. The dose of fasudil used in the intravenous group (2.5 mg/kg over 15 minutes) is the same we used previously in our studies 9 and similar to that used in early-phase clinical studies in Japan, 10,12 as well as in studies in dogs, 31 rabbits, 32 and rats. 33,34 Dose–response experiments with U-46619-induced PH in adult rats indicate that an intravenous dose of 2.5 mg/kg is not likely to have maximal effects. 34 However, extrapolation of results from monocrotaline-induced PH model in adult rats to thromboxane-induced PH model in newborn lambs is admittedly tenuous, particularly considering (1) interspecies differences, (2) the possibility that newborn animals may have heightened responses to fasudil, 18 and (3) acute models of PH do not display similar degree of vascular remodeling as that seen in chronic PH models. Dose–response studies comparing newborns and adults in various species in acute and chronic PH models are necessary to adequately address this.
The average time to peak plasma fasudil concentrations was similar between the 2 routes of administration, suggesting inhaled fasudil enters the systemic circulation with kinetics similar to that of an intravenous infusion. Although plasma concentrations were not sampled frequently enough to enable determination of the elimination half-life, the finding that plasma concentrations had fallen by >90% at the 90-minute time point suggests that the circulation half-life of fasudil in newborn lambs is comparable to or shorter than that of rats (∼0.39 hours) 33 and humans (0.5 hours) 20 following both inhaled and intravenous administration. The lack of an obviously prolonged half-life of inhaled versus intravenous fasudil, as has been reported in adult rats, 33 indicates that the comparable systemic effects at lower Cmax cannot be explained by a prolonged systemic exposure following inhaled fasudil.
The observation of comparable systemic effects despite lower peak plasma concentrations could be explained by differences in the metabolic pathways of fasudil for the 2 routes of administration. For example, hydroxyfasudil, a major metabolite of fasudil with a much longer half-life (3.6 hours), 20 has even more potent Rho-kinase antagonist activity than fasudil. 35 Thus, greater systemic effects relative to plasma fasudil concentrations may have been due to a greater portion of fasudil conversion to hydroxyfasudil following inhaled versus intravenous administration. Further metabolite profiling work is necessary to fully explore this possibility. It is however important to note that the time to peak plasma concentration for both inhaled and intravenous fasudil groups was similar at 20 minutes (Figure 3A). Thus, difference in rate of systemic absorption of fasudil per se does not necessarily explain the difference in hemodynamic effects between the 2 methods of delivery.
Fasudil has major effects on the systemic circulation as shown in multiple animal and human studies. 6 -8 One study suggests that fasudil can vasodilate U-46619-induced constriction of coronary arteries. 32 It is known that the bioactive metabolite hydroxyfasudil has possible nonselective effects by inhibiting other serine/threonine kinases and acts on the endothelium in addition to smooth muscle cells. 23,36 Fasudil metabolism is different among species 37 and may also vary with developmental age. Thus, without measurement of hydroxyfasudil in our experiments, it is difficult to ascertain whether it plays a role in the systemic vascular and cardiac effects we observed.
Limitations
Our study has several limitations. First, we did not measure concentrations of fasudil metabolites or Rho-kinase activity. Based on our results, future studies should incorporate measurement of hydroxyfasudil and some form of quantification of Rho-kinase activity such as myosin light chain phosphorylation and levels of total and phosphorylated ERM proteins (ezrin, radixin, and moesin). 18,34
Secondly, although the mean PAP was not significantly different prior to the start of saline/fasudil administration (35.3 ± 2.5 mm Hg, 36.9 ± 1.2 mm Hg, and 41.6 ± 1.0 for controls, infused, and inhaled, respectively, P = .2), it is possible that the higher starting pressures in the inhaled fasudil group resulted in an enhanced efficacy of inhaled fasudil despite lower peak plasma concentrations, a phenomenon reported in other acute PH animal models using thromboxane. 38 In addition, the dose of U46619 required to achieve the target mean PAP varied significantly from animal to animal, which may have been due to interanimal variation in hydration status and volume of distribution which we did not assess in our study.
Finally, use of thromboxane/U46619 to elicit acute PH does not mimic the vascular remodeling associated with chronic PH. In adult rats, fasudil is reported to prevent and reverse vascular remodeling associated with PH induced by chronic hypoxia, 39 and thus, studies in newborn lamb models of chronic PH may be warranted. Our study did not evaluate vascular remodeling effects of fasudil. Furthermore, our study was conducted in newborn lambs with highly elastic arteries and a forgiving right ventricle and does not replicate chronic PH encountered in clinical setting.
Conclusions
The present study suggests that in a model of thromboxane-induced acute PH in newborn sheep, PAP was significantly reduced by inhaled and intravenous fasudil. However, inhaled fasudil was not selective to the pulmonary vasculature and reduced systemic artery pressure as well. Further studies to enhance pulmonary specificity of inhaled fasudil are required.
Footnotes
Acknowledgment
The authors acknowledge the Department of Internal Medicine at Loma Linda University School of Medicine for awarding this study with the Research Innovation Grant (681206-2967) and Shannon Bragg for providing skillful surgical assistance for the experiments.
Authors’ Contribution
S. F. L. Hanson contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. D. T. Moretta contributed to design, analysis, and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. M. H. Terry contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. G. G. Power contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. S. M. Wilson contributed to conception and design, contributed to analysis, and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. F. Alam contributed to design, analysis, and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. F. Ahsan contributed to design, analysis, and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. A. B. Blood contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. P. C. Giri contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Internal Medicine at Loma Linda University School of Medicine for awarding this study with the Research Innovation Grant (681206-2967) This work was supported in part by an NIH grant (R01HL114677) awarded to Fakhrul Ahsan.
